Why Fat Tissue Is an Endocrine Organ — And What It Means for Your Health
Discover how fat tissue functions as an endocrine organ, influencing insulin resistance, inflammation, visceral fat risk, and metabolic disease.
OBESITYMETABOLISM
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/2/202611 min read
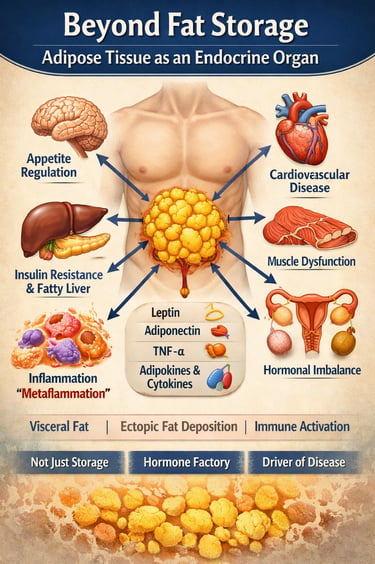
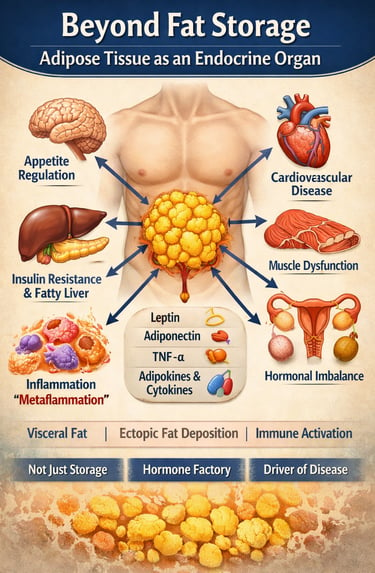
For many years, body fat was described as “extra weight” — something that simply stored unused calories. We now know that the idea is outdated. Fat tissue is not inactive or silent. It behaves like a hormone-producing organ that constantly communicates with the brain, liver, muscles, pancreas, blood vessels, and immune system. In other words, your fat is biologically active and deeply involved in regulating hunger, blood sugar, inflammation, and overall metabolic health (Clemente-Suárez et al., 2023).
Different types of fat behave differently. Subcutaneous fat (the fat under the skin), visceral fat (fat around the abdominal organs), and even brown fat each have unique roles. These tissues release chemical messengers called adipokines — including leptin, adiponectin, and inflammatory cytokines — that help control appetite, insulin sensitivity, immune responses, and energy use (Tilg et al., 2025). When fat tissue expands beyond its healthy storage capacity, however, its behavior changes. Instead of supporting balance, it promotes low-grade inflammation, insulin resistance, and fat buildup in organs such as the liver and pancreas (Dobre et al., 2025; Pan et al., 2023).
Visceral fat deserves special attention. Because it drains directly to the liver through the portal circulation, it delivers high concentrations of free fatty acids and inflammatory signals. Over time, this can contribute to fatty liver disease (now called MASLD), type 2 diabetes, and cardiovascular disease. Researchers call this process “metaflammation” — a chronic metabolic inflammation linked to excess adiposity (Nadolsky et al., 2025).
Understanding fat in this way changes the conversation. Obesity is not simply about willpower or appearance. It reflects changes in how adipose tissue functions hormonally and immunologically. To understand modern metabolic disease, we must first understand fat — not as stored weight, but as an active endocrine organ influencing nearly every system in the body.
Clinical pearls
1. The "Waist-to-Height" Rule
Scientific Perspective: BMI is a crude proxy that fails to account for visceral adipose tissue (VAT). VAT is uniquely dangerous because it drains directly into the portal vein, exposing the liver to high concentrations of inflammatory cytokines and free fatty acids.
Where you carry your fat matters more than how much you weigh. Fat deep inside your belly acts like an "angry" organ that sends stress signals to your liver. A simple goal: keep your waist circumference less than half your height.
2. The Power of "Small Wins" (The 5% Threshold)
Scientific Perspective: Metabolic dysfunction is driven by ectopic fat—lipids stored in the liver, muscle, and pancreas. Clinical trials like DiRECT show that losing just 5–10% of total body weight disproportionately reduces this "misplaced" fat, often restoring beta-cell function in the pancreas.
