mTORC2 Explained: How This Metabolic Thermostat Regulates Insulin Resistance & Aging After 50
Discover how mTORC2 regulates insulin sensitivity, metabolism, and aging. Learn science-backed strategies to improve metabolic health and prevent disease.
DIABETESAGING
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/22/202613 min read
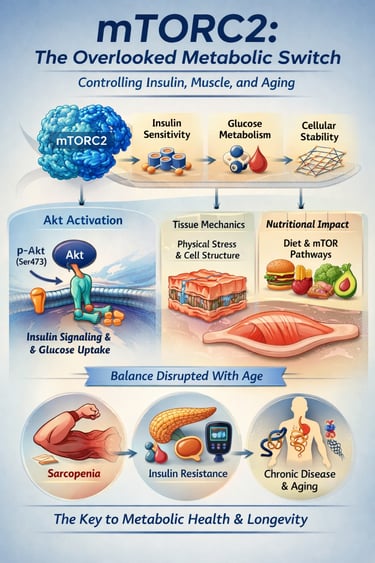
mTORC2 (mechanistic target of rapamycin complex 2) is a key cellular pathway that regulates insulin sensitivity, glucose metabolism, and cellular stability. It activates Akt signaling, enabling efficient glucose uptake and metabolic balance. Declining mTORC2 activity—especially after age 50—contributes to insulin resistance, sarcopenia, and metabolic disease.
Inside every cell in your body, a highly coordinated signaling system is constantly deciding how you respond to food, exercise, and metabolic stress. At the center of this system is mTOR (mechanistic Target of Rapamycin), a master regulator of cellular growth and metabolism. While most attention has focused on mTORC1—known for driving muscle growth and protein synthesis—its lesser-known counterpart, mTORC2, may be even more critical for long-term metabolic health (Panwar et al., 2023).
mTORC2 plays a central role in insulin sensitivity, glucose metabolism, and cellular stability, primarily through activation of Akt, the key mediator of insulin signaling (Taylor et al., 2026). When mTORC2 functions properly, cells efficiently uptake glucose, maintain structural integrity, and adapt to metabolic demands. When it is impaired, the consequences ripple across the body—contributing to insulin resistance, type 2 diabetes, and accelerated aging.
Emerging research reveals that mTORC2 does far more than regulate biochemistry—it also integrates mechanical signals from the cellular environment, linking tissue stiffness and structural stress to metabolic demand (Collins et al., 2026). At the same time, nutritional patterns directly influence mTOR signaling, with chronic overnutrition disrupting this delicate balance and impairing metabolic control (Wang et al., 2025).
With aging, this system drifts further off balance—mTORC1 becomes chronically overactive, while mTORC2 activity declines, creating a metabolic environment that promotes sarcopenia, insulin resistance, and chronic disease (Zhang et al., 2024).
Understanding mTORC2 is no longer optional—it is central to preserving metabolic health, muscle function, and longevity in modern clinical practice.
How to Support mTORC2 for Better Metabolic Health
Perform resistance training 2–3 times per week
Maintain a whole-food, balanced diet
Avoid chronic overnutrition
Prioritize 7–9 hours of sleep
Incorporate intermittent fasting (if appropriate)
Manage stress and maintain circadian rhythm
Role in insulin sensitivity, cytoskeletal integrity, and chronic disease prevention — the science your cells have been waiting for
Clinical pearls.
1. The "Glucose Sink" Principle
Scientific: mTORC2-mediated Akt phosphorylation (Ser473) is the primary driver of GLUT4 translocation. Without this "second complex" signaling, peripheral insulin sensitivity collapses regardless of insulin levels.
Think of your muscles as a giant sponge for sugar. mTORC2 is the handle that turns the faucet on. If that handle is broken, the sugar stays in your blood, no matter how much insulin your body pumps out.
2. Pulsatile vs. Chronic Activation
Scientific: Chronic mTORC1 hyperactivation via overnutrition leads to a negative feedback loop that suppresses IRS-1 signaling, effectively "shunting" mTORC2’s homeostatic control.
Your growth switch (mTOR) is like a light—it’s meant to be turned on when you eat or exercise and off when you rest. Leaving it "on" 24/7 by overeating actually breaks the circuit, leading to faster aging and "clogged" cells.
