Leucine Threshold Explained: How Much Protein Per Meal to Build Muscle (Science-Based Guide)
Discover the leucine threshold for muscle growth and how much protein per meal you actually need. Learn how leucine activates mTOR, triggers muscle protein synthesis, and why protein timing matters.
EXERCISENUTRITION
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/30/202621 min read
What is the leucine threshold for muscle growth?
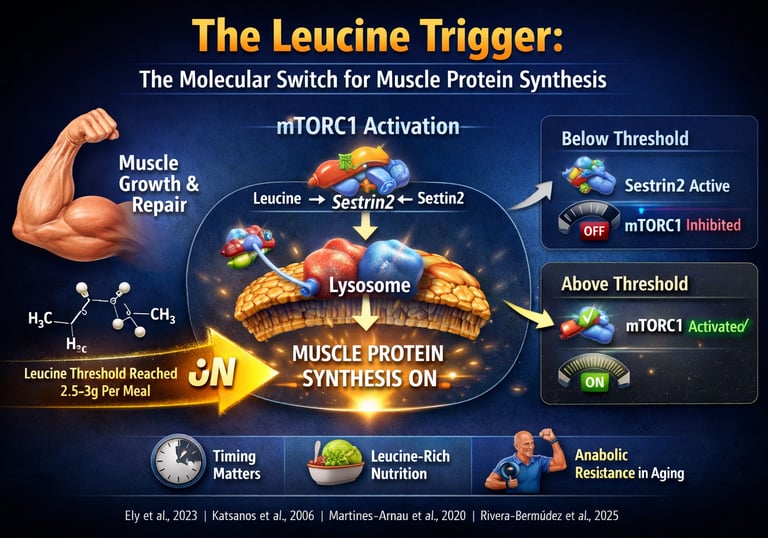
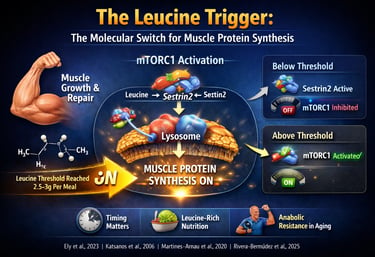
The leucine threshold is the minimum amount of leucine (~2.5–3 grams per meal) required to activate muscle protein synthesis (MPS) via the mTORC1 pathway. If this threshold is not reached, muscle-building signals remain inactive, regardless of total daily protein intake. This makes per-meal protein quality and distribution critical for muscle growth, aging, and metabolic health. Muscle growth is triggered per meal—not per day—by crossing the leucine threshold.
How to optimize protein intake for muscle growth
Combine with resistance training
Leucine threshold: Aim for 2.5–3g leucine per meal to activate muscle protein synthesis
Protein per meal: Consume 25–40g of high-quality protein per serving
Meal frequency: Target 3 main protein-rich meals per day
Meal spacing: Allow 3–4 hours between meals for optimal anabolic signaling
Older adults: Require higher leucine intake (~3–4g per meal) due to anabolic resistance
Best protein sources: Prioritize whey protein, dairy, eggs, and lean animal proteins for superior leucine content
Clinical Pearls: The Leucine Trigger
1. The "All-or-Nothing" Threshold
Muscle Protein Synthesis (MPS) is a non-linear, gated process. Sub-threshold leucine intake fails to disengage Sestrin2-mediated inhibition of mTORC1, resulting in negligible anabolic signaling regardless of total nitrogen balance.
Think of your muscle-building machinery like a motion-sensor light. If you don't walk past it with enough "force" (2.5–3g of leucine), the light stays off. Eating a little bit of protein won't turn the switch halfway; you have to hit the target to see results.
2. Overcoming Anabolic Resistance in Aging
Aging is characterized by a rightward shift in the leucine dose-response curve. To achieve fractional synthetic rates comparable to younger cohorts, older adults require a higher bolus (3–4g) to overcome age-related "noise" in the mTORC1 pathway.
As we get older, our muscles become "hard of hearing." To get them to listen and start building tissue, you have to "shout" louder by putting more protein on your plate at every single meal—especially breakfast.
3. The Danger of "Anabolic Blurring" (Grazing)
Continuous low-level amino acidemia prevents the necessary "reset" of Rag GTPases. This refractory period is essential for maintaining cellular sensitivity to nutrient pulses; constant grazing leads to a state of "anabolic deafness."
Snacking on tiny bits of protein all day long is actually worse than eating three square meals. Your muscles need a break to "reset" so they can respond to the next big protein hit. If you graze, the signal gets blurry and your body stops paying attention.
4. The "Synergy of Stress" (Exercise + Leucine)
Resistance exercise acts as a potent sensitizer to the leucine trigger. Mechanical loading upregulates the translocation of mTORC1 to the lysosomal membrane, significantly lowering the leucine threshold required to initiate MPS for up to 24–48 hours.
