The Cardiometabolic Cascade: How Disease Starts—and How to Reverse It (2026 Science)
Can cardiometabolic disease be reversed? Discover the latest 2026 science on diabetes remission, GLP-1 drugs, inflammation, and how to reduce heart risk naturally and medically.
HEARTMETABOLISM
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/24/202616 min read
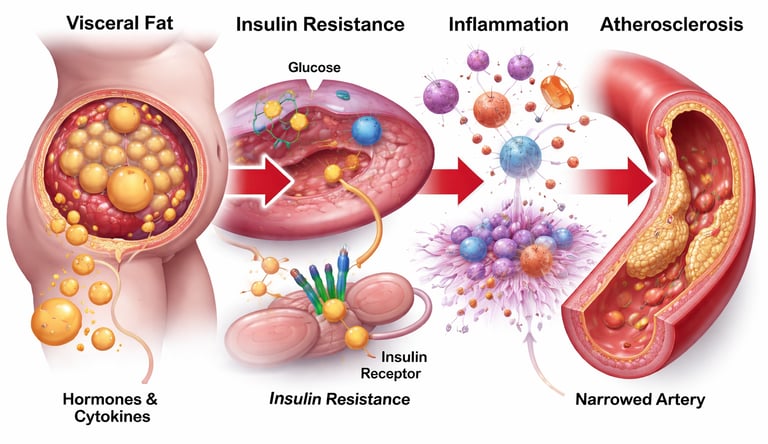
Cardiometabolic disease is a cluster of conditions—including diabetes, obesity, and heart disease—driven by shared mechanisms such as insulin resistance, chronic inflammation, and metabolic dysfunction. Emerging evidence shows that early-stage disease can often be reversed through targeted lifestyle changes and modern therapies, especially when intervention occurs before irreversible vascular damage develops.
Cardiometabolic disease does not begin with a heart attack, a diabetes diagnosis, or a number on a lab report. It begins silently—years, often decades earlier—at the level of cellular metabolism. Long before glucose rises or arteries narrow, a complex biological network starts to shift. Muscle cells become resistant to insulin. Fat tissue transforms into an inflammatory organ. Mitochondria lose efficiency. Vascular function begins to deteriorate. What emerges is not a single disease, but a systems-level failure of metabolic regulation.
This is the essence of the modern cardiometabolic epidemic: a deeply interconnected pathology driven by shared mechanisms rather than isolated risk factors. Increasingly, research shows that conditions such as type 2 diabetes, atherosclerosis, fatty liver disease, and hypertension are not separate entities, but different clinical expressions of the same underlying biological disturbance—centered around insulin resistance, chronic inflammation, and energy surplus (Khanna et al., 2026; Yang et al., 2026).
Recent work has fundamentally reshaped how we understand this process. Rather than viewing disease progression as linear, evidence now supports a network model, where disruptions in one system amplify dysfunction in others. For example, excess visceral fat not only worsens insulin resistance but also drives inflammatory signaling that accelerates vascular damage. Simultaneously, mitochondrial dysfunction reduces metabolic flexibility, further compounding energy imbalance and cellular stress (Ren et al., 2025).
Crucially, these processes begin early—and are, at least initially, reversible. Advances in molecular biomarkers now allow detection of cardiometabolic dysfunction at a stage when traditional diagnostics still appear normal, opening a window for timely intervention (Macvanin & Klisic, 2026).
Understanding this unified pathophysiology is not just an academic exercise. It is the key to moving beyond symptom management toward true disease modification—targeting root causes rather than downstream consequences
Clinical pearls
1. The "Muscle as Medicine" Principle
From a scientific view, skeletal muscle is your body’s largest glucose sink. When you perform resistance training, you aren't just building "vanity" muscle; you are expanding your metabolic reservoir.
The Pearl: Think of your muscles as a specialized sponge for blood sugar. Every bit of lean mass you maintain or build makes it easier for your body to manage a carbohydrate load, even without an increase in insulin.
2. The Postprandial Power Walk
Biology dictates that blood glucose peaks roughly 30 to 60 minutes after a meal. A short bout of movement during this window utilizes "GLUT4 translocation"—a process where muscles pull sugar from the blood without needing a signal from the pancreas.
