Brown Fat and Thermogenesis: The Hidden Metabolic System That May Transform Weight Loss
Discover how thermogenesis and brown fat influence metabolism, weight loss, and obesity treatment, including new research on GLP-1 drugs and metabolic pathways.
OBESITY
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/13/202616 min read
The Hidden Metabolic Furnace Behind Weight Loss
Millions of people follow strict diets, track calories meticulously, and exercise regularly — yet their weight loss eventually slows or stops. For decades, this frustrating plateau was explained by a simple equation: calories in versus calories out.
But modern metabolic research suggests something far more complex is happening inside the body. Hidden within our fat tissue is a biological system capable of burning calories without movement, generating heat instead of stored energy. Scientists call this process thermogenesis, and it may help explain why metabolism — not just diet — plays a powerful role in long-term weight regulation.
Over the past few years, scientific interest in thermogenesis has surged as researchers uncover its potential role in obesity, metabolic health, and weight-loss therapies. Studies show that people with obesity often have reduced brown fat activity, which may partly explain why some individuals burn fewer calories at rest and struggle with persistent weight-loss plateaus (Brown & Yoneshiro, 2024). At the same time, new research suggests that thermogenesis interacts with hormones, the nervous system, dietary nutrients, and even modern medications such as GLP-1 receptor agonists used for obesity treatment (Soliman et al., 2025).
Recent discoveries have made this field even more intriguing. A landmark study in Nature Metabolism found that altering the availability of a single amino acid — cysteine — can dramatically trigger fat browning and thermogenic energy expenditure in experimental models, opening entirely new avenues for metabolic research (Lee et al., 2025). Meanwhile, emerging neuroscience research shows that the nervous system controls brown fat through complex sympathetic circuits that influence both heat production and glucose metabolism (Alves, 2026).
Together, these discoveries reveal a powerful truth: weight regulation is not simply about willpower or calorie counting — it is deeply influenced by hidden metabolic systems inside our own fat tissue.
Key Insights
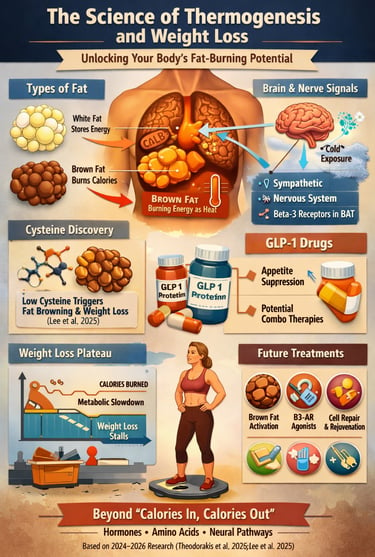
• Thermogenesis is the body’s heat-producing metabolic process, where specialized fat cells burn fuel to generate warmth rather than store energy. This mechanism can increase overall energy expenditure.
• Brown adipose tissue (BAT) plays a central role in thermogenesis. Unlike white fat that stores calories, brown fat contains mitochondria that burn energy and help regulate metabolism.
• Obesity is associated with reduced thermogenic activity, which may contribute to lower resting energy expenditure and weight-loss plateaus.
• Modern obesity treatments, including GLP-1–based medications, primarily reduce appetite but may also interact with metabolic pathways affecting thermogenesis.
• Future therapies may combine appetite control with thermogenic activation to improve long-term weight management and metabolic health.
What Is Thermogenesis and Why Should You Care?
Thermogenesis is the process by which the body burns calories to produce heat rather than storing that energy as fat. This metabolic mechanism is largely driven by brown adipose tissue (brown fat) — a specialised type of fat that contains mitochondria capable of converting fuel directly into heat through a protein called uncoupling protein-1 (UCP1). Unlike white fat, which primarily stores energy, brown fat actively increases energy expenditure and helps regulate body temperature and metabolism.
Researchers now believe thermogenesis plays an important role in weight regulation and obesity. Studies show that people with higher brown fat activity tend to have better insulin sensitivity, improved glucose metabolism, and lower body fat levels compared with individuals whose thermogenic systems are less active (Brown & Yoneshiro, 2024).
Modern obesity treatments are also beginning to interact with these pathways. GLP-1 receptor agonists, such as semaglutide and tirzepatide, primarily promote weight loss by reducing appetite, but emerging research suggests they may also influence brain signaling pathways that regulate thermogenesis and metabolic activity (Soliman et al., 2025).
