Why Blood Sugar Spikes Overnight — Even When You Eat Healthy All Day
High fasting blood sugar despite a healthy lifestyle? Learn how nocturnal gluconeogenesis and liver metabolism influence morning glucose levels.
DIABETES
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/9/202619 min read
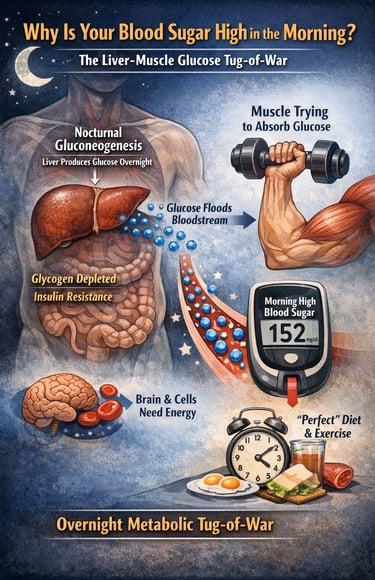
What is nocturnal gluconeogenesis?
Nocturnal gluconeogenesis is the liver's overnight production of glucose from non-carbohydrate molecules such as amino acids, lactate, and glycerol during the fasting period of sleep. In people with insulin resistance or type 2 diabetes, this process may become exaggerated, contributing to elevated fasting blood glucose levels in the morning.
Why Is My Blood Sugar High in the Morning Without Eating
You follow the rules. Your diet is clean, rich in lean protein, fibre, and nutrient-dense vegetables. You exercise regularly, prioritising resistance training to build skeletal muscle and improve insulin sensitivity. Your carbohydrate intake is controlled, your weight is stable, and your lifestyle appears metabolically sound. Yet when you check your fasting blood glucose in the morning, the number is stubbornly elevated.
For many individuals living with type 2 diabetes mellitus (T2DM), prediabetes, insulin resistance, or metabolic syndrome, this frustrating pattern is surprisingly common. The problem often has little to do with what you ate at dinner. Instead, it may reflect a powerful metabolic process occurring while you sleep: nocturnal hepatic gluconeogenesis — the liver’s overnight production of glucose from non-carbohydrate substrates such as amino acids, lactate, and glycerol.
Under normal physiological conditions, hepatic gluconeogenesis is a tightly regulated survival mechanism. During the overnight fast, the liver maintains stable blood glucose levels by balancing glycogenolysis (the breakdown of stored liver glycogen) with gluconeogenesis. This process ensures a steady glucose supply to glucose-dependent tissues such as the brain and red blood cells. However, in insulin-resistant states, this regulatory system becomes dysregulated. The liver continues producing glucose even when circulating glucose levels are already sufficient, contributing to fasting hyperglycaemia and poor glycaemic control (Barroso et al., 2024).
Recent metabolic research has revealed an additional layer of complexity. Studies using stable isotope tracing show that individuals with T2DM often exhibit excessive overnight gluconeogenesis, particularly when hepatic glycogen stores are depleted (Unni et al., 2025). Meanwhile, cutting-edge spatial transcriptomics research demonstrates that gluconeogenesis is regulated by specialised hepatocyte zones within the liver, highlighting the remarkable metabolic plasticity of hepatic glucose production (Okada et al., 2025).
The result is a subtle but powerful liver–muscle metabolic competition: while your muscles are trying to improve glucose uptake through exercise and nutrition, the liver may simultaneously be flooding the bloodstream with newly produced glucose. Understanding this overnight metabolic tug-of-war is essential for explaining why even a “perfect” muscle-focused diet sometimes fails to control morning blood sugar
Key Points
1. The liver produces glucose overnight
During sleep, the body relies on the liver to maintain blood glucose through gluconeogenesis and glycogen breakdown.
2. Insulin resistance weakens this control
In type 2 diabetes, insulin’s ability to suppress hepatic glucose production becomes impaired, allowing excessive overnight glucose release.
3. Protein can contribute to glucose production
Certain gluconeogenic amino acids (such as alanine and glutamine) from dietary protein or muscle turnover can serve as substrates for glucose synthesis.