Lifting weights prepares the soil, and leucine is the seed. When you exercise, your muscles become much more sensitive to the protein you eat afterward. It makes every gram of leucine work twice as hard for you.
5. Bioavailability vs. Total Content
The kinetics of "leucinemia" (the rate of leucine appearance in the blood) dictate the magnitude of the anabolic peak. Rapidly digested proteins (whey) generate a superior signal to slow-digesting proteins (wheat or collagen) even when leucine totals are matched.
Speed matters. A protein shake that gets into your system quickly creates a "spike" that wakes up your muscles. Slow-digesting plant proteins are healthy, but you often need to eat more of them—or add a leucine supplement—to get that same wake-up call.
6. The Overnight Safety Net (Casein)
While daytime strategy focuses on "pulses," the overnight fast is a period of unavoidable catabolism. Pre-sleep ingestion of slow-release casein provides a sustained "trickle" of leucine that attenuates muscle breakdown without blunting the morning's anabolic sensitivity.
Your body tries to "eat" its own muscle for energy while you sleep. Having a slow-digesting protein like cottage cheese or a casein shake before bed acts like a slow-release IV drip, protecting your hard-earned muscle until breakfast.
The Leucine Trigger: The Molecular Switch That Controls Muscle Protein Synthesis
What if muscle growth isn’t determined by how much protein you eat—but by whether your body receives a precise molecular signal strong enough to activate muscle protein synthesis (MPS)?
At the cellular level, skeletal muscle is governed by a highly regulated nutrient-sensing system centered on mTORC1 (mechanistic target of rapamycin complex 1)—a master regulator of anabolic metabolism that integrates signals from amino acids, insulin, and energy status. Among all nutritional inputs, one signal stands above the rest: leucine, an essential branched-chain amino acid that functions not merely as a substrate, but as a metabolic trigger for muscle growth (Ely et al., 2023).
This process is not gradual—it is threshold-dependent. Unless circulating leucine levels rise sufficiently to activate intracellular sensors such as Sestrin2, the mTORC1 pathway remains functionally silent, and muscle protein synthesis does not meaningfully increase. In contrast, once the leucine threshold (~2.5–3 g per meal) is exceeded, mTORC1 is rapidly activated, initiating a cascade of translational signaling that drives muscle repair, hypertrophy, and metabolic adaptation (Katsanos et al., 2006; Rivera-Bermúdez et al., 2025).
This “all-or-nothing” phenomenon fundamentally reframes modern protein nutrition. It explains why total daily protein intake alone is insufficient—and why protein timing, leucine-rich foods, and per-meal protein dosing are critical determinants of anabolic response. These principles are especially relevant in the context of aging, sarcopenia, insulin resistance, and metabolic health, where impaired anabolic sensitivity—often termed anabolic resistance—demands a stronger leucine signal to achieve the same physiological effect (Martínez-Arnau et al., 2020; Guo et al., 2022).
Understanding the leucine trigger is therefore not just an academic exercise—it is a clinically actionable framework for optimizing protein distribution, resistance training adaptation, and long-term muscle preservation. In an era where muscle mass is increasingly recognized as a key determinant of longevity and cardiometabolic health, mastering this molecular switch may be one of the most important nutritional strategies in modern medicine.
Leucine is not just a building block—it is the primary trigger for muscle protein synthesis.
mTORC1 activation depends more on leucine availability than total protein intake.
Without sufficient leucine, muscle-building pathways remain switched off.
Leucine, Insulin & Net Protein Balance
Muscle growth is not determined by muscle protein synthesis (MPS) alone. It is the result of net protein balance, defined as:
MPS (building)
Muscle protein breakdown (MPB) (degradation)
Leucine plays a dominant role in stimulating MPS via mTORC1 activation. However, insulin — released in response to mixed meals — plays a complementary role by suppressing MPB.
This creates a dual-signal system:
Leucine → activates synthesis (MPS)
Insulin → suppresses breakdown (MPB)
Importantly, leucine alone can stimulate MPS transiently, but without adequate insulin (or energy availability), the suppression of breakdown is incomplete. This is why mixed meals containing protein + some carbohydrate often produce a more favorable net anabolic environment than isolated amino acid intake.
Clinical implication:
For older adults or catabolic patients, pairing leucine-rich protein with modest carbohydrate intake may optimize net muscle retention, not just synthesis signaling.
The BCAA Hierarchy
Leucine belongs to the branched-chain amino acid (BCAA) family, alongside isoleucine and valine. All three are essential amino acids, meaning your body cannot synthesise them — they must come entirely from dietary sources. But within this trio, leucine occupies a uniquely dominant position.
While all three BCAAs contribute to protein structure and energy metabolism, only leucine has been consistently shown to independently stimulate muscle protein synthesis (MPS). Isoleucine plays a modest supporting role; valine has minimal direct anabolic effect. The dose-response relationship between leucine concentration in the blood — what scientists call leucinaemia — and the activation of mTORC1 is remarkably steep and specific (Rivera-Bermúdez et al., 2025).