Because the body can adapt to calorie restriction by slowing metabolism — a phenomenon known as adaptive thermogenesis — understanding how to maintain or stimulate thermogenic activity has become a major focus of metabolic research.
Scientists are now exploring strategies that combine appetite regulation, thermogenic activation, and metabolic health improvements to create more effective long-term treatments for obesity and weight management.
The Two Types of Fat — and Why Brown Fat Is Special
Most people know they have white fat — the energy-storing tissue that accumulates around the waist, hips, and thighs. But humans also have brown adipose tissue (BAT), a completely different type of fat whose entire purpose is to burn energy and release it as heat. Brown fat is packed with mitochondria (the energy-processing engines of the cell), which give it its characteristic dark colour.
When BAT is activated — usually by cold exposure or by signals from the nervous system — a remarkable protein called Uncoupling Protein 1 (UCP1) essentially “short-circuits” the normal energy-production pathway. Instead of generating ATP (the cellular energy currency), the mitochondria simply release energy as heat. In practical terms, this means your body is burning calories without you having to move a muscle.
A third type of fat, called beige adipose tissue, sits somewhere between white and brown. Under the right conditions, ordinary white fat depots can remodel themselves, sprouting beige cells capable of UCP1-driven thermogenesis in a process known as “browning.” This is an area of intense scientific interest, because it suggests that, in theory, you could activate heat-producing capacity within fat stores that currently just sit there.
Key Metabolic Mechanisms in Thermogenesis and Weight Regulation
• Brown adipose tissue (Brown fat)
What it does: Burns calories to produce heat through mitochondrial thermogenesis.
Why it matters: Increases total energy expenditure and may help regulate body weight and metabolic health.
• Branched-chain amino acid (BCAA) metabolism
What it does: Active brown fat helps clear circulating BCAAs such as leucine, isoleucine, and valine from the bloodstream.
Why it matters: Lower BCAA levels are associated with improved insulin sensitivity and a reduced risk of metabolic disorders like type 2 diabetes.
• Cysteine signaling pathway
What it does: The amino acid cysteine can act as a metabolic signal influencing the browning of white adipose tissue.
Why it matters: Alterations in cysteine availability may activate thermogenic pathways, making it a potential target for future obesity therapies.
• Sympathetic nervous system activation
What it does: Nerve signals from the brain stimulate brown fat through β3-adrenergic receptors, triggering thermogenesis.
Why it matters: Proper neural signaling is essential for activating the body’s natural calorie-burning systems.
• GLP-1–related metabolic signaling
What it does: GLP-1–based medications primarily reduce appetite but may also influence brain pathways that regulate thermogenesis.
Why it matters: Combining appetite suppression with thermogenic activation may improve long-term weight-loss outcomes.
How the Nervous System Controls Brown Fat Thermogenesis
Two Neural Pathways That Activate Brown Fat (2026 Discovery)
For many years, scientists believed that a simple biological pathway controlled brown adipose tissue (BAT). Cold exposure activates the brain, which sends signals through the sympathetic nervous system, and those nerves stimulate brown fat to produce heat via uncoupling protein-1 (UCP1).
However, new research published in Nature Reviews Endocrinology shows that the neural control of brown fat thermogenesis is far more complex than previously thought (Alves, 2026).
Researchers have now identified two distinct types of sympathetic nerve fibers that regulate BAT:
• Parenchymal sympathetic fibers
These nerves connect directly to brown fat cells and activate thermogenesis, allowing mitochondria to burn calories and produce heat.
• Vascular sympathetic fibers
These nerves control blood flow within adipose tissue and influence systemic metabolism, including glucose regulation, rather than directly generating heat.
This discovery has major implications for future obesity therapies. It suggests that the metabolic effects of brown fat — such as increased calorie burning and improved glucose metabolism — may be controlled by separate neural circuits.
As a result, drugs designed to target BAT could potentially:
• increase energy expenditure and fat burning,
• improve blood sugar regulation,
• or ideally do both simultaneously.
Designing therapies that activate the correct neural pathways is now an important goal in metabolic medicine and obesity research (Alves, 2026).
An easy way to understand this is to imagine the electrical wiring of a house. The heating system and lighting may both use electricity, but they run on separate circuits. Turning on one switch does not necessarily activate the other.
How Obesity Weakens Brown Fat Activation
Obesity can damage the neural signaling that activates fat-burning thermogenesis.
