The Cardiometabolic Continuum: How Insulin Resistance Leads to Heart Disease (2026 Update)
Your heart disease may begin years before symptoms. Explore how insulin resistance drives endothelial dysfunction and atherosclerosis—and what tests and lifestyle strategies can stop it early.
DIABETESHEART
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/25/202615 min read
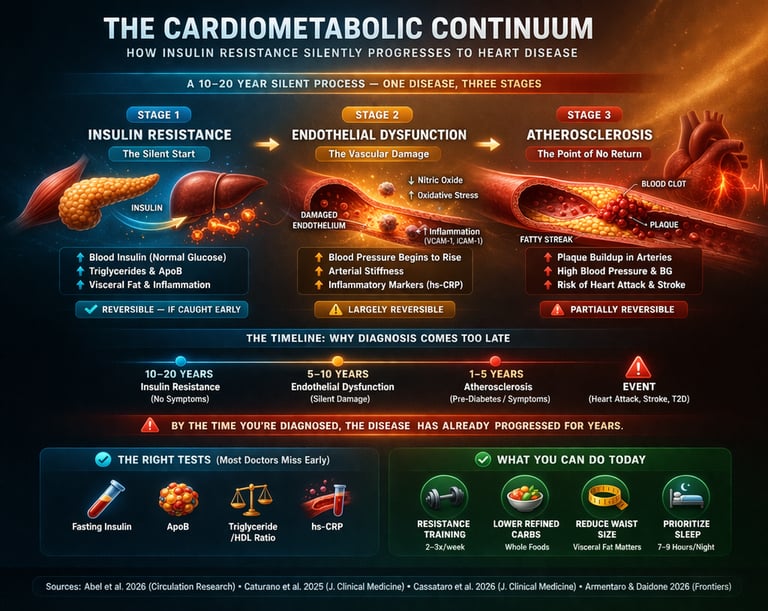
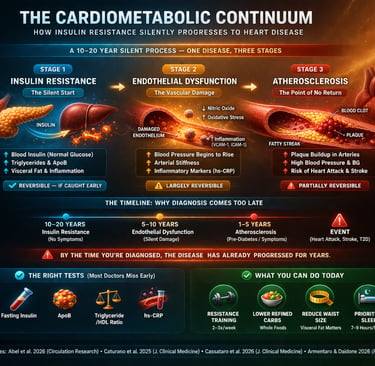
The cardiometabolic continuum is a progressive disease model in which insulin resistance leads to endothelial dysfunction and ultimately atherosclerosis and heart disease. This process can begin 10–20 years before diagnosis, often with normal blood sugar levels. Early detection using markers like fasting insulin, ApoB, and inflammatory biomarkers allows intervention before irreversible vascular damage occurs. The Cardiometabolic Continuum: How Insulin Resistance Silently Progresses to Heart Disease
Stages of the Cardiometabolic Continuum:
Insulin Resistance – Elevated insulin with normal glucose (early, silent stage)
Endothelial Dysfunction – Reduced nitric oxide, increased inflammation
Atherosclerosis – Plaque formation and arterial narrowing
Clinical Disease – Diabetes, heart attack, or stroke
Heart disease rarely begins with chest pain. It begins quietly—years, often decades earlier—inside your metabolism.
Long before a diagnosis of diabetes or a blocked artery, a silent process is already underway: insulin resistance. In this early phase, your blood sugar may still appear “normal,” but your body is compensating by producing higher levels of insulin to keep it that way. This hidden state can persist for 10–20 years, during which metabolic damage accumulates beneath the surface (Caturano et al., 2025).
What makes this process dangerous is not just elevated glucose, but what high insulin and metabolic dysfunction do to your blood vessels. Insulin resistance disrupts critical signaling pathways, impairing nitric oxide production and promoting oxidative stress within the endothelium—the delicate inner lining of your arteries. This marks the transition from a metabolic disturbance to early vascular disease (Abel et al., 2026).