3. The Mechanosensing Mandate
Scientific: The SKT–mTORC2 coupling proves that cellular metabolism is a downstream effector of extracellular matrix (ECM) tension. Myocellular loading is a non-pharmacological ligand for metabolic flux.
Your cells "feel" the weight you lift. When you do resistance training, the physical tension tells your cells to speed up their metabolism. It’s not just about burning calories; it’s about using physical force to "signal" your cells to stay young.
4. The First-Phase Secretion Failure
Scientific: mTORC2 deficiency impairs F-actin remodeling in pancreatic beta cells. This creates a structural bottleneck for insulin vesicle trafficking, specifically compromising the immediate (first-phase) insulin response to glucose.
Your pancreas doesn't just need "orders" to release insulin; it needs a clear "highway system" inside the cell to move the insulin out. mTORC2 keeps those highways clear so your body can respond to a meal instantly.
5. The "Sarcopenia-Diabetes" Axis
Scientific: Aging-related decline in mTORC2 activity in skeletal muscle isn't just a pro-inflammatory state; it is a primary driver of anabolic resistance. This reduces the "metabolic margin" for glucose disposal.
Losing muscle as you age (sarcopenia) isn't just about weakness—it’s a metabolic emergency. Every pound of muscle you lose is a piece of your "sugar-burning engine" gone. Maintaining muscle is your best insurance policy against diabetes.
6. The Rapamycin Paradox
Scientific: While rapalogs are potent autophagy inducers via mTORC1 inhibition, their long-term use can "leak" into mTORC2 inhibition, leading to the "Rapamycin-induced Diabetes" phenotype.
There are "longevity drugs" that help clean out cellular junk, but they can be a double-edged sword. If used incorrectly, they can accidentally turn off your body’s ability to manage blood sugar. Precision and timing are everything.
What Is mTORC2 and Why It Matters for Insulin Resistance, Metabolism & Aging
Inside virtually every cell, a powerful signaling system is constantly regulating how your body processes glucose, responds to insulin, and adapts to metabolic stress. At the center of this system is mTOR (mechanistic target of rapamycin)—a master kinase that integrates nutrition, energy status, and environmental signals to control cellular function (Panwar et al., 2023).
Most discussions focus on mTORC1, the complex responsible for muscle growth and protein synthesis. However, a growing body of research shows that mTORC2 is equally critical—particularly for metabolic health, insulin sensitivity, and aging.
What Is mTORC2?
mTORC2 (mechanistic target of rapamycin complex 2) is a structurally distinct protein complex that regulates:
Insulin signaling via Akt activation
Glucose uptake in muscle and fat tissue
Cell survival and metabolic stability
Cytoskeletal organization and cellular structure
Unlike mTORC1, mTORC2 is not acutely inhibited by rapamycin and operates as a metabolic stabilizer, fine-tuning how cells respond to nutrients, hormones, and physical stress.
Key Concept:
mTORC2 functions as a metabolic thermostat—continuously adjusting insulin sensitivity, cellular structure, and energy utilization to maintain metabolic balance.
How mTORC2 Connects Mechanics, Metabolism, and Disease
1. Mechanosensing: How Cells “Feel” Their Environment
Recent research highlights a critical but underappreciated function of mTORC2: linking physical forces to metabolic demand.
A landmark study by Collins et al. (2026) demonstrated that cell-matrix mechanosensing is directly coupled to metabolism through SKT kinase and mTORC2. In simple terms:
Cells sense tissue stiffness (e.g., fat vs. fibrotic tissue)
This mechanical signal alters cellular energy demand
mTORC2 acts as the bridge between structure and metabolism
Clinical Relevance
Increased tissue stiffness (fibrosis, atherosclerosis, cancer) disrupts mTORC2 signaling
Leads to metabolic misregulation, abnormal growth, and insulin resistance
SEO Takeaway:
Metabolic disease is not just biochemical—it is also mechanical, with mTORC2 at the center of this interaction.
2. Nutrition and mTORC2: How Diet Shapes Metabolic Health
mTORC2 activity is highly responsive to nutrient signals, including glucose, amino acids, and fatty acids.
According to Wang et al. (2025), chronic overnutrition leads to persistent mTOR activation, triggering feedback mechanisms that impair insulin signaling.
