The 4 Key Drivers of Type 2 Diabetes: Insulin Resistance, Obesity, Genetics, and Lifestyle
Discover the key causes of type 2 diabetes, including insulin resistance, obesity, genetics, and lifestyle habits, and how these factors affect metabolic health.
DIABETES
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/10/202618 min read
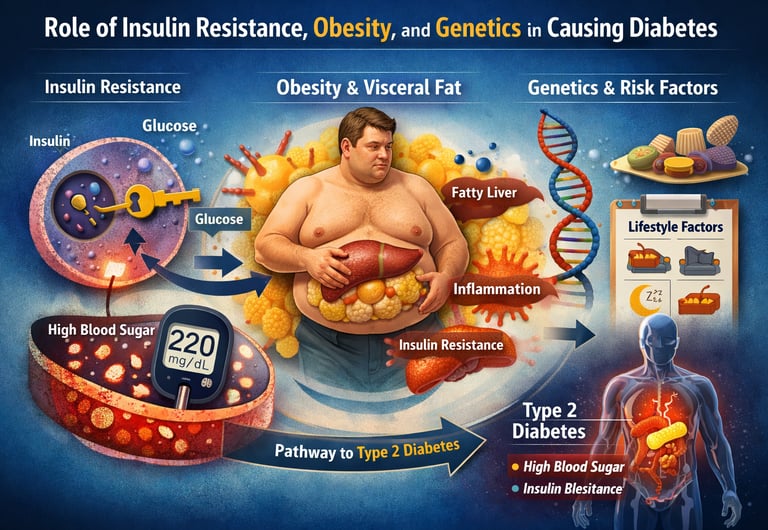
Type 2 diabetes develops when the body becomes insulin-resistant and cannot effectively regulate blood glucose. The condition is driven by a combination of insulin resistance, excess body fat—especially visceral obesity—genetic susceptibility, and lifestyle factors such as poor diet, physical inactivity, inadequate sleep, and chronic stress.
Type 2 diabetes does not develop overnight. Long before blood sugar levels rise high enough to trigger a diagnosis, a silent metabolic process is already unfolding inside the body. At the center of this process is insulin resistance—a condition in which the body’s cells gradually lose their ability to respond effectively to insulin, the hormone responsible for allowing glucose to enter cells and be used for energy. When insulin signaling becomes impaired, glucose accumulates in the bloodstream, setting the stage for chronic hyperglycemia and eventually type 2 diabetes mellitus (T2DM) (Accili et al., 2025).
But insulin resistance rarely acts alone. Modern research shows that obesity, particularly visceral or abdominal fat, plays a critical role in disrupting normal metabolic regulation. Excess adipose tissue is not just a passive energy store; it behaves like an active endocrine organ, releasing inflammatory cytokines and free fatty acids that interfere with insulin signaling pathways in muscle, liver, and fat cells (Ruze et al., 2023). This chronic low-grade inflammation contributes to metabolic dysfunction, hepatic glucose overproduction, and worsening insulin sensitivity, all of which accelerate the progression toward diabetes.
Genetics also shapes an individual’s susceptibility to the disease. Large population studies demonstrate that certain genetic variants, metabolic biomarkers, and circulating metabolites can predict a higher risk of developing T2DM years before symptoms appear (Li et al., 2026). However, genes rarely act in isolation. Lifestyle factors such as diet quality, physical activity, body weight, and sleep patterns strongly influence whether genetic risk actually translates into disease.
Understanding how insulin resistance, obesity, and genetic predisposition interact is essential for explaining why type 2 diabetes has become one of the fastest-growing metabolic disorders worldwide. More importantly, it highlights an encouraging reality: although genetics may increase vulnerability, many of the biological pathways leading to diabetes are strongly influenced by modifiable lifestyle and metabolic factors. This means that prevention—and in some cases even remission—is possible when these mechanisms are addressed early and effectively.
Key Mechanisms Behind Type 2 Diabetes
1. Insulin Resistance
Biological mechanism: Impaired insulin signaling through the IRS–PI3K–Akt pathway reduces the ability of muscle and fat cells to absorb glucose from the bloodstream.
Clinical impact: Leads to elevated blood glucose levels and compensatory hyperinsulinemia as the pancreas produces more insulin to overcome resistance.
2. Obesity (Visceral Fat)
Biological mechanism: Visceral adipose tissue acts as an endocrine organ, releasing pro-inflammatory cytokines such as TNF-α and IL-6, along with excess free fatty acids that disrupt insulin signaling.