Studies show that individuals with obesity often have reduced sympathetic nerve density in adipose tissue. In simple terms, fewer nerve signals reach the fat cells that normally trigger heat production.
This creates a metabolic feedback loop:
Excess fat reduces nerve signaling to brown fat.
Reduced signaling lowers thermogenic calorie burning.
Lower energy expenditure promotes further fat accumulation.
Understanding how to restore sympathetic signaling to brown fat may therefore become an important strategy for treating obesity.
Brown Fat Does More Than Burn Calories
The Surprising Role of Brown Fat in BCAA Metabolism
Brown adipose tissue does more than generate heat. It also plays an important role in metabolic health and amino acid regulation.
A 2024 review in the Endocrine Journal highlighted the relationship between brown fat activity and branched-chain amino acids (BCAAs) — including leucine, isoleucine, and valine (Brown & Yoneshiro, 2024).
BCAAs are essential nutrients found in many protein-rich foods, including:
• meat
• dairy products
• eggs
• protein supplements
For decades, researchers observed that elevated BCAA levels in the blood are strongly associated with obesity and type 2 diabetes, but the reason remained unclear.
Recent studies suggest that active brown fat helps clear BCAAs from the circulation by metabolising them within mitochondria.
When BAT activity declines, which commonly occurs in obesity, BCAAs accumulate in the bloodstream and may contribute to insulin resistance and metabolic dysfunction.
A landmark study published in Cell demonstrated that this BCAA-clearing function of brown fat may protect against type 2 diabetes, even when thermogenesis itself is modest (Brown & Yoneshiro, 2024).
In other words, healthy brown fat improves metabolic health in ways that extend beyond calorie burning.
Why Dieting Can Slow Your Metabolism
Adaptive Thermogenesis During Calorie Restriction
When people reduce calorie intake to lose weight, fat tissue undergoes significant biological remodelling.
A 2025 review in Discover Medicine examined how calorie restriction affects brown fat and beige fat development (Mısırlıoğlu, 2025).
The research shows several beneficial changes:
• reduced chronic inflammation in adipose tissue
• increased anti-inflammatory immune cells
• stimulation of beige fat formation
• increased expression of thermogenic proteins such as UCP1
However, prolonged dieting can also activate adaptive thermogenesis, a biological survival mechanism.
During adaptive thermogenesis:
• metabolic rate slows
• energy expenditure declines
• brown fat activity may decrease
This is one of the main reasons why many people experience a weight-loss plateau despite continued dieting.
The Cysteine Discovery: A New Metabolic Switch
How an Amino Acid May Regulate Fat Burning
One of the most surprising discoveries in thermogenesis research involves the amino acid cysteine.
A 2025 study in Nature Metabolism analyzed adipose tissue samples from participants in the CALERIE-II clinical trial, where healthy adults reduced calorie intake by about 14% over two years (Lee et al., 2025).
Researchers found that caloric restriction significantly reduced cysteine levels in fat tissue.
Cysteine is commonly found in protein-rich foods, including:
• meat
• eggs
• dairy
• legumes
• nuts
Traditionally, cysteine was known mainly as a protein building block and precursor of glutathione, an important antioxidant.
However, new research suggests cysteine may also function as a metabolic signal regulating thermogenesis.
What Happens When Cysteine Levels Fall
To test this hypothesis, scientists experimentally depleted cysteine in obese mice.
The results were striking:
• rapid browning of white adipose tissue
• increased energy expenditure
• about 30% body-weight reduction
• reduced metabolic inflammation
When cysteine was reintroduced into the diet, the thermogenic effects reversed, confirming that cysteine availability was controlling the process (Lee et al., 2025).
Researchers concluded that cysteine acts as a metabolic brake on fat-burning thermogenesis.
When cysteine levels drop, adipose tissue appears to switch from energy storage mode to energy-burning mode.
A New Thermogenic Pathway
The most important discovery was the mechanism behind this effect.
Cysteine restriction activates thermogenesis through the sympathetic nervous system, triggering noradrenaline signaling and β3-adrenergic receptor activation in fat cells.
Interestingly, this process occurred independently of two previously known thermogenic regulators:
• UCP1
• FGF21
This suggests that multiple parallel thermogenic pathways exist, many of which are only beginning to be understood.
For obesity medicine, this means future treatments may target several different metabolic switches rather than relying on a single pathway.
Why Cellular Health Determines Thermogenesis
The “Cellular Competence” Problem
Even when the right hormones, nerves, and signals are present, thermogenesis may still fail.