Key Effects of Poor Dietary Patterns
Chronic mTOR dysregulation
Reduced insulin sensitivity
Increased inflammation and metabolic stress
Metabolic Insight
Diets high in refined carbohydrates and saturated fats overactivate mTOR pathways
Caloric moderation, intermittent fasting, and whole-food diets help restore mTOR balance
Patient Insight:
Every meal influences mTOR signaling. Long-term dietary patterns can either enhance insulin sensitivity or drive metabolic disease.
3. mTORC2 in Chronic Disease and Aging
mTOR signaling plays a central role in nearly all major chronic diseases (Panwar et al., 2023):
Type 2 Diabetes: Impaired mTORC2–Akt signaling → insulin resistance
Cancer: Dysregulated mTOR promotes uncontrolled growth
Cardiovascular Disease: Alters vascular and cardiac cell function
Neurodegeneration: Impaired autophagy and protein clearance
Aging: Decline in mTORC2 → metabolic dysfunction and frailty
Aging-Specific Insight
With aging:
mTORC1 becomes overactive
mTORC2 activity declines
This imbalance drives:
Insulin resistance
Sarcopenia
Reduced metabolic flexibility (Zhang et al., 2024)
Key Concept:
Healthy aging depends not on suppressing mTOR entirely, but on restoring balance between mTORC1 and mTORC2.
4. mTORC2 and Insulin Secretion: The Cytoskeleton Connection
Beyond insulin sensitivity, mTORC2 also regulates insulin secretion at the pancreatic level.
Blandino-Rosano et al. (2022) demonstrated that mTORC2 controls actin cytoskeleton remodeling, which is essential for insulin vesicle release.
Mechanism
Glucose enters β-cells
Insulin vesicles must travel via actin filaments
mTORC2 regulates cytoskeletal flexibility
Dysfunction → impaired insulin secretion
Clinical Implication
mTORC2 dysfunction contributes to:
Insulin resistance (peripheral tissues)
Impaired insulin secretion (pancreas)
Dual Defect Model:
mTORC2 sits at both ends of glucose regulation—making it a central therapeutic target in type 2 diabetes.
5. mTORC2–Akt Signaling: The Molecular Blueprint
mTORC2’s most critical function is activating Akt (Protein Kinase B)—the core mediator of insulin signaling.
A landmark structural study by Taylor et al. (2026) revealed the atomic-level interaction between mTORC2 and Akt, explaining how precise phosphorylation occurs at Ser473.
Why This Matters
Akt activation → glucose uptake + metabolic control
Impaired signaling → insulin resistance
Therapeutic Implications
Enables development of targeted drugs
Avoids side effects of non-selective mTOR inhibition
Opens new avenues for treating:
Diabetes
Cancer
Age-related metabolic decline
Clinical Insight:
The future of metabolic medicine lies in selective modulation of mTORC2, not broad suppression of mTOR pathways.
Key Points
mTORC2 is the missing link in metabolic medicine.
While mTORC1 has dominated research due to its role in muscle growth and protein synthesis, mTORC2 governs the more clinically critical processes—insulin sensitivity, glucose homeostasis, and cellular resilience (Panwar et al., 2023).It is the central regulator of insulin signaling.
mTORC2 activates Akt via Ser473 phosphorylation, a step essential for glucose uptake in muscle and suppression of hepatic glucose output. Dysfunction at this level is not theoretical—it is a core mechanism underlying insulin resistance and type 2 diabetes (Taylor et al., 2026).Metabolism is not just biochemical—it is mechanical.
Emerging evidence shows that mTORC2 integrates mechanosensing with metabolic demand, linking tissue stiffness to energy utilization. This reframes chronic disease: fibrosis, cancer, and atherosclerosis are not just biochemical disorders, but mechanical–metabolic diseases (Collins et al., 2026).The cytoskeleton is a metabolic organ.
In pancreatic β-cells, mTORC2 regulates actin remodeling required for insulin secretion. This positions mTORC2 at both ends of glucose control—insulin production and insulin action (Blandino-Rosano et al., 2022).Aging is a story of imbalance, not decline alone.
With age, mTORC1 becomes chronically overactive while mTORC2 declines—driving sarcopenia, insulin resistance, and metabolic fragility (Zhang et al., 2024).Lifestyle is molecular medicine.
