You’re Missing This: Why mTOR Activation Is the Real Goal of Strength Training
Discover why mTOR activation—not just lifting weights—is the real driver of muscle growth, fat loss, and metabolic health. Learn how resistance training reprograms your body at the cellular level to improve insulin sensitivity, build lean muscle, and slow aging.
EXERCISEMETABOLISM
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/29/202617 min read
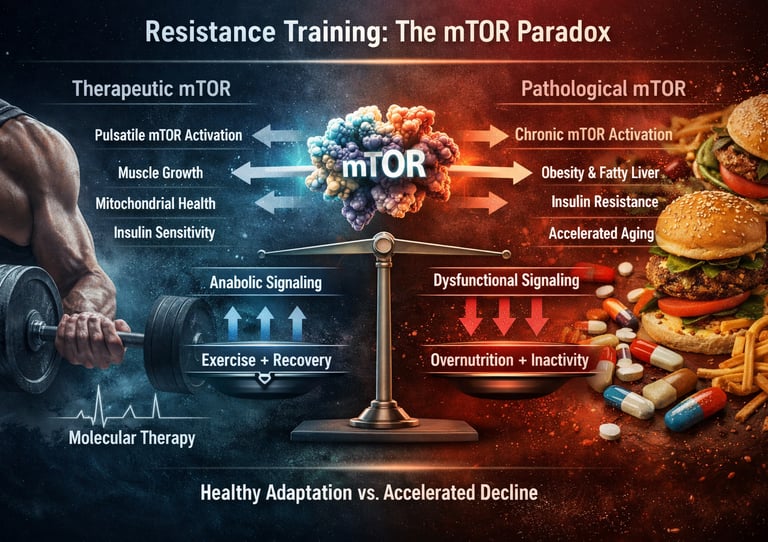
Resistance training is a form of anabolic medicine that activates key molecular pathways—particularly mTOR (mechanistic target of rapamycin)—to stimulate muscle protein synthesis, improve insulin sensitivity, and enhance mitochondrial function. Through mechanotransduction, mechanical load applied to muscle fibers is converted into biochemical signals that promote hypertrophy and metabolic adaptation. When performed with proper intensity, progression, and recovery, resistance training produces pulsatile mTOR activation, which supports muscle growth while preserving metabolic health. This makes it a powerful, evidence-based intervention for preventing and managing conditions such as sarcopenia, type 2 diabetes, and cardiovascular disease.
How Resistance Training Works as Anabolic Medicine:
Activates mTORC1 to stimulate muscle protein synthesis
Converts mechanical load into cellular signals via mechanotransduction
Improves insulin sensitivity and glucose uptake (GLUT4 activation)
Enhances mitochondrial function and metabolic flexibility
Produces pulsatile (not chronic) anabolic signaling
Prevents sarcopenia and metabolic disease
How to Use Resistance Training as Anabolic Medicine
Train 3–4 times per week using compound movements
Apply progressive overload (increase weight, reps, or volume)
Maintain moderate to high intensity (6–12 rep range for hypertrophy)
Consume 1.6–2.2 g/kg/day protein, with leucine-rich sources
Prioritise post-workout protein intake (20–40 g)
Combine with aerobic training for mitochondrial health
Ensure 7–9 hours of sleep for optimal mTOR signaling and recovery
Clinical Pearls
1. The Anabolic Resistance Threshold
The Science: Aging is characterized by "Anabolic Resistance," where the skeletal muscle’s mTORC1 response to both mechanical tension and amino acids is blunted. To overcome this, the "Leucine Threshold" must be higher than in younger cohorts.
As we get older, our muscles' "ears" get a bit hard of hearing. To get the message across that it’s time to grow and repair, we have to "speak louder" by eating more high-quality protein and lifting slightly heavier weights than a 20-year-old might need to.
2. The "Metabolic Sink" Effect
The Science: Skeletal muscle is the primary site for postprandial glucose disposal (up to 80%). Increasing GLUT4 translocation through mechanotransduction creates a "metabolic sink," drastically improving insulin sensitivity independent of insulin signaling.
Think of your muscles as a giant sponge for blood sugar. Every time you lift weights, you’re "wringing out" the sponge so it can soak up more sugar from your blood. This keeps your energy steady and protects your heart and organs.
