Think Insulin Controls Weight? The Liver Is the Real Metabolic Powerhouse
Learn how hepatic metabolism drives insulin resistance, fatty liver, weight gain, and low energy—and why the liver is central to metabolic health
METABOLISM
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/2/202613 min read
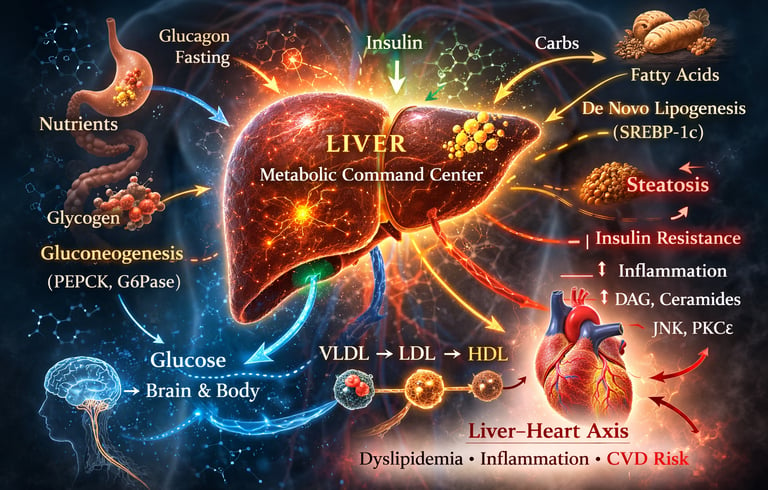
Every time you eat, skip a meal, or experience stress, one organ quietly decides what happens next — your liver.
Before glucose reaches your brain.
Before fats circulate to your muscles.
Before your energy rises or crashes.
Your liver has already made the metabolic decision.
Positioned between the digestive system and the rest of the body through the portal circulation, the liver functions as a real-time metabolic command center — integrating nutrient signals, insulin, glucagon, inflammatory mediators, and neural inputs within minutes (Zhang et al., 2019). It does not merely “process” food. It interprets it.
After a meal, insulin signals the liver to store glucose as glycogen and convert excess carbohydrates into fatty acids through de novo lipogenesis. During fasting, glucagon activates glycogen breakdown and gluconeogenesis to maintain stable blood glucose levels — a tightly regulated transcriptional program involving FOXO1, PGC-1α, PEPCK, and G6Pase (Zhang et al., 2019). This dynamic flexibility protects the brain from hypoglycemia and prevents dangerous swings in blood sugar.
But what happens when this command center becomes metabolically dysregulated?
In metabolic dysfunction-associated steatotic liver disease (MASLD), insulin signaling becomes selectively resistant. The liver may fail to suppress glucose production while continuing to drive fat synthesis — producing the paradox of hyperglycemia alongside hepatic fat accumulation (Truong & Lee, 2025). At the same time, altered VLDL secretion and impaired LDL receptor regulation reshape circulating lipoproteins into a pro-atherogenic profile, increasing cardiovascular risk (Winterfeldt & Siddiqi, 2026).
The consequences extend beyond the liver. Through hepatokine secretion, chronic low-grade inflammation, and dyslipidemia, a metabolically dysfunctional liver actively contributes to cardiovascular pathology — a relationship increasingly described as the liver–heart axis (Capone et al., 2025).
To understand modern cardiometabolic disease, we must look beyond insulin alone.
We must begin with the organ that decides, moment by moment, whether nutrients are stored safely — or transformed into disease.
Clinical pearls
1. The "Selective" Insulin Resistance Paradox
In the insulin-resistant liver, the AKT-FOXO1 pathway (suppressing gluconeogenesis) is impaired, while the SREBP-1c pathway (promoting lipogenesis) remains sensitive. This results in simultaneous fasting hyperglycemia and accelerated de novo lipogenesis.
Think of your liver like a radio with two stations. In a healthy state, insulin turns the "Sugar Production" station down and the "Fat Building" station up only when needed. In a diseased state, the "Sugar" knob gets stuck on high, while the "Fat" knob stays sensitive—so your body produces too much sugar and too much fat at the same time.
