Leucine and Insulin: How Protein Controls Blood Sugar and Muscle Growth
Discover how leucine, a key amino acid in protein, stimulates insulin, regulates blood sugar, and supports muscle growth. Learn the science behind protein’s metabolic effects, ideal intake, and what it means for diabetes, fat loss, and long-term health.
DIABETESNUTRITION
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/31/202618 min read
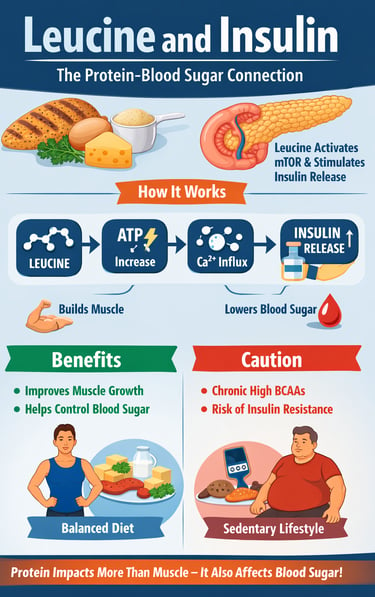
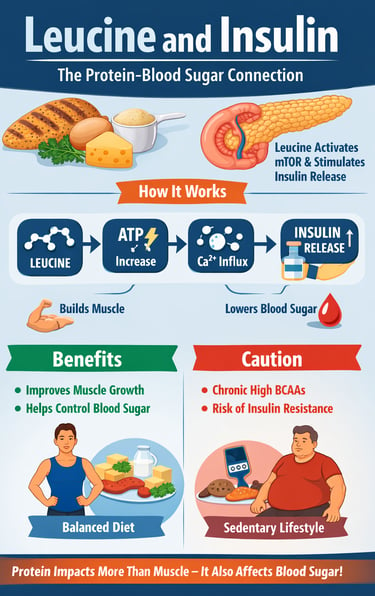
Leucine and insulin response refers to the ability of the amino acid leucine to stimulate insulin secretion and regulate blood sugar. Unlike carbohydrates, leucine produces a moderate insulin response but works synergistically with glucose to enhance metabolic control and muscle protein synthesis.
Leucine stimulates insulin (moderately)
Works best with carbs
Threshold: ~2.5–3g
Exercise determines outcome
Key Benefits of Leucine for Metabolism
Supports muscle protein synthesis via mTOR activation
Improves post-meal glucose control
Enhances nutrient partitioning
Helps preserve lean muscle during weight loss
Clinical pearls
1. The "Dual-Key" Secretagogue Mechanism
Scientific Perspective: Leucine acts as an independent insulin secretagogue by generating ATP to close KATP channels and by allosterically activating Glutamate Dehydrogenase (GDH). This creates a potent "double-signal" for insulin release within the pancreatic beta cell.
Think of leucine as a second key to the "insulin door." While sugar is the main key, leucine can also unlock that door. When you eat them together, the door opens wider, helping your body process nutrients more efficiently after a workout.
2. The Anabolic Resistance Threshold
Scientific Perspective: To overcome age-related anabolic resistance, the "leucine trigger" threshold shifts. While 2.5g may suffice for a 25-year-old, adults over 60 often require 3.5g to 4g per bolus to stimulate mTORC1 and protein synthesis maximally.
As we age, our muscles become "hard of hearing" to protein signals. To get the message through, you need a louder signal. Aiming for a slightly larger portion of high-quality protein (like a large chicken breast or a leucine-fortified shake) ensures your muscles actually "hear" the instruction to grow.
3. The Sedentary "Overflow" Paradox
Scientific Perspective: In a sedentary state, high BCAA levels can lead to mitochondrial overload. Incomplete oxidation of these amino acids creates metabolic intermediates that trigger a negative feedback loop, inhibiting IRS-1 (Insulin Receptor Substrate-1) and promoting insulin resistance.
Protein is a building block, but if you aren't "building" anything (through movement), those blocks just clutter the workspace. In a body that doesn't move, excess leucine can actually get in the way of how your insulin works. Exercise clears the "clutter" so leucine can do its job.
4. The Post-Exercise "Nutrient Sink"
Scientific Perspective: Post-resistance exercise, muscle cells increase GLUT4 translocation to the membrane. Leucine-stimulated insulin secretion at this specific window acts as a "partitioning agent," driving glucose and amino acids into the muscle "sink" rather than into adipose tissue.