3. Myokine Signaling (The Pharmacy Within)
The Science: Contracting muscle acts as an endocrine organ, secreting "myokines" like IL-6 (which, in an exercise context, is anti-inflammatory) and Irisin. These cross the blood-brain barrier, stimulating BDNF and improving cognitive resilience.
Your muscles are actually a natural pharmacy. When you exercise, they release specialized "messenger chemicals" that travel to your brain to improve your mood, sharpen your memory, and reduce inflammation throughout your entire body.
4. The Concurrent Training "Interference" Myth
The Science: The "Interference Effect" (where cardio kills gains) is largely overstated in clinical populations. Low-to-moderate intensity "Zone 2" aerobic work actually enhances recovery by increasing capillary density, which improves the delivery of nutrients to the mTOR-activated satellite cells.
You don't have to choose between a strong heart and strong muscles. Doing some light walking or cycling alongside your weightlifting actually helps your muscles recover faster by improving blood flow—bringing in the "building materials" your body needs to heal.
5. The "Sarcopenic Obesity" Paradox
The Science: In metabolic disease, weight loss via caloric restriction alone often leads to a disproportionate loss of Lean Body Mass (LBM), worsening the basal metabolic rate. Resistance training is the only non-pharmacological intervention that preserves LBM during a fat-loss phase.
If you focus only on the number on the scale by eating less, you might lose muscle instead of fat. This actually makes it easier to gain the weight back later. Lifting weights ensures that the weight you lose is the "bad" weight (fat) while keeping the "good" weight (muscle) that keeps your metabolism running hot.
Why Resistance Training Is the Most Powerful Metabolic Therapy
What if one intervention could simultaneously treat diabetes, prevent aging-related decline, and improve cardiovascular health
Resistance training is often celebrated for building muscle—but its true power lies far deeper, in the cellular signalling networks that govern metabolism, ageing, and disease risk. At the center of this network is the mechanistic target of rapamycin (mTOR), a master regulator that integrates mechanical load, nutrient availability, and hormonal cues to determine whether skeletal muscle grows, repairs, or adapts metabolically (Zhao, 2025). When activated in a pulsatile, exercise-driven manner, mTOR promotes muscle protein synthesis, enhances insulin sensitivity, and supports mitochondrial remodeling—key processes in preventing sarcopenia and metabolic disease (Zhao & Gao, 2024).
However, this same pathway represents a double-edged sword. Chronic, non-physiological mTOR activation—driven by persistent hyperinsulinemia, excess caloric intake, and physical inactivity—has been implicated in accelerated aging, impaired autophagy, and increased risk of insulin resistance and cardiometabolic disease (Zhao, 2025). This paradox underscores a critical principle: it is not mTOR activation per se, but the pattern, context, and stimulus that determine whether the outcome is therapeutic or pathological.
Key Clinical Insight
Loss of muscle mass is directly associated with increased insulin resistance, greater frailty risk, and higher all-cause mortality. Muscle is not optional tissue — it is metabolic infrastructure.
What Actually Happens When You Lift: The Science of Muscle Hypertrophy
Three Interacting Pathways Drive Muscle Growth
Muscle hypertrophy — the increase in muscle fiber size — does not happen in the gym. It happens afterwards, during recovery, as your body responds to the mechanical and metabolic signals generated during exercise. Three overlapping pathways drive this process:
Mechanical Tension is the primary driver. When you apply load to a muscle — through squatting, pressing, pulling, or any resistance-based movement — specialised proteins within the muscle fiber sense that tension. This triggers a process called mechanotransduction, activating enzymes such as focal adhesion kinase (FAK) and ultimately recruiting satellite cells (muscle stem cells) to the site of adaptation.
Metabolic Stress — characterized by lactate accumulation, cellular swelling, and controlled production of reactive oxygen species — provides a secondary anabolic signal. This is why high-rep, moderate-weight training can also stimulate growth, even without very heavy loads.
Muscle Damage and Repair, the third mechanism, involves microscopic trauma to muscle fibers during eccentric contractions (the lowering phase of a lift). The subsequent inflammatory response and cellular repair process contributes to long-term adaptation.