At the same time, the liver begins overproducing atherogenic lipoproteins, increasing the number of ApoB-containing particles that infiltrate the arterial wall. Inflammation rises, macrophages are activated, and the earliest stages of atherosclerosis begin—often without a single symptom (Cassataro et al., 2026).
By the time blood sugar crosses the threshold for diabetes—or a patient presents with angina or myocardial infarction—the disease has already progressed through multiple biological stages. This is why cardiovascular events frequently occur soon after a diabetes diagnosis: the underlying vascular damage has been evolving silently for years (Armentaro & Daidone, 2026).
This is the essence of the cardiometabolic continuum—a unified disease process linking insulin resistance, vascular dysfunction, and heart disease. And understanding it may be the most important step in preventing both.
Clinical Pearls
1. The "Pancreatic Overtime" Rule
Scientific Perspective: Hyperinsulinemia can maintain euglycemia (normal blood sugar) for up to 20 years by compensating for peripheral insulin resistance. Relying solely on HbA1c or Fasting Glucose for screening ignores the early, pathological rise in endogenous insulin production.
Your blood sugar test is like a "final grade," but it doesn't show how hard you had to study. You can have a "passing grade" (normal sugar) while your pancreas is working "triple overtime" to keep it there. Eventually, the pancreas burns out. We want to catch the struggle before the burnout.
2. The "Back Door" to Muscle Cells
Scientific Perspective: Muscle contraction triggers GLUT4 translocation to the cell membrane through an AMP-activated protein kinase (AMPK) pathway. This process is insulin-independent, meaning exercise can clear glucose from the blood even in a highly insulin-resistant state.
Think of insulin as the "Front Door Key" to your muscles. In insulin resistance, that key is broken. However, exercise acts like a "Back Door." When you move your muscles, they open up to take in sugar automatically, without needing the broken insulin key at all.
3. The "Traffic Count" vs. "Passenger Weight"
Scientific Perspective: Apolipoprotein B (ApoB) provides a direct measure of the total number of atherogenic particles (LDL, VLDL, IDL). LDL-Cholesterol (LDL-C) only measures the mass of cholesterol within those particles. In metabolic syndrome, patients often have "Discordance"—normal LDL-C but high ApoB—increasing their risk for plaque formation.
To predict a car crash, you don't weigh the passengers; you count the number of cars on the road. LDL-C is the weight of the passengers, but ApoB is the number of cars. Even if the passengers are "light," too many cars on the road lead to more accidents in your arteries.
4. The "Internal Rust" of the Endothelium
Scientific Perspective: Chronic hyperinsulinemia and hyperglycemia increase the production of Reactive Oxygen Species (ROS), which "quench" Nitric Oxide (NO). This leads to endothelial dysfunction—a state where blood vessels lose their ability to dilate and become pro-inflammatory and pro-thrombotic.
High sugar and high insulin act like "biological rust" inside your pipes. This rust eats away at the "Teflon coating" (Nitric Oxide) of your arteries. Once that coating is gone, your blood vessels get stiff and "sticky," making it easy for cholesterol to latch on and form clogs.
5. The "Personal Fat Threshold"
Scientific Perspective: Every individual has a genetically determined "Personal Fat Threshold" (PFT). Once subcutaneous adipose tissue capacity is exceeded, lipids begin to deposit ectopically in the liver and pancreas, triggering the "Twin Cycle" of Type 2 Diabetes regardless of the patient's Body Mass Index (BMI).
Everyone has a different-sized "fat suitcase." Some people have a huge suitcase and can stay metabolically healthy at a heavier weight. Others have a tiny suitcase; if they gain even 5 pounds, the "clothes" (fat) spill out into their liver and heart. This is why "thin" people can still get metabolic disease.
What Is the Cardiometabolic Continuum?
The term "cardiometabolic continuum" describes the idea that cardiovascular disease and metabolic disorders (especially type 2 diabetes) share a common biological origin and progress through overlapping, interconnected stages.