Clinical impact: Promotes systemic inflammation, insulin resistance, and metabolic syndrome, increasing the risk of developing type 2 diabetes.
3. Genetic Predisposition
Biological mechanism: Certain genetic variants influence β-cell function, insulin secretion, fat distribution, and glucose metabolism, increasing susceptibility to metabolic dysfunction.
Clinical impact: Individuals with these variants have a higher lifetime risk of developing type 2 diabetes, especially when combined with environmental risk factors.
4. Lifestyle Factors
Biological mechanism: Unhealthy lifestyle behaviors—including poor diet, physical inactivity, sleep deprivation, and chronic psychological stress—alter metabolic signaling pathways that regulate insulin sensitivity and energy balance.
Clinical impact: These factors can accelerate insulin resistance and diabetes progression, but improving lifestyle habits can also reduce risk and support prevention or remission of type 2 diabetes.
Clinical Pearls
1. The "Broken Key" Phenomenon (Insulin Resistance)
Scientific Perspective: Insulin resistance is characterized by impaired signaling through the IRS-1/PI3K/Akt pathway. Even when insulin binds to its receptor, the intracellular "second messengers" fail to trigger the translocation of GLUT4 transporters to the cell membrane, preventing glucose uptake.
Think of insulin as a key and your cells as a locked door. In diabetes, the lock is jammed with "cellular rust." You have plenty of keys (insulin), but they can’t turn the lock, so the energy (sugar) stays stuck out in the hallway (your bloodstream) instead of fueling the room.
2. Visceral Fat as an Endocrine Organ
Scientific Perspective: Unlike subcutaneous fat, visceral adipose tissue is immunologically active. It secretes pro-inflammatory cytokines like TNF-$\alpha$ and IL-6, which directly interfere with insulin signaling and promote systemic low-grade inflammation.
Not all fat is the same. The fat stored deep around your organs acts like a "chemical factory" that sends out stress signals. These signals tell your body to ignore insulin, making your blood sugar harder to control even if your total weight hasn't changed much.
3. The Liver’s "Night Shift" Error
Scientific Perspective: In a healthy state, insulin suppresses gluconeogenesis (the liver’s production of sugar). In T2DM, the liver becomes insulin-resistant and fails to "read" the signal to stop, leading to excessive hepatic glucose output, especially overnight.
Your liver is like a backup battery that releases sugar when you haven't eaten. In diabetes, the "off switch" breaks. Even while you sleep, your liver keeps pumping sugar into your system, which is why your blood sugar can be high in the morning before you’ve even had breakfast.
4. Genetics: Susceptibility vs. Fate
Scientific Perspective: While GWAS (Genome-Wide Association Studies) have identified over 500 genetic loci associated with T2DM risk, these primarily affect beta-cell function and fat distribution. However, epigenetic modifications (driven by environment) determine whether these genes are actually expressed.
Your DNA is like a blueprint, not a finished house. You might have a family "blueprint" that makes you more sensitive to sugar or weight gain, but your daily habits—like walking and eating fiber—act as the construction crew that can choose not to build the disease.
5. The "Tipping Point" (Beta-Cell Exhaustion)
Scientific Perspective: Early T2DM is often marked by hyperinsulinemia (the pancreas overworking to compensate for resistance). Clinical diabetes usually manifests only when the pancreatic beta-cells can no longer keep up with the demand and begin to fail or undergo apoptosis (cell death).
Your pancreas is like a marathon runner. Early on, it runs twice as fast to keep your sugar normal. But eventually, the runner gets exhausted and slows down. The goal of early treatment is to "give the runner a break" before they stop running entirely.
6. Muscle as the Primary "Glucose Sink"
Scientific Perspective: Skeletal muscle is responsible for approximately 80% of postprandial (after-meal) glucose clearance. Muscle contraction can trigger glucose uptake through insulin-independent pathways, bypassing some aspects of insulin resistance.
Your muscles are your body’s biggest "sugar sponges." When you move your muscles, they can soak up sugar from your blood even without needing extra insulin. This is why a simple 10-minute walk after a meal is one of the most powerful "medicines" you have.
Type 2 diabetes is not a life sentence. With the right knowledge and support, you have the power to manage — and in many cases reverse — the course of this disease.