2. The Liver-Heart Axis & Systemic Risk
MASLD is an independent risk factor for CVD. The liver-heart axis is mediated by pro-atherogenic dyslipidemia (small, dense LDL) and the dysregulation of hepatokines (e.g., increased Fetuin-A, decreased FGF21 sensitivity), leading to myocardial inflammation and remodeling.
Your liver and heart are in a constant "group chat." When the liver is stressed and fatty, it sends out "emergency signals" (inflammatory proteins) that travel through the blood and irritate the lining of your heart and arteries. Protecting your liver is actually one of the best ways to protect your heart.
3. The "Silent" Progression of Fibrosis
Simple steatosis (NAFLD/MASLD) can progress to NASH (MASH) via a "multi-hit" model of oxidative stress and mitochondrial dysfunction. Fibrosis stage—not just the amount of fat—is the primary predictor of liver-related and all-cause mortality.
Liver damage is a "silent" process. You can’t feel fat or even early scarring in your liver because the organ has no pain receptors. By the time someone feels "liver pain" or turns yellow, the damage is often advanced. This is why using blood tests and scans to catch "silent" scarring early is life-saving.
4. Gluconeogenesis: The "Night Shift" Factory
During fasting (6–12+ hours), the liver shifts from glycogenolysis to gluconeogenesis, using precursors like lactate, glycerol, and alanine to maintain normoglycemia. In T2D, this "night shift" production is overactive due to high glucagon and cortisol levels.
Even if you don't eat sugar before bed, your liver "manufactures" its own sugar overnight to keep your brain fueled. If your liver is insulin resistant, it overproduces this sugar while you sleep, which is why many people wake up with high blood sugar levels even after fasting all night.
5. Non-Invasive Staging (The End of the Biopsy Era)
Clinical models (like the Liang & Ren 2026 model) and scores like FIB-4 or NAFLD Fibrosis Score use routine biomarkers (ALT, AST, Age, Platelets) to accurately exclude advanced fibrosis, reducing the need for invasive liver biopsies.
You don’t always need a painful needle biopsy to check your liver health. Modern math and specialized "vibration" ultrasounds (like FibroScan) can now look at your regular blood work and tell your doctor if your liver is becoming stiff or scarred with surprising accuracy.
1. Hepatic Glucose Production: Why Fasting Blood Sugar Spikes
The liver’s primary job between meals is maintaining normoglycemia to fuel the brain. It does this via two distinct pathways:
Glycogenolysis: The rapid breakdown of stored glycogen (active for the first 6–12 hours of fasting).
Gluconeogenesis: The "de novo" synthesis of glucose from non-carbohydrate sources like lactate and amino acids.
A 2019 study by Zhang et al. highlights that insulin suppresses glucose production by excluding the transcription factor FOXO1 from the nucleus. In metabolic disease, this suppression fails, leading to the high fasting glucose levels typical of pre-diabetes.
2. Lipid Metabolism: When the Liver Becomes a "Fat Factory"
When we consume excess carbohydrates, the liver initiates De Novo Lipogenesis (DNL)—converting sugar into fat. This fat is either stored (leading to a "fatty liver") or exported as VLDL (Very Low-Density Lipoprotein).
According to Winterfeldt & Siddiqi (2026), insulin resistance disrupts this balance. Instead of acting as a safety valve, the liver overproduces VLDL, leading to a "pro-atherogenic" profile: high triglycerides, low HDL, and dangerous small, dense LDL particles that clog arteries.
3. The Paradox of Selective Insulin Resistance
Why do patients with Type 2 Diabetes often have high blood sugar and a fatty liver? The answer lies in Selective Hepatic Insulin Resistance.
In a diseased liver, insulin signaling is "broken" for glucose but "hyper-active" for fat.
The Glucose Branch: The liver "ignores" insulin and keeps pumping out sugar.
The Fat Branch: The liver remains sensitive to insulin, which continues to drive fat production.