4. Glycogen stores act as a metabolic brake
Adequate hepatic glycogen helps regulate overnight glucose production, while depleted glycogen may push the liver toward greater gluconeogenesis.
5. Hormonal rhythms amplify the effect
Early-morning hormones such as cortisol, glucagon, and growth hormone may further increase hepatic glucose output.
6. Medications can influence this pathway
Common diabetes therapies like metformin reduce hepatic glucose production, while SGLT2 inhibitors may trigger a mild compensatory increase in gluconeogenesis due to urinary glucose loss.
Clinical pearls
1. The "Empty Tank" Paradox
Scientific Perspective: Low hepatic glycogen concentrations (measured via MRS) are inversely correlated with gluconeogenic rates. In T2DM, the liver fails to sense "sufficient" systemic glucose, and a depleted glycogen buffer removes the final metabolic brake on the PEPCK/G6Pase axis.
Skipping carbs at dinner to "fix" your morning sugar often backfires. If your liver’s "fuel tank" (glycogen) is empty, it panics and spends the whole night manufacturing new sugar from scratch, leading to a higher reading than if you had eaten a small, healthy carb.
2. Protein is Not "Free" Energy
Scientific Perspective: Glucogenic amino acids (alanine/glutamine) serve as primary substrates for the $de novo$ glucose pathway. In insulin-resistant states, the conversion of protein to glucose is poorly suppressed by endogenous insulin.
While protein is great for muscles, your body can turn "steak into sugar" overnight. If you are eating massive amounts of protein late at night without any fibre or healthy fats to slow things down, your liver treats that protein like raw material for its midnight sugar factory.
3. The Evening Workout "Debt"
Scientific Perspective: High-intensity resistance training acutely depletes both muscular and hepatic glycogen. Without post-exercise glucose replenishment, the body enters a state of "metabolic urgency" that upregulates FOXO1 activity overnight.
An intense gym session at 7:00 PM is great, but if you don't "refuel" your liver afterwards with a tiny bit of complex carbs, your body stays in "emergency mode" all night. This leads to the frustrating "Dawn Phenomenon" where you wake up with high sugar despite a hard workout.
4. Metabolic "Zoning" and Timing
Scientific Perspective: Spatial transcriptomics (Okada et al., 2025) shows that different "zones" of the liver handle sugar at different times. Chronic hyperglycemia "blurs" these lines, making the liver constitutively active (always on).
Your liver has an internal clock and a floor plan. It’s designed to do different jobs at different hours. Consistency in when you eat and sleep helps "re-train" the liver cells so they know when to be a storage unit and when to be a factory, rather than trying to do both at once.
5. Front-Loading the Day
Scientific Perspective: Insulin sensitivity and the thermic effect of food follow a circadian rhythm, typically peaking in the biological morning. Aligning bolus protein intake with these peaks minimizes the substrate availability for nocturnal gluconeogenesis.
Eat like a king in the morning and a rhythmic sleeper at night. By eating most of your protein and calories earlier in the day, you give your body plenty of time to use that energy for movement rather than leaving it sitting around for the liver to process while you sleep.
6. The "Brake Pedal" Effect of Metformin
Scientific Perspective: Metformin acts as a mild mitochondrial inhibitor (Complex I), which increases the AMP: ATP ratio, activating AMPK and naturally suppressing the expression of gluconeogenic genes like PEPCK.
Think of your medication not just as "lowering sugar," but as a way to "fix the stuck gas pedal" in your liver. It helps the liver listen to the "stop" signal again, making your dietary efforts (like eating more protein) much more effective.
1. The Liver: Your Body's Secret Overnight Sugar Factory
Most people think of the liver as a detox organ — a filter for alcohol and medications. While that reputation is not entirely wrong, the liver's most metabolically consequential night job is far less glamorous: it manufactures glucose from scratch, a process called gluconeogenesis (literally, 'the birth of new glucose').