Importantly, a 2025 study by Jerez-Martínez & Romero-Arenas using near-infrared spectroscopy demonstrated that hypertrophy-focused resistance training also produces significant improvements in mitochondrial capacity — an observation that challenges the long-held view that aerobic exercise alone is responsible for mitochondrial adaptation (Jerez-Martínez & Romero-Arenas, 2026).
mTOR: The Master Switch of Muscle Anabolism
Understanding the mTOR Pathway
At the center of the entire hypertrophy cascade sits one of the most important molecular complexes in human biology: the mechanistic target of rapamycin complex 1, or mTORC1. Think of mTOR as the master switch that determines whether your body is in a building state or a breakdown state. When mTOR is activated, protein synthesis accelerates, ribosomes multiply, and muscle grows. When it is suppressed, the opposite occurs.
What Turns mTOR On?
Three primary inputs activate mTORC1: mechanical load from resistance exercise, amino acids (particularly leucine, found in protein-rich foods and supplements), and hormonal signals from insulin and insulin-like growth factor-1 (IGF-1). This is why the combination of resistance training and protein intake is so powerfully synergistic — both pathways converge on the same molecular target.
Once activated, mTORC1 phosphorylates downstream targets, including p70S6 kinase, increasing ribosomal biogenesis and enhancing translation initiation — in plain language, it accelerates the machinery responsible for building new proteins inside your muscle cells.
A comprehensive 2025 review by Zhao et al. documented mTOR's dual role in skeletal muscle: not only does it coordinate hypertrophic adaptation, but it also promotes mitochondrial biogenesis — making it a central therapeutic target for the management of chronic diseases, including type 2 diabetes, obesity, and sarcopenia (Zhao, 2025).
The mTOR–AMPK Balance: Why Training Type Matters
mTOR does not operate in isolation. It exists in a dynamic balance with AMPK (AMP-activated protein kinase), the body's cellular energy sensor. During prolonged aerobic exercise, AMPK rises, and mTOR is suppressed. During resistance training, mTOR predominates. This is not a flaw in human physiology — it is a feature. The appropriate balance of AMPK and mTOR signals determines whether adaptation trends toward endurance capacity or muscle mass.
A 2025 study by Mingzheng and You explored this AMPK/mTOR balance specifically in the context of aging muscle and insulin resistance, concluding that age-related disruption of this balance contributes meaningfully to sarcopenia and metabolic dysfunction — and that resistance training can partially restore it (Mingzheng & You, 2025).
Key Clinical Insight
Combining resistance training with adequate protein intake (~25–40g per meal, with at least 2.5–3g of leucine) produces maximal mTOR activation. Either stimulus alone is less effective than both together.
Anabolic Signals and the Broader Landscape of Hypertrophy Science
The field of anabolic signaling is evolving rapidly. A 2025 paper by Behringer, Heinrich, and Franz provided an updated synthesis of anabolic signals relevant to strength training in sports medicine, emphasizing that the interaction between mechanical stimuli, hormonal milieu, and nutritional status determines the magnitude and quality of hypertrophic adaptation (Behringer et al., 2025).
Meanwhile, a large-scale scientometric analysis by Chen (2025), covering over three decades of nutritional intervention research in the context of resistance training (1992–2025), confirmed that protein supplementation strategies remain the most studied and most consistently effective nutritional adjunct to resistance training for muscle hypertrophy, with leucine-rich sources consistently outperforming alternatives.
Sarcopenia: The Silent Epidemic of Aging Muscle
What Is Sarcopenia?
Sarcopenia — from the Greek for 'poverty of flesh' — is defined by the progressive, generalized loss of skeletal muscle mass and strength that accompanies aging. According to current diagnostic criteria, sarcopenia is identified by three features: low muscle mass (assessed by DEXA scan or bioelectrical impedance), reduced grip strength (below established thresholds), and impaired physical performance (such as slow gait speed or difficulty rising from a chair).
Sarcopenia affects an estimated 10–27% of community-dwelling older adults globally, rising to over 50% in institutionalized populations. Beyond its impact on quality of life, it is an independent predictor of hospitalization, disability, and death.
The Molecular Drivers of Muscle Loss
At the cellular level, sarcopenia is characterized by increased protein breakdown via the ubiquitin-proteasome pathway, overexpression of myostatin (a protein that inhibits muscle growth), and chronic low-grade inflammation — sometimes called 'inflammaging.' Satellite cell function declines with age, impairing the regenerative capacity of muscle tissue.