Dietary Essentiality: The Absolute Bottleneck
Because the body cannot manufacture leucine, every molecule available for signalling must come from what you eat. This creates a hard dietary constraint: if leucine intake at any given meal is insufficient, the anabolic signal will not fire — regardless of how much total protein you have consumed over the day. Ijaz et al. (2025) emphasise this in their comprehensive review of leucine and whey protein in sarcopenic individuals, concluding that leucine adequacy at each eating occasion is a non-negotiable prerequisite for maintaining muscle mass, particularly in older adults.
Patient Takeaway
Think of leucine as the key to a car's ignition. Protein is the fuel, but without the key — enough leucine — the engine won't start. This is why the type and timing of your protein matters as much as the amount.
The Sestrin2 Mechanism: How Your Cells "See" Leucine
For decades, the "missing link" in nutritional science was how a single amino acid could dictate cellular growth. The discovery of Sestrin2—a high-affinity leucine sensor—provided the landmark breakthrough that explained exactly how your body "interrogates" its nutritional environment.
The Molecular "Gating" System: How It Works
Think of Sestrin2 as a sensitive molecular brake. When leucine is absent, the brake is clamped down. When leucine levels rise, the brake is released.
Low Leucine State (Brakes ON): In a fasted or low-protein state, Sestrin2 binds to a protein complex called GATOR2. This interaction sends a "stop" signal to mTORC1 (the body's master growth regulator), preventing the expensive metabolic investment of building new muscle.
High Leucine State (Brakes OFF): Once blood leucine levels cross the critical threshold, leucine molecules bind directly to Sestrin2. This causes Sestrin2 to change shape and "let go" of GATOR2.
mTORC1 Activation: Freed from the inhibitory grip of Sestrin2, mTORC1 is finally "unleashed" to begin the process of muscle protein synthesis
.
Lysosomal Recruitment: Where the Magic Happens
The signaling process isn't just chemical; it’s physical. For mTORC1 to actually start building muscle, it must physically move to the surface of the lysosome (the cell's recycling and signaling hub).
The Rag GTPase Shuttle: This relocation is managed by a family of proteins called Rag GTPases. They act as the "security guards" that escort mTORC1 to the lysosomal membrane.
Full Catalytic Power: Only once mTORC1 is docked at the lysosome can it reach full power to drive tissue repair and growth.
Clinical Context: Beyond Muscle Biology
While we primarily discuss Sestrin2 in the context of muscle, new research (Zhang et al., 2024) shows this leucine-mediated threshold effect also governs macrophage signaling and cardiovascular risk. This suggests that the way we "pulse" our leucine doesn't just impact our strength—it may influence our systemic metabolic health and longevity.
Key Takeaway: Your cells aren't just passively absorbing protein; they are actively counting leucine molecules. If you don't provide enough to "disengage" the Sestrin2 brake, the anabolic machinery stays dormant regardless of your total calorie intake.
The Leucine Threshold: The "All-or-Nothing" Muscle Growth Switch
Most people fail to build muscle because they never reach the leucine threshold per meal.
Protein “grazing” leads to weak, ineffective anabolic signals.
More protein is not always better—timing and threshold matter more.
Aging muscles require a stronger leucine signal to respond.
Anabolic resistance increases the leucine threshold to ~3–4g per meal.
Strategic protein timing can slow or reverse sarcopenia.
The 2.5g–3g Rule: Why Quality Beats Quantity
In nutritional science, the Leucine Threshold is the most critical factor for stimulating Muscle Protein Synthesis (MPS). Unlike other nutrients that provide a linear, dose-dependent response, leucine operates like a biological toggle switch.
The Threshold Mechanism: If a meal contains less than ~2.5g of leucine, the mTORC1 signal remains "OFF," and muscle repair is negligible.
The "All-or-Nothing" Principle: Once you cross the specific leucine threshold, the anabolic signal fires robustly. Consuming more than the required amount (e.g., 10g) does not result in more muscle; it simply reaches a physiological plateau.
Total Protein vs. Leucine: A meal can be "high protein" (e.g., 20g of collagen or wheat protein) but still fail to trigger growth if it lacks sufficient leucine density.
Anabolic Resistance: The Age-Adjusted Threshold
As we age, our cellular machinery becomes less sensitive to nutrient signals—a clinical phenomenon known as Anabolic Resistance. This means the "trigger" required to start muscle construction moves higher as we get older.
Leucine Signal Strength by Age Group:
Young Adults (< 50 years): * The Trigger: ~2.5g leucine per meal.
The Result: Reliable activation of the mTORC1 pathway and tissue repair.
Older Adults (> 50 years): * The Trigger: ~3g to 4g leucine per meal.
The Result: Requires a "louder" nutritional signal to overcome cellular resistance and prevent sarcopenia (age-related muscle wasting).
Evidence-Based Insight: The Katsanos Study
The clinical importance of this threshold was proven in the landmark Katsanos et al. (2006) study. Researchers compared two groups of elderly subjects:
Group A: Received a standard essential amino acid (EAA) mixture.