You don’t need to reach a "perfect" weight to see massive health gains. Losing just 5% of your weight (e.g., 10 lbs if you weigh 200 lbs) "vacuums" the dangerous fat out of your internal organs first, which can help reverse conditions like Type 2 Diabetes.
3. Muscles Are the "Sponge" for Blood Sugar
Scientific Perspective: Skeletal muscle handles roughly 80% of insulin-stimulated glucose disposal. However, when fat leaks into muscle cells (intramyocellular lipids), it "clogs" the insulin receptors (IRS-1 signaling), leading to systemic insulin resistance.
Think of your muscles as a sponge for the sugar in your blood. When you have too much internal fat, that sponge gets "greasy" and stops soaking up sugar. Exercise—especially strength training—helps "wring out" the grease so the sponge can work again.
4. Inflammation Is a "Slow Burn," Not a Fever
Scientific Perspective: Obesity is a state of metaflammation—chronic, low-grade, sterile inflammation. Unlike the high fever of an infection, this is driven by M1 macrophages in the fat tissue. This "slow burn" eventually scars the arteries and damages DNA, increasing cancer risk.
Excess belly fat isn't just sitting there; it’s constantly "leaking" inflammatory chemicals into your blood, similar to a silent, internal sunburn that never heals. This is why weight management is actually a form of anti-inflammatory medicine.
5. Hunger Is Hormonal, Not a Character Flaw
Scientific Perspective: Leptin resistance means the brain is "blind" to the satiety signals sent by fat tissue. High levels of circulating leptin fail to cross the blood-brain barrier effectively, leaving the hypothalamus in a perceived state of starvation despite high energy stores.
If you struggle with constant hunger, it isn't a lack of willpower—it’s a "broken thermostat." Your brain isn't receiving the "I’m full" memo because the signal is being blocked by inflammation. Medical treatments (like GLP-1 medications) help clear that communication line so your brain can finally "see" the energy you already have.
Section I: Types of Adipose Tissue — Not All Fat Is Equal
1. Subcutaneous Adipose Tissue (SAT)
SAT is the fat found directly beneath the skin (the "pinchable" fat on arms and thighs). In moderate amounts, it is relatively metabolically benign. Lower-body subcutaneous fat (gluteo-femoral) may even offer protective metabolic effects (Dobre et al., 2025). However, in severe obesity, SAT can become dysfunctional, triggering hypoxia and systemic insulin resistance.
2. Visceral Adipose Tissue (VAT)
VAT accumulates deep within the abdominal cavity, surrounding vital organs. Unlike SAT, visceral fat drains directly into the portal circulation, sending inflammatory signals straight to the liver. High VAT mass is a primary driver of:
Type 2 Diabetes
MASLD (formerly NAFLD)
Cardiovascular Disease
3. Brown and Beige Fat: The "Calorie Burners"
Brown Adipose Tissue (BAT): Rich in mitochondria and UCP1, BAT burns energy to generate heat (thermogenesis).
Beige Fat: White fat cells that undergo "browning" in response to cold or exercise.
Stimulating these fats is a top therapeutic target for increasing energy expenditure without caloric restriction.
Section II: Adipokines — The Endocrine Language of Fat
Adipose tissue communicates via adipokines. While over 600 have been identified, three are most critical to human health:
Leptin: The Satiety Signal
Leptin tells your brain you have enough energy. In obesity, however, leptin resistance occurs. The brain becomes "blind" to the signal, leading to persistent hunger despite high fat stores (Clemente-Suárez et al., 2023).
Adiponectin: The "Good" Hormone
Adiponectin is anti-inflammatory and insulin-sensitizing. Unlike other adipokines, adiponectin levels drop as fat mass increases. Low levels are a major biomarker for cardiometabolic risk (Tilg et al., 2025).
TNF-α: The Inflammatory Trigger
Tumor Necrosis Factor-alpha (TNF-α) links obesity to systemic inflammation. It directly interferes with insulin signaling, creating a state of "metaflammation" (metabolic inflammation) that damages arteries and organs.
Section III: Ectopic Fat — Fat in the Wrong Places
When standard fat depots "overflow," lipids accumulate in organs not designed for storage. This ectopic fat deposition is a critical driver of disease:
Liver (Hepatic Steatosis): Leads to MASLD and impaired glucose production.