The Pearl: A 15-minute walk after dinner is metabolically worth an hour of exercise before dinner. It "blunts" the sugar spike, protecting your blood vessels from the oxidative stress that causes long-term damage.
3. Visceral Fat vs. Subcutaneous Fat
Not all fat is created equal. Subcutaneous fat (under the skin) is largely an energy storage depot. Visceral fat (around the organs), however, acts like an independent, pro-inflammatory endocrine organ, secreting "cytokines" that drive systemic insulin resistance.
The Pearl: Your waist-to-height ratio is often more predictive of health than your weight. Focusing on "shrinking the middle" is not about aesthetics; it is about "turning off" a chemical factory that is actively working against your heart and liver.
4. The "Lag Time" of Biomarkers
Chronic diseases like cardiometabolic syndrome develop over decades, but standard blood tests (like Fasting Glucose) are often the last markers to break. Scientific models now look at "upstream" signals like Fasting Insulin or hs-CRP (inflammation) to catch the "smolder" before the "fire."
The Pearl: A "normal" blood sugar reading doesn't always mean perfect health; it might just mean your body is working overtime to keep it there. Asking for an insulin or inflammation check can provide a 10-year "early warning" for your metabolic health.
5. Circadian Metabolic Rhythm
Our metabolism is not a flat line; it follows a clock. Insulin sensitivity is generally higher in the morning and lower in the evening. Processing a heavy meal at 9:00 PM requires significantly more effort from the body than the same meal at 1:00 PM.
The Pearl: Eat with the sun. By front-loading your calories earlier in the day and closing the "kitchen" early, you align your nutrition with your body’s natural hormonal peaks, allowing for better repair and fat oxidation during sleep.
Why Cardiometabolic Disease Is So Hard to Catch Early
One of the biggest challenges with cardiometabolic disease is that it develops silently — over years, sometimes decades — before obvious symptoms appear. By the time most people are diagnosed, significant damage has already accumulated.
This is why the science of molecular biomarkers has become so important. Biomarkers are measurable signals in your blood, urine, or tissues that indicate what's happening inside your body long before symptoms surface.
Macvanin and Klisic (2026), in their editorial published in Frontiers in Endocrinology, highlight the expanding role of molecular biomarkers in cardiometabolic disease. Their work underscores a critical shift in medicine: instead of waiting for disease to fully manifest, scientists are working to detect subtle molecular changes early — in the inflammation pathways, in lipid metabolism, in hormonal signaling — so that intervention can happen at a point when reversal is far more achievable.
Some of the most exciting biomarkers currently being studied include:
High-sensitivity C-reactive protein (hsCRP) — a marker of low-grade systemic inflammation linked to cardiovascular risk
Adiponectin — a hormone secreted by fat cells; low levels signal metabolic dysfunction
Trimethylamine N-oxide (TMAO) — a gut-derived compound associated with plaque buildup in arteries
MicroRNAs — tiny RNA molecules that regulate gene expression and can indicate early cellular stress in the heart and liver
Fibroblast growth factor 21 (FGF21) — a hormonal signal tied to fat metabolism and insulin sensitivity
The ability to detect these signals early fundamentally changes the reversal equation. The earlier you catch the problem, the more biology is on your side.
The Reversal Question: What Does Science Actually Say?
Let's be direct. "Reversal" in medicine is a loaded word. It does not always mean a complete cure. In the context of cardiometabolic disease, reversal typically means:
Restoring normal blood sugar levels (remission of type 2 diabetes)
Reducing or eliminating the need for medications
Normalizing blood pressure and cholesterol without drugs
Reversing early-stage damage to the arteries, heart, or liver
Dramatically reducing the risk of future heart attack or stroke
And yes — for many people, particularly those in the early to moderate stages of disease, all of the above is achievable.