Recent research by Caturano et al. (2025) in the Journal of Clinical Medicine argues powerfully for "rethinking the diabetes-cardiovascular disease continuum," calling for integrated care models that treat these conditions as one unified disease process rather than two separate problems. Their work highlights that the conventional siloed approach — treating blood sugar in one clinic and heart risk in another — leads to late detection and fragmented therapy.
Similarly, Armentaro and Daidone (2026) described the continuum as spanning hypertension, diabetes, and cardiovascular disease — all driven by the same underlying physiological disruptions.
This is not a minor academic reshuffling. It changes everything about how we should screen for, think about, and manage chronic disease.
Stage 1: Insulin Resistance — The Silent Starting Point
What Is Insulin Resistance?
Insulin is the hormone that acts like a key, unlocking your cells so they can absorb glucose from the bloodstream for energy. Insulin resistance happens when your cells stop responding to that key efficiently. Your pancreas compensates by producing more insulin. Blood glucose stays normal — for a while — but the system is under strain.
This compensatory phase can last 10 to 20 years before blood sugar rises to diabetic levels. During this entire time, damage is accumulating.
Why It Matters So Much
Insulin resistance affects three critical organs simultaneously:
Skeletal muscle: Takes up less glucose, leaving more circulating in the blood.
Liver: Increases its production of glucose (gluconeogenesis) and pumps out more triglyceride-rich particles.
Adipose tissue: Releases excess free fatty acids, which worsen insulin signaling in muscle and liver — a vicious cycle.
The molecular machinery breaks down at the level of IRS-1 signaling and the PI3K-Akt pathway, which normally drives glucose uptake. Intramyocellular lipid accumulation and ceramide buildup further impair these pathways.
How to Detect It Early
Traditional testing misses insulin resistance almost entirely. Fasting glucose and HbA1c only become abnormal once the pancreas begins to fail — meaning the disease has been progressing for years before the test turns positive.
Better early markers include:
Fasting insulin detects compensatory hyperinsulinemia early
HOMA-IR Calculated score of insulin resistance
Triglyceride/HDL ratio: Surrogate marker of insulin resistance
ApoB Reflects atherogenic particle burden
The groundbreaking AHA-funded research network described by Abel et al. (2026) in Circulation Research maps precisely why insulin resistance is the foundational driver — establishing a biological road map from metabolic dysfunction to full cardiovascular disease.
Stage 2: Endothelial Dysfunction — When the Vascular System Starts to Fail
The Endothelium: Your Vascular Shield
The endothelium is a thin, single-cell layer lining every blood vessel in your body. It is not just passive plumbing. It actively regulates:
Vascular tone (dilating and constricting blood vessels through nitric oxide production)
Inflammation (deciding when to recruit immune cells)
Clotting (controlling the balance between bleeding and thrombosis)
A healthy endothelium is powerfully anti-atherogenic — it keeps vessels clean, flexible, and inflammation-free.
How Insulin Resistance Damages the Endothelium
Here is where the story becomes clinically urgent. Insulin normally signals the endothelium to produce nitric oxide (NO), which relaxes blood vessels and reduces inflammation. When insulin signaling is impaired:
Nitric oxide production drops (via reduced eNOS activation)
Oxidative stress rises (reactive oxygen species damage vascular cells)
Endothelin-1 increases (a powerful vasoconstrictor)
Adhesion molecules such as VCAM-1 and ICAM-1 appear on the vessel wall, recruiting inflammatory cells
What makes this particularly insidious is a paradoxical split in insulin's effects: the metabolic pathway (glucose uptake) becomes impaired, but the mitogenic pathway (cell growth and proliferation) remains active. This means high insulin levels drive vascular smooth muscle cell proliferation — actively contributing to arterial stiffening and early plaque formation.
Why This Stage Is Critical — and Hopeful
Endothelial dysfunction is the pivot point of the continuum. It represents the transition from a metabolic problem to a structural vascular problem. But here is the hopeful truth: at this stage, the damage is still largely reversible.