Throughout this article, we will explore the molecular roots of insulin resistance, the powerful connection between body weight and blood sugar, the role your genetics and lifestyle play together, how diabetes differs between urban and rural populations, and how self-care and social support influence your HbA1c — the gold-standard measure of long-term blood sugar control.
Section 1: What Is Insulin Resistance — And Why Does It Happen?
The Body's Master Regulator
Insulin is a hormone produced by your pancreas, and its job is essentially to act as a key — unlocking your cells so that glucose (sugar from food) can enter and be used for energy. In type 2 diabetes, that key no longer works properly. This is called insulin resistance: your cells stop responding to insulin's signal, leaving glucose stranded in the bloodstream instead of powering your body.
According to a 2025 review published in Nature Reviews Endocrinology, insulin resistance is not a single malfunction — it is the result of a complex, interconnected web of disruptions happening at the cellular and molecular level (Accili et al., 2025). Understanding these disruptions helps explain why no two people with diabetes are exactly alike, and why treatment must be personalized.
The Molecular Chain Reaction
Inside your cells, insulin works by activating a signalling pathway — essentially a series of chemical messages that tell the cell what to do with glucose. Research summarised by Ahn (2025) in the International Journal of Molecular Sciences identifies several key points where this pathway can break down in type 2 diabetes:
IRS (Insulin Receptor Substrate) proteins become impaired, disrupting the initial signal.
PI3K-Akt pathway dysfunction reduces the cell's ability to absorb glucose.
Chronic inflammation — often driven by excess body fat — interferes with insulin signalling at multiple levels.
Mitochondrial dysfunction reduces the efficiency of energy metabolism in muscle and liver cells.
Endoplasmic reticulum (ER) stress causes cellular malfunction in key metabolic tissues.
What this means practically: insulin resistance is not simply about "too much sugar" in your diet. It is a systems-level problem involving your immune system, your fat tissue, your liver, and even the tiny energy factories (mitochondria) inside every cell. This is why effective management requires a whole-body approach.
Key takeaway: Insulin resistance develops gradually, over years, as multiple biological systems become disrupted — and that means multiple strategies can help reverse the process.
Promising New Treatment Targets
The identification of these molecular pathways has opened exciting doors for new therapies. Ahn's (2025) review highlights emerging therapeutic targets including AMPK activators (which improve cellular energy sensing), GLP-1 receptor agonists (medications like semaglutide that improve insulin secretion and reduce appetite), and novel anti-inflammatory strategies. These treatments work with your biology — not against it — to restore insulin sensitivity at the molecular level.
Section 2: The Obesity-Diabetes Connection — More Than Just Weight
How Excess Fat Drives Blood Sugar Dysregulation
Obesity is the single most powerful modifiable risk factor for type 2 diabetes. But the relationship is more nuanced than most people realise. It is not simply that carrying extra weight "causes" diabetes. Rather, obesity triggers a cascade of biological processes that progressively undermine the body's ability to regulate blood sugar.
A comprehensive 2023 review in Frontiers in Endocrinology found that the epidemiological and pathological links between obesity and type 2 diabetes are so tightly intertwined that some researchers use the combined term "diabesity" to describe them together (Ruze et al., 2023). Globally, over 90% of people with type 2 diabetes are either overweight or obese.
Visceral Fat: The Hidden Culprit
Not all body fat is equal. Visceral fat — the fat stored deep in the abdomen around internal organs — is metabolically active in harmful ways. Ruze et al. (2023) describe how visceral adipose tissue releases excessive free fatty acids and inflammatory cytokines (chemical signals of inflammation), which directly impair insulin signalling in the liver and muscle. This is why waist circumference is often a better predictor of diabetes risk than overall body weight.
The liver, in particular, plays a central role. When fat accumulates in the liver (a condition called non-alcoholic fatty liver disease, or NAFLD), it becomes resistant to insulin's instruction to stop producing glucose, leading to chronically elevated fasting blood sugar levels even before a person eats anything.
The Good News: Weight Loss Has Disproportionate Benefits
Here is something remarkable: you do not need to reach your "ideal" body weight to see dramatic improvements in blood sugar. Research consistently shows that a 5–10% reduction in body weight can significantly improve insulin sensitivity, reduce HbA1c, and in some cases lead to remission of type 2 diabetes entirely. For a person weighing 90 kg, that is just 4.5 to 9 kg. The biology of fat loss is powerfully on your side when you start moving in the right direction.