Muscle (Intramyocellular Lipid): Blocks insulin's ability to move sugar into cells.
Pancreas: Fat accumulation here impairs insulin secretion and can lead to beta-cell failure.
Section IV: The Immune Connection — Macrophage Polarization
Obese adipose tissue is heavily infiltrated by immune cells, specifically macrophages. Their behavior determines your metabolic health:
M2 Macrophages (The Healers): Predominant in lean tissue; these are anti-inflammatory.
M1 Macrophages (The Attackers): Predominant in obese tissue; these secrete inflammatory cytokines that drive insulin resistance (Pan et al., 2023).
This shift from M2 to M1 creates a self-amplifying cycle of tissue death and chronic inflammation.
Section V: Clinical Integration — Obesity as an Endocrine Disorder
Here is what every patient should understand about fat — clearly, simply, and without stigma:
1. Fat Is Not Inactive — It’s Hormonal
Body fat is not just stored energy. It acts like a hormone-producing gland. It releases chemical messengers called adipokines that influence hunger, blood sugar, inflammation, blood pressure, and even fertility (Clemente-Suárez et al., 2023). This means fat tissue is constantly communicating with the rest of your body.
2. Not All Fat Behaves the Same
There are different types of fat:
Subcutaneous fat (under the skin)
Visceral fat (around internal organs)
Brown fat (helps burn energy for heat)
Visceral fat is more metabolically active and more inflammatory than subcutaneous fat. That’s why waist circumference often predicts health risk better than weight alone (Dobre et al., 2025).
3. Fat Talks to Your Brain About Hunger
Fat tissue produces leptin, a hormone that signals fullness to the brain. In people with obesity, leptin levels are high — but the brain stops responding properly. This is called leptin resistance. It explains why weight regulation is biological, not simply about willpower (Tilg et al., 2025).
4. Healthy Fat Supports Metabolism
Fat also produces adiponectin, a hormone that improves insulin sensitivity and reduces inflammation. As visceral fat increases, adiponectin levels fall — raising the risk of type 2 diabetes and heart disease (Tilg et al., 2025).
5. Inflammation Is the Real Problem
When fat cells enlarge beyond healthy limits, immune cells called macrophages move in. This creates chronic low-grade inflammation — sometimes called metaflammation — which interferes with insulin action and damages organs over time (Pan et al., 2023).
6. Obesity Is an Endocrine Condition, Not a Moral Failure
Modern medical guidelines describe obesity as an adiposity-based chronic disease because excess fat disrupts hormonal and immune balance (Nadolsky et al., 2025). It is a medical condition requiring structured, long-term management — not judgment.
The Bottom Line
Fat is not the enemy. Dysfunctional fat is. When adipose tissue becomes inflamed and hormonally imbalanced, metabolic disease follows. Understanding this biology empowers smarter, kinder, and more effective treatment approaches.
Takeaways for each study:
1. Dobre et al. (2025) — Current Issues in Molecular Biology Fat type matters clinically. Brown fat burns energy thermogenically, subcutaneous fat is relatively benign in moderation, and visceral fat is the most metabolically dangerous — directly driving insulin resistance through portal lipid overflow, dysfunctional adipokine secretion, and hypoxia-induced inflammation.
2. Clemente-Suárez et al. (2023) — Biomedicines Adipokines are the chemical language of fat. Leptin, adiponectin, TNF-α and others regulate appetite, immunity, cardiovascular tone, and metabolism. In obesity, this language becomes dysregulated — leptin resistance silences satiety signals while falling adiponectin and rising TNF-α fuel systemic inflammation and insulin resistance.
3. Tilg et al. (2025) — Nature Reviews Immunology Adipokines are masterminds of "metaflammation" — a chronic, low-grade, sterile inflammatory state distinct from classical infection-driven inflammation. This state, sustained by visceral fat-derived cytokines and macrophage activation, mechanistically underlies type 2 diabetes, atherosclerosis, NAFLD, and obesity-related cancers.
4. Pan et al. (2023) — Frontiers in Immunology. In lean adipose tissue, anti-inflammatory M2 macrophages dominate. In obesity, the balance flips to pro-inflammatory M1 macrophages that form crown-like structures around dying fat cells and release TNF-α and IL-1β — directly impairing insulin signalling and creating a self-amplifying cycle of metabolic deterioration.