Truong & Lee (2025) identify lipotoxicity (DAGs and ceramides) and ER stress as the primary triggers for this molecular "short circuit."
4. MASLD: The New Frontier of Fatty Liver Disease
The medical community has officially rebranded Non-Alcoholic Fatty Liver Disease (NAFLD) as Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD). This change emphasizes that liver fat is a metabolic issue, not just a weight issue.
Early Identification Without Biopsy
Liver biopsies are invasive and risky. However, Liang & Ren (2026) have validated a machine-learning predictive model that uses routine biomarkers:
BMI and Waist Circumference
ALT/AST (Liver Enzymes)
Fasting Glucose & Triglycerides
This allows clinicians to catch liver disease in the "window of opportunity" before it progresses to irreversible fibrosis or cirrhosis.
5. The Liver-Heart Axis: Why Liver Health is Heart Health
The liver and heart are connected by a "bidirectional communication network." A diseased liver doesn't just sit there; it actively harms the heart through:
Hepatokine Dysregulation: Over-secretion of Fetuin-A (which causes heart insulin resistance) and loss of FGF21 (a cardio-protective protein).
Systemic Inflammation: The fatty liver releases inflammatory cytokines that accelerate plaque buildup in the coronary arteries.
RAAS Activation: Liver stress triggers hormonal imbalances that lead to hypertension.
The landmark Capone et al. (2025) review in Circulation Research confirms that MASLD is an independent risk factor for heart failure and atrial fibrillation.
Synthesis: A Unified View of Hepatic Metabolic Dysfunction
1️⃣ Your Liver Decides What Happens After Every Meal
The moment you eat, nutrients travel directly to the liver. It decides:
How much sugar to store
How much fat to make
How much energy to release
It acts before your brain even knows food has arrived.
2️⃣ Insulin Doesn’t “Lower Sugar” — It Talks to the Liver
When insulin rises after a meal, it tells the liver:
Stop producing glucose
Store extra sugar as glycogen
Convert excess carbohydrates into fat for later use
If the liver responds properly, blood sugar remains stable.
3️⃣ During Fasting, the Liver Protects You
When you skip meals or sleep:
The liver releases stored glucose
It makes new glucose to keep levels steady
It prevents dangerous drops in blood sugar
This flexibility is what keeps your energy consistent.
4️⃣ When the Liver Becomes Insulin Resistant, Chaos Begins
In metabolic dysfunction-associated steatotic liver disease (MASLD):
The liver continues producing glucose even when sugar is high
It keeps making fat at the same time
Blood sugar rises
Liver fat accumulates
This is why fatty liver and diabetes often appear together.
5️⃣ A Diseased Liver Affects the Heart
A stressed liver:
Produces abnormal cholesterol particles
Releases inflammatory proteins
Increases cardiovascular risk
The liver–heart connection is now a major focus of cardiometabolic research.
6️⃣ Weight Gain Is Not Just About Calories
If the liver is metabolically dysregulated:
More nutrients are converted into fat
Energy regulation becomes inefficient
Fat storage increases even without extreme overeating
7️⃣ True Metabolic Health Starts With the Liver
To understand insulin resistance, fatty liver, abnormal cholesterol, or unexplained weight gain — we must begin with liver function.
The liver is not a passive organ.
It is your metabolic command center.
And when it struggles, the entire system feels the consequences.
Frequently Asked Questions (FAQs)
1. What is the difference between glycogenolysis and gluconeogenesis?
Glycogenolysis is the breakdown of stored glycogen (a glucose polymer) already present in the liver, releasing glucose rapidly into the bloodstream. It is the first-line response to falling blood sugar, typically active within the first 6–12 hours of fasting. Gluconeogenesis, by contrast, synthesizes entirely new glucose molecules from non-carbohydrate precursors (lactate, amino acids, and glycerol). It becomes the dominant hepatic glucose-producing pathway during prolonged fasting, extended exercise, or in states of chronic insulin resistance. Both are regulated by glucagon, insulin, and cortisol, but through distinct enzymatic machinery.
