Why Your Muscles Aren’t Growing: The mTOR Mistake That Blocks Strength, Metabolism & Longevity
Discover how to activate mTOR for muscle growth, metabolic health, and longevity—science-backed strategies for protein, exercise, and aging.
AGINGSARCOPENIA
Dr. T.S. Didwal, M.D.(Internal Medicine)
5/7/202612 min read
Why are your muscles not growing despite exercise and high-protein diets? One of the most overlooked reasons is poor regulation of mTOR signaling—the master cellular pathway responsible for muscle protein synthesis, strength adaptation, metabolic health, and healthy aging (Bodine, 2022; Zhao, 2025). mTOR acts as the body’s “growth switch,” integrating signals from resistance training, amino acids (especially leucine), insulin, and cellular energy status to determine whether the body builds muscle or shifts into repair and recycling mode (Jiang et al., 2024).
Current research shows that resistance training combined with adequate leucine-rich protein intake is the most effective physiological strategy to activate mTOR for muscle growth, metabolic resilience, and preservation of lean mass during aging (Bodine, 2022; Zhao, 2025). However, aging, physical inactivity, chronic stress, poor sleep, and insufficient protein intake can blunt mTOR responsiveness—a phenomenon known as anabolic resistance—contributing to sarcopenia, insulin resistance, frailty, impaired recovery, and metabolic decline (Mingzheng & You, 2025).
This article explains how strategic “pulsatile” activation of mTOR signaling—through resistance exercise, optimized protein timing, recovery, and intermittent fasting—may help preserve lean muscle mass, improve glucose metabolism, support mitochondrial function, and promote healthy aging (Zhao, 2025). It also explores the critical relationship between mTOR and autophagy, showing why chronically elevated mTOR activity from overnutrition and sedentary behavior may impair insulin signaling, suppress cellular repair pathways, and accelerate metabolic dysfunction and aging-related disease (Jiang et al., 2024; Li et al., 2025).
Key evidence-based strategies discussed include:
Resistance training 3–4 times weekly to stimulate mTOR-mediated muscle protein synthesis (Zhao, 2025)
Consuming 25–40 g of high-quality protein per meal to maximize anabolic signaling
Reaching the leucine threshold (~2.5–3 g per meal) required for robust mTORC1 activation
Combining post-workout protein intake with progressive overload to amplify hypertrophic adaptation
Using intermittent fasting or recovery windows to restore autophagy and metabolic flexibility (Li et al., 2025)
Prioritizing sleep, recovery, and omega-3 fatty acids to optimize muscle repair and cellular signaling
Preventing anabolic resistance after age 50 through higher protein intake and consistent resistance exercise (Mingzheng & You, 2025)
The article also examines the relationship between mTOR signaling, insulin signaling, mitochondrial health, autophagy, type 2 diabetes, obesity, cardiovascular disease, and longevity—making it highly relevant for clinicians, aging adults, exercise professionals, and patients seeking evidence-based strategies to preserve muscle, maintain metabolic health, and support healthy aging throughout life (Bodine, 2022; Jiang et al., 2024; Zhao, 2025).
What Is mTOR signaling? The Science Behind Muscle Growth, Metabolism, and Healthy Aging
mTOR (mechanistic Target of Rapamycin) is one of the most important cellular pathways involved in muscle growth, strength, metabolism, exercise recovery, and longevity. Often called the body’s “master growth regulator,” mTOR acts like a molecular switch that tells your cells when to build, repair, conserve energy, or recycle damaged components.
This pathway continuously monitors your nutritional and metabolic environment by sensing:
Amino acids (especially leucine)
Insulin and IGF-1
Cellular energy levels (ATP)
Oxygen availability
Mechanical stress from exercise
When nutrients and energy are abundant—such as after resistance training or a high-protein meal—mTOR activates anabolic processes that stimulate:
Muscle protein synthesis
Muscle repair and hypertrophy
Ribosome production
Cellular growth
Metabolic adaptation
When nutrients are scarce during fasting, caloric restriction, illness, or prolonged stress, mTOR activity declines. This allows the body to shift toward cellular maintenance and autophagy, the body’s natural recycling and repair system.