A 2026 perspective in Frontiers in Cell and Developmental Biology introduced the concept of cellular competence (Brown, 2026).
Cellular competence refers to whether fat cells possess the internal machinery required to generate heat efficiently.
Healthy adipocytes require:
• functional mitochondria
• intact cell signaling pathways
• efficient vesicle trafficking systems
In obesity and aging, these systems can deteriorate.
As a result:
• mitochondria become dysfunctional
• metabolic signaling becomes inefficient
• fuel delivery to thermogenic pathways declines
The outcome is that the thermogenic signal is received but cannot be executed efficiently.
In simple terms, the problem is not just the signal — the metabolic engine itself is damaged.
Future obesity treatments may therefore focus on restoring mitochondrial health and cellular metabolism, rather than simply increasing thermogenic stimulation.
The Bigger Picture: Energy Balance Is Personal
• Obesity is more than a calorie imbalance.
For decades, weight management advice focused on the simple equation of “calories in versus calories out.” While energy balance remains fundamental, modern metabolic research shows that hormonal regulation, neural signaling, gut microbiota, and thermogenic capacity strongly influence how the body stores and expends energy (Theodorakis & Nikolaou, 2025).
• Brown adipose tissue is a metabolic regulator, not just a heat generator.
Brown fat was once considered relevant mainly for infant temperature regulation. Today, studies show that active brown adipose tissue improves glucose metabolism, clears circulating branched-chain amino acids (BCAAs), and may protect against insulin resistance, even when thermogenesis itself is modest (Brown & Yoneshiro, 2024).
• The nervous system plays a central role in activating the body’s metabolic furnace.
Recent neuroscience research demonstrates that brown fat receives distinct sympathetic nerve inputs controlling thermogenesis and vascular metabolism separately, highlighting new targets for therapeutic intervention (Alves, 2026).
• Dietary signals may influence thermogenic pathways.
One of the most intriguing discoveries in metabolic research is that the amino acid cysteine may function as a metabolic switch. Experimental evidence suggests that reducing cysteine availability can trigger fat browning and increase energy expenditure through sympathetic activation (Lee et al., 2025).
• GLP-1 medications represent a major advance—but not a complete solution.
Drugs such as semaglutide and tirzepatide primarily reduce appetite. Their effects on thermogenesis are modest and variable, which may explain why many patients eventually reach weight-loss plateaus despite continued therapy (Soliman et al., 2025).
• Future obesity treatments will likely be combination therapies.
The most promising strategy may combine appetite regulation, thermogenic activation, and cellular metabolic repair. This integrated approach could address the multiple biological drivers of obesity rather than targeting a single pathway.
• Personalized metabolic medicine is the next frontier.
As researchers better understand the roles of brown fat activity, neural signaling, amino-acid metabolism, and cellular competence, clinicians may eventually tailor obesity treatments to each patient’s unique metabolic profile.
What You Can Do Today
While the research continues and new treatments develop, there are evidence-supported lifestyle strategies that influence thermogenic activity:
• Cold exposure: Even brief cold showers or cooler ambient temperatures can activate BAT in adults. The evidence for meaningful metabolic impact is modest but real.
• Exercise: Physical activity improves BAT innervation and function over time, and promotes the browning of white adipose tissue, partly through hormones released by muscle during exercise.
• Dietary quality matters: Excess saturated fat can impair the GLP-1 and GIP receptor pathways that help regulate thermogenesis. A varied, nutritionally dense diet protects these signalling systems.
• Moderate caloric restriction: As the Mısırlıoğlu (2025) review showed, moderate — rather than severe — energy restriction tends to promote more favourable adipose remodelling without triggering the aggressive adaptive suppression of thermogenesis seen with crash dieting.
• Work with your healthcare team: If you are on GLP-1 medication and have plateaued, ask your provider about emerging combination strategies or clinical trials investigating thermogenic agents.
Frequently Asked Questions
Q1. What exactly is brown adipose tissue and do adults actually have it?
Yes — adults do have brown adipose tissue (BAT), although in smaller amounts than infants or rodents. In adults, it is typically found around the collarbone, neck, and along the spine. Functional BAT has been confirmed in adults using PET imaging, and its amount is inversely correlated with body mass index. People with more active BAT tend to have better metabolic health, including lower rates of type 2 diabetes.
Q2. If I take semaglutide or tirzepatide, am I already activating brown fat?