Timing matters. After a workout, your muscles are like a dry sponge. Consuming leucine-rich protein right then uses your body's natural insulin response to soak up nutrients where they are needed most—in your muscles, not your fat cells.
5. Plant-Based Density Requirements
Scientific Perspective: Due to the lower leucine concentration in plant proteins (e.g., ~6–8% in soy vs. ~11% in whey), patients must consume 20–30% more total volume of plant protein or utilize targeted fortification to reach the 3g leucine "anabolic floor."
If you prefer plant-based proteins, you simply need a bigger plate. Because lentils or beans have less "muscle-building power" per gram than steak or eggs, you need to eat a larger portion or mix different types (like rice and peas) to hit the target that triggers muscle repair.
6. The mTOR/S6K1 Feedback Loop
Scientific Perspective: Chronic, nutrient-driven overactivation of mTORC1/S6K1 can paradoxically blunt insulin signaling. This highlights the importance of pulsatile protein intake (3–4 distinct peaks) rather than continuous "grazing," which may lead to sustained, maladaptive signaling.
Your muscle-building machinery needs "rest" just like you do. Instead of sipping on protein all day long, eat distinct, high-protein meals. This gives your body a clear "start" and "stop" signal, which keeps your metabolism sharp and prevents your cells from becoming "numb" to insulin.
Leucine: Protein’s Secret Weapon for Insulin Secretion, Muscle Growth, and Diabetes Management
Most people associate insulin with carbohydrates, blood sugar spikes, and diabetes. But emerging metabolic research shows that certain amino acids—especially leucine—can directly stimulate insulin secretion, even in the absence of glucose. This shifts how we think about protein, not just as a building block for muscle, but as an active regulator of blood sugar control, metabolism, and energy balance (De Bandt et al., 2023).
Leucine, a key branched-chain amino acid (BCAA) found in foods like whey protein, eggs, and meat, plays a dual role in the body. It activates muscle growth through mTOR signaling while also influencing pancreatic beta cells to release insulin. In simple terms, leucine helps your body decide when to build muscle and when to move glucose out of the bloodstream into cells (Jiang et al., 2021).
For patients managing type 2 diabetes, prediabetes, or insulin resistance, this is highly relevant. Protein-rich meals—especially those high in leucine—can produce a moderate insulin response without causing the sharp glucose spikes seen with high-glycemic carbohydrates. When combined with carbohydrates in a balanced meal, leucine enhances insulin secretion and improves post-meal glucose control.
However, context matters. While leucine can support metabolic health, chronically elevated BCAA levels—often seen in obesity and sedentary lifestyles—are linked to insulin resistance and cardiometabolic risk (McGarrah & White, 2023).
Understanding how leucine affects insulin helps you make smarter dietary choices—whether your goal is better blood sugar control, improved muscle health, or long-term metabolic fitness.
How Leucine Stimulates Insulin Secretion — The Science Explained Simply
To appreciate how leucine affects insulin, it helps to understand what happens when leucine enters the pancreatic beta cell — the specialised cell responsible for producing and releasing insulin.
1. Direct Beta-Cell Effects: The ATP–Potassium Channel Pathway
When leucine is metabolised inside beta cells, it generates ATP (energy). This rise in ATP causes potassium channels (KATP channels) to close. Closure of these channels triggers a change in the cell's electrical charge — called membrane depolarisation — which then opens calcium channels. Calcium rushes into the cell, and this calcium influx is the direct trigger for insulin release.
This is the same fundamental pathway that glucose uses to stimulate insulin, which explains why leucine can act as an independent insulin secretagogue (a substance that stimulates insulin release) — even without any carbohydrates present.
2. Allosteric Activation of Glutamate Dehydrogenase (GDH)
Leucine has a second, more nuanced mechanism. It allosterically activates an enzyme called glutamate dehydrogenase (GDH). This activation increases the conversion of glutamate to alpha-ketoglutarate, which then feeds directly into the TCA (Krebs) cycle — the cell's energy-producing machinery.
The practical result: more fuel for beta cells, which amplifies glucose-stimulated insulin secretion (GSIS). This means that when leucine and glucose are present together, insulin secretion can be significantly higher than with either stimulus alone.
3. Synergy With Glucose — The Mixed Meal Effect
Here is a clinically important nuance that is often overlooked: leucine alone produces only a modest insulin response. Its real power emerges when combined with carbohydrates. This is why mixed meals — protein plus carbohydrates — produce greater insulin responses than either macronutrient consumed separately.