Modern research shows that balanced mTOR activation is essential for preserving muscle mass, improving insulin sensitivity, supporting mitochondrial health, and slowing age-related metabolic decline.
mTORC1 vs mTORC2: Understanding the Two Major mTOR Pathways
mTORC1 and mTORC2 are the two major protein complexes that make up the mTOR system. Although closely related, they perform different physiological functions.
mTORC1: The Primary Muscle-Building Pathway
mTORC1 is the best-studied form of mTOR and the major regulator of skeletal muscle hypertrophy. It responds rapidly to:
Dietary protein
Leucine intake
Resistance exercise
Insulin signaling
Mechanical loading
Once activated, mTORC1 stimulates muscle growth by activating downstream targets involved in protein synthesis, including:
S6K1 (ribosomal protein synthesis)
4E-BP1 (mRNA translation initiation)
This process accelerates muscle repair, recovery, and hypertrophy after exercise.
Research also shows that mTORC1 suppresses excessive muscle breakdown by inhibiting protein degradation systems such as:
the ubiquitin-proteasome pathway
the autophagy-lysosome system
This makes mTORC1 one of the most important regulators of lean muscle preservation during aging.
mTORC2: The Metabolic and Insulin-Sensitivity Regulator
Unlike mTORC1, mTORC2 functions more as a metabolic stabilizer. It regulates:
Insulin sensitivity
Glucose uptake
GLUT4 translocation
Cellular survival
Cytoskeletal organization
Healthy mTORC2 signaling supports metabolic flexibility and helps maintain normal glucose metabolism. Impaired mTORC2 activity has been associated with:
insulin resistance
obesity
type 2 diabetes
metabolic aging
Importantly, chronically elevated mTORC1 activity from overeating or inactivity may impair mTORC2 signaling through negative feedback mechanisms, potentially worsening insulin resistance over time.
This is why modern longevity research emphasizes balanced—not constant—mTOR activation.
How mTOR Senses Nutrients and Protein Intake
One of the most fascinating aspects of mTOR signaling is its ability to detect nutrient availability with remarkable precision.
Leucine: The Key Amino Acid for Muscle Growth
Among all amino acids, leucine is the strongest activator of mTORC1. Leucine-rich foods include:
Whey protein
Eggs
Lean meat
Fish
Greek yogurt
Soy protein
Legumes
After a high-protein meal, intracellular leucine levels rise and activate mTORC1 at the lysosomal surface, initiating an anabolic window of muscle protein synthesis that may last 90–120 minutes.
Current evidence suggests that most adults require approximately:
2.5–3g leucine per meal to maximally stimulate mTOR-mediated muscle protein synthesis.
This “leucine threshold” becomes especially important after age 50 due to anabolic resistance.
The Lysosome: The Hidden Control Center of mTOR Activation
Recent research has identified the lysosome as the primary activation hub for mTORC1 signaling.
A landmark 2026 study demonstrated that lysosomal membrane lipid composition may influence how efficiently muscles respond to dietary protein and exercise. This suggests that healthy fats—particularly omega-3 fatty acids—may indirectly improve anabolic signaling by optimizing lysosomal membrane function.
In practical terms:
protein quality matters,
but membrane health may matter too.
This emerging area of research may help explain why omega-3 fatty acids improve muscle quality and metabolic health in aging adults.
How Resistance Exercise Activates mTOR
Resistance training is the most powerful natural activator of mTOR in skeletal muscle.
Mechanical loading stimulates mTORC1 through mechanosensitive pathways independent of dietary protein, including:
phosphatidic acid signaling
focal adhesion kinase (FAK)
MAP4K3 activation
This means that strength training alone can stimulate muscle protein synthesis—even during fasting—although the response is significantly amplified when combined with post-workout protein intake.
Research increasingly shows that resistance training activates both:
hypertrophic pathways (muscle growth)
mitochondrial biogenesis pathways (metabolic health)
This dual effect explains why strength training improves:
muscle mass
insulin sensitivity
glucose disposal
mitochondrial function
metabolic flexibility
For aging adults and patients with obesity or type 2 diabetes, resistance exercise functions as both a muscle-building and metabolic therapy.
mTOR, Aging, and Anabolic Resistance
One of the defining features of aging is anabolic resistance—a reduced ability of muscle tissue to respond to protein intake and exercise.