2. What is selective hepatic insulin resistance, and why does it matter clinically?
Selective hepatic insulin resistance refers to a paradoxical state in which hepatic insulin signaling is impaired in one branch (gluconeogenesis suppression, via AKT-FOXO1) but preserved in another (lipogenesis activation, via SREBP-1c). The clinical consequence is devastating: the liver simultaneously overproduces glucose — contributing to fasting hyperglycemia — and overproduces fat — contributing to steatosis and dyslipidemia. This paradox explains why patients with type 2 diabetes and NAFLD often have elevated fasting glucose, elevated triglycerides, and fatty liver at the same time, and why treating one without the other is rarely sufficient.
3. What are the main risk factors for developing NAFLD?
The principal risk factors for NAFLD include obesity (especially visceral/central adiposity), type 2 diabetes and insulin resistance, metabolic syndrome (hypertension, dyslipidemia, hyperglycemia), excessive dietary intake of refined carbohydrates and saturated fats, physical inactivity, and genetic polymorphisms (notably in PNPLA3 and TM6SF2). Emerging research also points to gut microbiome dysbiosis and sleep-disordered breathing as contributory factors. Importantly, NAFLD can occur in non-obese individuals, particularly in those with visceral fat accumulation or genetic predisposition, highlighting the importance of metabolic phenotyping rather than BMI-based screening alone.
4. How does NAFLD progress to more serious liver disease?
NAFLD progression follows a multi-hit model. The first hit is hepatic fat accumulation (steatosis), driven by excess free fatty acid influx, insulin-stimulated de novo lipogenesis, and impaired lipid export. The second and subsequent hits involve oxidative stress, lipid peroxidation, endoplasmic reticulum stress, mitochondrial dysfunction, and inflammatory cytokine release (from Kupffer cells and adipose-derived macrophages). These insults cause hepatocyte injury and death, triggering stellate cell activation and fibrogenesis. Over years to decades, progressive fibrosis can lead to cirrhosis, portal hypertension, hepatocellular carcinoma, and liver failure — representing the most severe end of the NAFLD spectrum.
5. Why is NAFLD considered a cardiovascular risk factor?
NAFLD is now recognized as an independent cardiovascular risk factor, beyond the contribution of its associated conditions (obesity, diabetes, dyslipidemia). The steatotic liver actively promotes cardiovascular disease through multiple mechanisms: it secretes excess VLDL (raising triglycerides and generating atherogenic small, dense LDL), it releases pro-inflammatory hepatokines and cytokines into systemic circulation (accelerating atherosclerosis), it reduces cardioprotective factors such as FGF21, and it disrupts RAAS balance (contributing to hypertension). The Capone et al. (2025) review in Circulation Research demonstrates that patients with MASLD have a substantially elevated risk of coronary artery disease, heart failure, and cardiac arrhythmias, independent of traditional risk factors.
6. What are hepatokines, and how do they link the liver and heart?
Hepatokines are liver-secreted proteins that exert endocrine effects on distant organs, including the heart. Key hepatokines in the liver-heart axis include FGF21 (fibroblast growth factor 21), which under normal conditions improves insulin sensitivity and has cardioprotective properties; fetuin-A, which promotes insulin resistance and myocardial lipotoxicity when over-secreted by the steatotic liver; selenoprotein P, which impairs insulin action across multiple tissues; and angiopoietin-like proteins (ANGPTLs), which regulate lipoprotein lipase activity and triglyceride clearance. In MASLD, the hepatokine secretory profile shifts toward a pro-inflammatory, pro-atherogenic pattern — illustrating how a diseased liver can drive cardiovascular disease even at a distance.