Pathophysiology Model of Cardiometabolic Disease
Central Driver: Insulin Resistance (IR)
The primary initiating defect in skeletal muscle, liver, and adipose tissue
Leads to hyperinsulinemia, impaired glucose uptake, and increased hepatic gluconeogenesis
Visceral Adiposity as an Endocrine Organ
Excess visceral fat releases pro-inflammatory adipokines (TNF-α, IL-6)
Reduced protective adipokines (e.g., adiponectin)
Drives systemic inflammation and worsens IR
Chronic Low-Grade Inflammation
Activation of innate immune pathways (e.g., NF-κB signaling)
Promotes endothelial dysfunction, plaque formation, and beta-cell stress
Mitochondrial Dysfunction
Impaired oxidative phosphorylation and fatty acid oxidation
Increased reactive oxygen species (ROS) production
Contributes to metabolic inflexibility and cellular damage
mTOR–AMPK Imbalance
Overactivation of mTORC1 (nutrient excess, leucine signaling) → anabolic resistance, impaired autophagy
Suppressed AMPK (energy sensing pathway) → reduced fatty acid oxidation and glucose uptake
Endothelial Dysfunction
Reduced nitric oxide (NO) bioavailability
Increased vascular stiffness and impaired vasodilation
Early step in atherogenesis
Lipotoxicity & Ectopic Fat Deposition
Accumulation of toxic lipid intermediates (DAGs, ceramides) in the liver, muscle, and pancreas
Drives insulin resistance and beta-cell dysfunction
Gut Microbiome Dysbiosis
Increased gut permeability (“leaky gut”)
Elevated metabolites like TMAO contribute to atherosclerosis
Modulates systemic inflammation and metabolism
Neurohormonal Dysregulation
Chronic activation of the sympathetic nervous system and RAAS
Elevates blood pressure, worsens insulin resistance
Progressive Beta-Cell Dysfunction
Compensatory hyperinsulinemia → eventual beta-cell exhaustion
Transition from insulin resistance → overt type 2 diabetes
Cardiometabolic disease is not a collection of isolated conditions but a network disorder driven by energy surplus
The Stages Matter: What Research Tells Us About Risk and Timing
A critical piece of the puzzle comes from a 2026 systematic review and meta-analysis published in Ageing Research Reviews by Yang et al. Their study examined stage-specific risk factors of cardiometabolic multimorbidity — meaning they looked at different risk factors depending on whether a patient was at the stage of disease onset (incidence), disease progression, or death (mortality).
This research matters enormously because it reveals something counterintuitive: the risk factors driving disease onset are not the same as those driving disease progression or death. This means that what reverses early-stage disease may be different from what slows advanced disease.
Key takeaways from Yang et al. (2026) include:
At the incidence stage, modifiable lifestyle factors — diet, exercise, smoking, sleep — are the dominant risk drivers and the most powerful targets for reversal
At the progression stage, systemic inflammation and metabolic dysfunction become more prominent, requiring both lifestyle change and targeted medical intervention
At the mortality stage, cardiovascular complications, kidney damage, and multiorgan involvement dominate, where reversal is more limited but management can still dramatically extend life and quality
This stage-specific framework is a game-changer for how patients and doctors should be thinking about treatment. It's not "one strategy fits all." It's about meeting the biology where it is right now — and choosing the right tools accordingly.
Emerging Therapies: What's Changing the Game in 2025–2026
This is where things get genuinely exciting. The therapeutic landscape for cardiometabolic disease has transformed dramatically in recent years. It is no longer just about statins and metformin, though these remain important.
Ren, Chen, and Wang (2025), writing in the British Journal of Pharmacology, outline the emerging therapeutic frontiers in cardiometabolic and inflammatory diseases. Their editorial identifies several key directions that are reshaping how doctors treat — and potentially reverse — these conditions.
Here are the most important emerging areas:
1. GLP-1 Receptor Agonists (Semaglutide, Tirzepatide) Originally developed for type 2 diabetes, drugs like semaglutide (Ozempic/Wegovy) and tirzepatide (Mounjaro/Zepbound) have demonstrated remarkable results not just for blood sugar control but for weight loss, blood pressure reduction, and cardiovascular event prevention. Clinical trials have shown that these agents reduce the risk of major adverse cardiovascular events even in people without diabetes.
2. SGLT-2 Inhibitors (Empagliflozin, Dapagliflozin) These medications were initially diabetes drugs but are now approved for heart failure and chronic kidney disease as well. They work by causing the kidneys to excrete excess glucose, while simultaneously reducing inflammation, lowering blood pressure, and protecting the heart and kidneys through mechanisms still being fully understood.