Evidence-backed insight: Even modest weight loss of 5–10% of body weight can substantially improve insulin sensitivity and may help reverse early-stage type 2 diabetes.
Treatment Strategies at the Obesity-Diabetes Interface
Given the deep connection between obesity and T2DM, Ruze et al. (2023) highlight that treatments targeting both conditions simultaneously show the greatest promise. These include lifestyle interventions (diet and exercise), GLP-1 receptor agonists such as semaglutide and tirzepatide, which produce both weight loss and improved blood sugar control, and, in some cases, bariatric surgery, which has achieved diabetes remission rates of 40–80% in clinical studies. The message is clear: treating obesity is treating diabetes.
Section 3: Your Metabolic Profile and Genetic Risk — What Science Can Now Predict
Metabolomics: Reading the Body's Chemical Signals
One of the most exciting frontiers in diabetes research is metabolomics — the study of small chemical molecules (metabolites) circulating in your blood. These molecules reflect your body's metabolic state with remarkable precision, and researchers are now using them to predict who will develop type 2 diabetes years before any symptoms appear.
A landmark 2026 study published in Nature Medicine examined circulating metabolites, genetic variants, and lifestyle factors in a large population to understand their combined effect on future T2DM risk (Li et al., 2026). The findings were striking: specific metabolite patterns in the blood, combined with genetic and lifestyle data, could predict diabetes risk with significantly greater accuracy than traditional risk factors like BMI and family history alone.
Key Metabolites Linked to Diabetes Risk
The study by Li et al. (2026) identified several metabolite clusters associated with elevated risk, including:
Branched-chain amino acids (BCAAs) such as valine, leucine, and isoleucine — elevated levels are consistently linked to insulin resistance.
Lipid metabolites — abnormal profiles of certain fats in the blood preceded diabetes onset by years.
Glucose and glycolysis-related metabolites — early dysregulation in sugar metabolism is detectable before clinical prediabetes.
What makes this finding especially meaningful is that many of these metabolite patterns are modifiable through lifestyle. A diet lower in processed foods and higher in fibre, combined with regular physical activity, can shift your metabolic profile in a protective direction.
The Gene-Lifestyle Interaction
Genetics does influence your risk — but perhaps not as much as you might fear. Li et al. (2026) found that lifestyle factors significantly modified genetic risk. In other words, even people with a high genetic predisposition for type 2 diabetes could substantially reduce their risk through healthy behaviours. This finding is empowering: your genes load the gun, but your lifestyle largely pulls the trigger.
Empowering fact: Healthy lifestyle choices can significantly reduce diabetes risk even in people with a high genetic predisposition. Your choices matter more than your DNA.
Section 4: Urban vs. Rural — Does Where You Live Affect Your Risk?
Geography as a Health Factor
Type 2 diabetes and hypertension are not distributed equally across populations. Where you live — urban or rural — shapes your exposure to risk factors in profound ways. A 2025 comparative study published in the International Journal of Innovations and Research Analysis examined lifestyle factors influencing the onset of both T2DM and hypertension across urban and rural Indian populations (Kumari, 2025).
Urban Risk Factors
Urban populations showed higher rates of both conditions, driven by factors that will sound familiar to many city-dwellers:
Sedentary occupations and reduced physical activity — desk jobs, long commutes, and screen time.
Ultra-processed food consumption — fast food, packaged snacks, and sugar-sweetened beverages.
Chronic psychosocial stress — work pressure, financial anxiety, noise, and social isolation.
Disrupted sleep patterns — irregular schedules and light pollution disrupting circadian rhythms.
These urban-specific risk factors compound one another: stress leads to poor sleep, poor sleep increases appetite for high-calorie foods, poor diet contributes to weight gain, and weight gain worsens insulin resistance.
Rural Risk Factors
Rural populations, however, were not without risk. Kumari (2025) found that limited healthcare access, lower health literacy, and reduced awareness of early diabetes symptoms meant that rural residents were often diagnosed later — when the disease had already progressed. Physical labour, while protective against obesity, did not eliminate risk when dietary quality was poor or healthcare was unavailable.
The takeaway is important: diabetes prevention and management must be context-sensitive. What works for a sedentary urban professional requires a different approach than what works for someone in a rural community with limited access to fresh produce or specialist care.
Health equity insight: Effective diabetes care must account for where people live and the resources available to them. A one-size-fits-all approach fails the most vulnerable.