With aging:
mTOR responsiveness declines
AMPK signaling increases
muscle protein synthesis becomes blunted
sarcopenia accelerates
This contributes to:
frailty
insulin resistance
reduced metabolic rate
weakness
impaired recovery
Research suggests that older adults may require:
1.2–1.6 g/kg/day protein, along with progressive resistance training to overcome age-related anabolic resistance.
The evidence strongly supports combining:
higher protein intake,
leucine-rich meals,
and resistance exercise
to restore healthy mTOR signaling in aging muscle.
mTOR, Autophagy, and Longevity
mTOR is also a master regulator of autophagy, the cellular repair and recycling process that removes damaged proteins and dysfunctional mitochondria.
When mTOR is highly active:
muscle growth increases,
but autophagy decreases.
When mTOR activity falls during fasting, caloric restriction, or aerobic exercise:
autophagy increases,
cellular repair accelerates,
and metabolic cleanup improves.
This creates an important biological balance:
excessive mTOR suppression may impair muscle maintenance,
but chronic overactivation may accelerate aging and metabolic dysfunction.
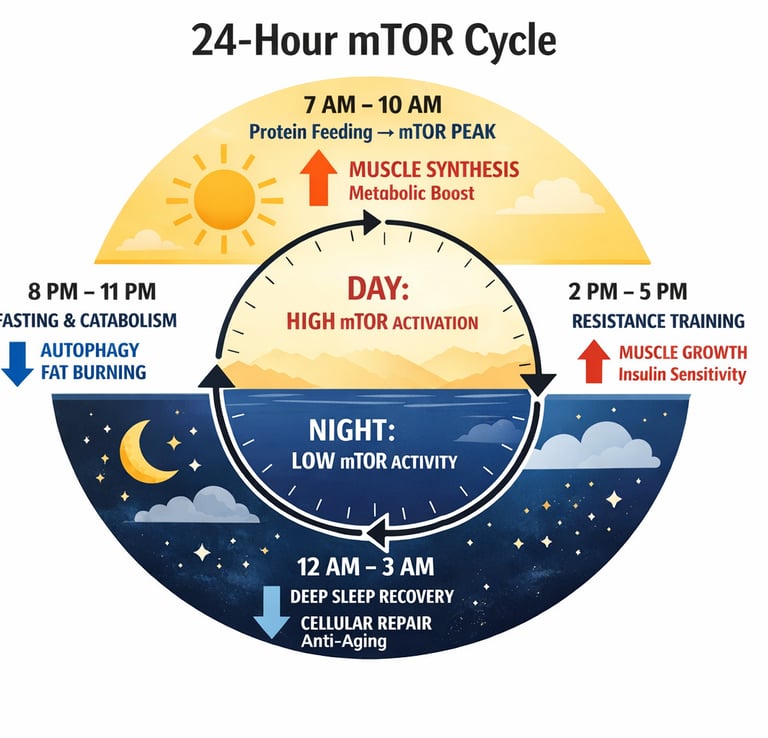
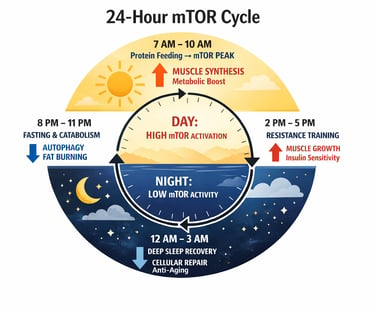
Modern longevity science increasingly supports a pulsatile strategy:
activate mTOR during feeding and resistance training,
suppress mTOR during fasting and recovery periods.
This approach may optimize both:
muscle preservation,
and long-term cellular health
.
Practical Strategies to Optimize mTOR for Muscle and Longevity
Evidence-Based mTOR Activation Strategies
Resistance Training
Train 3–4 times weekly
Focus on progressive overload
Prioritize compound movements
Protein Intake
Consume 25–40 g protein per meal
Prioritize leucine-rich proteins
Distribute protein evenly across meals
Strategic Fasting
Use overnight fasting windows
Consider intermittent fasting when appropriate
Allow periodic mTOR suppression for autophagy
Sleep and Recovery
Aim for 7–9 hours nightly
Deep sleep supports mTOR-mediated repair
Recovery is essential for muscle adaptation
Omega-3 Fatty Acids
Consume fatty fish regularly
Consider EPA/DHA supplementation
Support membrane and mitochondrial health
Why mTOR Matters for Metabolic Health and Chronic Disease
Balanced mTOR signaling may play a major role in preventing:
sarcopenia
insulin resistance
obesity
type 2 diabetes
cardiovascular disease
metabolic aging
Properly regulated mTOR activity helps preserve lean muscle mass, improve glucose metabolism, maintain mitochondrial quality, and support healthy aging.