Group B: Received a leucine-enriched EAA mixture (leucine increased from 26% to 41%).
The Result: Despite consuming the same amount of total amino acids, only the leucine-enriched group showed significant improvements in muscle protein synthesis. This confirms that leucine concentration, not total protein volume, is the primary driver of muscle health in aging populations.
Clinical Pearl
For patients over 60, don't just prescribe "more protein." Prescribe leucine-dense boluses. To hit the 3.5g threshold, a patient typically needs ~40g of high-quality whey or lean meat per meal.
Protein Distribution vs. Anabolic Pulsing
Muscle growth is threshold-driven, not calorie-driven.
Hitting the leucine threshold is more important than daily protein totals alone.
Each meal must independently trigger an anabolic signal to build muscle.
Muscle protein synthesis operates in pulses, not continuously.
Eating protein every 1–2 hours can blunt anabolic signaling.
The optimal pattern is 3 leucine-rich meals spaced 3–4 hours apart.
Why Even Distribution Wins
Most people in Western eating patterns consume the vast majority of their daily protein at dinner. From a muscle health standpoint, this is suboptimal. Distributing protein intake across multiple meals — each exceeding the leucine threshold — produces superior anabolic signalling compared to consuming the same total protein in one or two large doses.
Practical prescription: 30g of complete protein three times daily (breakfast, lunch, dinner) consistently outperforms 10g/10g/70g patterns for stimulating muscle protein synthesis across the full day. Each meal must independently cross the leucine threshold to count as an anabolic event.
The Refractory Period: Timing Your "Pulses"
After a leucine pulse successfully activates mTORC1, the pathway enters a refractory period — typically 3–4 hours — during which it becomes temporarily desensitised. The Rag GTPases and associated regulators essentially need time to reset before they can respond to the next stimulus.
This has profound practical implications. Eating two leucine-sufficient meals within 90 minutes of each other does not produce double the anabolic response. The second "pulse" largely falls on deaf ears. Optimal spacing between leucine-rich meals is therefore 3–4 hours minimum (Ely et al., 2023).[2]
The Danger of "Anabolic Blurring"
The refractory period also explains a counterintuitive clinical finding: protein "grazing" — consuming small amounts of protein every 60–90 minutes throughout the day — can actually impair net muscle protein synthesis compared to the three-meal approach.
When leucine is chronically elevated at low-to-moderate levels (never crossing the threshold confidently, never fully falling to baseline), mTORC1 enters a state of persistent low-level activation that paradoxically makes it less responsive to clear anabolic signals. The pathway is neither fully on nor fully off. This blurring of the signal — a kind of nutritional noise — partially explains why elderly patients in hospital who receive small, frequent sip-feeds throughout the day can still lose significant muscle mass despite adequate caloric intake.
Common Mistake
Encouraging patients to "graze on protein" throughout the day in small amounts (5–10g per sitting) is not the same as adequate protein intake. Without crossing the leucine threshold, the anabolic switch never fires — regardless of total daily protein consumed.
Sources: Supplements vs. Whole Foods
Not all proteins are equal—leucine density determines anabolic potential.
Whey protein produces the fastest and strongest leucine spike.
Plant proteins often require higher doses or supplementation to match effects.
Bioavailability & Kinetics: The "Leucinaemia" Profile
Not all proteins trigger leucinaemia — the rapid spike in blood leucine concentrations — with equal efficiency. The kinetics of how quickly leucine enters the bloodstream after a meal profoundly affects the anabolic response. A sharp, high leucinaemia spike is more effective at activating mTORC1 than the same total leucine delivered slowly over hours.
To help your readers choose the right fuel for their goals, here is the breakdown of protein sources categorized by their leucine density and absorption kinetics:
High-Leucine "Fast" Proteins (Post-Exercise Gold Standard)
Whey Isolate
Leucine: ~3.0g (per 25g protein)
Speed: Rapid (45–90 min peak)
Anabolic Profile: Excellent. The rapid spike in blood leucine ("leucinaemia") makes it the most effective trigger for mTORC1 immediately following a workout.
High-Quality Whole Foods (Mid-Day Foundation)
Chicken / Lean Meats
Leucine: ~2.5g (per 25g protein)
Speed: Moderate (2–3 hr peak)
Anabolic Profile: Very Good. Provides a steady, reliable pulse that usually hits the threshold for most adults in a standard serving size.
Eggs
Leucine: ~2.2g (per 25g protein)
Speed: Moderate (2–3 hr peak)
Anabolic Profile: Very Good. While slightly lower in leucine per gram than whey, eggs have the highest biological value of any whole food.
Slow" Proteins (Overnight Recovery)
Casein / Dairy
Leucine: ~2.4g (per 25g protein)
Speed: Slow (4–7 hr sustained)
Anabolic Profile: Excellent at Night. Casein "clots" in the stomach, creating a slow-release "drip" of leucine that prevents muscle breakdown during sleep.