Exercise — particularly aerobic training — directly improves endothelial function by upregulating eNOS and increasing nitric oxide bioavailability. Dietary changes, weight loss, and medications such as SGLT2 inhibitors and GLP-1 receptor agonists have all shown benefit at this stage.
Cassataro et al. (2026) in the Journal of Clinical Medicine specifically emphasize that personalized cardiometabolic management must identify and address these intermediate stages, noting that clinical phenotyping — understanding which metabolic abnormalities are present and how severe — is essential to effective intervention.
Stage 3: Atherosclerosis — Structural Vascular Disease
The Birth of a Plaque
Once the endothelium is dysfunctional and inflamed, the door opens for atherosclerosis — the buildup of fatty plaques inside arterial walls. This is not just "clogging" as the old metaphor suggested. It is an active, inflammatory disease.
The process unfolds in stages:
LDL cholesterol infiltrates the damaged endothelium and enters the arterial wall (intima).
Oxidation of LDL (oxLDL) triggers an immune response — macrophages are recruited.
Macrophages engulf oxLDL and become foam cells, forming early fatty streaks.
Smooth muscle cells proliferate, a necrotic core develops, and a fibrous cap forms over it.
Plaque instability develops over time — the fibrous cap can rupture, triggering acute clotting and causing a heart attack or stroke.
How Insulin Resistance Accelerates Atherosclerosis
Insulin resistance specifically accelerates this process by:
Increasing ApoB-containing lipoprotein particles (the particles that carry LDL into the artery wall)
Promoting small, dense LDL — the most atherogenic form
Sustaining chronic low-grade inflammation (elevated TNF-α, IL-6, hs-CRP)
Impairing reverse cholesterol transport
The AHA road map by Abel et al. (2026) connects these molecular dots with particular clarity, showing how the inflammatory milieu of insulin resistance feeds directly into plaque biology.
The Interconnected Web: Why These Diseases Reinforce Each Other
The cardiometabolic continuum is not a straight line — it is a web of reinforcing feedback loops.
Insulin resistance drives inflammation → inflammation worsens insulin resistance
Hyperglycemia damages the endothelium → damaged endothelium increases cardiovascular risk
Lipotoxicity from excess free fatty acids → further impairs pancreatic beta-cell function
We are treating the wrong disease at the wrong time.
Modern medicine still waits for glucose to rise or arteries to block before acting. Yet the real pathology—insulin resistance—begins silently decades earlier, long before diagnostic thresholds are crossed (Caturano et al., 2025).Cardiovascular disease is not a “late complication” of diabetes—it is part of the same disease.
The traditional separation between endocrinology and cardiology is increasingly obsolete. The cardiometabolic continuum reframes these conditions as a single, progressive biological process (Abel et al., 2026).Hyperinsulinemia—not just hyperglycemia—is a central driver.
Elevated insulin levels promote endothelial dysfunction, vascular smooth muscle proliferation, and pro-atherogenic signaling—well before glucose becomes abnormal.The endothelium is the battlefield where metabolism meets cardiology.
Impaired nitric oxide signaling, oxidative stress, and inflammatory activation mark the critical transition from metabolic dysfunction to structural vascular disease (Cassataro et al., 2026).ApoB, not LDL-C, defines true atherogenic risk.
Particle number—not cholesterol content—determines arterial infiltration. Many “normal” lipid panels miss this entirely, delaying intervention.By the time diabetes is diagnosed, vascular disease is often advanced.
This explains why cardiovascular events cluster early after diagnosis—the pathology has been evolving silently for years (Armentaro & Daidone, 2026).Lifestyle intervention is not adjunct therapy—it is first-line disease modification.
Exercise, particularly resistance and aerobic training, directly targets insulin sensitivity, endothelial function, and mitochondrial health.Pharmacotherapy has evolved—but remains underutilized early.