During the day, your liver happily stores glucose as glycogen — a compact, branching chain of sugar molecules that acts like a metabolic battery. But as the sun sets and your last meal recedes into memory, the liver begins liquidating those glycogen stores through glycogenolysis and, once those stores run low, it ramps up gluconeogenesis to keep your blood glucose from dropping dangerously low overnight.
This mechanism is ancient, essential, and beautifully designed. In healthy individuals, it maintains blood glucose within a tight range (roughly 70–100 mg/dL) throughout the night. The problem arises when the system loses its sensitivity to insulin's 'stop manufacturing' signal — a hallmark of type 2 diabetes.
Key Research Spotlight
Unni et al. (2025) studied 38 individuals — both with and without T2DM — using a sophisticated stable isotope technique to measure how much of overnight glucose came from gluconeogenesis versus glycogenolysis. The findings were striking: in participants with T2DM, the liver continued producing glucose via gluconeogenesis at abnormally elevated rates even when glycogen stores were far from depleted.
What this means in plain language: the diabetic liver does not wait until it needs to make sugar — it overproduces glucose all night long, flooding the bloodstream and setting up elevated fasting glucose by morning
2. Molecular Mischief: The Pathway Behind the Problem
Gluconeogenesis does not happen randomly. It is an orchestrated, enzyme-driven process that runs through a cascade of molecular signals. Understanding the key players helps explain why, in people with insulin resistance, the liver essentially ignores the body's attempts to put on the brakes.
The PEPCK and G6Pase Axis
Two enzymes sit at the top of the gluconeogenesis command chain: phosphoenolpyruvate carboxykinase (PEPCK) and glucose-6-phosphatase (G6Pase). In T2DM, both are chronically overexpressed, meaning the liver is permanently primed to overproduce glucose (Barroso et al., 2024). Think of it as a car with a stuck accelerator — even when insulin says 'slow down,' these enzymes keep the glucose pipeline flowing.
FOXO1 and PGC-1α: The Gene-Level Governors
Transcription factors FOXO1 and the co-activator PGC-1α act as the master switches that turn gluconeogenic genes on or off. In healthy insulin signalling, insulin activates AKT, which phosphorylates and deactivates FOXO1 — effectively switching off the gluconeogenesis programme. In insulin-resistant states, this signalling cascade breaks down: FOXO1 remains active, PGC-1α continues driving gluconeogenic gene expression, and the liver keeps making glucose regardless of ambient glucose levels (Barroso et al., 2024).
Spatial Zonation: Not All Liver Cells Behave the Same
A remarkable 2025 study by Okada and colleagues added an entirely new dimension to our understanding of this process. Using cutting-edge spatial transcriptomics — a technology that maps gene expression within specific physical zones of the liver — the research team revealed that gluconeogenesis is not uniformly distributed across liver tissue. Instead, different hepatocyte zones (pericentral vs. periportal) take turns dominating gluconeogenesis depending on whether the body is fed, fasted, or in prolonged starvation (Okada et al., 2025).
In the fed state, pericentral hepatocytes dominate glucose uptake and glycogen synthesis. As fasting progresses, the periportal zone takes over gluconeogenesis. Under prolonged starvation, this switches back toward a broader pattern. This spatial 'metabolic plasticity' means that targeting gluconeogenesis therapeutically is not a single-point problem — the liver's flexibility makes blanket suppression approaches both difficult and potentially dangerous
Imagine your liver as a factory with different production floors. In health, these floors take turns managing glucose. In type 2 diabetes, the cells responsible for making new glucose stay permanently on — even on overnight shifts when they should be resting.
3. The Glycogen Buffer: Your Liver's Brake Pedal
Here is where dietary strategy meets liver biology in a particularly instructive way. Hepatic glycogen — the sugar stored in the liver — plays a crucial role in suppressing gluconeogenesis. Think of it as a buffer: a well-stocked glycogen reserve signals to the liver, 'We have plenty of fuel. No need to manufacture more.'