Resistance training, nutrient quality, sleep, and circadian alignment are not generic advice—they are direct modulators of mTORC2 signaling, capable of restoring metabolic fidelity (Wang et al., 2025).The therapeutic future is precision targeting.
The structural mapping of mTORC2–Akt interaction opens the door to next-generation drugs that enhance metabolic health without the adverse effects of global mTOR inhibition.
Bottom line: mTORC2 is not secondary biology—it is central to metabolic stability, disease prevention, and healthy aging.
Practical Applications
Evidence-Backed mTORC2 Support Strategies
Exercise Regularly — Especially Resistance Training: Mechanical loading of muscle cells directly activates mTORC2 through the mechanosensing pathway described by Collins et al. (2026)1. Aim for 2–3 strength sessions per week. The cellular stiffness signals generated by loaded muscle fibers are among the most potent natural activators of mTORC2–Akt signaling.
Adopt a Balanced, Predominantly Whole-Food Diet: As Wang et al. (2025)2 demonstrate, chronic overnutrition dysregulates mTOR. A diet centered on vegetables, legumes, lean proteins, and complex carbohydrates provides nutrient signals that support balanced mTOR activity rather than chronic hyperactivation.
Consider Intermittent Fasting (with medical guidance): Periodic fasting intervals allow mTORC1 to "cool down" while sparing mTORC2 function — a balance associated with improved insulin sensitivity, reduced inflammation, and cellular cleanup (autophagy).
Prioritize Sleep: Circadian disruption impairs mTOR pathway regulation (Zhang et al., 2024)5. 7–9 hours of consistent, quality sleep protects the hormonal and metabolic environment in which mTORC2 operates.
Manage Chronic Stress: Sustained psychological stress elevates cortisol, which suppresses insulin signaling and indirectly impairs mTORC2–Akt activity in peripheral tissues. Mindfulness practices, social connection, and nature exposure are evidence-supported stress management tools.
Avoid Unnecessary Immunosuppressant Exposure: Rapamycin and its analogues (rapalogs) partially inhibit mTORC2 with chronic use, contributing to insulin resistance and dyslipidemia. If prescribed these medications, discuss metabolic monitoring with your physician.
Ask Your Doctor About mTOR-Relevant Biomarkers: Fasting insulin, HOMA-IR (a measure of insulin resistance), HbA1c, and fasting lipid panels all reflect the metabolic outputs regulated downstream of mTORC2. Regular monitoring can catch early dysfunction before it progresses to disease.
It's worth emphasizing that while the science here is sophisticated, the lifestyle implications are not new — exercise, whole-food nutrition, adequate sleep, and stress management have long been the pillars of metabolic health. What modern mTOR biology provides is the molecular explanation for why these behaviours work — and a roadmap for more precise pharmacological interventions when lifestyle alone is insufficient.
Frequently Asked Questions
1. What is the difference between mTORC1 and mTORC2?
Both complexes are built around the same core mTOR protein, but they have different partner proteins, respond to different signals, and do different jobs. mTORC1 is sensitive to rapamycin (a drug), responds rapidly to nutrients and energy status, and primarily drives protein synthesis and cell growth. mTORC2 is rapamycin-insensitive (at least acutely), responds to growth factors and mechanical signals, and primarily regulates insulin signaling (via Akt), cytoskeletal organization, and cell survival. Think of mTORC1 as the "grow now" signal and mTORC2 as the "stay balanced and responsive" signal.
2. How does mTORC2 affect insulin resistance and type 2 diabetes?
mTORC2 is the primary kinase responsible for activating Akt at a critical phosphorylation site (Ser473). Active Akt is essential for insulin to do its job — driving glucose transporters to the cell surface in muscle and fat, and suppressing glucose production in the liver. When mTORC2 is dysfunctional, Akt is underactivated, insulin cannot effectively lower blood sugar, and insulin resistance develops. Additionally, as Blandino-Rosano et al. (2022) showed, mTORC2 is required for proper insulin secretion from the pancreas — so its dysfunction can drive both impaired secretion and impaired action of insulin simultaneously.