Agents such as SGLT2 inhibitors and GLP-1 receptor agonists act at multiple points along the continuum, yet are often introduced too late.The future is early detection and integrated care.
Measuring fasting insulin, ApoB, and inflammatory markers must become routine—not optional.The paradigm must shift from reaction to prevention.
The cardiometabolic continuum is not just a theory—it is a clinical imperative.
Therapeutic Strategies Across the Continuum
Lifestyle: Still the Most Powerful Medicine
Exercise remains the single most effective intervention across the entire continuum.
Resistance training improves insulin sensitivity independent of weight loss by increasing GLUT4 transporter expression in muscle cells and enhancing mitochondrial density.
Aerobic exercise directly improves endothelial function by upregulating nitric oxide production.
HIIT (High-Intensity Interval Training) has shown superior effects on insulin sensitivity and VO₂ max compared to moderate continuous exercise in several head-to-head trials.
Nutrition strategies with strong evidence include:
Reducing refined carbohydrates (lowers postprandial glucose and insulin spikes)
Increasing dietary protein (preserves lean mass, promotes satiety)
Omega-3 fatty acids (reduce triglycerides and systemic inflammation)
Mediterranean-style dietary patterns (protective endothelial effects)
Sleep and circadian rhythm are increasingly recognized as metabolic regulators. Chronic sleep deprivation raises cortisol, impairs insulin sensitivity, and increases inflammatory markers.
Key Pharmacological Agents
Metformin
Mechanism: Improves insulin sensitivity and reduces the amount of glucose produced by the liver.
Continuum Benefit: Primarily targets the early metabolic stage, helping to arrest the progression of insulin resistance before structural damage occurs.
SGLT2 Inhibitors
Mechanism: Promotes glycosuria (excreting excess sugar through urine) and reduces cardiac preload (the "stretch" on the heart).
Continuum Benefit: Provides powerful heart failure and renal (kidney) protection, addressing the middle-to-late stages where organ strain becomes critical.
GLP-1 Receptor Agonists
Mechanism: Reduces appetite, supports weight loss, and directly lowers systemic inflammation.
Continuum Benefit: Effective across multiple stages, from reversing obesity-driven insulin resistance to reducing the risk of major cardiovascular events.
Statins
Mechanism: Lowers ApoB-containing particles and strengthens the "cap" on existing arterial plaques to prevent them from bursting.
Continuum Benefit: Essential for atherosclerosis prevention and regression, targeting the structural vascular stage of the disease.
The Move Toward Personalized Management
The modern clinical consensus emphasizes that these drugs should not be used in a "one-size-fits-all" manner. Instead, the approach must be:
Phenotype-Driven: Identifying exactly where a patient sits on the continuum (e.g., Are they still in the silent insulin-resistance phase, or is there established plaque?).
Integrated Combinations: Using these agents together to provide "synergistic protection" for the heart, kidneys, and metabolism simultaneously.
Proactive Intervention: Using these tools to stabilize the "biological soil" of the body before a major clinical event (like a heart attack) occurs.
.
Practical Applications: What You Can Do Starting Today
Understanding the cardiometabolic continuum is not just for doctors. Here is what it means for you:
Ask for the Right Tests
Standard fasting glucose and HbA1c miss the early stages. Request:
Fasting insulin level
HOMA-IR calculation
ApoB (not just LDL)
hs-CRP (high-sensitivity C-reactive protein)
Triglyceride/HDL ratio
Start Resistance Training
Even 2–3 sessions per week of resistance exercise significantly improves insulin sensitivity. You do not need to run marathons. Lifting weights, doing bodyweight exercises, or using resistance bands all count.
Reduce Your Processed Carbohydrate Load
This does not mean eliminating all carbs. It means replacing refined grains, sugary drinks, and ultra-processed foods with whole foods, vegetables, legumes, and lean proteins.