The Unni et al. (2025) study found an inverse relationship between hepatic glycogen content and the rate of nocturnal gluconeogenesis. Participants with higher liver glycogen stores showed reduced gluconeogenic activity overnight. In contrast, those with depleted hepatic glycogen — a state that can be induced by prolonged fasting, very low-carbohydrate diets, or heavy exercise without adequate carbohydrate refuelling — showed amplified gluconeogenesis, particularly pronounced in the T2DM group.
This finding carries a nuanced and often overlooked implication: aggressively carbohydrate-restricting your diet without addressing underlying insulin resistance may inadvertently accelerate the very gluconeogenic process you are trying to tame. The liver, deprived of its glycogen buffer, ramps up glucose manufacturing overnight — and in a metabolically dysregulated liver, that acceleration is exaggerated.
⚠️ Important Note
This does not mean people with T2DM should eat high amounts of simple carbohydrates. Rather, it highlights that dietary carbohydrate management in T2DM is nuanced and should be guided by a qualified healthcare professional or registered dietitian with experience in metabolic conditions.
4. The Muscle Diet Paradox: Where Your Best Efforts Fall Short
Now let us bring this directly to the experience many fitness-conscious individuals with T2DM describe: following what appears to be an optimal muscle-building diet, only to find morning blood glucose remains elevated and overall glucose control disappoints.
High-Protein Diets and Gluconeogenesis
Protein is a cornerstone of muscle-focused nutrition, and for good reason — amino acids are the building blocks of muscle tissue, and adequate protein intake is essential for muscle protein synthesis, particularly in older adults and people with insulin resistance, who may have impaired anabolic responses. However, amino acids — especially glucogenic amino acids such as alanine, glutamine, and glycine — are potent substrates for gluconeogenesis.
When you consume a high-protein meal, the liver extracts many of these amino acids and, particularly in the context of insulin resistance, preferentially channels them into glucose production rather than other metabolic pathways (Barroso et al., 2024). This means that the protein you eat to build muscle may partially 'feed' the liver's nocturnal glucose factory. Importantly, this does not negate the value of dietary protein — but it does mean that the glucose-raising effect of high protein intake is often underappreciated in standard dietary counselling for T2DM.
Resistance Training: A Double-Edged Sword Overnight
Resistance exercise powerfully depletes muscle glycogen and creates a demand for glucose uptake by muscle cells. This is enormously beneficial for insulin sensitivity and long-term glucose management. However, intense evening resistance training without adequate carbohydrate provision can leave both muscle and liver glycogen stores depleted overnight.
As Unni et al. (2025) demonstrated, hepatic glycogen depletion is a key driver of nocturnal gluconeogenesis. A well-intentioned evening workout, followed by a high-protein, low-carbohydrate recovery meal (a classic muscle-focused approach), may set up the metabolic conditions for an amplified overnight gluconeogenic surge — resulting in elevated fasting glucose the next morning, undermining the very discipline you are trying to maintain.
The Muscle Fuel Competition
There is also a direct competition at play between the liver and muscle for circulating glucose. In states of hepatic insulin resistance — where the liver overproduces glucose but peripheral tissues (including muscle) have reduced glucose uptake — blood glucose accumulates not because muscles are receiving too much fuel, but because the liver is flooding the bloodstream faster than insulin can direct that glucose into muscle cells. This 'tug-of-war' means even active, muscular individuals with T2DM can struggle with glycaemic control.
5. The Science of Hepatic Plasticity: Why One-Size-Fits-All Approaches Fail
The spatial transcriptomics research by Okada et al. (2025) fundamentally challenges the idea that hepatic gluconeogenesis can be simply 'switched off' with a single dietary or pharmacological intervention. The liver is not a static glucose-producing organ — it is a dynamic, spatially organised tissue that adjusts its metabolic programming based on nutritional state, hormonal signals, and even circadian rhythm cues.
This means that what works to suppress gluconeogenesis in a fasted morning state may differ from what is needed to suppress it in a post-exercise state or a prolonged caloric restriction state. The clinical implication is that personalised, state-aware metabolic management strategies are needed — cookie-cutter diet plans applied uniformly across patients with T2DM are unlikely to achieve consistent glycaemic control precisely because they do not account for this hepatic plasticity.