For patients managing blood sugar or athletes timing their nutrition, this synergy matters enormously. A high-protein meal without carbohydrates will have a different insulin profile than the same meal eaten alongside rice, bread, or fruit.
Leucine on its own produces a modest insulin response, but when combined with carbohydrates, it significantly amplifies insulin secretion. This nutrient synergy enhances post-exercise recovery, which is why protein shakes paired with a small amount of carbohydrates are more metabolically effective.
Clinical Application: For patients with sarcopenia or athletes seeking recovery, the goal is pulsatile delivery—distinct spikes of leucine and glucose—rather than constant grazing, to maintain the sensitivity of the insulin signaling pathway.
Also read: AMPK vs mTOR: The Molecular Switch That Controls Muscle, Fat Loss, and Aging | DR T S DIDWAL
How Much Leucine Does It Take? Understanding the Dose–Response
One of the most practical questions clinicians and patients ask is: how much leucine is needed to trigger a meaningful insulin response and activate muscle protein synthesis?
Research consistently points to a threshold effect. Approximately 2 to 3 grams of leucine per meal appears necessary to stimulate a meaningful insulin and mTORC1 response. Below this threshold, the anabolic and metabolic signals are sub-optimal. Beyond 3 to 4 grams per meal, there are diminishing returns — more leucine does not proportionally increase the response.
To contextualise this: a standard 25-gram serving of whey protein contains approximately 2.5 to 3 grams of leucine. A chicken breast (approximately 100 grams) provides roughly 2.2 grams. For plant-based protein sources, leucine content is significantly lower, which has implications for both muscle building and insulin response.
Context matters: in a fasted state, the beta cells and muscle tissue are primed to respond more sensitively to leucine. In someone with insulin resistance, the same dose may produce an attenuated effect.
Leucine, mTOR, and Insulin Signalling — An Important Crosstalk
mTORC1 Activation and Insulin Pathways
One of the most remarkable features of leucine is that it activates mTORC1 — the central regulator of muscle protein synthesis — independently of insulin. This means even in a fasted state, with minimal circulating insulin, leucine can stimulate anabolic processes in muscle.
However, insulin and leucine work synergistically. Insulin enhances amino acid uptake into muscle cells and amplifies the mTOR signalling cascade. When both are present — as in a post-workout meal containing protein and carbohydrates — the anabolic response is maximised. This is the biochemical rationale behind post-exercise nutrition strategies that combine protein with moderate carbohydrates.
Chronic mTOR Activation and the Risk of Insulin Resistance
Here is where the science becomes more cautionary. While acute mTOR activation is beneficial, persistent nutrient-driven overactivation of mTOR — particularly the downstream kinase S6K1 — can paradoxically impair insulin sensitivity.
The mechanism involves a negative feedback loop: sustained S6K1 activation phosphorylates and inhibits insulin receptor substrate-1 (IRS-1), a critical node in the insulin signalling cascade. This effectively blunts the cell's response to insulin — the very definition of insulin resistance.
This is not a reason to avoid leucine; rather, it underscores the importance of context. Chronic overfeeding with high BCAA intake in sedentary individuals — without the metabolic "sink" provided by exercise — may contribute to insulin resistance over time. De Bandt and colleagues (2023) provided important mechanistic insights into how branched-chain amino acids can contribute to insulin resistance, particularly in the context of diet-induced obesity, emphasising that the harm is not from leucine itself but from the broader metabolic environment.
Important Nuance:
Acute leucine supplementation = beneficial for insulin secretion and muscle.
Chronic excess in sedentary, overfed individuals = potential contributor to insulin resistance. Context is everything.
Acute vs. Chronic Effects on Insulin Sensitivity
Acute Effects — Beneficial and Well-Documented
In the short term, leucine exerts broadly positive metabolic effects. Acutely, it improves postprandial insulin secretion and enhances glucose uptake into muscle tissue. Jiang and colleagues (2021) demonstrated in animal models that leucine supplementation combined with exercise produced distinct and complementary improvements in systemic insulin sensitivity — a finding with clear implications for exercise and nutrition timing strategies.
After exercise, muscle cells express increased GLUT4 transporter activity — the protein that ferries glucose from the bloodstream into muscle. Leucine-stimulated insulin release at this window further amplifies glucose uptake and glycogen replenishment, accelerating recovery and improving metabolic outcomes.