Measure Your Waist Circumference
Visceral adiposity is a stronger cardiometabolic risk marker than BMI. In general:
Men: Waist >94 cm (37 inches) = elevated risk
Women: Waist >80 cm (31.5 inches) = elevated risk
Prioritize Sleep
Aim for 7–9 hours of quality sleep per night. Poor sleep increases cortisol, raises blood sugar, and drives inflammation — three direct inputs into the cardiometabolic continuum.
Work With an Integrated Care Team
If you have diabetes or heart disease risk, advocate for a care model that addresses both. Ask your doctor whether an endocrinologist and cardiologist are coordinating your care. The integrated approach is now supported by top-level research (Caturano et al., 2025).
Frequently Asked Questions
What is the cardiometabolic continuum, in simple terms? It is the idea that insulin resistance, type 2 diabetes, and cardiovascular disease are not separate conditions — they are different stages of the same underlying disease process. One leads to the other, often over many years.
Can you have cardiovascular disease risk even if your blood sugar is normal? Absolutely. Insulin resistance — the earliest stage — can exist for 10–20 years with completely normal blood glucose. This is why standard glucose testing misses early cardiometabolic risk. Fasting insulin, HOMA-IR, and ApoB are more sensitive early markers.
Is eindothelial dysfunction reversible? Yes — and this is one of the most clinically important facts in this field. Endothelial dysfunction, identified early, responds well to aerobic exercise, dietary change, weight loss, and certain medications. It becomes progressively harder to reverse as atherosclerosis advances.
What is the difference between ApoB and LDL-C, and why does it matter? LDL-C measures the cholesterol content inside LDL particles. ApoB measures the number of atherogenic particles directly. In insulin-resistant individuals, LDL-C can appear normal even when ApoB is elevated — meaning standard cholesterol panels can be falsely reassuring. ApoB is a superior marker of cardiovascular risk.
Does losing weight fix insulin resistance? Weight loss — especially loss of visceral fat — significantly improves insulin sensitivity. However, exercise improves insulin sensitivity independent of weight loss, which means you do not have to achieve your goal weight to start reducing your risk. Every step counts.
How do SGLT2 inhibitors and GLP-1 agonists help the cardiometabolic continuum? These newer drug classes work at multiple points in the continuum simultaneously. SGLT2 inhibitors reduce cardiovascular events, heart failure hospitalizations, and kidney disease progression — effects that go beyond glucose control. GLP-1 receptor agonists reduce weight, inflammation, blood pressure, and cardiovascular events. Both are now considered first-line for high-risk cardiometabolic patients.
At what age should I start thinking about cardiometabolic health? The earlier, the better — but the answer is: now, regardless of age. Atherosclerosis begins in adolescence. Insulin resistance can develop in the 20s and 30s. The lifestyle habits you build in your 30s and 40s determine your cardiovascular health in your 60s and beyond. Early biomarker testing is appropriate for anyone with a family history of diabetes, heart disease, or obesity.
Can you have heart disease risk with normal blood sugar?
Yes. Insulin resistance can exist for 10–20 years before glucose rises. During this time, vascular damage and atherosclerosis may already be developing.
What is the earliest sign of cardiometabolic disease?
Elevated fasting insulin and HOMA-IR are among the earliest detectable markers, often appearing years before abnormal glucose levels.
Call to Action: Take Your Cardiometabolic Health Into Your Own Hands
The research is clear: the best time to intervene in the cardiometabolic continuum is before symptoms appear. Waiting for a diabetes diagnosis or a cardiac event means waiting until the disease has already advanced through its most treatable stages.
Here is your action plan:
✅ This week: Schedule a blood test that includes fasting insulin and ApoB — not just glucose and LDL.
✅ This month: Add two sessions of resistance training to your weekly routine.
✅ This year: Have a frank conversation with your doctor about integrated cardiometabolic risk — using the language of the continuum, not just individual risk factors.