Furthermore, Okada et al. (2025) showed that chronic metabolic stressors — including obesity and persistent hyperglycaemia — can disrupt the normal spatial zonation of hepatic gluconeogenesis, causing zones that should be metabolically dormant during fed states to become constitutively active. This constitutive activation is a key mechanism driving fasting hyperglycaemia in long-standing T2DM.
6. Practical Strategies: Working With Your Liver, Not Against It
Understanding the science of nocturnal gluconeogenesis is empowering because it identifies specific, addressable targets. While none of the following should replace personalised medical advice, the evidence base offers several clinically grounded directions:
1. Strategic Carbohydrate Timing
Rather than eliminating carbohydrates wholesale, consider distributing complex carbohydrates (such as legumes, whole grains, and root vegetables) strategically to maintain hepatic glycogen stores. An evening meal that includes a moderate portion of low-glycaemic-index carbohydrates may provide enough substrate for the liver's glycogen buffer, partially suppressing nocturnal gluconeogenesis. This aligns with the glycogen-gluconeogenesis relationship identified by Unni et al. (2025).
2. Prioritise Morning and Midday Protein
Distributing protein intake more heavily toward morning and midday meals may reduce the amino acid substrate available for hepatic gluconeogenesis during the overnight fasting period. Front-loading protein also aligns with circadian metabolic principles, as insulin sensitivity is generally higher in the morning.
3. Post-Exercise Carbohydrate Refuelling
If resistance training occurs in the evening, including a small post-workout carbohydrate portion — even a modest serve of fruit, a small sweet potato, or a portion of low-fat dairy — may help replenish hepatic glycogen stores before the overnight fast begins, reducing the liver's gluconeogenic drive.
4. Optimise Sleep and Circadian Alignment
The liver's gluconeogenic activity follows circadian rhythms. Disrupted sleep and circadian misalignment (common in shift workers, for example) have been shown to amplify nocturnal gluconeogenesis. Prioritising consistent sleep timing can support healthier hepatic metabolic cycling.
5. Work with Your Healthcare Team on Pharmacological Support
Medications such as metformin directly inhibit hepatic gluconeogenesis via the AMPK pathway and the reduction of mitochondrial complex I activity. For many people with T2DM, optimising pharmacological therapy alongside dietary and lifestyle changes offers a more comprehensive approach to managing nocturnal glucose excursions. This is particularly relevant given the molecular complexity
Dawn Phenomenon vs Nocturnal Gluconeogenesis: What’s the Difference?
Both dawn phenomenon and nocturnal gluconeogenesis can cause elevated morning fasting blood glucose, but they arise from slightly different physiological mechanisms.
Nocturnal gluconeogenesis refers to the liver’s overnight production of glucose from non-carbohydrate sources such as amino acids, lactate, and glycerol during the fasting period of sleep. In healthy individuals this process is tightly controlled by insulin and hepatic glycogen levels. However, in insulin resistance and type 2 diabetes, the liver may continue producing excessive glucose throughout the night, leading to higher fasting glucose levels.
The dawn phenomenon, on the other hand, is primarily driven by the body’s early-morning hormonal surge. Between roughly 4:00 AM and 8:00 AM, circadian hormones such as cortisol, growth hormone, glucagon, and catecholamines increase in preparation for waking. These hormones stimulate the liver to release glucose into the bloodstream. In people with normal insulin sensitivity, insulin secretion rises simultaneously to keep glucose stable. But in type 2 diabetes or impaired insulin secretion, this hormonal surge can lead to a noticeable rise in morning blood sugar.
In simple terms:
Nocturnal gluconeogenesis: excessive overnight glucose production due to hepatic insulin resistance or depleted glycogen stores.
Dawn phenomenon: early-morning rise in blood glucose caused by circadian hormone release.
In many people with type 2 diabetes, both mechanisms can occur together, making fasting hyperglycaemia more pronounced. Understanding the difference helps clinicians tailor strategies such as meal timing, medication adjustments, and post-exercise nutrition to better control morning blood glucose levels.