Chronic Excess — Where the Risk Emerges
The long-term picture is more complex. Elevated BCAA levels in the circulation — often called BCAA dyslipidaemia — have been consistently associated with insulin resistance and increased type 2 diabetes risk in epidemiological studies. McGarrah and White (2023) reviewed the role of branched-chain amino acids in cardiovascular disease, highlighting that elevated circulating BCAAs are increasingly recognised as independent markers of cardiometabolic risk — a reflection of underlying metabolic dysfunction rather than simply a cause.
The proposed mechanisms include mitochondrial overload from incomplete BCAA oxidation, leading to the accumulation of toxic metabolic intermediates that interfere with insulin signalling. It is worth emphasising that high circulating BCAAs in insulin-resistant individuals may reflect impaired BCAA catabolism — a consequence of the disease — rather than a primary driver.
Clinical Applications — Who Benefits Most From Understanding This?
Athletes and Active Individuals — Post-Exercise Nutrition
For people who exercise regularly, the leucine-insulin connection provides clear practical guidance. Consuming a leucine-rich protein source (25 to 40 grams of high-quality protein) alongside moderate carbohydrates within the post-exercise window maximises both muscle protein synthesis and glycogen replenishment. The insulin response acts as a "nutrient delivery signal" — shuttling both amino acids and glucose into muscle tissue at a time when the cells are primed to receive them.
Weight Management and Body Composition
Leucine's ability to stimulate insulin secretion — without the large glucose surge of high-carbohydrate foods — contributes to improved nutrient partitioning. High-protein meals rich in leucine tend to produce a moderate, sustained insulin response, which supports better body composition outcomes compared to high-carbohydrate, low-protein approaches.
Additionally, leucine has a well-documented role in preserving lean muscle mass during caloric restriction — a critical factor in sustainable fat loss that protects metabolic rate.
Aging and Sarcopenia — A Therapeutic Role
Older adults experience a well-characterised phenomenon called anabolic resistance — a blunted muscle protein synthesis response to both protein ingestion and exercise. Leucine supplementation has been proposed as a strategy to partially overcome this resistance by providing a stronger anabolic signal and a more robust insulin secretory response.
Wunderle et al.(2025) examined associations between leucine and other BCAAs with clinical outcomes in malnourished hospitalised patients. Their secondary analysis of the EFFORT randomised controlled trial found associations between BCAA status and clinical trajectories — underscoring the relevance of leucine in vulnerable populations where muscle preservation is not merely a cosmetic concern but a matter of survival and recovery.
Type 2 Diabetes — Friend or Foe?
This is the most clinically nuanced application. In early or uncomplicated type 2 diabetes, leucine's ability to stimulate insulin secretion can be genuinely beneficial — it provides an additional secretagogue signal that supports glucose disposal without the glycemic load of carbohydrates.
However, in advanced insulin resistance with chronically elevated BCAAs, the picture is more complicated. Dash and Mishra (2026) studied combined leucine and glutamine supplementation in a Drosophila model of type 2 diabetes and found that this combination could alleviate hyperglycaemia and oxidative stress by targeting insulin signalling genes — suggesting promising therapeutic directions, though translation to human clinical practice requires further study.
The key clinical principle: leucine is not a diabetic medication, but its metabolic effects are relevant to dietary management — particularly in the context of protein quality, meal composition, and physical activity.
For People With Type 2 Diabetes: Leucine-rich protein sources can support insulin secretion and muscle preservation. Always discuss dietary protein changes with your healthcare provider, especially if you are on insulin or sulfonylureas.
Leucine Across Different Dietary Patterns
High-Protein Diets
Higher protein intake naturally increases leucine consumption. Paradoxically, research suggests that high-protein diets — when not accompanied by excessive caloric intake — are associated with improved insulin sensitivity, better glycemic control, and enhanced body composition. The moderate, sustained insulin response to protein-rich meals appears metabolically preferable to the rapid insulin spikes produced by high-glycemic carbohydrates.
Plant-Based Diets
Plant proteins generally contain less leucine per gram compared to animal proteins. Legumes, tofu, and grains have lower leucine concentrations, meaning that plant-based eaters may need to consume larger total quantities of protein to reach the leucine threshold for optimal muscle protein synthesis and insulin stimulation.