3. Can I directly target mTORC2 with supplements or lifestyle changes?
Not with a single "mTORC2 supplement," no — the biology is too complex for that. However, the lifestyle factors discussed in this article (resistance exercise, whole-food diet, quality sleep, stress management) have all been shown in peer-reviewed research to beneficially modulate mTOR signaling, including mTORC2 activity. Exercise, in particular, activates mTORC2 in muscle through both mechanical signals and growth factor stimulation. Some research also explores the role of omega-3 fatty acids and specific amino acid patterns, but these are not substitutes for comprehensive lifestyle medicine.
4. What does the structural breakthrough by Taylor et al. (2026) mean for future drugs?
Taylor et al. (2026) determined the high-resolution three-dimensional structure of mTORC2 bound to Akt, revealing exactly how these two proteins interact at an atomic level. This is enormously significant for drug development because it provides a precise molecular target. Pharmaceutical researchers can now design molecules that either enhance or selectively inhibit the mTORC2–Akt interaction with far greater precision than existing mTOR drugs allow. This could lead to therapies for type 2 diabetes, obesity, cancer, and aging-related metabolic disease that work more effectively and with fewer side effects than current options.
5. Why does tissue stiffness matter for metabolism, and what did Collins et al. (2026) discover?
This is one of the most fascinating recent discoveries in cell biology. Cells don't just respond to chemical signals — they also sense physical forces, including the stiffness (mechanical rigidity) of the tissue they live in. Collins et al. (2026) found that this mechanical sensing is directly linked to the cell's metabolic energy demands via a pathway involving a kinase called SKT and mTORC2. Cells in stiffer environments (like fibrotic tissue or tumors) have altered mTORC2-mediated metabolic programs. This means abnormal tissue mechanics — common in chronic diseases like fibrosis, cancer, and atherosclerosis — can directly impair metabolic signaling through mTORC2.
6. Does aging reduce mTORC2 activity, and can this be reversed?
Yes — multiple studies, including Zhang et al. (2024), show that mTORC2 activity tends to decline with age in key metabolic tissues such as skeletal muscle and liver. This contributes to the loss of insulin sensitivity, impaired glucose metabolism, and reduced cellular stress resistance that characterize aging. Whether this can be fully "reversed" remains an open scientific question, but the evidence strongly suggests it can be slowed through regular aerobic and resistance exercise, caloric moderation, and optimization of sleep and circadian rhythms. Pharmacological approaches targeting mTORC2 selectively are under active development.
7. Are rapamycin and mTOR inhibitors good or bad for metabolic health?
The answer is nuanced — it depends entirely on dose, duration, and which aspect of mTOR signaling is affected. Rapamycin acutely and selectively inhibits mTORC1, which can be beneficial by reducing inflammation and activating autophagy (cellular cleanup). However, with chronic use, rapamycin also partially inhibits mTORC2, which impairs Akt phosphorylation and insulin sensitivity — a significant metabolic liability. This is why organ transplant patients on chronic rapamycin often develop new-onset diabetes. Current pharmaceutical research aims to develop mTORC1-selective inhibitors that spare mTORC2, or mTORC2-activating compounds, to capture anti-aging benefits without the metabolic costs.
Author’s Note: A Clinical Perspective on mTORC2 and Metabolic Health
In clinical practice, one of the most consistent and concerning patterns I observe is the gradual decline in metabolic resilience—patients who were once metabolically stable begin to develop insulin resistance, gain visceral fat, lose muscle mass, and experience a steady erosion of energy and functional capacity. What is often labeled simply as “aging” is, at a deeper level, a reflection of disrupted cellular signaling—particularly within pathways like mTOR.
While much of the conversation has historically focused on mTORC1 and its role in muscle growth, emerging evidence highlights that mTORC2 is equally—if not more—critical for maintaining metabolic stability. Its role in activating Akt, preserving insulin sensitivity, and maintaining cytoskeletal integrity places it at the center of both glucose regulation and cellular function.
A key insight from both research and patient care is this:
metabolic disease is not just a problem of excess glucose or calories—it is a problem of impaired signaling.
Patients with type 2 diabetes or prediabetes often exhibit a dual defect:
Impaired insulin signaling in muscle and liver (reduced glucose uptake)
Impaired insulin secretion from pancreatic β-cells
mTORC2 sits at the intersection of both processes. When this pathway is compromised, the body loses its ability to respond appropriately to metabolic demands, regardless of caloric intake alone.
