The Fat Inside Your Muscles: More Dangerous Than We Thought?
Muscle fat may be the hidden driver of obesity, insulin resistance, and sarcopenia. Here’s what the latest science reveals about IMAT and IMCL.
EXERCISEMETABOLISM
Dr. T.S. Didwal, M.D.(Internal Medicine)
2/27/202614 min read
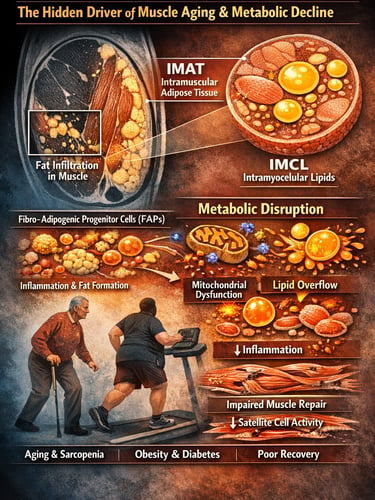
Intramuscular Fat: The Hidden Driver of Muscle Aging, Obesity, and Metabolic Decline
When we think about body fat, we usually picture subcutaneous fat or visceral adiposity. But an increasingly important form of fat is attracting attention in 2025 research: intramuscular fat — fat that accumulates within and between skeletal muscle fibres. Known as intramuscular adipose tissue (IMAT) and intramyocellular lipids (IMCL), this hidden fat depot is emerging as a critical regulator of muscle quality, insulin sensitivity, aging-related sarcopenia, osteoarthritis progression, and post-injury recovery.
Unlike subcutaneous fat, intramuscular fat directly infiltrates the contractile machinery of skeletal muscle. MRI-based population studies now show that IMAT increases significantly with age and body mass index, with sex-specific spatial distribution patterns that may influence musculoskeletal risk (Joseph et al., 2025). Mechanistic research further reveals that fibro-adipogenic progenitor (FAP) cells play a central role in pathological fat infiltration, especially under conditions of chronic inflammation, aging, or impaired regeneration (Jones et al., 2025). Even more striking, experimental models demonstrate that IMAT is not merely a passive marker of poor muscle health — it actively suppresses satellite cell function and impairs muscle repair (Norris et al., 2025).
At the metabolic level, excess IMCL acts as a biochemical bridge between obesity and skeletal muscle insulin resistance, mediated through lipid oversupply, mitochondrial dysfunction, and inflammatory signaling cascades (Razi et al., 2025). Yet the story is nuanced: endurance athletes also possess high IMCL content, highlighting that lipid compartmentalization and metabolic flexibility, not fat quantity alone, determine risk.
Intramuscular fat is no longer a secondary observation in imaging studies. It is a multidimensional biological actor at the intersection of metabolic syndrome, sarcopenic obesity, musculoskeletal degeneration, and regenerative medicine. Understanding where this fat hides — and how it behaves — may redefine how we assess and treat muscle health in aging and obesity
Clinical pearls
1. Muscle "Quality" Trumps "Quantity"
For years, the focus was solely on muscle mass (sarcopenia). The latest science shows that you can have large muscles that are "marbled" with fat (IMAT), rendering them less effective. Think of your muscles like an engine: it doesn't just matter how big the engine is, but how clean the fuel lines are.
Clinical Pearl: Don't just step on a scale or measure limb circumference; focus on how that muscle performs in real-world movements like standing up from a chair or balancing on one leg.
2. The "Stiff Ankle" Warning Sign
Study 1 (Zhao et al.) shows that fat doesn't deposit evenly; it tends to accumulate in the ankle plantar flexors. Because these muscles are the "powerhouse" for walking and balance, fat infiltration here is a primary predictor of falls in older adults.
Tip: If you feel a loss of "spring" in your step or reduced power when pushing off the ground, it may be an early indicator of age-related intramuscular fat accumulation.
3. Fat is an Active Blockade, Not a Passive Bystander
One of the most profound shifts in 2025 research (Norris et al.) is the realization that muscle fat actively "talks" to your stem cells. It secretes inflammatory signals that tell your muscle not to repair itself after an injury.
Tip: Treating muscle fat is a prerequisite for successful physical therapy. If IMAT is high, traditional rehab may be less effective because the cellular environment is "pro-fat" rather than "pro-repair."