3. Anti-Inflammatory Therapies Because cardiometabolic disease is fundamentally an inflammatory condition, targeted anti-inflammatory treatments are showing promise. Colchicine, originally used for gout, has demonstrated cardiovascular benefits in large trials. Researchers are also exploring IL-6 inhibitors and other biologics to tame the chronic low-grade inflammation that drives plaque formation and insulin resistance.
4. Gut Microbiome Interventions The gut microbiome — the trillions of bacteria living in your intestines — plays a profound role in metabolic health. Disrupted gut bacteria (dysbiosis) contribute to insulin resistance, systemic inflammation, and even cardiovascular disease. Emerging therapies targeting the microbiome through dietary interventions, probiotics, prebiotics, and fecal microbiota transplant (FMT) are showing early but promising results.
5. Precision Medicine and Genomic Approaches. Not everyone responds to the same treatment. Precision medicine uses genetic, biomarker, and lifestyle data to tailor therapy to the individual. This approach is increasingly being applied to cardiometabolic disease to identify who will respond best to which treatment — dramatically improving outcomes.
6. RNA-Based Therapeutics Small interfering RNA (siRNA) drugs — like inclisiran, which silences a gene responsible for excess LDL cholesterol production — represent a new frontier. These can lower LDL by up to 50% with just two injections per year, completely changing medication adherence for high-risk patients.
Practical Applications: What You Can Do Right Now
Science is clear that lifestyle change remains the most powerful lever for reversing early-to-moderate cardiometabolic disease — more powerful, in many cases, than medication alone. Here's what the evidence supports:
Dietary Changes
Shift toward a whole-food, plant-forward diet rich in vegetables, legumes, nuts, whole grains, and olive oil
Dramatically reduce ultra-processed foods, added sugars, refined carbohydrates, and trans fats
Consider time-restricted eating (intermittent fasting) — evidence supports its role in improving insulin sensitivity and reducing visceral fat
Reduce sodium intake to support blood pressure control
Increase fiber intake — soluble fiber feeds beneficial gut bacteria and directly lowers LDL cholesterol
Physical Activity
Aim for at least 150 minutes of moderate aerobic activity per week — walking, cycling, swimming
Add resistance training at least twice per week — muscle mass is metabolically active and improves insulin sensitivity significantly
Reduce prolonged sitting — even breaking up sitting time with short walks every 30–45 minutes measurably improves blood sugar regulation
Consider high-intensity interval training (HIIT) if medically cleared — powerful metabolic benefits in shorter time frames
Sleep and Stress
Prioritize 7–9 hours of quality sleep per night — poor sleep directly worsens insulin resistance, blood pressure, and appetite hormones
Practice evidence-based stress reduction: mindfulness meditation, yoga, or cognitive behavioral therapy (CBT) all show measurable improvements in cardiometabolic markers
Address sleep apnea if present — untreated sleep apnea is a major but underdiagnosed driver of cardiovascular risk
Monitoring and Knowing Your Numbers
Know your HbA1c (blood sugar average), LDL, HDL, triglycerides, blood pressure, and waist circumference
Ask your doctor about hsCRP — a key inflammation marker that is inexpensive and highly informative
Track trends, not just snapshots — changes over time tell a richer story than a single reading
Medication Adherence
If prescribed medication, take it as directed — lifestyle change works best in combination with optimized medical therapy, not as a replacement for it
Discuss newer agents like GLP-1 agonists or SGLT-2 inhibitors with your doctor if you haven't already — evidence now strongly supports their use for many patients
The Asia Focus: A Warning and an Opportunity
The findings of Khanna et al. (2026) in JACC: Asia deserve special attention. Asia is not just experiencing an increase in cardiometabolic disease — it is experiencing a structurally different form of it. South Asians, East Asians, and Southeast Asians tend to develop metabolic dysfunction at lower body weight and younger ages than Western populations. The traditional BMI cutoffs used to assess obesity risk are less accurate for many Asian populations.