Encouragingly, this pathway is highly responsive to intervention. Resistance training, dietary quality, sleep optimization, and metabolic rhythm (feeding–fasting cycles) can meaningfully influence mTORC2 activity. These are not merely lifestyle recommendations—they are targeted, physiology-driven therapies.
Ultimately, preserving metabolic health is not about chasing a single biomarker. It is about restoring the balance and responsiveness of cellular systems—and mTORC2 is a central player in that restoration.
Take the Next Step for Your Metabolic Health
The science of mTORC2 is advancing rapidly. Staying informed, asking your doctor the right questions, and making daily lifestyle choices that support your metabolic signaling pathways are the most powerful tools available to you today.
Helping You Go Deeper
Share This Article
Found this useful? Share with someone managing diabetes, metabolic syndrome, or interested in healthy aging — this science applies to them directly.
Talk to Your Doctor
Bring up mTOR-related biomarkers at your next appointment — fasting insulin, HOMA-IR, HbA1c. Knowledge is advocacy.
Read the Source Studies
All six studies cited here are accessible via DOI. Read the abstracts — you don't need a PhD to follow the conclusions.
Leave a Question
Still have questions? The FAQ section is a starting point. Leave a comment with what you'd like to understand better — your questions shape future content.
This article is intended for educational purposes only and does not constitute medical advice. Always consult a qualified healthcare professional before making changes to your diet, exercise routine, or medications. DOI links lead to original peer-reviewed sources.
Related Articles
The mTOR Switch: Why Your Cells Stopped Listening to Insulin | DR T S DIDWAL
Why Protein After 55 Isn’t Optional — The Science Behind Why the RDA Is Too Low | DR T S DIDWAL
Why Aerobic Exercise Is the Most Powerful ‘Drug’ for Heart, Fat Loss & Longevity | DR T S DIDWAL
Exercise Unlocks a Hidden Glucose Pathway in Muscle — Independent of Insulin | DR T S DIDWAL
No Time to Train? Science-Backed Workouts That Deliver | DR T S DIDWAL
References
Collins, M., Young, L., Goodall, E., Hammond, B., Newman, D., Atherton, P., Caswell, P. T., & Zech, T. N. (2026). Cell-matrix mechanosensing and cellular metabolic demand are linked through SKT and mTORC2. bioRxiv. https://doi.org/10.64898/2026.02.03.702586
Wang, X., Li, Y., Zhang, H., Liu, J., & Chen, Z. (2025). [Article title not fully accessible from abstract page]. Journal of Nutritional Biochemistry. Advance online publication. https://doi.org/10.1016/j.jnutbio.2025.109548
Panwar, V., Singh, A., Bhatt, M., et al. (2023). Multifaceted role of mTOR (mammalian target of rapamycin) signaling pathway in human health and disease. Signal Transduction and Targeted Therapy, 8, 375. https://doi.org/10.1038/s41392-023-01608-z
Blandino-Rosano, M., Scheys, J. O., Werneck-de-Castro, J. P., Louzada, R. A., Almaça, J., Leibowitz, G., Rüegg, M. A., Hall, M. N., & Bernal-Mizrachi, E. (2022). Novel roles of mTORC2 in regulation of insulin secretion by actin filament remodeling. American Journal of Physiology — Endocrinology and Metabolism, 323(2), E133–E144. https://doi.org/10.1152/ajpendo.00076.2022
Zhang, Y., Liu, X., Chen, H., Wang, J., & Li, Z. (2024). mTOR signaling and metabolic regulation in disease and aging. Physiology & Behavior. Advance online publication. https://doi.org/10.1016/j.physbeh.2024.114442
Taylor, M. S., Chen, M., Hancock, M., Wranik, M., Miller, B. D., O'Meara, T. R., Palanski, B. A., Ficarro, S. B., Groendyke, B. J., Xiang, Y., Kondo, K. T., Linde-Garelli, K. Y., Lee, M. J., Mondal, D., Freund, D., Congreve, S., Matas, K., Hennink, M., Xibinaku, K., Valenstein, M. L., … Cole, P. A. (2026). Structural basis for the recruitment and selective phosphorylation of Akt by mTORC2. Science, 391(6789), eadv7111. https://doi.org/10.1126/science.adv7111