4. The Athlete’s Paradox: Good vs. Bad Fat
Not all fat inside a muscle cell (IMCL) is a villain. In elite athletes, these fats are organized, high-quality fuel tanks used for endurance. In sedentary individuals, they become "toxic" intermediates (ceramides) that break the insulin pump.
Tip: The goal isn't to reach 0% muscle fat, but to create a "metabolic vacuum" where your mitochondria are efficient enough to burn that fat for energy rather than letting it sit and rot.
5. FAP Cells: The Body’s "Switch"
We now know that Fibro-adipogenic progenitor (FAP) cells are the architects of muscle fat. Under healthy conditions, they help repair; under stress or chronic inflammation, they turn into fat cells.
Tip: Chronic, low-grade inflammation (often caused by poor diet or lack of sleep) flips the switch on these FAP cells, telling your body to replace muscle with fat. Keeping systemic inflammation low is key to keeping your muscle "clean."
6. Actionable Tips: The "Metabolic Clean-Up" Protocol
To prevent or reverse the accumulation of intramuscular fat, you must attack it from two angles: Mechanical and Metabolic.
Zone 2 Stability (Aerobic): Engage in low-intensity steady-state exercise (like brisk walking where you can still hold a conversation). This trains your mitochondria to oxidize (burn) the intramyocellular lipids (IMCL) that cause insulin resistance.
Eccentric Loading (Resistance): Slow, controlled "lowering" phases of strength exercises (like slowly sitting down into a squat) specifically stimulate the satellite cells and can help "re-program" FAP cells toward muscle growth rather than fat storage.
The 10-Minute Post-Meal Walk: This helps clear circulating fatty acids from your bloodstream before they can "overflow" into your muscle tissue.
Antioxidant-Rich Recovery: Since oxidative stress (Study 5) drives the insulin-muscle fat connection, a diet high in polyphenols (berries, leafy greens, green tea) can protect the signaling proteins in your muscle.
Study 1: Where Fat Hides in Aging
Age-Related Differences in Intramuscular Fat Distribution
Intramuscular fat in the ankle plantar flexors increases substantially with age and is spatially non-uniform — accumulating preferentially in specific compartments — which may disproportionately impair the force-generating capacity and neuromuscular coordination of older adults.
Zhao et al (2025) tackled a question that had previously been difficult to answer with precision: where exactly does intramuscular fat accumulate in the muscles that power ankle plantar flexion — the movement essential for walking, balance, and rising from a seated position?
Using advanced MRI-based spatial quantification techniques, the research team mapped fat distribution within the ankle plantar flexor muscle group in participants across different age categories. Rather than simply measuring the total volume of intramuscular fat, the study examined how that fat was distributed within the muscle — a distinction that turns out to have profound functional implications.
The findings revealed that older adults did not simply have more intramuscular fat distributed uniformly throughout their muscles. Instead, the fat accumulated in specific spatial patterns, concentrating in particular fascicles and connective tissue regions. This uneven distribution is critical because it means that some parts of the muscle may be more compromised than others, with fat infiltration potentially disrupting the precise coordination needed for effective force transmission.
These results have significant implications for understanding falls risk in older adults. The ankle plantar flexors play a vital role in propulsion during walking and postural correction during balance perturbations. If intramuscular fat clusters in these muscles in ways that impair rapid force generation, even individuals who appear to have adequate overall muscle mass may face elevated functional deficits. The study highlights that assessing muscle quality — not just quantity — is essential in aging populations.
Spatial fat mapping, rather than aggregate fat volume measurements, may offer superior predictive value for functional impairment in clinical assessments of older adults.
Study 2: The Biology of Muscle Fat — From Cell to Pathology
Intramuscular Adipose Tissue: From Progenitor to Pathology
Jones et al.(2025) studied. the cellular origins of intramuscular adipose tissue are now more clearly understood: fibro-adipogenic progenitor (FAP) cells are the primary architects of pathological muscle fat accumulation, and their dysregulation is a mechanistic linchpin connecting muscle injury, disease, and aging with fat infiltration.
While most research on intramuscular fat focuses on population-level outcomes and imaging measurements, Jones, Kopinke, and Meyer take us inside the cell to answer a more fundamental question: where does this fat actually come from, and what goes wrong when it accumulates pathologically?