This has practical implications: plant-based individuals may benefit from strategic leucine supplementation or from combining complementary protein sources (e.g., rice and peas) to achieve the leucine threshold per meal.
Intermittent Fasting
During an extended fast, insulin levels are low, and cells become sensitised to nutrient signals. When leucine-rich protein is consumed at the end of a fasting window, the insulin and mTOR response can be notably robust. This makes the first meal after a fast a high-leverage opportunity — consuming sufficient leucine at this point maximises anabolic and metabolic effects. However, the total daily protein target must still be met, as fasting windows alone do not compensate for chronically low protein intake.
Common Misconceptions: Setting the Metabolic Record Straight
"Leucine spikes insulin the same way sugar does."
The Clinical Reality: Inaccurate. Leucine is an insulin secretagogue, meaning it triggers release, but the magnitude is moderate compared to high-glycemic carbohydrates. A massive insulin surge typically only occurs through leucine-glucose synergy (the "Mixed Meal Effect"), where the two nutrients amplify each other's response.
"BCAAs are a direct cause of insulin resistance."
The Sedentary Nuance: Oversimplified. High circulating BCAAs are often a marker of metabolic dysfunction in sedentary, overfed individuals. Without the "metabolic sink" of exercise to oxidize these amino acids, incomplete metabolic byproducts accumulate and interfere with insulin signaling. In active individuals, this risk is virtually non-existent.
"Dietary protein has no impact on insulin levels."
The Clinical Reality: Incorrect. Protein—especially leucine-dense sources like whey, eggs, and dairy—is highly insulinogenic. This is a physiological advantage; it provides the "nutrient delivery signal" required to shuttle amino acids into muscle tissue without needing a high carbohydrate load.
"The more leucine you consume, the more muscle you build."
Quantitative Clarity: There is a definitive "ceiling" known as the Leucine Trigger. For most, 2.5g–3g per meal is the "anabolic floor." In older adults facing anabolic resistance, this threshold shifts upward to 3.5g–4g. Consuming 10g in one sitting does not yield 4x the growth; the excess is simply oxidized for energy.
"Leucine supplementation is a shortcut to metabolic health."
The Clinical Reality: Context-dependent. Leucine is a metabolic "key," but physical activity determines how wide the door actually opens. Chronic, high-dose leucine intake without resistance training can overactivate the mTOR/S6K1 pathway, which may paradoxically blunt insulin sensitivity over time.
"All protein sources trigger the same insulin and muscle response."
The Clinical Reality: Highly variable. The response is proportional to leucine density and absorption speed. Whey protein, being rapidly digested and leucine-rich, is significantly more insulinogenic and anabolic than slower-digesting or leucine-poor proteins like collagen or wheat.
Practical Applications — What You Can Do Today
Understanding the leucine-insulin connection is valuable only if it translates into actionable guidance. Here are evidence-informed strategies:
Optimise Post-Workout Nutrition
• Consume 25 to 40 grams of high-quality protein after exercise, ensuring at least 2.5 to 3 grams of leucine
• Pair with 30 to 50 grams of moderate-glycemic carbohydrates (e.g., banana, oats, rice) to amplify insulin secretion and glycogen replenishment
• Whey protein is optimal due to its high leucine content and rapid absorption kinetics
Structure Your Meals Strategically
• Aim for 25 to 35 grams of protein per meal across 3 to 4 meals daily, rather than concentrating all protein in one sitting
• Each meal should ideally meet the leucine threshold of 2.5 to 3 grams to maximise the mTOR and insulin signal
• Plant-based eaters: consider combining protein sources or using leucine-enriched plant protein blends
Contextualise Supplementation
• Leucine supplements (free-form or as part of BCAA products) may benefit older adults with anabolic resistance or those struggling to reach protein targets through food alone
• Avoid chronic high-dose BCAA supplementation without regular exercise — this combination may have adverse metabolic effects over time
• Those with type 2 diabetes should discuss any supplementation changes with their healthcare provider
Prioritise Exercise as the Foundation
• Regular resistance and aerobic exercise is the most potent modifier of leucine's metabolic effects
• Exercise increases muscle GLUT4 expression, insulin sensitivity, and BCAA utilisation — transforming leucine from a potential metabolic burden into a powerful anabolic and metabolic tool
Think of leucine as a key that unlocks the metabolic door — but exercise determines how wide that door opens and how long it stays open.