A related and increasingly recognized condition is sarcopenic obesity — the simultaneous presence of excess fat and reduced muscle mass. This combination carries particularly high cardiometabolic risk, as the metabolic dysfunction of adipose tissue compounds the insulin resistance driven by muscle loss.
Anabolic Resistance: Why Older Muscles Don't Respond as Well
Central to the biology of sarcopenia is the concept of anabolic resistance — a blunted muscle protein synthesis response to both protein intake and exercise. An older adult consuming the same protein as a younger person may experience significantly less mTOR activation and less muscle building as a result. The mechanisms behind this include impaired mTOR signaling, hormonal decline (testosterone, growth hormone, IGF-1), mitochondrial dysfunction, and elevated systemic inflammation.
This does not mean older adults cannot build muscle — they absolutely can. It means they typically require higher protein intakes, higher leucine content per meal, and carefully structured resistance training to achieve comparable results.
Resistance Training Reverses Sarcopenia — Here Is the Evidence
Resistance training directly addresses the molecular underpinnings of sarcopenia. It restores mTOR signaling, increases muscle protein synthesis, improves neuromuscular function, and stimulates satellite cell activity — even in octogenarians. This is not theoretical: controlled trials in older adults consistently demonstrate measurable gains in muscle mass, strength, and physical performance in response to progressive resistance training programs.
A scoping review by Sharma et al(2025) systematically examined the effects of different resistance exercise variants on sarcopenia, concluding that both traditional and alternative resistance modalities (including elastic band training and water-based resistance exercise) are effective, though progressive overload remains the key principle regardless of the equipment or setting (Sharma et al., 2025).
Complementing this, a comprehensive 2025 scoping review by Govindasamy et al. examined the physiological mechanisms through which resistance training reduces sarcopenia risk in healthy older adults. The review highlighted improvements in muscle fiber type composition, neuromuscular recruitment, hormonal responsiveness, and markers of systemic inflammation — collectively constituting a multi-mechanism therapeutic effect (Govindasamy et al., 2025).
Metabolic Benefits Beyond Muscle
The benefits of resistance training extend well beyond the muscle fiber itself. Regular resistance training improves whole-body insulin sensitivity, reduces visceral adiposity, enhances glucose disposal, and reduces circulating inflammatory markers. For patients with type 2 diabetes or metabolic syndrome, these effects are clinically meaningful and comparable — in some domains — to pharmacological interventions.
💡 Clinical Bottom Line
Resistance training is not an adjunct to metabolic therapy — it is a primary intervention. Its effects on insulin resistance, body composition, and inflammatory biology are mechanistically distinct from and complementary to those of aerobic exercise.
How to Train for Hypertrophy and Sarcopenia Reversal: A Practical Guide
The Core Training Variables
Effective resistance training for muscle hypertrophy and sarcopenia reversal requires attention to four primary variables: intensity, volume, frequency, and progression.
Intensity: Working at 60–85% of your one-repetition maximum (1RM) is the evidence-supported range for hypertrophy. In practical terms, this means selecting a weight that allows you to complete 8–12 repetitions with proper technique, where the final 2–3 repetitions feel genuinely challenging.
Volume: 2–4 sets per exercise, with 8–12 repetitions per set, performed across 2–3 sessions per week, represents the gold-standard prescription for most adults. Higher volumes may be tolerated by trained individuals.
Frequency: Each major muscle group should be trained at least twice per week, with 48 hours of recovery between sessions targeting the same muscles.
The Principle of Progressive Overload
Progressive overload — the gradual, systematic increase in training stimulus over time — is the single most important training principle for long-term muscle adaptation. Without progressive overload, adaptation plateaus. This does not always mean adding weight; it can mean increasing reps, sets, range of motion, or reducing rest periods. The key is that the stimulus must continue to challenge the muscle beyond its current capacity.
Special Considerations for Older Adults
For older adults or those new to resistance training, the principle of 'start low, progress slowly' applies with particular importance. Initial sessions should prioritize technique, balance, and safety over load. Unilateral exercises (single-leg or single-arm movements) are particularly valuable for identifying and correcting strength imbalances. Rest periods may be longer. The goal is consistent, long-term engagement — not short-term intensity.