The paper provides a rigorous mechanistic review of intramuscular adipose tissue biology, focusing on fibro-adipogenic progenitor cells — often abbreviated FAPs. These are resident mesenchymal stromal cells found within skeletal muscle that, under normal circumstances, play supportive roles in muscle repair. When muscle is healthy and regeneration proceeds efficiently, FAPs are kept in check by signals from immune cells and muscle stem cells (satellite cells). However, when this regulatory balance is disrupted — as it is in chronic disease, aging, muscular dystrophy, and persistent inflammation — FAPs shift toward adipogenic differentiation, producing fat cells that progressively replace functional muscle tissue.
The review synthesises evidence showing that FAP behaviour is exquisitely sensitive to the microenvironment. Key regulatory factors discussed include macrophage-derived signals, transforming growth factor-beta (TGF-β) signalling, and hedgehog pathway activity, all of which influence whether FAPs promote muscle repair or fat deposition. The paper also explores how these pathways are altered with aging and in disease states such as Duchenne muscular dystrophy and obesity.
From a translational perspective, this study is invaluable. By establishing FAPs as the primary cellular source of pathological intramuscular fat, it identifies them as prime therapeutic targets. Interventions that modulate FAP activity — whether pharmacologically, via exercise, or through nutritional strategies — could potentially slow or reverse muscle fat accumulation in vulnerable populations.
FAPs are not passive bystanders in muscle fat accumulation — they are active decision-makers whose regulatory environment can be therapeutically targeted.
Study 3: Mapping Thigh Fat in Thousands of Patients via MRI
This study by Joseph et al. (2025) leverages the rich imaging database of the Osteoarthritis Initiative (OAI) — one of the largest musculoskeletal cohort studies ever undertaken — to characterise thigh intramuscular fat across thousands of participants. The OAI enrolled over 4,000 men and women aged 45 to 79 years, providing an unparalleled opportunity to examine how intramuscular fat relates to age, sex, and body mass index (BMI) at a population level.
The researchers used a combination of T1-weighted MRI sequences and semi-automated segmentation protocols to quantify IMAT in the thigh region. Their analysis revealed a clear, graded relationship between advancing age and increasing intramuscular fat content in both men and women. However, the study uncovered important sex-specific differences in how this accumulation occurs. Women tended to show different spatial patterns of fat infiltration compared to men, even when total fat volumes were similar — a finding with potential implications for understanding sex-based differences in knee osteoarthritis prevalence and severity.
BMI emerged as an independent predictor of intramuscular fat above and beyond age, consistent with the hypothesis that excess systemic adiposity promotes fat infiltration into muscle tissue through mechanisms including lipid overflow, inflammatory signalling, and altered hormonal milieu. Importantly, the association between BMI and IMAT was not linear in all analyses, suggesting that other metabolic factors may modulate the relationship between obesity and muscle fat.
This study's particular strength lies in its statistical power and population representativeness. By demonstrating these associations across a large, well-characterised cohort, it provides a robust foundation for clinical risk stratification tools and reinforces the case for routine IMAT assessment in patients with or at risk of osteoarthritis.
Thigh IMAT measured by MRI may serve as an important addition to clinical osteoarthritis risk models, particularly given its sensitivity to both aging and obesity-related changes.
Study 4: Fat That Blocks Recovery After Muscle Injury
Experimentally induced intramuscular adipose tissue directly impairs functional muscle regeneration by physically and biochemically obstructing satellite cell activity and myofibre repair, establishing IMAT not merely as a marker of poor recovery, but as an active mechanical and biochemical barrier to it.
Norris et al. (2025) tested a hypothesis that has long been debated in the field: Does intramuscular fat merely accompany poor muscle recovery, or does it actively cause it? The distinction matters enormously for therapeutic strategy. If IMAT is a passive marker, treatment efforts can focus on underlying causes. But if IMAT is itself an active barrier, then removing or preventing it becomes an independent therapeutic goal.
The research team used a combination of mouse genetic models and pharmacological interventions to selectively induce IMAT accumulation within skeletal muscle and then assess regeneration outcomes following standardised injury protocols. By manipulating the FAP compartment — consistent with the cellular biology described by Jones et al. — the group was able to create muscles with varying degrees of fat infiltration and compare their regenerative capacity under controlled conditions.