Partially. GLP-1 receptor agonists do stimulate BAT thermogenesis to some degree, primarily through the brain’s hypothalamus, increasing sympathetic nerve signals to fat tissue. However, this effect is inconsistent across individuals, depends on the dose and route of administration, and tends to diminish over time. Researchers are exploring whether adding a dedicated thermogenic agent (such as a beta-3 adrenergic receptor agonist) to GLP-1 therapy could provide more robust, sustained calorie-burning effects.
Q3. Why do I always seem to hit a weight-loss plateau even when I am doing everything right?
This is a very real and scientifically documented phenomenon. When you restrict calories, your body interprets this as a signal of scarcity and activates adaptive mechanisms to conserve energy — including reducing the thermogenic activity of brown fat. Research published in 2025 by Mısırlıoğlu confirmed that sustained energy restriction remodels adipose tissue in ways that can both help (promoting beige fat formation) and hinder (suppressing thermogenic output). A plateau is not a personal failure; it is your biology trying to protect itself.
Q4. What are branched-chain amino acids (BCAAs) and how do they relate to obesity?
BCAAs — leucine, isoleucine, and valine — are amino acids found in protein-rich foods like meat, dairy, and many protein supplements. Elevated blood levels of BCAAs have long been associated with type 2 diabetes and obesity. Recent research has revealed that healthy, active brown adipose tissue plays a crucial role in clearing BCAAs from the bloodstream. When BAT activity declines (as it does in obesity), BCAAs accumulate, contributing to insulin resistance. This means that restoring BAT function could improve metabolic health through mechanisms completely separate from calorie burning.
Q5. Can cold exposure really help with weight loss?
Cold exposure does activate brown fat and can increase energy expenditure, and this effect has been confirmed in adult humans. However, the practical contribution to weight loss is modest by itself — you would need very prolonged or intensive cold exposure to generate a clinically meaningful caloric deficit through BAT activation alone. That said, regular mild cold exposure (cooler environments, cold showers) may help maintain BAT function and improve metabolic markers, particularly as part of a broader healthy lifestyle. The real excitement in this area is pharmacological: drugs that mimic cold’s effect on brown fat — without the discomfort of actually being cold — are in active development.
Q6. What does “cellular competence” mean and why does it matter for weight loss treatments?
Cellular competence, as described by Aaron Brown (2026), refers to the internal readiness of fat cells to actually carry out thermogenesis when stimulated. Think of it as the difference between a car that has fuel and a functioning engine versus one with fuel but a broken engine. In obesity and aging, the internal machinery of fat cells — including their mitochondria, signalling pathways, and trafficking systems — degrades. This means stronger stimulation (higher drug doses, more aggressive interventions) may not work if the cell itself cannot respond. Future treatments may need to repair this cellular infrastructure, not just push harder on existing pathways.
Q7. Is there a single “best” treatment for obesity coming in the future?
Probably not a single one, but rather a more personalised approach. The emerging scientific consensus is that obesity involves multiple overlapping mechanisms — appetite regulation, thermogenic capacity, BCAA metabolism, neural wiring of fat tissue, cellular health of adipocytes, and now amino acid sensing (like the cysteine pathway) — that vary significantly between individuals. The most promising future therapies are likely to combine GLP-1 based appetite suppression with thermogenic activators (such as beta-3 AR agonists), cellular repair strategies, and lifestyle interventions tailored to each person’s specific biological profile. The goal is not to find one magic bullet, but to match the right combination of tools to each patient’s unique biology.
Q8. Should I reduce cysteine in my diet to lose weight?
Not based on current evidence, and this would be premature and potentially risky. The dramatic thermogenic effects of cysteine depletion in Lee et al. (2025) were observed in animal models using complete systemic cysteine removal — an extreme condition far beyond what dietary changes could safely achieve in humans. Cysteine plays vital roles throughout the body, including in antioxidant defence (as a precursor to glutathione) and protein synthesis. The value of this research is not as a dietary prescription, but as a scientific breakthrough revealing a new molecular switch controlling thermogenesis. Pharmaceutical researchers may one day develop targeted ways to modulate this pathway safely. For now, focus on balanced dietary protein and speak with your healthcare provider about evidence-based weight management strategies.