The Nutritional Partner: Protein, Leucine, and Meal Timing
Resistance training provides the stimulus for muscle growth, but protein provides the raw material. Without adequate dietary protein, mTOR activation from exercise cannot be fully translated into new muscle tissue.
Current evidence supports protein intakes of 1.2–1.6 g per kilogram of body weight per day for general muscle maintenance and health, rising to 1.6–2.0 g/kg/day in older adults and those actively trying to increase muscle mass. Per-meal leucine content is increasingly recognized as a critical variable: a threshold of approximately 2.5–3 g of leucine per meal appears necessary for robust mTOR activation. This is typically achieved through 25–40 g of high-quality protein (meat, fish, eggs, dairy, or leucine-enriched plant-based blends).
Distributing protein intake evenly across three to four meals — rather than consuming the majority at dinner — maximizes the number of mTOR activation events per day and appears to optimize muscle protein synthesis over 24 hours. Post-exercise protein consumption within a 2-hour window is supportive, though the overall daily intake remains the most important variable.
Three Common Myths — Corrected by Science
Myth 1: 'Older adults cannot build muscle'
FALSE. While anabolic resistance means the response is blunted relative to younger adults, multiple randomized controlled trials have demonstrated meaningful gains in muscle mass and strength in adults well into their 80s. The stimulus must be appropriate, protein intake must be adequate, and progression must be consistent — but the capacity for adaptation does not disappear with age.
Myth 2: 'Light weights are enough'
PARTIALLY TRUE. Light loads can stimulate hypertrophy if sets are taken to near-muscular failure. However, loads of 60% 1RM or higher produce more reliable results across most populations, particularly for older adults who benefit from the additional neuromuscular and bone-loading stimulus of heavier training.
Myth 3: 'Protein alone can reverse sarcopenia'
FALSE. Protein intake without the mechanical stimulus of resistance training is insufficient to reverse established sarcopenia. Protein requires a training-induced anabolic environment to be effectively utilized for muscle synthesis. The combination of both is substantially more effective than either alone.
Resistance Training as Standard of Care
Across multiple chronic disease contexts, the evidence now supports resistance training not as a lifestyle recommendation but as a clinical intervention — with defined mechanisms, dosing parameters, and expected outcomes. In type 2 diabetes, it improves insulin sensitivity comparably to metformin in some patient groups. In metabolic-associated steatotic liver disease (MASLD, formerly NAFLD), it reduces hepatic fat and systemic inflammation. In aging, it is the most effective single intervention available for the prevention and reversal of sarcopenia.
It is worth noting one pharmacological nuance: metformin, while beneficial for glycemic control, has been shown in some studies to partially blunt the hypertrophic adaptation to resistance training — likely via its activation of AMPK, which competes with mTOR. Clinicians managing patients on metformin who are also pursuing resistance training should be aware of this potential interaction, though it does not negate the value of either intervention.
The Core Message
Resistance training is first-line anabolic therapy. It should be prescribed with the same specificity and seriousness as any pharmaceutical intervention — because its biological effects are just as real, just as measurable, and just as consequential.
Summary: What the Science Is Telling Us
Reframing Exercise as Molecular Therapy
Resistance training should no longer be viewed as optional lifestyle advice but as a targeted, dose-dependent intervention that modulates key signaling pathways governing metabolism, aging, and disease. At the center of this paradigm is mTOR, which integrates mechanical, nutritional, and hormonal inputs to regulate muscle protein synthesis and cellular adaptation (Deldicque et al., 2005; Zhao, 2025).Mechanotransduction: The Primary Therapeutic Trigger
Mechanical tension generated during resistance exercise activates integrin-mediated signaling cascades (FAK → PI3K/Akt → mTORC1), initiating anabolic processes within minutes. This position loads—not just movement—as the critical variable in therapeutic exercise prescription.The Dual Role of mTOR in Modern Medicine
Emerging evidence challenges the outdated view of mTOR as purely anabolic. Instead, it functions as a context-sensitive regulator, coordinating both hypertrophy and mitochondrial biogenesis depending on training stimulus and metabolic demand (Zhao & Gao, 2024).Resolving the Hypertrophy vs Metabolic Health Debate
The traditional divide between strength and endurance training is physiologically artificial. Properly designed concurrent training activates both mTORC1 and PGC-1α, enabling simultaneous improvements in muscle mass and mitochondrial function—critical for managing insulin resistance and cardiometabolic disease.The Four Pillars of Anabolic Signaling
Effective resistance training converges four stimuli: mechanical tension, metabolic stress, muscle damage, and hormonal signaling. Among these, progressive overload remains the dominant driver, with nutrition and recovery acting as amplifiers (Behringer et al., 2025).Clinical Translation: From Gym to Guidelines
Resistance training meets the criteria of a first-line therapy for sarcopenia, type 2 diabetes, and metabolic syndrome. When prescribed using structured frameworks (e.g., FITT-VP), it delivers reproducible, measurable outcomes across diverse populations.The Central Principle: Pulsatile mTOR Activation
Health outcomes depend not on chronic activation but on cyclical stimulation followed by recovery—a pattern uniquely achieved through resistance training, aligning anabolic signaling with metabolic resilience.