Plant-Based Sources (The Leucine Challenge)
Soy Protein
Leucine: ~2.1g (per 25g protein)
Speed: Moderate
Anabolic Profile: Good. The highest-quality plant protein, though it requires slightly larger servings to hit the 3g threshold.
Pea Protein
Leucine: ~1.8g (per 25g protein)
Speed: Moderate
Anabolic Profile: Moderate. Often used in blends; requires fortification or larger doses to maximize signaling.
Rice Protein & Legumes
Leucine: ~1.2g – 1.6g (per 25g protein)
Speed: Slow to Moderate
Anabolic Profile: Below Threshold (Solo). When eaten alone, these rarely trigger the "leucine switch." They must be combined with other sources or a leucine supplement to be effective for muscle growth.
Whey protein's exceptional anabolic profile is directly attributable to its rapid digestion and high leucine content — making it the gold standard for post-exercise nutrition. Verreijen et al. (2015) demonstrated in a double-blind RCT that a high-whey, high-leucine, vitamin D-enriched supplement significantly preserved lean mass during intentional weight loss in obese older adults — a clinically challenging scenario in which standard weight-loss approaches routinely cause unacceptable muscle loss alongside fat reduction.
Also Read: Leucine Foods Chart: Exactly How Much You Need Per Meal for Muscle Growth | DR T S DIDWAL
The Vegan Challenge: Closing the Leucine Gap
Plant proteins present a genuine clinical challenge. Almost all plant protein sources — legumes, grains, nuts, seeds — have meaningfully lower leucine density than animal proteins. A vegan patient consuming adequate total daily protein (e.g., 100g) from diverse plant sources may still fail to trigger sufficient leucine pulses at any individual meal.
The evidence-based solutions are practical and effective:
Blend strategically: Soy + pea protein combination offers the most complete and leucine-rich plant protein profile available.
Increase volume: Consuming larger servings of plant protein per meal (40–45g vs. 25–30g) compensates for lower leucine density.
Supplement leucine directly: 2–3g of free leucine added to a plant-protein meal effectively raises its anabolic potential to match animal-protein levels.
Choose high-leucine plants: Edamame, tofu, tempeh, and lentils offer the most leucine per gram among plant foods.
Clinical Applications & Practical Prescriptions
Leucine acts as a nutritional switch for metabolic health and muscle preservation.
Optimizing leucine intake improves insulin sensitivity via increased muscle mass.
Protein distribution is a therapeutic tool in aging and chronic disease.
Sarcopenia Management
For adults over 60, implement leucine-enriched protein pulses (≥3g leucine per meal, ×3 daily) spaced 3–4 hours apart. Target 1.2–1.6g/kg/day total protein. Resistance exercise within the anabolic window amplifies the leucine response synergistically. Guo et al. (2022) meta-analysis of 17 RCTs confirmed leucine supplementation significantly improves muscle mass and physical performance in sarcopenic older adults.
Type 2 Diabetes & Obesity
Skeletal muscle is the body's primary site of insulin-stimulated glucose disposal. Leucine-optimised nutrition — by preserving and building muscle mass — expands the "metabolic sink" for glucose, improving insulin sensitivity independently of weight loss. Prioritising leucine-rich meals before exercise maximises both anabolic and metabolic benefit simultaneously. Wenz et al. (2009) demonstrated how muscle-targeted interventions profoundly affect systemic metabolic disease trajectories.
Bedtime Protein Strategy
During overnight fasting (typically 8–10 hours), the body enters net negative protein balance — muscle breakdown modestly exceeds synthesis. Consuming 30–40g of casein protein before sleep provides a slow, sustained release of leucine that maintains low-level mTORC1 signalling through the night, significantly attenuating overnight muscle catabolism without causing the full anabolic refractory effect that would blunt morning breakfast signalling.
Post-Surgical Recovery
Surgical trauma induces a profound catabolic state — elevated inflammatory cytokines including IL-6 and TNF-α directly suppress muscle protein synthesis and accelerate muscle wasting. Bian et al. (2017) documented the powerful relationship between these inflammatory factors and sarcopenic decline. Leucine-optimised nutritional support — starting pre-operatively where possible — meaningfully attenuates surgical muscle loss and accelerates functional recovery.
The 24-Hour Leucine Signaling Protocol
07:00 AM | The Morning Anabolic Pulse
Meal: 3 eggs + Greek yogurt + 30g whey protein.
Protein/Leucine: ~50g Protein | ~4.2g Leucine.
Clinical Goal: To terminate the overnight catabolic (breakdown) state and aggressively "flip the switch" for muscle protein synthesis (MPS) to start the day.
12:00 PM | The Midday Anabolic Pulse
Meal: 150g Chicken breast + legumes + quinoa.
Protein/Leucine: ~40g Protein | ~3.3g Leucine.