SGLT2 Inhibitors and Gluconeogenesis: A Brief Clinical Note
SGLT2 inhibitors such as empagliflozin, dapagliflozin, and canagliflozin lower blood glucose primarily by blocking glucose reabsorption in the proximal renal tubule, leading to urinary glucose excretion (glycosuria). While this mechanism effectively reduces plasma glucose, it also triggers several counter-regulatory metabolic responses, including a modest increase in hepatic gluconeogenesis.
Mechanism Behind the Effect
When glucose is lost in the urine (typically 60–80 g/day), the body perceives this as a mild energy deficit. To compensate, hormonal signals shift toward a fasting-like metabolic state. Two important changes occur:
Glucagon levels rise while insulin levels fall slightly.
This altered glucagon-to-insulin ratio stimulates the liver to increase gluconeogenesis and endogenous glucose production.
Studies using tracer techniques have shown that endogenous glucose production can increase modestly during SGLT2 inhibitor therapy, largely through enhanced gluconeogenesis.
Why Blood Glucose Still Falls
Despite this increase in hepatic glucose production, overall blood glucose still declines because the amount of glucose lost through the kidneys usually exceeds the extra glucose produced by the liver. In other words, the renal glucose loss outweighs the compensatory gluconeogenesis.
Metabolic Implications
Interestingly, this mild increase in gluconeogenesis is part of a broader metabolic shift produced by SGLT2 inhibitors, which includes:
increased fat oxidation
increased ketone production
modest weight loss
improved metabolic flexibility
This “fasting-mimicking” state is thought to contribute to the cardiovascular and renal benefits observed with these medications.
Clinical Takeaway
SGLT2 inhibitors may increase hepatic gluconeogenesis slightly, but this is a compensatory physiological response to urinary glucose loss. The net effect remains lower blood glucose levels, improved metabolic control, and significant cardiovascular and renal protection in people with type 2 diabetes.
Key Takeaways: What This Means for Your Blood Sugar
Understanding the liver–muscle glucose tug-of-war can help explain why blood sugar sometimes remains elevated even when you are eating well and exercising regularly. The key insight is that overnight liver metabolism plays a major role in morning blood glucose levels, especially in people with type 2 diabetes, insulin resistance, or prediabetes.
First, remember that the liver naturally produces glucose overnight through a process called gluconeogenesis. This is a normal survival mechanism that prevents blood sugar from dropping too low during sleep. However, in insulin-resistant states, the liver may keep producing glucose even when your body already has enough circulating sugar. This excess overnight glucose production can lead to elevated fasting blood glucose in the morning.
Second, liver glycogen acts as a natural brake on this process. When the liver has adequate glycogen stores, it is less likely to ramp up gluconeogenesis. But if glycogen becomes depleted — for example, after very low-carbohydrate meals, prolonged fasting, or intense evening exercise without carbohydrate refuelling — the liver may compensate by producing more glucose overnight.
Third, high-protein diets can unintentionally provide raw material for glucose production. Certain amino acids from protein can be converted into glucose by the liver. In a healthy metabolism, this process is tightly controlled, but in type 2 diabetes, the regulatory signals may not work properly, allowing more glucose to be produced than necessary.
Fourth, this creates a metabolic tug-of-war. Your muscles — strengthened by exercise and protein intake — are trying to improve glucose uptake, while the liver may simultaneously be releasing glucose into the bloodstream. The result can be persistently high fasting glucose despite good lifestyle habits.
The practical message is not to eliminate protein or carbohydrates completely. Instead, balanced carbohydrate timing, adequate post-exercise refuelling, good sleep, and appropriate medical therapy can help restore a healthier overnight glucose balance. Understanding how the liver behaves during the night is an important step toward achieving better long-term metabolic control.
Frequently Asked Questions (FAQs)
❓ Q1: What exactly is nocturnal gluconeogenesis, and is it always harmful?
Nocturnal gluconeogenesis is the liver's process of making new glucose from non-sugar molecules (such as amino acids, lactate, and glycerol) during the overnight fasting period. In healthy individuals, it is a normal and necessary process that prevents blood glucose from dropping too low during sleep. It only becomes harmful when it is dysregulated — as in type 2 diabetes — where the liver overproduces glucose despite adequate or high circulating glucose levels, leading to elevated fasting blood sugar each morning.