Frequently Asked Questions
Q1. How quickly can I expect to see results from resistance training?
Initial improvements in strength typically appear within 2–4 weeks of beginning a consistent program, largely due to neurological adaptations (your nervous system learning to recruit muscle fibers more efficiently). Measurable changes in muscle size generally require 6–12 weeks of progressive training. In older adults, functional improvements such as better balance, easier stair climbing, and reduced fatigue often precede visible changes in body composition.
Q2. I am over 65 — is it safe for me to start resistance training?
Yes, and it is one of the most evidence-supported interventions available to you at this life stage. Multiple clinical trials have demonstrated that resistance training is safe and beneficial for adults in their 70s, 80s, and even 90s. Begin with bodyweight exercises or low-resistance bands, prioritize technique over load, and consider working with a physiotherapist or certified trainer initially. Your doctor can help identify any specific considerations relevant to your health history.
Q3. Do I need to take protein supplements, or can I get enough from food?
Whole food sources of protein — meat, fish, eggs, dairy, legumes — are entirely sufficient for most people. Protein supplements (whey, casein, pea protein) are a convenient way to increase intake but are not superior to food sources with equivalent protein and leucine content. If meeting your protein targets through diet alone is difficult (particularly for older adults or those with reduced appetite), a protein supplement can be a practical addition.
Q4. What is the difference between resistance training and aerobic exercise for metabolic health?
Both are beneficial and mechanistically complementary. Aerobic exercise primarily improves cardiovascular fitness, mitochondrial density in muscle, and fat oxidation. Resistance training builds muscle mass, improves insulin sensitivity through glucose uptake in muscle, strengthens bone, and elevates resting metabolic rate. Current evidence supports the combination of both — with resistance training being the primary driver of structural metabolic improvement, particularly in the context of sarcopenia and insulin resistance.
Q5. Can resistance training help if I have type 2 diabetes?
Yes — meaningfully so. Resistance training improves insulin sensitivity by increasing the mass and metabolic activity of skeletal muscle, which is the body's primary site of glucose disposal. In some clinical studies, the glycemic benefits of resistance training are comparable to those of first-line medications. It is important to discuss exercise planning with your diabetes care team, as blood glucose monitoring and medication adjustments may be warranted.
Q6. I have heard that sarcopenia is inevitable with aging — is that true?
Sarcopenia is common with aging, but it is not inevitable — and it is substantially reversible. The rate of muscle loss accelerates after age 60, but this trajectory can be slowed, halted, or reversed with consistent resistance training and adequate protein intake. Studies have documented significant muscle mass and strength gains in adults in their 70s and 80s through structured exercise programs. Age is not a barrier to adaptation; it is simply a context that requires appropriate programming.
Q7. How does nutrition interact with resistance training for muscle building?
Nutrition and resistance training are synergistic — neither is fully effective without the other. Resistance training creates the anabolic environment (via mTOR activation and satellite cell recruitment), while protein provides the amino acid building blocks that are assembled into new muscle tissue. Carbohydrates support training performance and glycogen replenishment. Total caloric adequacy matters: muscle building is difficult in a significant caloric deficit. Distributing protein evenly across meals — with at least 2.5–3g of leucine per serving — appears to maximize the anabolic response across the day.