Clinical Goal: To provide a second robust leucine signal. By spacing this 5 hours after breakfast, you allow the "refractory period" to end, making the cells sensitive to the signal once again.
04:00 PM | The Post-Exercise "Window" (Optional)
Meal: Whey isolate shake.
Protein/Leucine: ~25g Protein | ~3.0g Leucine.
Clinical Goal: To take advantage of increased post-exercise insulin sensitivity and mechanical loading. Whey's rapid kinetics ensure a sharp, fast spike in blood leucine levels.
07:00 PM | The Evening Anabolic Pulse
Meal: Salmon + green vegetables + side of cottage cheese.
Protein/Leucine: ~45g Protein | ~3.6g Leucine.
Clinical Goal: To maintain muscle mass through the evening. Using a whole-food source like salmon provides a slower release of amino acids compared to a shake, keeping the system "fed" for longer.
09:30 PM | The Overnight Anti-Catabolic Shield
Meal: Casein shake or 1 cup of cottage cheese.
Protein/Leucine: ~30g Protein | ~2.4g Leucine.
Clinical Goal: Not intended as a full anabolic "pulse," but rather a slow-release "drip" to minimize muscle breakdown during the 8-hour sleep fast.
Protocol Notes for Patients
Consistency: The goal isn't just "high protein"—it's hitting that 3g leucine target at least three times a day.
Spacing: Ensure at least 3–4 hours between pulses to avoid "anabolic blurring."
Flexibility: If you miss the post-workout shake, simply ensure your next whole-food meal is rich in leucine.
Key Takeaways for Clinical Practice
Dose Matters Per Meal, Not Per Day Total daily protein intake is a secondary metric. To trigger muscle growth, every individual eating occasion must independently cross the leucine threshold (approx. 2.5–3g). If a meal falls below this "trigger" point, the anabolic switch remains "OFF."
Frequency Requires Strategic Spacing. While multiple pulses are necessary, they must be spaced 3–4 hours apart. This honors the "refractory period," allowing the cellular machinery (mTORC1) to reset. Constant grazing prevents this reset, leading to "anabolic blurring."
Age-Adjusted Prescriptions Aging creates anabolic resistance, requiring a "louder" nutritional signal. For patients over 60, the threshold increases; aim for 35–40g of high-quality protein per meal, or use 2–3g of supplemental leucine to "fortify" smaller meals.
Source-Dependent Kinetics Match the protein's digestion speed to the clinical goal:
Whey: Rapid delivery for post-exercise recovery.
Casein: Slow, sustained release for overnight muscle protection.
Whole Foods: Steady amino acid supply for standard daily meals.
Exercise as the Synergistic Amplifier: Nutrition and training are partners, not alternatives. Resistance exercise dramatically increases the muscle's sensitivity to leucine for up to 48 hours, making every gram of protein more "potent" after a workout.
The Power of the Pulse Consistent, high-quality "pulses" of leucine are superior to a single large dose at dinner. Distributing protein evenly across three main meals ensures the body spends more time in an anabolic (building) state rather than a catabolic (breaking down) state.
Why Most Protein Strategies Fail
“I eat enough protein per day”
→ But no single meal crosses leucine thresholdProtein grazing
→ Blunts mTOR responsivenessOver-reliance on plant proteins without adjustment
→ Chronic sub-threshold leucine intakeMega-dosing leucine
→ No added benefit beyond plateauIgnoring resistance training
→ Leucine signal remains underutilized
Frequently Asked Questions
Here are the evidence-based answers to the most common questions regarding the leucine trigger and muscle metabolism.
1. How much leucine do I actually need per meal to build muscle?
For most healthy young adults, 2.5g of leucine per meal is the "threshold" required to fully activate the mTORC1 pathway. However, this is not a one-size-fits-all number. If you are over the age of 50, you likely require 3–4g per meal to achieve the same muscle-building signal due to age-related anabolic resistance.
2. Can I just take leucine supplements instead of eating more protein?
No. While leucine is the "ignition key" that starts the engine, you still need the "fuel"—the other eight essential amino acids (EAAs)—to actually build the tissue. Taking leucine in isolation without a complete protein source is like turning the car's key but having an empty gas tank; the signal fires, but there are no raw materials to construct new muscle.
3. Is whey protein actually superior to whole food protein sources?
In terms of kinetics, yes. Whey is a "fast" protein, meaning it causes a rapid spike in blood leucine levels (leucinaemia), which is ideal for triggering the mTORC1 switch, especially post-workout. Whole foods like steak or eggs are excellent but digest more slowly, creating a lower, longer "hump" of leucine rather than a sharp "spike." For maximum muscle growth, a combination of both is usually best.
4. I’m vegan. Is it really possible to get enough leucine for muscle growth?
Absolutely, but it requires more strategy. Plant proteins (like beans or rice) are naturally lower in leucine than animal proteins. To hit the 2.5–3g threshold, a vegan athlete usually needs to consume a larger volume of protein (e.g., 40g of plant protein vs. 25g of whey) or strategically use leucine-enriched plant blends like Soy + Pea to mimic the amino acid profile of dairy.