7. Can NAFLD be detected early without a liver biopsy?
Yes, and this is an area of active and exciting research. The 2026 study by Liang and Ren in Frontiers in Physiology demonstrates that a clinical predictive model built from routinely available metabolic variables — including BMI, waist circumference, fasting blood glucose, liver enzymes (ALT, AST), and lipid levels — can identify NAFLD with high discriminatory accuracy. Additionally, non-invasive imaging modalities such as hepatic ultrasound, controlled attenuation parameter (CAP) on FibroScan, and MRI-PDFF (proton density fat fraction) allow for quantification of hepatic steatosis without biopsy. Blood-based biomarkers such as the FIB-4 index, NAFLD Fibrosis Score, and emerging ELF (enhanced liver fibrosis) panel are validated tools for assessing fibrosis stage non-invasively, enabling clinicians to triage patients for biopsy and prioritize intervention appropriately.
Key Research Summaries: The Future of Liver & Metabolic Health (2025-2026)
Understanding the liver's role as a metabolic command center is essential for managing Type 2 Diabetes, MASLD (formerly NAFLD), and Cardiovascular Disease. Below are the definitive summaries of five landmark studies that are reshaping modern cardiometabolic medicine.
Study 1: The Molecular Drivers of Fasting Hyperglycemia
Source: Zhang et al. (2019) | Frontiers in Endocrinology
This study maps the complex regulatory network of hepatic gluconeogenesis (the liver's production of sugar). It identifies how rate-limiting enzymes, PEPCK and G6Pase, are controlled by transcription factors like FOXO1 and PGC-1α.
The Breakthrough: The research explains how insulin normally suppresses sugar production via the AKT-FOXO1 pathway. When this "off switch" fails, the result is chronic fasting hyperglycemia.
Key Takeaway: Hepatic glucose production is governed by both genetic signals and post-translational changes (like acetylation). This architecture provides new therapeutic targets for treating high morning blood sugar in diabetics.
Study 2: The Liver’s Role in Cholesterol & Lipoprotein Dynamics
Source: Winterfeldt & Siddiqi (2026) | Livers
The liver is the body's primary "logistics hub" for fats. This 2026 review explores how the liver assembles VLDL particles, clears LDL through hepatic receptors, and manages HDL recycling.
The Breakthrough: It explains the "atherogenic lipid triad"—high triglycerides, low HDL, and small, dense LDL—seen in fatty liver patients. It specifically highlights ApoB-100 and PCSK9 as the most critical targets for preventing heart attacks in metabolic patients.
Key Takeaway: Insulin resistance breaks the liver’s lipid safety valves, shifting metabolism into a pro-inflammatory, artery-clogging state.
Study 3: The "Selective Resistance" Paradox in MASLD
Source: Truong & Lee (2025) | Diabetes & Metabolism Journal
This paper solves a medical mystery: why does an insulin-resistant liver stop regulating sugar but continue making fat? This is known as Selective Hepatic Insulin Resistance.
The Breakthrough: The authors link lipotoxicity (DAGs and ceramides) and ER stress to a "short circuit" in cellular signaling. While the path to stop sugar production is blocked, the path to create new fat (lipogenesis) remains wide open.
Key Takeaway: This bidirectional cycle of fat and insulin resistance is why GLP-1 agonists and SGLT2 inhibitors are so effective—they help break the molecular loop.
Study 4: Machine Learning for Early Fatty Liver Detection
Source: Liang & Ren (2026) | Frontiers in Physiology
As MASLD becomes a global epidemic, invasive liver biopsies are no longer practical for everyone. This study validates a high-performance clinical predictive model for early detection.
The Breakthrough: Using machine learning to analyze routine data (BMI, waist circumference, ALT/AST enzymes, and triglycerides), the researchers created a tool that identifies at-risk patients without expensive imaging or needles.
Key Takeaway: Early identification is the only way to prevent progression to cirrhosis and liver cancer. This model makes population-level screening accessible and accurate.
Study 5: Decoding the Liver-Heart Axis
Source: Capone et al. (2025) | Circulation Research
The most comprehensive map of the Liver-Heart Axis to date. This landmark review proves that what happens in the liver directly dictates the health of the heart.
The Breakthrough: The study identifies Hepatokines (liver-secreted proteins) like Fetuin-A and FGF21 as the "messengers" that cause heart inflammation and rhythm issues (Atrial Fibrillation) when the liver is diseased.