5. Nadolsky et al. (2025) — Endocrine Practice (AACE Consensus). Obesity must be managed as Adiposity-Based Chronic Disease (ABCD) — not a lifestyle failing but a relapsing endocrine condition. The 2025 algorithm calls for complication-centred risk stratification and tiered treatment: lifestyle therapy, GLP-1/dual agonist pharmacotherapy, and metabolic bariatric surgery where indicated.
Frequently Asked Questions (FAQs)
1. What does it mean to say adipose tissue is an 'endocrine organ'?
An endocrine organ is one that secretes hormones into the bloodstream to regulate distant tissues. Adipose tissue does exactly this through its secretion of adipokines — bioactive proteins including leptin, adiponectin, resistin, chemerin, visfatin, and many others. These hormones regulate appetite, insulin sensitivity, immune function, bone metabolism, blood pressure, and reproductive function. Far from being an inert energy store, fat is in constant hormonal conversation with the rest of the body (Clemente-Suárez et al., 2023).
2. Why is visceral fat more dangerous than subcutaneous fat?
Visceral fat is anatomically positioned to drain into the hepatic portal circulation, exposing the liver to a concentrated stream of pro-inflammatory adipokines and free fatty acids. It is more metabolically active, more lipolytic, and more immunologically active than subcutaneous fat, secreting higher levels of TNF-α, IL-6, and other inflammatory mediators. It is also more heavily infiltrated by M1 macrophages. These features make visceral adiposity a far stronger predictor of metabolic disease than body mass index alone (Dobre et al., 2025).
3. What is leptin resistance and can it be reversed?
Leptin resistance occurs when the hypothalamus fails to respond to leptin despite high circulating levels. It is caused by impaired leptin transport across the blood-brain barrier, reduced receptor expression, and inflammatory interference with intracellular STAT3 and PI3K signalling pathways. Partial reversal of leptin resistance can occur with weight loss and with reductions in adipose tissue inflammation, but it is not completely normalised in all individuals. Emerging pharmacological strategies targeting hypothalamic inflammation and leptin receptor sensitisation are under investigation (Tilg et al., 2025).
4. What is the role of macrophages in obesity?
In obesity, adipose tissue becomes infiltrated by large numbers of M1 (pro-inflammatory) macrophages that form 'crown-like structures' around dying adipocytes and release TNF-α, IL-1β, IL-6, and reactive oxygen species. These signals impair insulin signalling, promote further adipocyte inflammation, and sustain a cycle of metabolic deterioration. The shift from an M2-dominant (anti-inflammatory, tissue-remodelling) to an M1-dominant macrophage profile in adipose tissue is a critical molecular event linking obesity to insulin resistance and metabolic syndrome (Pan et al., 2023).
5. Can the pancreas recover if ectopic fat is removed?
Evidence from the Diabetes Remission Clinical Trial (DiRECT) and related studies suggests that substantial weight loss — sufficient to reduce pancreatic and hepatic fat — can restore near-normal beta-cell insulin secretion in some individuals with type 2 diabetes. The reversibility appears to depend on the duration of diabetes and the extent of residual beta-cell mass. This finding supports the concept that pancreatic fat-induced dysfunction is not inevitably permanent and reinforces the importance of early, intensive metabolic intervention (Nadolsky et al., 2025).
6. What is 'metaflammation' and how does it differ from classical inflammation?
Metaflammation (metabolic inflammation) is a chronic, low-grade, sterile inflammatory state arising from metabolic excess — particularly excess nutrient availability and lipid overload — rather than from infection or injury. Unlike classical acute inflammation, it does not resolve but instead persists for years, driven by adipose-derived cytokines, macrophage activation, and pattern recognition receptor signalling triggered by saturated fatty acids and damage-associated molecular patterns. Metaflammation underpins the pathogenesis of type 2 diabetes, atherosclerosis, NAFLD, and adiposity-associated cancers (Tilg et al., 2025).