Author’s Note: A Clinician’s Perspective on the Cardiometabolic Continuum
As a practicing physician, one of the most consistent and concerning patterns I observe is how late we encounter cardiometabolic disease in its trajectory. Patients often present with a new diagnosis of type 2 diabetes, angina, or even an acute coronary event—and yet, when you step back and examine their metabolic history, it becomes clear that the disease process has been evolving silently for years, if not decades.
What is particularly striking is that many of these patients had “normal” routine blood tests for years. Fasting glucose was within range. LDL cholesterol did not raise alarm. And yet, beneath that apparent normalcy, insulin resistance, hyperinsulinemia, and endothelial dysfunction were already at work. By the time traditional markers become abnormal, we are often dealing with an advanced stage of a much older disease.
This is where the concept of the cardiometabolic continuum fundamentally changes clinical thinking. It shifts our focus from reactive disease management to proactive risk identification. In my view, this means moving beyond glucose-centric models and incorporating earlier, more sensitive markers—such as fasting insulin, ApoB, and inflammatory biomarkers—into routine clinical practice, particularly for high-risk individuals.
Equally important is the recognition that lifestyle interventions are not merely preventive—they are therapeutic at every stage of the continuum. Structured exercise, nutritional optimization, sleep regulation, and weight management are not adjuncts to pharmacology; they are foundational treatments with measurable effects on insulin sensitivity, vascular function, and long-term outcomes.
From a clinical standpoint, the future of cardiometabolic care lies in integration. The artificial divide between metabolic and cardiovascular disease must be replaced with a unified, patient-centered approach. Early identification, combined with targeted lifestyle and pharmacological strategies, offers a genuine opportunity to alter disease trajectories—before irreversible vascular damage occurs.
Ultimately, the goal is simple but powerful: detect earlier, intervene sooner, and prevent what we currently treat too late.
Share this article with someone who has been told their blood sugar is "borderline" or their cholesterol is "a little high." Understanding the connection could change the trajectory of their health.
Comment below: Have you or someone you know been diagnosed with both diabetes and heart disease? What was your experience with the healthcare system's approach to treating them together (or separately)?
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
The Cardiometabolic Cascade: How Disease Starts—and How to Reverse It (2026 Science) | DR T S DIDWAL
Bone as an Endocrine Organ: Does Osteocalcin Influence Weight Regulation? | DR T S DIDWAL
The Metabolic Engine: Why Lower Body Strength Is Central to Fat Oxidation | DR T S DIDWAL
References
Abel, E. D., Ahima, R. S., Anderson, E. J., Berg, D. D., Berger, J. S., Das, S., Feinberg, M. W., Fisher, E. A., Garshick, M. S., Giannarelli, C., Goldberg, I. J., Hamburg, N. M., Kim, S. F., Moura, F. A., Ndumele, C. E., Newman, J. D., Sabatine, M. S., Selvin, E., & Shah, R. (2026). A road map to understanding cardiovascular disease in diabetes: From the AHA Strategically Focused Research Network in cardiometabolic health and type 2 diabetes. Circulation Research, 138(2), e325798. https://doi.org/10.1161/CIRCRESAHA.125.325798
Armentaro, G., & Daidone, M. (2026). Editorial: The cardiovascular continuum between hypertension, diabetes and cardiovascular disease. Frontiers in Cardiovascular Medicine, 13, 1813448. https://doi.org/10.3389/fcvm.2026.1813448
Cassataro, G., Geraci, G., Giusti, M. A., Maida, C., Maggio, V., Rizzo, M., & Mattina, A. (2026). The cardiovascular burden of diabetes: Risk factors, clinical phenotypes, and personalized cardiometabolic management. Journal of Clinical Medicine, 15(6), 2358. https://doi.org/10.3390/jcm15062358
Caturano, A., Morciano, C., Zielińska, K., Russo, V., Perrone, M. A., Berra, C. C., & Conte, C. (2025). Rethinking the diabetes-cardiovascular disease continuum: Toward integrated care. Journal of Clinical Medicine, 14(18), 6678. https://doi.org/10.3390/jcm14186678