Section 5: Self-Care, Lifestyle, and Social Support — The Human Factors in HbA1c
HbA1c: Your Three-Month Blood Sugar Report Card
HbA1c (glycated haemoglobin) is the most important measure of long-term blood sugar control in type 2 diabetes. It reflects your average blood glucose over the past two to three months and is the primary target for diabetes management. But HbA1c is not just about medication — it is profoundly shaped by human factors: how well you care for yourself, how you live, and who supports you.
A 2025 study published in Clinical Medicine Insights: Endocrinology and Diabetes examined the impact of diabetes self-care behaviours, healthy lifestyle, social support, and demographic variables on HbA1c outcomes in patients with T2DM (Chen & Lin, 2025).
The Power of Diabetes Self-Care Behaviours
Self-care in diabetes encompasses a range of daily behaviours: monitoring blood glucose, taking medications as prescribed, following a balanced eating plan, engaging in regular physical activity, proper foot care, and attending regular healthcare check-ups. Chen and Lin (2025) found that patients who engaged consistently in structured self-care behaviours achieved significantly better HbA1c outcomes compared to those with low self-care engagement.
Importantly, the study found that self-care adherence was a stronger predictor of HbA1c than medication type alone. In other words, what you do every day matters more than which pill you take. This is an empowering finding — it means that meaningful control of your condition is substantially in your own hands.
Social Support: The Underrated Medicine
One of the most striking findings from Chen and Lin's (2025) research was the significant positive impact of social support on HbA1c control. Patients with strong social support networks — whether from family, friends, community groups, or peer support programs — showed better self-care adherence and better metabolic outcomes.
Why does this matter? Because diabetes management is a daily effort that can feel exhausting and isolating. Having people around you who understand your condition, encourage healthy choices, and provide practical support makes a measurable biological difference. The people in your life are part of your treatment plan.
Human truth: You don't have to manage diabetes alone — and science shows that having strong social support improves your blood sugar control. Reach out and build your team.
Lifestyle Pillars That Move the Needle
Across all the studies reviewed, certain lifestyle pillars emerged consistently as the most impactful for managing and preventing type 2 diabetes:
Nutrition: A diet rich in whole foods, vegetables, lean proteins, and healthy fats — and low in ultra-processed foods, refined carbohydrates, and sugar-sweetened drinks — is foundational.
Physical activity: At least 150 minutes per week of moderate-intensity exercise improves insulin sensitivity in muscle tissue. Strength training is particularly beneficial for glucose regulation.
Sleep: Chronic sleep deprivation impairs insulin signalling and increases cortisol, directly raising blood sugar. Aim for 7–9 hours per night.
Stress management: Chronic psychological stress elevates cortisol, a counter-regulatory hormone that raises blood glucose. Mindfulness, relaxation techniques, and adequate rest are clinically meaningful.
Smoking cessation: Smoking directly worsens insulin resistance and increases cardiovascular risk, which is already elevated in T2DM.
Section 6: Emerging Treatments and the Road Ahead
The treatment landscape for type 2 diabetes has transformed dramatically in the past decade, and the pace of innovation is accelerating. Beyond metformin — still the gold-standard first-line oral medication — several classes of newer drugs have shown remarkable benefits that extend well beyond blood sugar control:
GLP-1 receptor agonists (e.g., semaglutide, liraglutide): promote weight loss, improve insulin secretion, reduce cardiovascular risk, and, in some patients, facilitate remission of diabetes.
SGLT-2 inhibitors (e.g., empagliflozin, dapagliflozin): Lower blood sugar by causing the kidneys to excrete excess glucose; they also show powerful cardioprotective and renoprotective effects.
GIP/GLP-1 dual agonists (e.g., tirzepatide): Among the most powerful agents yet for both weight loss and glucose control.
Ahn (2025) and Accili et al. (2025) both highlight that the future of diabetes treatment will increasingly involve precision medicine: matching therapies to an individual's specific metabolic profile, genetic variants, and gut microbiome composition. What works best for one person may not be optimal for another, and science is getting much better at predicting this.
Looking ahead to Li et al.'s (2026) metabolomics findings, the possibility of identifying at-risk individuals a decade before diagnosis — and intervening early with targeted lifestyle and pharmaceutical strategies — represents a paradigm shift. Prevention, not just treatment, is the next frontier.
The future is bright: Precision medicine, metabolomics-based risk prediction, and new drug classes are converging to make type 2 diabetes not only treatable but, for many, preventable and reversible
Clinical Takeaways
• Insulin resistance is the earliest metabolic abnormality in most cases of type 2 diabetes.