Author’s Note
This article reflects an evolving shift in how we understand exercise—not as a supplementary lifestyle choice, but as a primary, mechanism-driven therapeutic intervention. The concepts discussed here are drawn from a synthesis of contemporary research in exercise physiology, molecular biology, and metabolic medicine, with particular emphasis on the role of mTOR signaling and mechanotransduction in human health and disease.
While the science of resistance training continues to advance, it is important to recognize that much of the mechanistic evidence originates from controlled laboratory settings. Translating these findings into real-world clinical practice requires careful consideration of individual variability, including age, comorbidities, training history, and nutritional status. Therefore, the programme and recommendations provided in this article should be interpreted as evidence-informed frameworks rather than rigid prescriptions.
For clinicians, this represents an opportunity to integrate structured resistance training into standard care models for conditions such as type 2 diabetes, sarcopenia, and metabolic syndrome. For individuals, it offers a scientifically grounded pathway to improve strength, metabolic resilience, and long-term health outcomes.
Finally, it is worth emphasizing that consistency, progression, and recovery remain the foundational principles that determine success—far more than any single nutrient, supplement, or training variable. The molecular pathways described throughout this article are not theoretical constructs; they are highly responsive to daily behavior. When applied correctly, resistance training becomes more than exercise—it becomes medicine. If you are looking for a simplified 4-week action plan to start your training today, see my Practical Guide for Patients here. Why Your Muscles Aren’t Growing: The mTOR Mistake That Blocks Strength, Metabolism & Longevity | DR T S DIDWAL
This article is intended for educational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider before beginning a new exercise or nutrition program, especially if you have an existing medical condition.
Related Articles
mTOR, Protein, and Autophagy: How to Build Muscle Without Compromising Longevity
The mTOR Switch: Why Your Cells Stopped Listening to Insulin | DR T S DIDWAL
Can Resistance Training Reverse Diabetes? The Muscle–mTOR Link | DR T S DIDWAL
Why Aerobic Exercise Is the Most Powerful ‘Drug’ for Heart, Fat Loss & Longevity | DR T S DIDWAL
Exercise Unlocks a Hidden Glucose Pathway in Muscle — Independent of Insulin | DR T S DIDWAL
No Time to Train? Science-Backed Workouts That Deliver | DR T S DIDWAL
References
Behringer, M., Heinrich, C., & Franz, A. (2025). Anabolic signals and muscle hypertrophy: Significance for strength training in sports medicine. Sports Orthopaedics and Traumatology, 41(1), 9–18. https://doi.org/10.1016/j.orthtr.2025.01.002
Chen, W. (2025). Nutritional interventions in muscle hypertrophy research: A scientometric analysis within the context of resistance training (1992–2025). Journal of Health, Population and Nutrition, 44, 272. https://doi.org/10.1186/s41043-025-01031-w
Govindasamy, K., Rao, C. R., Chandrasekaran, B., Parpa, K., & Granacher, U. (2025). Effects of resistance training on sarcopenia risk among healthy older adults: A scoping review of physiological mechanisms. Life, 15(5), 688. https://doi.org/10.3390/life15050688
Jerez-Martínez, A., & Romero-Arenas, S. (2026). Impact of hypertrophy-focused resistance training on mitochondrial capacity: A new perspective through near-infrared spectroscopy. Research Quarterly for Exercise and Sport, 97(1), 60–67. https://doi.org/10.1080/02701367.2025.2542816
Mingzheng, X., & You, W. (2025). AMPK/mTOR balance during exercise: Implications for insulin resistance in aging muscle. Molecular and Cellular Biochemistry, 480, 5941–5953. https://doi.org/10.1007/s11010-025-05362-4
Sharma, S., Sharma, M., Tripathi, D., et al. (2025). Impact of resistance exercise variants on sarcopenia: A scoping review. Discover Public Health, 22, 474. https://doi.org/10.1186/s12982-025-00866-2
Zhao, Y.-C. (2025). Dual roles of mTOR in skeletal muscle adaptation: Coordinating hypertrophic and mitochondrial biogenesis pathways for exercise-induced chronic disease management. Frontiers in Medicine, 12, 1635219. https://doi.org/10.3389/fmed.2025.1635219
Zhao, Y. C., & Huang, T. (2025). [Title in English not available]. Sheng Li Xue Bao: Acta Physiologica Sinica, 77(3), 573–586. https://doi.org/10.13294/j.aps.2025.0048