This means:
Cardiometabolic risk can be significantly elevated even in people who appear lean by standard measures
Abdominal fat (visceral adiposity), even without obvious obesity, is a major risk factor in Asian populations
Genetic predisposition to insulin resistance is higher in certain South Asian populations
The transition from traditional diets to Westernised ultra-processed food diets is happening rapidly across the continent
For patients of South Asian, East Asian, or Southeast Asian descent living anywhere in the world, this research is a reminder to ask your doctor about ethnicity-specific risk thresholds and to not be lulled into a false sense of security by a "normal" BMI.
Quick summary of the "reversal" potential
Weight Loss (Target: 10–15% of Body Weight)
Mechanism: Primarily works by clearing ectopic fat (fat stored in the wrong places) from the liver and pancreas. This restores the organs' ability to regulate insulin and glucose.
Reversal Potential: High. This remains the "gold standard" for achieving Type 2 Diabetes remission and reversing Non-Alcoholic Fatty Liver Disease (NAFLD).
GLP-1 and SGLT-2 Inhibitor Therapies
Mechanism: These "twin-pillar" medications improve systemic insulin signaling and allow the kidneys to excrete excess glucose. They also provide significant "cardioprotection" by reducing the workload on the heart.
Reversal Potential: Moderate to High. While often viewed as "management," these drugs are increasingly shown to move patients back into "normal" metabolic ranges, effectively reversing early-stage dysfunction.
Resistance Training (Strength Training)
Mechanism: Expands your "Glucose Sink." By increasing lean muscle mass, you create more surface area (via GLUT4 translocation) to pull sugar out of the bloodstream, even at rest.
Reversal Potential: High. This is the most effective way to restore metabolic flexibility—the body's ability to switch efficiently between burning carbs and burning fat.
Targeted Anti-Inflammatory Strategies
Mechanism: Focuses on lowering systemic "smolder" (measured by hs-CRP). This stabilises arterial plaques, making them less likely to rupture, and improves the health of the endothelium (the inner lining of blood vessels).
Reversal Potential: Moderate (Risk Reduction). While it may not "delete" existing calcified plaque, it is highly effective at stopping the progression of atherosclerosis and reducing the risk of a primary cardiac event.
Elite Editorial: The Unified Biology of Cardiometabolic Disease
We are no longer dealing with isolated diseases—we are confronting a network disorder.
The traditional siloed view of type 2 diabetes, cardiovascular disease, and obesity is biologically outdated. Contemporary evidence demonstrates that these conditions arise from a shared pathophysiological core—anchored in insulin resistance, inflammatory signaling, and metabolic overload (Khanna et al., 2026).
Insulin resistance is not just a metabolic defect—it is the central organizing pathology.
It precedes hyperglycemia by years and drives a cascade of compensatory hyperinsulinemia, lipid dysregulation, and endothelial dysfunction. By the time glucose rises, vascular injury is often already underway (Yang et al., 2026).Adipose tissue has emerged as a key endocrine and immunologic organ.
Visceral fat is not inert storage—it actively secretes cytokines and adipokines that propagate systemic inflammation, disrupt insulin signaling, and accelerate atherogenesis.Mitochondrial dysfunction reframes the disease as one of energy failure.
Impaired oxidative capacity and excess reactive oxygen species create a state of metabolic inflexibility, linking nutrient excess directly to cellular stress and organ dysfunction (Ren et al., 2025).The mTOR–AMPK axis represents a critical therapeutic frontier.
Chronic nutrient abundance drives mTOR overactivation and suppresses AMPK, impairing autophagy and metabolic balance—mechanisms increasingly implicated in both metabolic and cardiovascular pathology.Endothelial dysfunction is the earliest clinically relevant manifestation.
Reduced nitric oxide bioavailability and vascular stiffness precede overt atherosclerosis, offering a window for early intervention before irreversible damage occurs.Stage-specific biology demands stage-specific intervention.
Lifestyle modification dominates early disease reversal, while advanced stages require integration of pharmacotherapy targeting inflammation, glucose handling, and cardiovascular risk (Yang et al., 2026).The clinical paradigm must shift from treatment to interception.