• Visceral adiposity promotes systemic inflammation and worsens metabolic dysfunction.
• Genetic susceptibility influences risk but does not determine disease outcome.
• Lifestyle factors—diet, exercise, sleep, and stress management—remain the most powerful tools for prevention and remission
Frequently Asked Questions (FAQs)
❓ FAQ 1: Can type 2 diabetes actually be reversed?
Yes — for many people, particularly those in the early stages of the disease, type 2 diabetes can go into remission. Remission means blood sugar levels return to the normal range without diabetes medication. This is most reliably achieved through significant weight loss (especially 10–15% of body weight), intensive dietary changes, and regular physical activity. The DiRECT trial and other studies have shown remission rates of up to 86% at one year with intensive dietary intervention. It is not a "cure" — the underlying susceptibility remains — but sustained lifestyle changes can maintain remission for years. Speak with your healthcare provider about whether remission is a realistic goal for you.
❓ FAQ 2: What is the difference between type 1 and type 2 diabetes?
Type 1 diabetes is an autoimmune condition in which the immune system attacks and destroys the insulin-producing cells in the pancreas, meaning the body produces little or no insulin. It typically develops in childhood or young adulthood and always requires insulin therapy. Type 2 diabetes, by contrast, develops when the body produces insulin but cells become resistant to it (insulin resistance), and over time the pancreas may also produce less insulin. Type 2 accounts for approximately 90–95% of all diabetes cases and is strongly linked to lifestyle factors, though genetics also play a role. Management ranges from lifestyle interventions to oral medications to insulin, depending on disease progression.
❓ FAQ 3: How does obesity cause type 2 diabetes?
Excess body fat — particularly visceral fat stored around the abdominal organs — triggers several harmful biological processes. Fat tissue releases inflammatory chemicals (cytokines) and excess free fatty acids that interfere with insulin signalling in muscles, liver, and fat cells. The liver, when infiltrated with fat (NAFLD), continues producing glucose even when it shouldn't, raising fasting blood sugar. Meanwhile, inflammatory signals from excess fat tissue impair the molecular pathways through which insulin communicates with cells. This is why weight loss — even modest amounts — dramatically improves insulin sensitivity and blood sugar control (Ruze et al., 2023).
❓ FAQ 4: What are the early warning signs of type 2 diabetes?
Type 2 diabetes often develops silently, with no symptoms in the early stages. When symptoms do appear, they may include: increased thirst and frequent urination (as the kidneys try to flush out excess glucose), unexplained fatigue (cells are starved of energy), blurred vision, slow-healing wounds or frequent infections, tingling or numbness in the hands and feet (early nerve damage), and darkened skin in skin folds (acanthosis nigricans — a sign of insulin resistance). Many people are diagnosed only through routine blood tests. If you have risk factors — overweight, family history, age over 45, sedentary lifestyle, or gestational diabetes history — ask your doctor about screening.
❓ FAQ 5: How important is diet compared to medication in controlling HbA1c?
Both matter, and they work best together. However, research by Chen and Lin (2025) found that consistent self-care behaviours — including dietary adherence, physical activity, and monitoring — were among the strongest predictors of HbA1c improvement, sometimes exceeding the impact of medication type alone. A well-chosen diet can reduce HbA1c by 1–2 percentage points on its own — comparable to many oral diabetes medications. The most evidence-supported dietary approaches for T2DM include the Mediterranean diet, low-glycaemic index diets, and very low-calorie or low-carbohydrate diets for those seeking remission. That said, never stop or adjust medication without your doctor's guidance.
❓ FAQ 6: Can my genes determine if I will get type 2 diabetes?
Genetics influences your risk, but it is far from destiny. Research by Li et al. (2026) demonstrated that lifestyle factors significantly modify genetic risk — meaning that even people with a high genetic predisposition to T2DM can substantially reduce their risk through healthy behaviours. Having a parent or sibling with type 2 diabetes roughly doubles your risk, but this is partly because families share both genes and lifestyle habits. Genetic risk scores are increasingly being used in research to identify high-risk individuals early, but for most people, the practical message is the same regardless of genetics: a balanced diet, regular physical activity, healthy weight, and good sleep are your most powerful protective factors.
❓ FAQ 7: How does stress affect blood sugar in type 2 diabetes?