The results were stark: muscles with elevated IMAT showed significantly impaired functional recovery across multiple metrics, including maximal force production, muscle fibre cross-sectional area, and activation of satellite cells — the resident stem cells responsible for muscle repair. Mechanistic investigation revealed that adipocytes within the muscle tissue were not simply taking up physical space; they were actively secreting factors that suppressed regenerative signalling and altered the extracellular matrix environment in ways that made myofibre repair less efficient.
For clinical medicine, these findings carry immediate relevance. Patients recovering from traumatic muscle injuries, orthopaedic surgeries, or periods of immobilisation who have pre-existing IMAT accumulation may face a systematically compromised recovery trajectory — not due to inadequate rehabilitation per se, but because the tissue environment itself has been rendered less conducive to healing. This argues for pre-rehabilitation strategies to assess and potentially reduce IMAT before recovery-critical interventions.
IMAT does not passively reside in recovering muscle — it actively suppresses regeneration, making it a direct therapeutic target in recovery-focused clinical care.
Study 5: The Obesity–Insulin Resistance–Muscle Fat Connection
From Obesity to Muscle Insulin Resistance: The Mediating Roles of Intramyocellular Lipids, Inflammation, and Oxidative Stress
Intramyocellular lipids (IMCL) serve as a critical mechanistic bridge between obesity and skeletal muscle insulin resistance — operating through a triad of lipid oversupply, chronic low-grade inflammation, and mitochondrial oxidative stress — identifying multiple pharmacological and lifestyle targets for interrupting this disease-promoting cascade.
The final study in our review by Razi et al. (2025) shifts focus from structural and imaging-based questions to molecular metabolism, examining how fat inside individual muscle cells — intramyocellular lipids (IMCL) — drives the development of insulin resistance in obese individuals. This comprehensive review by Razi and an international team of metabolic physiology experts provides one of the most up-to-date syntheses of the mechanistic literature on this topic.
The review begins by establishing the well-known epidemiological paradox: highly trained endurance athletes also have elevated IMCL, yet they are among the most insulin-sensitive individuals in the population. This so-called 'athlete's paradox' has been a source of productive scientific tension for two decades. The resolution lies not in IMCL quantity alone but in the metabolic quality and subcellular compartmentalisation of the lipids involved. In athletes, IMCL are efficiently mobilised and oxidised during exercise, with minimal accumulation of toxic intermediates. In obese, sedentary individuals, lipid oversupply overwhelms mitochondrial oxidative capacity, leading to accumulation of diacylglycerols (DAGs), ceramides, and acylcarnitines — bioactive lipid species that directly interfere with insulin signalling cascades.
The paper maps three parallel mechanistic pathways through which obesity drives IMCL-mediated insulin resistance. First, excess circulating fatty acids promote lipid overflow into muscle cells beyond their capacity for storage or oxidation. Second, adipose-derived inflammatory cytokines — including TNF-α and IL-6 — activate serine/threonine kinase cascades (particularly JNK and IKKβ) that phosphorylate and thereby deactivate key insulin signalling proteins. Third, mitochondrial dysfunction and resulting reactive oxygen species (ROS) generation further impair insulin signalling through oxidative modification of signalling enzymes.
From a translational standpoint, the review highlights that exercise remains the most powerful intervention for normalising IMCL metabolism, acting simultaneously on all three pathways. However, it also identifies promising pharmacological targets — including mitochondrial antioxidants, ceramide synthase inhibitors, and anti-inflammatory biologics — for individuals unable to achieve therapeutic levels of physical activity.
Targeting IMCL accumulation in skeletal muscle — not just adiposity broadly — may be essential to effectively break the cycle of obesity-driven insulin resistance.
Cross-Study Synthesis: What the Evidence Tells Us Together
1️⃣ Muscle Quality Has Overtaken Muscle Quantity
For decades, clinical assessment of skeletal muscle focused on mass — lean body mass, cross-sectional area, sarcopenia thresholds. That framework is now incomplete. Evidence from 2025 imaging and mechanistic studies makes it clear: intramuscular fat (IMAT and IMCL) fundamentally alters muscle quality, even when total muscle volume appears preserved. A patient can have “normal” muscle size yet possess metabolically and mechanically compromised tissue.