7. How does the new AACE 2025 framework change how obesity is treated?
The 2025 AACE Consensus Statement formally adopts the Adiposity-Based Chronic Disease (ABCD) model, which centres treatment on the identification and management of obesity-driven complications — cardiometabolic risk, mechanical complications, and mental health — rather than on BMI thresholds alone. It provides a structured clinical algorithm integrating lifestyle therapy, pharmacotherapy with approved anti-obesity medications (including GLP-1 and dual GLP-1/GIP receptor agonists), and metabolic bariatric surgery. The framework explicitly recognises obesity as a relapsing, chronic endocrine disease requiring long-term, multi-disciplinary management (Nadolsky et al., 2025).
Author’s Note
For much of modern medical history, adipose tissue was treated as an afterthought — a passive reservoir of stored energy, relevant primarily for its quantity rather than its biology. As our understanding of metabolic disease has evolved, that assumption has proven profoundly incomplete. This article reflects a shift in perspective that has taken place across endocrinology, immunology, and metabolic research over the past two decades: fat is not inert. It is active, communicative, and biologically decisive.
My goal in writing this piece was not merely to summarize the literature on adipokines, macrophage polarization, or visceral adiposity. It was to present adipose tissue as it truly functions — as an endocrine and immunologic organ engaged in constant dialogue with the brain, liver, muscle, pancreas, and vasculature. When this dialogue is balanced, metabolic health is maintained. When it becomes inflamed and dysregulated, the consequences reverberate systemically.
Importantly, reframing obesity as an adiposity-based chronic endocrine disease is not semantic. It is clinical. It shifts the conversation from blame to biology, from stigma to structured intervention. Understanding adipose tissue signaling helps explain why modest weight loss yields disproportionate metabolic benefit, why visceral fat carries greater risk than subcutaneous fat, and why modern pharmacotherapies targeting GLP-1 pathways are effective beyond simple calorie reduction.
If this article accomplishes anything, I hope it encourages clinicians, researchers, and patients alike to reconsider fat not as excess mass on a scale, but as tissue with endocrine power. The future of metabolic medicine will not be built solely on counting calories — it will be built on understanding cellular signaling, inflammatory networks, and organ cross-talk.
Adipose tissue is not silent. It is speaking continuously. The task before us is to listen carefully — and respond scientifically.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Tired All Day, Awake at Night? The Redox and Mitochondrial Link | DR T S DIDWAL
Obesity and Fatty Liver Disease: What Science Says About Risk and Health | DR T S DIDWAL
Intermittent Fasting: Metabolic Health Benefits and the Evidence on Longevity | DR T S DIDWAL
Activate Your Brown Fat: A New Pathway to Longevity and Metabolic Health | DR T S DIDWAL
References
Clemente-Suárez, V. J., Redondo-Flórez, L., Beltrán-Velasco, A. I., Martín-Rodríguez, A., Martínez-Guardado, I., Navarro-Jiménez, E., Laborde-Cárdenas, C. C., & Tornero-Aguilera, J. F. (2023). The role of adipokines in health and disease. Biomedicines, 11(5), 1290. https://doi.org/10.3390/biomedicines11051290
Dobre, M.-Z., Virgolici, B., & Timnea, O. (2025). Key roles of brown, subcutaneous, and visceral adipose tissues in obesity and insulin resistance. Current Issues in Molecular Biology, 47(5), 343. https://doi.org/10.3390/cimb47050343
Nadolsky, K., Garvey, W. T., Agarwal, M., Bonnecaze, A., Burguera, B., Chaplin, M. D., Griebeler, M. L., Harris, S. R., Schellinger, J. N., Simonetti, J., Srinath, R., & Yumuk, V. (2025). American Association of Clinical Endocrinology consensus statement: Algorithm for the evaluation and treatment of adults with obesity/adiposity-based chronic disease — 2025 update. Endocrine Practice, 31(11), 1351–1394. https://doi.org/10.1016/j.eprac.2025.07.017
Pan, D., Li, G., Jiang, C., Hu, J., & Hu, X. (2023). Regulatory mechanisms of macrophage polarization in adipose tissue. Frontiers in Immunology, 14, 1149366. https://doi.org/10.3389/fimmu.2023.1149366
Tilg, H., Ianiro, G., & Gasbarrini, A. (2025). Adipokines: Masterminds of metabolic inflammation. Nature Reviews Immunology, 25, 250–265. https://doi.org/10.1038/s41577-024-01103-8