Chronic psychological stress directly raises blood glucose through multiple pathways. When you are stressed, your body releases cortisol and adrenaline — "fight or flight" hormones that signal the liver to release stored glucose for immediate energy. In a person with insulin resistance, this extra glucose cannot be efficiently absorbed by cells, causing blood sugar to spike. Chronic stress also promotes unhealthy behaviours: poor sleep, emotional eating, reduced physical activity, and medication non-adherence. Studies show that stress management interventions — including mindfulness-based stress reduction, cognitive behavioural therapy, and regular moderate exercise — can measurably improve HbA1c. Managing your mental health is managing your diabetes.
Author’s Note
Type 2 diabetes mellitus is often discussed in terms of blood sugar numbers, medications, and laboratory values. However, the scientific literature increasingly shows that the condition is far more complex than elevated glucose alone. It is the result of interacting biological processes—including insulin resistance, excess adiposity, chronic low-grade inflammation, and genetic susceptibility—that gradually disrupt the body’s metabolic balance over many years.
This article was written to bridge the gap between advanced scientific research and practical understanding. Much of the current knowledge about diabetes comes from rapidly evolving fields such as molecular endocrinology, metabolomics, and precision medicine. While these discoveries are frequently presented in technical language within medical journals, their implications are highly relevant for clinicians, researchers, and patients alike.
The goal of this editorial is therefore twofold. First, it aims to summarize current scientific insights on how insulin resistance, obesity, and genetic factors interact in the development of type 2 diabetes. Second, it seeks to highlight an important and encouraging message from modern research: although genetic predisposition may increase vulnerability, many pathways to diabetes are strongly influenced by modifiable lifestyle factors, including nutrition, physical activity, sleep, and weight management.
Understanding these mechanisms can empower both healthcare professionals and individuals at risk to focus on early prevention and targeted intervention rather than reacting only after the disease has progressed. Advances in metabolic research are steadily moving toward more personalized approaches to diabetes prevention and treatment, offering new hope for reducing the global burden of this condition.
Remember: every healthy meal, every walk around the block, every good night's sleep, and every moment of connection with people who care about you is an act of medicine. Small steps, sustained over time, change everything.
Finally, this article is intended for educational and informational purposes. Readers are encouraged to consult qualified healthcare professionals for individualized medical advice and management strategies tailored to their specific health needs.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Exercise for Type 2 Diabetes: 3 Types of Exercise That Lower Blood Sugar | DR T S DIDWAL
The Fat Inside Your Muscles: More Dangerous Than We Thought? | DR T S DIDWAL
Your Body Fat Is an Endocrine Organ—And Its Hormones Shape Your Heart Health | DR T S DIDWAL
hsCRP Explained: What Inflammation Means for Your Heart | DR T S DIDWAL
References
Accili, D., Deng, Z., & Liu, Q. (2025). Insulin resistance in type 2 diabetes mellitus. Nature Reviews Endocrinology, 21, 413–426. https://doi.org/10.1038/s41574-025-01114-y
Ahn, B. (2025). Advances in insulin resistance — Molecular mechanisms, therapeutic targets, and future directions. International Journal of Molecular Sciences, 26(6), 2574. https://doi.org/10.3390/ijms26062574
Chen, W. J., & Lin, L. Y. (2025). The impact of diabetes self-care, healthy lifestyle, social support, and demographic variables on outcomes HbA1c in patients with type 2 diabetes. Clinical Medicine Insights: Endocrinology and Diabetes, 18, 11795514251331907. https://doi.org/10.1177/11795514251331907
Kumari, N. S. (2025). A comparative study of lifestyle factors influencing the onset of type 2 diabetes and hypertension among urban and rural populations. International Journal of Innovations & Research Analysis (IJIRA), 5(2-II), 93–100. https://doi.org/10.62823/IJIRA/5.2(II).7708
Li, J., Hu, J., Yun, H., et al. (2026). Circulating metabolites, genetics and lifestyle factors in relation to future risk of type 2 diabetes. Nature Medicine, 32, 660–670. https://doi.org/10.1038/s41591-025-04105-8
Ruze, R., Liu, T., Zou, X., Song, J., Chen, Y., Xu, R., Yin, X., & Xu, Q. (2023). Obesity and type 2 diabetes mellitus: Connections in epidemiology, pathogenesis, and treatments. Frontiers in Endocrinology, 14, 1161521. https://doi.org/10.3389/fendo.2023.1161521