Key Takeaway: MASLD is now considered an independent cardiovascular risk factor. Doctors must treat the liver and heart as a single, integrated system (Cardio-Hepatology).
Author’s Note
As a clinician trained in internal medicine and deeply engaged in metabolic research, I have come to view the liver not simply as an organ of detoxification or digestion, but as the strategic command center of human metabolism. In daily practice, I repeatedly encounter patients treated aggressively for glucose, cholesterol, or blood pressure — yet the upstream metabolic dysfunction driving these abnormalities often remains unaddressed. Writing this piece was an effort to reposition the liver where it belongs: at the center of the cardiometabolic conversation.
The science over the past decade has been transformative. We now understand hepatic gluconeogenesis at a transcriptional level, appreciate the paradox of selective insulin resistance, and recognize metabolic dysfunction-associated steatotic liver disease (MASLD) as a systemic condition with profound cardiovascular implications. The concept of the liver–heart axis, in particular, represents a paradigm shift — forcing collaboration between hepatology, endocrinology, and cardiology in ways that were not emphasized in prior generations of training.
This article is written for multiple audiences. For clinicians, it is an invitation to screen earlier, interpret metabolic markers more integratively, and treat fatty liver as a cardiovascular condition rather than an incidental imaging finding. For researchers, it is a reminder that organ systems do not fail in isolation — they communicate continuously through molecular and hormonal networks. And for patients and health-conscious readers, it underscores a simple but powerful truth: lifestyle decisions influence not just body weight or laboratory numbers, but the very metabolic control systems that determine long-term health.
If there is one message I hope endures, it is this: preventing cardiometabolic disease requires upstream thinking. The liver sits at that upstream intersection. When we protect hepatic metabolic integrity, we are not merely preventing steatosis — we are preserving systemic resilience.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Why Blood Sugar Spikes Overnight — Even When You Eat Healthy All Day | DR T S DIDWAL
Why Fat Tissue Is an Endocrine Organ — And What It Means for Your Health | DR T S DIDWAL
Tired All Day, Awake at Night? The Redox and Mitochondrial Link | DR T S DIDWAL
Obesity and Fatty Liver Disease: What Science Says About Risk and Health | DR T S DIDWAL
Intermittent Fasting: Metabolic Health Benefits and the Evidence on Longevity | DR T S DIDWAL
Activate Your Brown Fat: A New Pathway to Longevity and Metabolic Health | DR T S DIDWAL
Leptin vs. Adiponectin: How Your Fat Hormones Control Weight and Metabolic Health | DR T S DIDWAL
References
Capone, F., Vacca, A., Bidault, G., Sarver, D., Kaminska, D., Strocchi, S., Vidal-Puig, A., Greco, C. M., Lusis, A. J., & Schiattarella, G. G. (2025). Decoding the liver-heart axis in cardiometabolic diseases. Circulation Research, 136(11), 1335–1362. https://doi.org/10.1161/CIRCRESAHA.125.325492
Liang, T., & Ren, J. (2026). Development and validation of a high-performance clinical predictive model for early identification of non-alcoholic fatty liver disease. Frontiers in Physiology, 17, 1689882. https://doi.org/10.3389/fphys.2026.1689882
Truong, X. T., & Lee, D. H. (2025). Hepatic insulin resistance and steatosis in metabolic dysfunction-associated steatotic liver disease: New insights into mechanisms and clinical implications. Diabetes & Metabolism Journal, 49(5), 964–986. https://doi.org/10.4093/dmj.2025.0644
Winterfeldt, K., & Siddiqi, S. A. (2026). The liver at the crossroads: Orchestrating lipoprotein dynamics and lipid homeostasis. Livers, 6(1), 6. https://doi.org/10.3390/livers6010006
Zhang, X., Yang, S., Chen, J., & Su, Z. (2019). Unraveling the regulation of hepatic gluconeogenesis. Frontiers in Endocrinology, 9, 802. https://doi.org/10.3389/fendo.2018.00802