The emerging scientific consensus is clear:
the goal is not maximal mTOR activation—it is rhythmic, well-regulated activation aligned with exercise, nutrition, recovery, and metabolic health.
Frequently Asked Questions (FAQs)
Q1. What foods most powerfully activate mTOR for muscle growth? Leucine-rich proteins are the strongest dietary mTOR activators. Whey protein isolate, eggs, beef, chicken, salmon, and cottage cheese all provide the leucine threshold (~2.5–3 g per meal) needed for maximal mTORC1 stimulation. Plant sources like soy, lentils, and edamame are also effective, particularly when consumed in adequate quantities.
Q2. Can I activate mTOR without exercise? Yes — protein and amino acids alone can stimulate mTORC1, particularly after a period of fasting. However, exercise dramatically amplifies the mTOR response and opens a prolonged anabolic window. Without exercise, muscle protein synthesis increases only modestly and transiently. The combination of resistance training + protein intake produces far greater and more sustained mTOR activation than either alone.
Q3. Does mTOR cause cancer? mTOR itself does not cause cancer, but dysregulated, chronically elevated mTORC1 activity — as seen in obesity, PTEN mutations, or constitutively active Ras — contributes to tumor cell proliferation by promoting protein synthesis and suppressing autophagy. Jiang et al. (2024) review this extensively. This is one reason that caloric restriction and exercise — which periodically reduce mTOR activity — are associated with reduced cancer risk.
Q4. Is it bad to have high mTOR all the time? Yes. Chronically elevated mTOR suppresses autophagy (impairing cellular clean-up), generates IRS-1 inhibitory feedback that worsens insulin resistance, and may accelerate cellular aging. Healthy mTOR signaling is pulsatile — activated strongly after exercise and protein feeding, then allowed to return to baseline during fasting or rest periods.
Q5. How does aging affect mTOR signaling? Aging is associated with mTOR anabolic resistance — a blunted mTORC1 response to amino acids and exercise. Simultaneously, AMPK activation patterns change, disrupting the AMPK/mTOR balance that governs insulin sensitivity in muscle (Mingzheng & You, 2025). Practically, this means older adults need more protein (1.2–1.6 g/kg/day) and more mechanical stimulus (higher training volumes or frequencies) to achieve the same mTOR activation as younger individuals.
Q6. What is the relationship between mTOR and autophagy in disease? mTOR suppresses autophagy when active; autophagy activates when mTOR is inhibited. In metabolic diseases like diabetic kidney disease, insufficient autophagy — due in part to chronic mTOR overactivation — allows damaged proteins and organelles to accumulate, worsening oxidative stress and inflammation (Li et al., 2025). Therapeutic strategies that restore mTOR-autophagy balance can meaningfully improve outcomes.
Q7. Can rapamycin (an mTOR inhibitor) extend lifespan? In animal models, rapamycin — which selectively inhibits mTORC1 — consistently extends lifespan, even when started late in life. However, in humans, chronic mTOR inhibition carries significant risks: impaired immune function, insulin resistance (through mTORC2 inhibition with higher doses), and potential muscle loss. Research into intermittent low-dose rapamycin as a longevity intervention in humans is ongoing but not yet ready for routine clinical use. The safer alternative remains lifestyle: exercise, strategic fasting, and adequate-but-not-excessive protein intake.
Clinical Pearls: mTOR, Muscle, and Metabolic Health
1. Muscle Is Your Metabolic Organ—Protect It Aggressively
Scientific insight: Skeletal muscle is the largest site of insulin-mediated glucose disposal, and mTORC1 activation is essential for maintaining muscle protein synthesis and preventing atrophy (Bodine, 2022). Loss of muscle mass directly worsens insulin resistance.
“The more muscle you keep, the better your blood sugar control. Strength training is not optional—it’s medicine.”