5. Why does sarcopenia (muscle wasting) happen even when elderly people eat enough protein?
The issue is often distribution, not just total amount. Many older adults eat a low-protein breakfast and lunch, and a high-protein dinner. Because they have "anabolic resistance," their breakfast and lunch never cross the leucine threshold, meaning they spend 20 hours a day in a "muscle-wasting" state. To fight sarcopenia, every meal must cross the 3g leucine mark.
6. What is the "anabolic window" and how real is it?
The "30-minute window" is largely a myth. However, the anabolic opportunity is very real. Your muscles remain sensitized to leucine for 24–48 hours after resistance training. While you don’t need to chug a shake the second you drop the dumbbells, consuming a leucine-rich meal within 1–3 hours of exercise maximizes the synergistic effect of the workout and the nutrient signal.
7. Is it safe to take leucine supplements if I have type 2 diabetes or kidney disease?
Diabetes: Leucine actually helps improve glucose metabolism by building more muscle (your body's biggest "sugar sink").
Kidney Disease: This is a critical exception. Patients with Chronic Kidney Disease (CKD) must often limit total protein to reduce the workload on the kidneys.
Important: If you have a diagnosed kidney condition, never start a high-protein or leucine regimen without direct supervision from your nephrologist or renal dietitian.
8 .How much protein per meal to build muscle?
Most adults require 25–40g of high-quality protein per meal to reach the leucine threshold (~2.5–3g leucine), which is necessary to maximally stimulate muscle protein synthesis.
9 .Is 30g protein enough per meal?
For younger adults, ~30g protein per meal is typically sufficient. Older adults may require 35–40g per meal due to anabolic resistance.
10 .What are the best leucine-rich foods?
Top sources include:
Whey protein
Chicken and lean meats
Eggs
Dairy (especially Greek yogurt)
Soy protein
11 .Can you build muscle without hitting the leucine threshold?
Unlikely. Without sufficient leucine per meal, mTORC1 activation remains suboptimal, limiting muscle protein synthesis regardless of total daily protein.
Case Study: Reversing Anabolic Resistance
A 68-year-old male with early sarcopenia presented with:
Progressive muscle loss
Reduced grip strength
Protein intake ~60g/day
Despite “adequate” intake, distribution was skewed:
Breakfast: 10g protein
Lunch: 15g
Dinner: 35g
Problem: No meal reached leucine threshold consistently.
Intervention:
Restructured to 3 meals × 35–40g protein
Added 2g leucine to breakfast
Introduced resistance training (3×/week)
Outcome (12 weeks):
↑ Lean mass
↑ strength
Improved functional capacity
Key insight:
Total protein was not the issue. Signal strength per meal was.
Author’s Note (Clinician’s Perspective)
As a physician working at the intersection of metabolic health, aging, and nutrition science, I have seen a recurring and often underappreciated clinical pattern: patients who are “meeting their protein requirements” on paper, yet continue to lose muscle mass, strength, and functional capacity.
This disconnect is not a failure of patient compliance—it is a limitation of how we have historically prescribed protein.
Traditional dietary guidance focuses on total daily protein intake (e.g., 0.8–1.0 g/kg/day), but this approach overlooks a critical physiological reality: skeletal muscle does not respond to protein in a continuous, cumulative manner. Instead, it responds to discrete anabolic signals, each of which must reach a sufficient threshold—primarily driven by leucine—to activate muscle protein synthesis (MPS) via mTORC1.
In clinical practice, this insight has profound implications.
I routinely encounter:
Older adults consuming 60–70g protein/day but distributed as 10g breakfast, 15g lunch, 40g dinner
Hospitalized or post-surgical patients receiving frequent low-protein feeds that fail to trigger anabolic signaling
Patients with obesity or type 2 diabetes who are losing lean mass during weight loss despite “adequate” nutrition
In each case, the issue is not quantity—it is signal failure.
By restructuring protein intake into leucine-optimized pulses (≥2.5–3g leucine per meal) spaced across the day, the physiological response changes dramatically. When combined with resistance training, even modest interventions can restore anabolic sensitivity, improve muscle function, and alter long-term metabolic trajectories.
From a therapeutic standpoint, this reframes protein as more than a macronutrient—it becomes a precision metabolic tool.
It is equally important, however, to apply this knowledge judiciously. Patients with chronic kidney disease, advanced liver disease, or complex metabolic conditions require individualized assessment. Similarly, leucine supplementation should not be viewed as a shortcut, but as an adjunct to well-structured, whole-food-based nutrition.
Finally, as our understanding of muscle biology evolves, one principle is becoming increasingly clear:
muscle is not merely a structural tissue—it is a central organ of metabolic health, longevity, and resilience.
Optimizing leucine signaling is not just about improving muscle mass. It is about preserving independence, enhancing recovery, and fundamentally improving the quality of aging.