For decades, weight loss was framed as a simple matter of discipline — eat less, move more. But modern metabolic research is revealing a far more intricate biological landscape. Thermogenesis, brown adipose tissue, amino-acid signaling, neural circuits, and cellular metabolism all interact to determine how the body stores or burns energy. As scientists continue to map these hidden metabolic systems, the future of obesity treatment may shift away from one-size-fits-all advice toward personalized metabolic medicine — therapies designed not only to reduce appetite, but also to restore the body’s natural ability to burn energy.
Author’s Note
This article was written to bridge the gap between rapidly evolving metabolic science and the practical questions many patients ask about weight loss, obesity, and metabolic health. Over the past decade, research into thermogenesis, brown adipose tissue, and metabolic signaling pathways has expanded dramatically. Studies published in leading journals have revealed that body weight regulation is influenced not only by calorie intake and physical activity, but also by complex biological systems involving hormones, the nervous system, cellular metabolism, and nutrient signaling.
The goal of this article is to translate these emerging scientific insights into clear, patient-friendly explanations without oversimplifying the underlying biology. Each section draws on findings from peer-reviewed research published between 2024 and 2026, including studies exploring brown fat metabolism, branched-chain amino acid regulation, neural control of thermogenesis, and the metabolic effects of GLP-1–based obesity therapies.
At the same time, it is important to recognize that metabolic science is continually evolving. Many discoveries discussed here — such as the potential role of amino acids like cysteine in regulating thermogenesis — are still being investigated and should not yet be interpreted as direct clinical recommendations. The intention is not to promote specific treatments or diets, but rather to help readers understand how the body’s metabolic systems influence weight regulation and why sustainable weight management often requires a broader approach than calorie restriction alone.
Readers are encouraged to view this article as educational guidance grounded in current scientific evidence, rather than individualized medical advice. Anyone considering changes to their diet, lifestyle, or medications should discuss these decisions with a qualified healthcare professional.
By sharing this research in an accessible format, the hope is to encourage better understanding of metabolism, more informed conversations between patients and clinicians, and a deeper appreciation of the biological complexity underlying obesity and metabolic health
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Your Body Fat Is an Endocrine Organ—And Its Hormones Shape Your Heart Health | DR T S DIDWAL
hsCRP Explained: What Inflammation Means for Your Heart | DR T S DIDWAL
What’s New in the 2025 Blood Pressure Guidelines? A Complete Scientific Breakdown | DR T S DIDWAL
Low-Fat vs. Low-Carb: Which Diet is Best for Weight Loss? | DR T S DIDWA
References
All sources cited in this article are peer-reviewed academic publications. Readers are encouraged to consult the original papers for full methodological details.
Alves, J. M. (2026). Two roads to thermogenesis: vascular and parenchymal sympathetic innervation of brown adipose tissue. Nature Reviews Endocrinology, 22, 198. https://doi.org/10.1038/s41574-025-01224-7
Brown, A. C. (2026). Thermogenesis is limited by cellular competence. Frontiers in Cell and Developmental Biology, 14, 1784579. https://doi.org/10.3389/fcell.2026.1784579
Brown, Z., & Yoneshiro, T. (2024). Brown fat thermogenesis and branched-chain amino acids in metabolic disease. Endocrine Journal, 71(2), 89–100. https://doi.org/10.1507/endocrj.EJ23-0205
Lee, A. H., Orliaguet, L., Youm, Y. H., Maeda, R., Dlugos, T., Lei, Y., Coman, D., Shchukina, I., Andhey, P. S., Smith, S. R., Ravussin, E., Stadler, K., Chen, B., Artyomov, M. N., Hyder, F., Horvath, T. L., Schneeberger, M., Sugiura, Y., & Dixit, V. D. (2025). Cysteine depletion triggers adipose tissue thermogenesis and weight loss. Nature Metabolism, 7(6), 1204–1222. https://doi.org/10.1038/s42255-025-01297-8
Mısırlıoğlu, P. E. (2025). Energy restriction drives adipose tissue remodeling and thermogenic adaptation. Discover Medicine, 2, 295. https://doi.org/10.1007/s44337-025-00535-y
Soliman, S., Andrews-Dickert, R., Rocic, P., & Mitov, M. (2025). Thermogenic targets for obesity management in the era of incretin-based therapies. Pharmaceuticals, 18(10), 1519. https://doi.org/10.3390/ph18101519
Theodorakis, N., & Nikolaou, M. (2025). The human energy balance: Uncovering the hidden variables of obesity. Diseases, 13(2), 55. https://doi.org/10.3390/diseases13020055