2️⃣ Spatial Distribution Matters More Than Total Fat
Advanced MRI mapping demonstrates that fat infiltration is not uniform. It clusters in specific fascicles and connective tissue compartments, potentially disrupting force transmission and neuromuscular coordination. This reframes fall risk, gait instability, and osteoarthritis progression as partly micro-architectural problems, not merely strength deficits.
3️⃣ Fibro-Adipogenic Progenitors (FAPs) Are the Biological Switch
At the cellular level, FAPs emerge as the central decision-makers. Under regenerative conditions, they support repair. Under inflammatory, aging, or metabolic stress conditions, they pivot toward adipogenesis. Intramuscular fat is therefore not accidental accumulation — it is the product of a regulated but dysdirected cellular program.
4️⃣ IMAT Is Not Passive — It Actively Impairs Recovery
Experimental data now confirm that intramuscular fat suppresses satellite cell activation, alters extracellular matrix composition, and diminishes force recovery after injury. This has direct implications for orthopaedic surgery, immobilisation, and rehabilitation medicine. Pre-existing IMAT may predetermine recovery trajectories.
5️⃣ The Obesity–Insulin Resistance Bridge Runs Through Muscle
Intramyocellular lipid excess links systemic obesity to skeletal muscle insulin resistance through lipid intermediates, mitochondrial dysfunction, and inflammatory kinase activation. The critical distinction is not fat presence alone, but lipid handling capacity and metabolic flexibility — explaining the athlete’s paradox.
6️⃣ Clinical Implications Are Immediate
MRI-based IMAT assessment may refine osteoarthritis risk models.
Exercise prescriptions must target mitochondrial capacity, not just caloric expenditure.
Muscle fat reduction may become a prehabilitation objective.
Sarcopenic obesity should be reconceptualized as a disorder of muscle composition.
7️⃣ The Strategic Shift
Intramuscular fat should be treated as a modifiable, measurable, mechanistically active biomarker — not an incidental imaging observation.
Frequently Asked Questions (FAQs)
Q1: What is the difference between IMAT and IMCL?
IMAT (intramuscular adipose tissue) refers to fat cells — adipocytes — that accumulate between muscle fibres and fascicles, visible on MRI as discrete fat depots. IMCL (intramyocellular lipids) refers to lipid droplets stored within individual muscle fibre cells, detectable via spectroscopy or histology. Both are problematic when elevated in non-athletes, but they have distinct cellular origins and functional consequences. IMAT primarily impairs mechanical and regenerative function; IMCL primarily disrupts intracellular insulin signalling.
Q2: Does exercise reduce intramuscular fat?
Yes — both aerobic and resistance exercise are associated with reductions in IMAT and improved IMCL metabolism. Aerobic training enhances mitochondrial oxidative capacity, reducing the accumulation of toxic lipid intermediates. Resistance training promotes satellite cell activity and can reduce the adipogenic shift of FAPs. Razi et al. (2025) identify exercise as the most comprehensive intervention, acting on lipid oversupply, inflammation, and oxidative stress simultaneously.
Q3: Can you have high IMAT even with a normal BMI?
Yes. The relationship between BMI and IMAT is real but imperfect. Individuals with metabolically unhealthy normal weight — sometimes called TOFI (thin outside, fat inside) — can have elevated intramuscular fat without visible adiposity. Conversely, highly trained athletes may have elevated IMCL without negative metabolic consequences. Joseph et al. (2025) found that BMI predicts IMAT but not perfectly, suggesting additional metabolic determinants.
Q4: Is intramuscular fat accumulation reversible?
The reversibility of IMAT depends on its extent and underlying cause. Mild to moderate IMAT associated with deconditioning or obesity can be reduced through sustained exercise and weight loss. However, severe IMAT in chronic neuromuscular disease or advanced aging may be more refractory. Norris et al. (2025) suggest that reducing IMAT before or during recovery periods is particularly critical, as once established, IMAT may actively impair the regenerative process.
Q5: Why does intramuscular fat increase with age?
Aging is associated with a convergence of factors that promote IMAT accumulation: declining satellite cell activity reduces muscle repair capacity; chronic low-grade inflammation shifts FAPs toward adipogenesis; declining sex hormone levels alter fat distribution patterns; and reduced physical activity lowers energy demand and mitochondrial capacity. Jones et al. (2025) describe how the FAP regulatory environment becomes progressively dysregulated with aging, tipping the balance from repair to adipogenesis.