For a deep dive into the clinical research on how leucine specifically fights age-related muscle loss, read our full guide on Sarcopenia
Leucine and Muscle Health: The Essential Amino Acid for Fighting Sarcopenia | DR T S DIDWAL
Share your experience: Are you implementing leucine-threshold nutrition? Notice a difference with protein timing? Leave a comment below, share this article with someone managing sarcopenia.
Medical Disclaimer
This article is intended for educational purposes and does not constitute individualised medical advice. Nutritional interventions, particularly in the context of chronic disease, sarcopenia, post-surgical recovery, or kidney disease, should be implemented under the supervision of a qualified healthcare professional. Always consult your physician or registered dietitian before making significant changes to your diet or supplement regimen.
Content reviewed against peer-reviewed literature. Last updated 2026
Related Articles
Leucine and Insulin: How Protein Controls Blood Sugar and Muscle Growth | DR T S DIDWAL
Whey Protein vs Other Proteins: Which Builds More Muscle & Controls Insulin Best? | DR T S DIDWAL
AMPK vs mTOR: The Molecular Switch That Controls Muscle, Fat Loss, and Aging | DR T S DIDWAL
Intermittent Fasting: Metabolic Health Benefits and the Evidence on Longevity | DR T S DIDWAL
Activate Your Brown Fat: A New Pathway to Longevity and Metabolic Health | DR T S DIDWA
References
Ijaz, A., Ain, H. B. U., Tufail, T., Mariam, R., Noreen, S., Amjad, A., Ikram, A., Arshad, M. T., & Abdullahi, M. A. (2025). Enhancing muscle quality: Exploring leucine and whey protein in sarcopenic individuals.Journal of Cachexia, Sarcopenia and Muscle, 16(5), e70060.https://doi.org/10.1002/jcsm.70060
Ely, I. A., Phillips, B. E., Smith, K., Wilkinson, D. J., Piasecki, M., Breen, L., Larsen, M. S., & Atherton, P. J. (2023). A focus on leucine in the nutritional regulation of human skeletal muscle metabolism in ageing, exercise and unloading states.Clinical Nutrition, 42(10), 1849–1865.https://doi.org/10.1016/j.clnu.2023.08.010
Guo, Y., Fu, X., Hu, Q., Chen, L., & Zuo, H. (2022). The effect of leucine supplementation on sarcopenia-related measures in older adults: A systematic review and meta-analysis of 17 randomized controlled trials.Frontiers in Nutrition, 9, 929891.https://doi.org/10.3389/fnut.2022.929891
Martínez-Arnau, F. M., Fonfría-Vivas, R., Buigues, C., Castillo, Y., Molina, P., Hoogland, A. J., van Doesburg, F., Pruimboom, L., Fernández-Garrido, J., & Cauli, O. (2020). Effects of leucine administration in sarcopenia: A randomized and placebo-controlled clinical trial.Nutrients, 12(4), 932.https://doi.org/10.3390/nu12040932
Katsanos, C. S., Kobayashi, H., Sheffield-Moore, M., Aarsland, A., & Wolfe, R. R. (2006). A high proportion of leucine is required for optimal stimulation of the rate of muscle protein synthesis by essential amino acids in the elderly.American Journal of Physiology: Endocrinology and Metabolism, 291(2), E381–E387.https://doi.org/10.1152/ajpendo.00488.2005
Verreijen, A. M., Verlaan, S., Engberink, M. F., Swinkels, S., de Vogel-van den Bosch, J., & Weijs, P. J. (2015). A high whey protein-, leucine-, and vitamin D-enriched supplement preserves muscle mass during intentional weight loss in obese older adults: A double-blind randomized controlled trial.The American Journal of Clinical Nutrition, 101(2), 279–286.https://doi.org/10.3945/ajcn.114.090290
Bian, A. L., Hu, H. Y., Rong, Y. D., Wang, J., Wang, J. X., & Zhou, X. Z. (2017). A study on relationship between elderly sarcopenia and inflammatory factors IL-6 and TNF-α.European Journal of Medical Research, 22(1), 25.https://doi.org/10.1186/s40001-017-0266-9
Zhang, X., Kapoor, D., Jeong, S.-J., et al. (2024). Identification of a leucine-mediated threshold effect governing macrophage mTOR signalling and cardiovascular risk.Nature Metabolism, 6, 359–377.https://doi.org/10.1038/s42255-024-00984-2
Rivera-Bermúdez, G., Pizarro-Segura, M. F., Quesada-Quesada, D., et al. (2025). Effects of leucine intake on muscle growth, strength, and recovery in young active adults: A systematic review of randomized controlled trials.Nutrire, 50, 13.https://doi.org/10.1186/s41110-025-00311-z
Dr. Didwal. (n.d.).Leucine and muscle health: The essential amino acid for fighting sarcopenia.https://drdidwal.com/leucine-and-muscle-health-the-essential-amino-acid-for-fighting-sarcopenia