❓ Q2: Can a high-protein diet actually make my blood sugar worse at night?
It can contribute to higher overnight glucose in people with insulin resistance. Certain amino acids — particularly glucogenic ones such as alanine and glutamine — are converted to glucose by the liver through gluconeogenesis. In a metabolically healthy liver, insulin tightly regulates this conversion. But in insulin-resistant states, the liver's glucose-making activity is not adequately suppressed, meaning high protein intake may inadvertently provide more raw material for nocturnal glucose production. This does not mean you should avoid protein — it means protein timing and quantity should be part of a personalised metabolic management plan.
❓ Q3: Why does my fasting blood glucose go up even when I skip carbohydrates at dinner?
Skipping carbohydrates at dinner depletes hepatic (liver) glycogen stores more rapidly during the overnight fast. Research by Unni et al. (2025) found that lower liver glycogen is associated with higher rates of nocturnal gluconeogenesis. Additionally, without dietary carbohydrates, amino acids from your high-protein dinner become more readily available as gluconeogenic substrates. The result can be paradoxically higher fasting glucose — not because you ate the wrong thing, but because your liver ramped up glucose manufacturing during the night.
❓ Q4: Does exercise at night worsen the problem?
Intense evening exercise is not inherently harmful — it improves insulin sensitivity and glucose uptake in muscles over time. However, vigorous exercise without adequate carbohydrate refuelling can deplete hepatic glycogen stores, removing an important brake on overnight gluconeogenesis. If you are noticing elevated morning glucose after evening workouts, a small, low-glycaemic carbohydrate portion after exercise (e.g., a piece of fruit or a small serve of whole grains) may help replenish liver glycogen and dampen the overnight gluconeogenic response.
❓ Q5: What is hepatic glycogen, and how do I know if my liver stores are depleted?
Hepatic glycogen is glucose stored in liver cells, serving as a rapid-release reserve to maintain blood glucose during fasting. Stores can be measured with liver MRI or MR spectroscopy in research settings, but these are not standard clinical tests. Practically speaking, very low-carbohydrate diets, prolonged fasting, and high-intensity exercise all tend to deplete hepatic glycogen. If your fasting glucose is consistently elevated despite a clean diet and regular exercise, discussing hepatic glycogen dynamics with your endocrinologist or diabetes care team is worthwhile.
❓ Q6: Is this problem unique to type 2 diabetes, or can anyone experience it?
Elevated nocturnal gluconeogenesis is most clinically significant in type 2 diabetes mellitus, where insulin resistance prevents the liver from receiving and responding to the 'stop making glucose' signal. However, subthreshold insulin resistance — which can exist years before a formal T2DM diagnosis — may also produce milder versions of this effect. People with prediabetes, polycystic ovary syndrome (PCOS), non-alcoholic fatty liver disease (NAFLD), and metabolic syndrome may all experience some degree of nocturnal gluconeogenic dysregulation, even if they have not yet been diagnosed with T2DM.
❓ Q7: What is the most important takeaway for someone managing T2DM while trying to build or preserve muscle?
The most important insight is that your liver and muscles are competing for glucose, and that competition does not pause overnight. Building muscle through nutrition and exercise is absolutely beneficial for long-term metabolic health in T2DM — but achieving it while managing blood glucose requires understanding that the liver's overnight behaviour is a critical variable. Focus on strategic carbohydrate timing (not elimination), distribute protein throughout the day, ensure post-exercise glycogen replenishment, prioritise sleep quality, and work with your healthcare provider to ensure your medication regimen adequately addresses hepatic gluconeogenesis. A one-dimensional 'eat less, move more' approach is not enough — personalised, evidence-guided metabolic management is the gold standard.