Q6: How is intramuscular fat measured clinically?
The gold standard non-invasive method is MRI, particularly T1-weighted sequences that clearly distinguish fat from lean tissue based on signal intensity. Computed tomography (CT) is also used in research settings. Ultrasound imaging is an accessible and lower-cost alternative, though with less precision. For IMCL specifically, proton magnetic resonance spectroscopy (¹H-MRS) provides quantitative measures within individual muscle compartments. Zhao et al. (2025) and Joseph et al. (2025) both utilised MRI-based approaches.
Q7: What are the implications of intramuscular fat for osteoarthritis?
Osteoarthritis is traditionally understood as a joint disease, but muscle quality around the joint significantly influences biomechanical loading and functional outcomes. Joseph et al. (2025) used the Osteoarthritis Initiative cohort specifically because IMAT in thigh muscles — which include the quadriceps responsible for knee stabilisation — is increasingly recognised as a contributor to disease progression. Higher IMAT is associated with reduced muscle force, altered gait mechanics, and potentially accelerated cartilage degradation.
Author’s Note
Intramuscular fat is often dismissed as an incidental imaging observation — a radiologic curiosity rather than a clinical priority. The intention behind this review is to challenge that assumption. The 2025 evidence landscape suggests that fat within skeletal muscle is not merely a byproduct of aging or obesity, but an active participant in metabolic dysfunction, impaired regeneration, and biomechanical decline.
As clinicians and researchers, we have traditionally focused on visible adiposity and measurable muscle mass. Yet modern imaging, cellular biology, and metabolic research are converging on a more nuanced truth: muscle composition may matter more than muscle size alone. Intramuscular adipose tissue (IMAT) and intramyocellular lipids (IMCL) represent distinct yet interconnected phenomena that bridge endocrinology, geriatrics, orthopaedics, rehabilitation medicine, and exercise physiology.
This article synthesizes recent high-quality studies with the aim of translating complex mechanistic insights into clinically meaningful understanding. While the field is advancing rapidly, important questions remain — particularly regarding reversibility thresholds, optimal intervention strategies, and standardized clinical measurement protocols. The science is evolving, and recommendations should continue to adapt alongside emerging data.
Readers are encouraged to view intramuscular fat not as a niche research topic, but as a lens through which broader issues — sarcopenic obesity, insulin resistance, osteoarthritis progression, and post-injury recovery — can be better understood. The goal is not to generate alarm, but to promote precision: measure muscle quality, recognize modifiable risk factors, and integrate metabolic and musculoskeletal thinking.
The future of muscle health assessment will likely move beyond “how much muscle” toward “what kind of muscle.” This review is offered as a step in that direction.
References
Joseph, G. B., Akkaya, Z., Sims, W. M., et al. (2025). MRI-based analysis of thigh intramuscular fat and its associations with age, sex, and BMI using data from the osteoarthritis initiative data. Scientific Reports, 15, 6188. https://doi.org/10.1038/s41598-024-75005-z
Jones, H. G., Kopinke, D., & Meyer, G. A. (2025). Intramuscular adipose tissue: From progenitor to pathology. American Journal of Physiology–Cell Physiology, 329(4), C1268–C1282. https://doi.org/10.1152/ajpcell.00613.2025
Norris, A. M., Palzkill, V. R., Appu, A. B., Fierman, K. E., Noble, C. D., Ryan, T. E., & Kopinke, D. (2025). Intramuscular adipose tissue restricts functional muscle recovery. Cell Reports, 44(8), 116021. https://doi.org/10.1016/j.celrep.2025.116021
Razi, O., De Moraes, C., Zamani, N., Saeidi, A., Hadjicharalambous, M., Hackney, A. C., Del Coso, J., Laher, I., & Zouhal, H. (2025). From obesity to muscle insulin resistance: The mediating roles of intramyocellular lipids, inflammation, and oxidative stress. Diabetes/Metabolism Research and Reviews, 41(7), e70094. https://doi.org/10.1002/dmrr.70094
Zhao, Z., Smith, F. E., Dick, T. J. M., & Hodson-Tole, E. (2025). Age-related differences in intramuscular fat distribution: Spatial quantification in human ankle plantar flexors. Frontiers in Bioengineering and Biotechnology, 13, 1594557. https://doi.org/10.3389/fbioe.2025.1594557