Key Takeaways
Insulin isn’t just about sugar
Most people think insulin is only released when you eat carbohydrates. But your body is smarter than that—protein, especially leucine, can also trigger insulin release.Leucine = a “signal nutrient”
Leucine is an amino acid found in foods like milk, eggs, chicken, and whey protein. It doesn’t just build muscle—it sends signals that tell your body what to do with nutrients.How it works (simple version)
When leucine enters the pancreas (where insulin is made), it:Increases energy inside the cell (ATP)
Triggers calcium entry
Signals the release of insulin
Think of it like flipping a switch that tells the body: “Start using nutrients now.”
Protein can help control blood sugar
Unlike sugary foods, leucine-rich protein causes a moderate and controlled insulin response, helping move glucose into cells without sharp spikes.Better together: protein + carbs
Leucine works best when combined with carbohydrates.
This combination leads to a stronger, more efficient insulin response—useful after meals or exercise.Why this matters for diabetes
Can support insulin secretion in early type 2 diabetes
Helps preserve muscle (important for glucose control)
May improve how the body handles blood sugar when combined with exercise
But more is NOT always better
Chronically high levels of BCAAs (including leucine), especially in sedentary individuals:May be linked to insulin resistance
Often reflect poor metabolic health rather than causing it directly
Exercise changes everything
Physical activity makes your muscles more sensitive to insulin and better at using leucine.
Without exercise, the benefits are much smaller.Practical takeaway
Include protein in every meal
Aim for high-quality protein (rich in leucine)
Combine with balanced carbs when needed
Stay physically active
Bottom line: Leucine is not just about muscle—it’s a powerful metabolic messenger that helps regulate insulin, blood sugar, and overall health.
Frequently Asked Questions
Q: Does leucine spike insulin the same way sugar does?
No. Leucine does stimulate insulin secretion, but the magnitude is considerably smaller than that of high-glycemic carbohydrates. Leucine produces a moderate, sustained insulin response. The response is amplified when leucine is consumed alongside carbohydrates in a mixed meal, but leucine alone does not produce the rapid, large insulin spike associated with sugary foods.
Q: Is leucine safe for people with type 2 diabetes?
In the context of a balanced, high-quality protein diet, leucine-rich foods are generally safe and potentially beneficial for people with type 2 diabetes — they support insulin secretion, lean muscle preservation, and blood sugar management. However, isolated BCAA supplementation in the setting of advanced insulin resistance warrants caution and should be discussed with a healthcare provider, particularly if the individual is on insulin or insulin-stimulating medications.
Q: Can leucine improve insulin sensitivity?
Leucine supplementation combined with regular exercise has been shown to improve systemic insulin sensitivity in research models. Leucine alone, without the metabolic demand of physical activity, is less likely to confer this benefit and may even be counterproductive in chronically overfed, sedentary individuals. Exercise is the essential co-factor.
Q: Is whey protein insulinogenic (does it stimulate a lot of insulin)?
Yes, whey protein is among the most insulinogenic protein sources available — more so than casein, soy, or plant proteins — primarily because of its high leucine content and rapid absorption profile. This is one reason why whey protein is frequently recommended for post-exercise recovery. The insulin response to whey is moderate compared to carbohydrates, but it is meaningful and clinically relevant.
Q: Should you take leucine without carbs?
You can, and leucine will still produce a modest insulin response and activate mTOR independently. However, for maximising muscle protein synthesis and glycogen replenishment — particularly post-exercise — pairing leucine-rich protein with moderate carbohydrates produces superior metabolic outcomes. The synergy between leucine and glucose is well-established and clinically useful.
Q: Do plant-based diets provide enough leucine?
Most plant protein sources contain less leucine per gram than animal proteins, and meeting the per-meal leucine threshold (2.5 to 3 grams) can be challenging on a plant-exclusive diet without careful planning. Soy protein and pea protein are the highest-leucine plant options. Plant-based eaters may need to consume higher total protein volumes, combine complementary sources, or use leucine-enriched plant protein supplements to match the anabolic and metabolic effects of animal proteins.
Q: Can older adults benefit from higher leucine intake?
Yes — older adults experience anabolic resistance, meaning they require more leucine per meal (approximately 3 to 4 grams) to achieve the same muscle protein synthesis response that a younger person achieves with 2.5 grams. Higher leucine intake per meal, distributed across multiple meals rather than concentrated in one sitting, is an evidence-informed strategy for preserving muscle mass and metabolic health in aging populations. This is a clinically meaningful intervention for sarcopenia prevention.