Conclusion: Re-thinking the Overnight Hours
The science reviewed here tells a humbling but empowering story. For the millions of people living with type 2 diabetes who are doing their absolute best — eating thoughtfully, exercising consistently, managing stress — the liver's nocturnal glucose factory can silently undo those efforts in ways that are entirely invisible until morning. This is not a failure of willpower or discipline. It is a failure of a specific molecular signalling pathway — one that science is now illuminating with extraordinary precision.
The studies by Unni et al. (2025), Barroso et al. (2024), and Okada et al. (2025) collectively paint a picture of hepatic gluconeogenesis as a sophisticated, spatially organised, hormonally regulated process that does not respond to simple dietary fixes. Its dysregulation in T2DM is deeply mechanistic — rooted in transcription factor dysfunction, enzyme overexpression, impaired insulin signalling, and disrupted hepatic zonation. Addressing it requires an equally sophisticated, personalised approach.
The good news? Understanding the problem is the first step toward solving it. By working with your healthcare team to tailor carbohydrate timing, distribute protein intake strategically, manage exercise recovery nutrition, and optimise pharmacological support, you can begin to bring the liver's midnight sugar surge under meaningful control — and finally give your muscle-building efforts the glycaemic environment they deserve.
Author’s Note
Modern metabolic science is revealing that blood glucose regulation is far more complex than the simple advice of “eat less sugar and exercise more.” In clinical practice, many individuals with type 2 diabetes, prediabetes, and insulin resistance follow disciplined diets, remain physically active, and still struggle with elevated fasting glucose levels. This article was written to explain one often-overlooked reason for that experience: the liver’s powerful overnight glucose production through hepatic gluconeogenesis.
The goal of this piece is to translate recent advances in metabolic research—including studies on hepatic glycogen dynamics, transcriptional regulation of gluconeogenesis, and spatial liver metabolism—into clear, patient-friendly insights. Understanding the interaction between liver metabolism, skeletal muscle activity, diet composition, and circadian biology can help both clinicians and patients appreciate why metabolic regulation does not always respond to simple lifestyle changes alone.
Importantly, the science discussed here should not be interpreted as discouraging exercise, muscle-building nutrition, or thoughtful carbohydrate management. These remain foundational pillars of metabolic health. Rather, the emerging research highlights that timing, balance, and individual metabolic context matter greatly when designing dietary and lifestyle strategies for people living with metabolic disease.
As with all topics in diabetes and metabolism, individual responses vary, and no single approach works for everyone. The insights presented in this article are intended to support informed conversations between patients and healthcare professionals, encouraging more personalised approaches to metabolic care.
Continued research into hepatic glucose regulation, insulin signalling pathways, and metabolic flexibility will undoubtedly refine our understanding of these processes. As science evolves, the hope is that both clinicians and patients will gain more precise tools to manage fasting hyperglycaemia and improve long-term metabolic health.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Managing Diabesity: A Complete Guide to Weight Loss and Blood Sugar Control | DR T S DIDWAL
The BMI Paradox: Why "Normal Weight" People Still Get High Blood Pressure | DR T S DIDWAL
Breakthrough Research: Leptin Reduction is Required for Sustained Weight Loss | DR T S DIDWAL
References
Barroso, E., Jurado-Aguilar, J., Wahli, W., Palomer, X., & Vázquez-Carrera, M. (2024). Increased hepatic gluconeogenesis and type 2 diabetes mellitus. Trends in Endocrinology & Metabolism, 35(12), 1062–1077. https://doi.org/10.1016/j.tem.2024.05.006
Okada, J., Landgraf, A., Xiaoli, A. M., et al. (2025). Spatial hepatocyte plasticity of gluconeogenesis during the metabolic transitions between fed, fasted and starvation states. Nature Metabolism, 7, 1073–1091. https://doi.org/10.1038/s42255-025-01269-y
Unni, U. S., Bril, F., Mugler, J. P., 3rd, Carter, R. E., Basu, A., & Basu, R. (2025). Role of hepatic glycogen on nocturnal gluconeogenesis in type 2 diabetes mellitus. The Journal of Clinical Endocrinology and Metabolism, 110(10), 2790–2799. https://doi.org/10.1210/clinem/dgaf044