With the rise of molecular biomarkers and precision medicine, the opportunity is no longer just to manage disease—but to identify and reverse it at its biological inception (Macvanin & Klisic, 2026).
Bottom Line: Cardiometabolic disease is not inevitable—it is biologically orchestrated, clinically predictable, and, if addressed early, fundamentally modifiable.
Frequently Asked Questions (FAQs)
1. Can type 2 diabetes actually be reversed?
Yes — for many people, particularly those with relatively recent diagnosis and significant weight loss potential, type 2 diabetes can go into full clinical remission. This means blood sugar returns to normal levels without medication. The most powerful tool for this is sustained weight loss of 10–15% of body weight, combined with dietary change. GLP-1 receptor agonists like semaglutide are also showing remarkable reversal rates in clinical trials. Reversal is most achievable within the first few years of diagnosis.
2. Is cardiometabolic disease reversible if you have heart disease?
Reversal of established heart disease — hardened arteries, scarred heart muscle — is more limited. However, it is very well established that lifestyle changes and medications can slow progression, reduce inflammation, stabilize plaques, and dramatically reduce the risk of future heart attacks. Even in advanced disease, meaningful improvement in quality of life and longevity is achievable. Partial reversal of early arterial plaques has been demonstrated with intensive statin therapy combined with lifestyle change.
3. How long does it take to see measurable improvement in cardiometabolic markers?
Measurable improvements in blood sugar, blood pressure, cholesterol, and inflammatory markers can often be seen within 4–12 weeks of significant lifestyle change. More substantial structural changes — like reduction of visceral fat or liver fat — typically take 3–6 months of sustained effort. Consistency over time is the key variable.
4. What is the most important first step for someone newly diagnosed?
The most important first step is education and engagement. Understand your specific numbers, understand what they mean, and work with your healthcare team to build a personalized plan. Simultaneously, begin with the dietary and physical activity changes described above — these have the fastest impact and create momentum. Don't wait until the situation is more serious to act.
5. Are the new diabetes and obesity drugs (GLP-1s) right for everyone?
Not necessarily. GLP-1 receptor agonists like semaglutide are powerful tools and have strong evidence for cardiometabolic benefit. However, they come with costs, potential side effects (nausea, GI discomfort, rare risks), and are not appropriate for everyone. They work best as part of a comprehensive treatment plan that includes lifestyle change — not as a replacement for it. Speak honestly with your doctor about whether they are appropriate for your specific situation.
6. Can children and young adults develop cardiometabolic disease?
Absolutely — and this is a growing concern. Childhood obesity, early-onset insulin resistance, and even elevated cardiovascular risk markers are increasingly being detected in teenagers and young adults, particularly in high-risk populations. Early intervention is critical and far more effective in younger individuals, whose biology is significantly more responsive to lifestyle change.
7. Does stress really affect cardiometabolic health — or is that just a cliché?
It's not a cliché — it's well-established science. Chronic psychological stress drives cortisol production, which directly raises blood sugar, promotes visceral fat storage, elevates blood pressure, and worsens inflammatory markers. Stress also drives unhealthy coping behaviors (poor diet, alcohol, poor sleep) that compound the damage. Managing stress is not a soft add-on to a treatment plan — it is a core component of cardiometabolic care.
Call to Action: Your Next Steps Start Today
You've read the research. You understand the landscape. Now here's the truth: the window of greatest opportunity is right now — before disease progresses to its next stage.
Whether you've just received a diagnosis, you're worried about your family history, or you simply want to be proactive, here is a clear and realistic engagement strategy to move forward:
Step 1 — Get Your Baseline Numbers Book a comprehensive metabolic panel with your doctor. Ask specifically for: HbA1c, fasting glucose, full lipid panel, hsCRP, liver enzymes, blood pressure, and waist circumference. You cannot manage what you don't measure.
Step 2 — Commit to One Dietary Change This Week Not a complete overhaul — just one meaningful change. Replace your most frequently consumed ultra-processed food with a whole food alternative. Build from there.
Step 3 — Move Every Day Start with 20 minutes of walking after dinner. This single habit has measurable effects on post-meal blood sugar, blood pressure, and stress hormones. It is genuinely one of the most impactful interventions in all of medicine — and it's free.