Take the Next Step — Your Personalised Leucine Strategy
Whether you are an athlete optimising recovery, someone managing blood sugar, or a clinician advising patients on protein nutrition, the leucine-insulin relationship offers actionable, evidence-based leverage points.
Here is how to get started:
Calculate your current daily leucine intake using a food tracking app
Identify whether you are meeting the 2.5-3g leucine threshold per meal
Pair your largest protein meals with moderate carbohydrates for synergistic effect
Prioritise resistance exercise at least 3 times per week — it transforms leucine metabolism
Review this information with your dietitian or healthcare provider for personalised guidance
Author’s Note
This article was written to bridge a common gap in how we understand nutrition—particularly the role of protein in metabolic health. In both clinical practice and public discourse, insulin is often viewed almost exclusively through the lens of carbohydrates and blood sugar. However, physiology is rarely that simple. Amino acids—especially leucine—play a meaningful and often underappreciated role in regulating insulin secretion, muscle metabolism, and overall energy balance.
The goal here is not to promote leucine as a “magic nutrient,” but to place it in the correct biological context. Like many aspects of metabolism, its effects are highly dependent on the surrounding environment—diet quality, physical activity, energy balance, and underlying metabolic health. When interpreted without this context, even well-established scientific findings can be misunderstood or oversimplified.
This piece draws on current mechanistic and clinical research to present a balanced, evidence-based perspective that is relevant to both clinicians and patients. Particular care has been taken to distinguish between acute physiological benefits (such as improved insulin secretion and muscle protein synthesis) and chronic metabolic patterns (such as elevated circulating BCAAs in insulin resistance), which are often conflated in popular discussions.
For clinicians, I hope this serves as a practical framework to guide conversations around protein quality, meal composition, and metabolic health. For patients and readers, the aim is to provide clarity—so that dietary choices are informed not by trends or misconceptions, but by physiology.
As always, nutrition should be individualized. The insights presented here are most powerful when adapted to the person in front of you—not applied as a one-size-fits-all solution.Have questions about applying this to your specific health context? Share this article with your healthcare provider and use the insights here as a conversation starter. Science is most powerful when it moves from the page to a practical plan.
Share this article with someone who is managing blood sugar or focused on muscle health
Bookmark for reference when planning post-workout nutrition or structuring high-protein meals
Use the misconceptions table to fact-check common claims you encounter on social media or in supplement marketing
Clinicians: consider sharing the FAQ section directly with patients for easy-to-digest clinical communication
This article is intended for educational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider before beginning a new exercise or nutrition program, especially if you have an existing medical condition.
Related Articles
You’re Missing This: Why mTOR Activation Is the Real Goal of Strength Training | DR T S DIDWAL
The mTOR Switch: Why Your Cells Stopped Listening to Insulin | DR T S DIDWAL
Why Aerobic Exercise Is the Most Powerful ‘Drug’ for Heart, Fat Loss & Longevity | DR T S DIDWAL
References
1. Dash, K., & Mishra, M. (2026). Combined supplementation of leucine and glutamine acts as a novel therapeutic approach to alleviate hyperglycemia and oxidative stress by targeting insulin signalling genes in a drosophila model of type 2 diabetes. Molecular Biology Reports, 53, 8. https://doi.org/10.1007/s11033-025-11171-1
2. De Bandt, J.-P., Coumoul, X., & Barouki, R. (2023). Branched-chain amino acids and insulin resistance, from protein supply to diet-induced obesity. Nutrients, 15(1), 68. https://doi.org/10.3390/nu15010068
3. Jiang, X., Zhang, Y., Hu, W., Liang, Y., Zheng, L., Zheng, J., Wang, B., & Guo, X. (2021). Different effects of leucine supplementation and/or exercise on systemic insulin sensitivity in mice. Frontiers in Endocrinology, 12, 651303. https://doi.org/10.3389/fendo.2021.651303
4. McGarrah, R. W., & White, P. J. (2023). Branched-chain amino acids in cardiovascular disease. Nature Reviews Cardiology, 20, 77–89. https://doi.org/10.1038/s41569-022-00760-3
5. Wunderle, C., Ciobanu, C., Ritz, J., et al. (2025). Association of leucine and other branched chain amino acids with clinical outcomes in malnourished inpatients: A secondary analysis of the randomized clinical trial EFFORT. European Journal of Clinical Nutrition, 79, 42–49.