2. Protein Dose Matters More Than You Think
Scientific insight: mTORC1 activation requires crossing a leucine threshold (~2.5–3 g per meal) to maximally stimulate muscle protein synthesis; sub-threshold intake leads to a blunted anabolic response.
“It’s not just eating protein—it’s eating enough at each meal. A small serving won’t trigger muscle building.”
3. Timing Creates Synergy: Exercise + Nutrition
Scientific insight: Resistance exercise activates mTOR via mechanosensitive pathways, while protein and insulin amplify the response through PI3K–Akt signaling, producing a synergistic anabolic effect (Zhao, 2025).
“Your workout opens the door—protein after exercise locks in the gains.”
4. Constant Activation Is Harmful—Balance Is Key
Scientific insight: Chronic mTORC1 overactivation (overnutrition) triggers S6K1-mediated IRS-1 inhibition, promoting insulin resistance and suppressing autophagy (Jiang et al., 2024; Li et al., 2025).
Eating all the time—even healthy food—can backfire. Your body needs breaks to repair itself.”
5. Aging Blunts the Signal—You Must Push Harder (Safely)
Scientific insight: Aging muscle exhibits anabolic resistance, with reduced mTOR responsiveness to both amino acids and exercise, partly due to altered AMPK–mTOR signaling (Mingzheng & You, 2025).
“As you age, your body needs a stronger signal—more protein and regular strength training—to maintain muscle.”
Key Takeaways
Your body is constantly making a decision: build or break down. At the center of this decision is mTOR (mechanistic Target of Rapamycin)—a molecular regulator that determines whether you gain muscle, preserve strength, or drift toward metabolic decline.
mTOR is not just for athletes—it is central to clinical medicine. Impaired mTOR signaling is strongly linked to sarcopenia, insulin resistance, type 2 diabetes, and frailty, making it a critical pathway in aging and chronic disease (Bodine, 2022; Jiang et al., 2024).
When activated correctly, mTOR drives muscle protein synthesis. Resistance training and adequate protein intake—especially leucine-rich sources—activate mTORC1, initiating ribosomal activity and tissue repair. This is the biological foundation of strength, recovery, and metabolic health.
But mTOR is not simply “good” or “bad”—it is context-dependent. While pulsatile activation promotes hypertrophy and resilience, chronic overactivation—seen in overnutrition and sedentary lifestyles—can impair insulin signaling and suppress autophagy, accelerating aging (Li et al., 2025).
This creates a metabolic balancing act. The interplay between mTOR and AMPK determines whether your body is in growth mode or repair mode. Aging disrupts this balance, leading to anabolic resistance, where muscle becomes less responsive to protein and exercise (Mingzheng & You, 2025).
Exercise remains the most powerful modulator of mTOR. Resistance training activates mTOR independent of nutrients, while post-exercise protein intake amplifies the anabolic signal—making this combination essential for preserving muscle and reversing metabolic decline (Zhao, 2025).
The future of longevity lies in timing, not suppression. Strategic cycles of activation (feeding, training) and suppression (fasting, recovery) optimize mTOR signaling—supporting both muscle growth and cellular repair.
The Metformin/Berberine Conflict: If a patient is using Metformin or Berberine (AMPK activators), current evidence suggests taking them away from the resistance training window (e.g., in the morning or with the first meal) to avoid "blunting" the acute mTOR response triggered by the workout.
The Lipid Influence: Referencing the Picot et al. (2026) study, ensure the evening meal includes Omega-3s (EPA/DHA). Optimizing the lysosomal membrane's lipid composition makes the mTOR "machinery" more efficient at sensing the leucine from your dinner
.
The "Anabolic Threshold" in Aging: For patients over 65, the 12:00 "Trigger Meal" is non-negotiable. A low-protein breakfast followed by a low-protein lunch keeps the body in a catabolic state for too long, leading to the "drifting" sarcopenia you described.