Step 4 — Have an Honest Conversation With Your Doctor Bring this article if it helps. Ask about your stage-specific risk, your biomarker profile, and whether newer therapeutic options like GLP-1 agonists, SGLT-2 inhibitors, or anti-inflammatory strategies are appropriate for you.
Step 5 — Share This Information Cardiometabolic disease runs in families — and so do environments, diets, and habits. Share what you've learned with someone you care about. The conversation you have today might be the most important health intervention in someone's life.
Author’s Note (Clinician’s Perspective)
As a clinician working at the intersection of internal medicine and metabolic health, I have witnessed firsthand how cardiometabolic disease rarely presents as a single diagnosis. Patients do not walk in with “just diabetes” or “just hypertension.” What we see, more often than not, is a clustered biology—central obesity, rising glucose, dyslipidemia, fatty liver, and early vascular dysfunction—unfolding along a shared pathophysiological continuum.
What has changed in recent years is not merely our ability to treat these conditions, but our understanding of why they occur. The emerging evidence consistently points toward a unifying framework: insulin resistance, chronic low-grade inflammation, and energy dysregulation acting in concert across multiple organ systems (Khanna et al., 2026; Yang et al., 2026). This shift—from viewing disease in isolation to recognizing it as a network disorder—has profound clinical implications.
In practice, this means moving beyond symptom control toward early identification and targeted intervention. It means recognizing that a “normal” glucose level does not exclude metabolic dysfunction, that visceral adiposity carries risks independent of body weight, and that vascular injury often begins long before clinical disease becomes apparent. It also means embracing a more integrated therapeutic approach—combining lifestyle strategies with evidence-based pharmacology, including newer agents that address not just glucose, but cardiovascular and inflammatory pathways (Ren et al., 2025).
Perhaps most importantly, it reinforces a message I emphasize to every patient: timing matters. The earlier we intervene, the more reversible the biology. Waiting for overt disease is, in many cases, waiting too long.
This chapter is written with that urgency in mind—not to alarm, but to inform. Because when patients and clinicians understand the underlying biology, they are far better equipped to change its trajectory.
This article is intended for educational purposes and does not constitute medical advice. Always consult a qualified healthcare professional for diagnosis and treatment decisions.
Related Articles
Why Fat Tissue Is an Endocrine Organ — And What It Means for Your Health | DR T S DIDWAL
Tired All Day, Awake at Night? The Redox and Mitochondrial Link | DR T S DIDWAL
Obesity and Fatty Liver Disease: What Science Says About Risk and Health | DR T S DIDWAL
Intermittent Fasting: Metabolic Health Benefits and the Evidence on Longevity | DR T S DIDWAL
Activate Your Brown Fat: A New Pathway to Longevity and Metabolic Health | DR T S DIDWAL
References
Caturano, A. (2025). Cardiovascular and Metabolic Disease: New Treatment and Future Directions — The 3rd Edition. Biomedicines, 13(8), 1914. https://doi.org/10.3390/biomedicines13081914
Khanna, S., Gan, G. C. H., Sindone, A. P., Tromp, J., Butler, J., Foo, R., Nerlekar, N., & Bhat, A. (2026). Asia at the epicenter of the global cardiometabolic shift. JACC: Asia, 6(3), 269–283. https://doi.org/10.1016/j.jacasi.2026.01.001
Macvanin, M., & Klisic, A. (2026). Editorial: Molecular biomarkers of cardiometabolic disease — Volume II. Frontiers in Endocrinology, 17, 1792935. https://doi.org/10.3389/fendo.2026.1792935
Ren, J., Chen, X., & Wang, X. (2025). Emerging therapeutic frontiers in cardiometabolic and inflammatory diseases. British Journal of Pharmacology, 182(20), 4737–4740. https://doi.org/10.1111/bph.70159
Yang, L., Zhang, Z., Zhang, J., Miao, J., Zhang, H., Du, Y., & Ding, L. (2026). Stage-specific risk factors of cardiometabolic multimorbidity: A systematic review and meta-analysis from incidence to mortality. Ageing Research Reviews, 114, 102991. https://doi.org/10.1016/j.arr.2025.102991