To optimize the "Anabolic Phase" without risking chronic overnutrition, patients should prioritize Nutrient-Dense Proteins that maximize the Leucine-to-Calorie ratio. Choosing "Clean Anabolic" sources—such as whey protein isolate, egg whites, lean poultry, white fish, and fat-free Greek yogurt—allows the body to cross the critical 2.5 - 3 g leucine threshold required to flip the mTOR growth switch while keeping total caloric intake low. This precision prevents the "Always-On" mTOR state associated with high-calorie, fatty meats and processed foods, which can overwhelm the insulin-mTORC2 axis and trigger systemic inflammation. By focusing on these high-yield proteins, you ensure a powerful, pulsatile anabolic signal during feeding windows while leaving the metabolic "room" necessary for the AMPK/Autophagy phase to initiate later in the day, effectively balancing muscle preservation with cellular repair and longevity.
Author’s Note
As a clinician working at the intersection of internal medicine, metabolism, and exercise physiology, I have seen firsthand how rapidly patients lose muscle, strength, and metabolic resilience with age—and how powerfully these declines can be reversed with the right interventions.
The science of mTOR signaling is no longer confined to research laboratories. It is directly relevant to everyday clinical practice—from managing type 2 diabetes and sarcopenia to improving recovery, functional capacity, and long-term health outcomes. What was once considered a niche molecular pathway is now recognized as a central regulator of muscle biology, nutrient sensing, and aging.
This article was written with a dual purpose:
to translate complex molecular mechanisms into clinically actionable strategies, and to bridge the gap between bench research and bedside application. Every concept discussed here is grounded in current scientific evidence, but equally important is its practical relevance for patients navigating real-world challenges—loss of muscle mass, declining energy, and increasing metabolic risk.
It is important to emphasize that mTOR is not a pathway to be maximized indiscriminately, but one to be strategically modulated. Health lies not in constant activation, but in achieving the right physiological rhythm—periods of anabolic stimulation through nutrition and resistance training, balanced with phases of recovery and cellular repair.
As always, this content is intended for educational purposes. Individual responses to nutrition and exercise vary significantly, particularly in the presence of chronic disease. Clinical judgment and personalized care remain essential.
For a deeper clinical look at the molecular pathways of mechanotransduction, read my technical deep-dive here. You’re Missing This: Why mTOR Activation Is the Real Goal of Strength Training
This article is intended for educational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider before beginning a new exercise or nutrition program, especially if you have an existing medical condition.
Related Articles
The mTOR Switch: Why Your Cells Stopped Listening to Insulin | DR T S DIDWAL
Why Aerobic Exercise Is the Most Powerful ‘Drug’ for Heart, Fat Loss & Longevity | DR T S DIDWAL
Exercise Unlocks a Hidden Glucose Pathway in Muscle — Independent of Insulin | DR T S DIDWAL
No Time to Train? Science-Backed Workouts That Deliver | DR T S DIDWAL
References
Bodine, S. C. (2022). The role of mTORC1 in the regulation of skeletal muscle mass. Faculty Reviews, 11, 32. https://doi.org/10.12703/r/11-32
Jiang, C., Tan, X., Liu, N., Yan, P., Hou, T., & Wei, W. (2024). Nutrient sensing of mTORC1 signaling in cancer and aging. Seminars in Cancer Biology, 106–107, 1–12. https://doi.org/10.1016/j.semcancer.2024.08.001
Li, L., Zou, J., Zhou, T., Liu, X., Tan, D., Xiang, Q., & Yu, R. (2025). mTOR-mediated nutrient sensing and oxidative stress pathways regulate autophagy: A key mechanism for traditional Chinese medicine to improve diabetic kidney disease. Frontiers in Pharmacology, 16, 1578400. https://doi.org/10.3389/fphar.2025.1578400
Mingzheng, X., & You, W. (2025). AMPK/mTOR balance during exercise: Implications for insulin resistance in aging muscle. Molecular and Cellular Biochemistry, 480, 5941–5953. https://doi.org/10.1007/s11010-025-05362-4
Picot, M., Hifdi, N., Vaucourt, M., et al. (2026). Lysosomal phosphoinositide turnover acts upstream of RagGTPase–mTORC1 and controls muscle growth. Nature Metabolism. https://doi.org/10.1038/s42255-026-01484-1
Zhao, Y.-C. (2025). Dual roles of mTOR in skeletal muscle adaptation: Coordinating hypertrophic and mitochondrial biogenesis pathways for exercise-induced chronic disease management. Frontiers in Medicine, 12, 1635219. https://doi.org/10.3389/fmed.2025.1635219