GLP-1 Receptor Agonists and Cardiovascular Protection: Evidence from CVOT Trials Explained
Learn how GLP-1 receptor agonists lower cardiovascular risk through major clinical trials and real-world outcome
DIABETESHEART
Dr. T.S. Didwal, M.D.(Internal Medicine)
4/7/202615 min read
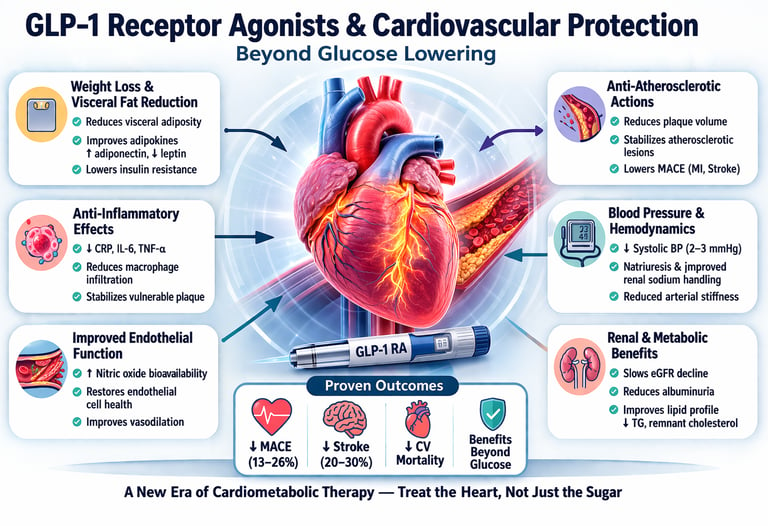
GLP-1 receptor agonists reduce cardiovascular risk not only by lowering blood glucose but by targeting the underlying drivers of heart disease, including visceral obesity, chronic inflammation, endothelial dysfunction, and atherosclerosis. Large cardiovascular outcome trials have shown significant reductions in major adverse cardiovascular events (MACE), particularly myocardial infarction and stroke. These benefits occur independently of HbA1c reduction, positioning GLP-1 receptor agonists as essential cardiometabolic therapies rather than simply glucose-lowering drugs.
Clinician’s Perspective: GLP-1 Receptor Agonists and the New Era of Cardiometabolic Medicine
We are no longer treating glucose — we are treating risk.
The traditional glucocentric model has failed to meaningfully reduce cardiovascular events despite achieving HbA1c targets. The shift toward cardiometabolic risk modification reflects a deeper understanding of disease biology—where insulin resistance, inflammation, and atherogenesis intersect.GLP-1 receptor agonists represent a therapeutic inflection point.
What distinguishes this class is not simply efficacy, but mechanistic breadth. These agents simultaneously target weight, endothelial dysfunction, inflammatory pathways, and lipid abnormalities—domains previously managed in isolation.Cardiovascular outcome trials have redefined prescribing priorities.
The consistent reduction in major adverse cardiovascular events (MACE) across trials has elevated GLP-1 RAs from second-line glucose-lowering drugs to first-line cardioprotective therapies in high-risk patients—independent of baseline HbA1c.Not all benefits are equal—and that matters clinically.
GLP-1 RAs predominantly reduce atherosclerotic events (MI, stroke), while SGLT2 inhibitors excel in heart failure and renal protection. Recognizing this divergence enables precision pharmacotherapy, rather than class-based prescribing.The most appropriate patient is not defined by glucose alone.
The ideal candidate is a patient with visceral adiposity, insulin resistance, and elevated atherosclerotic risk—even when glycemic markers appear controlled. This represents a fundamental shift in patient selection.Combination therapy is the future, not the exception.
The integration of GLP-1 RAs with SGLT2 inhibitors, statins, and lifestyle interventions reflects a multi-axis strategy targeting the full spectrum of cardiometabolic disease.Barriers remain—cost, access, and tolerability.
Despite robust evidence, real-world implementation is constrained by financial toxicity and gastrointestinal side effects. Advocacy and patient education are now part of clinical responsibility.We are entering the era of metabolic cardiology.
GLP-1 RAs are not the endpoint—they are the beginning. With dual and triple agonists emerging, the future lies in targeted, mechanism-driven therapy that addresses the root causes of cardiovascular disease, not just its biochemical markers.
GLP-1 Receptor Agonists and Cardiovascular Protection: Beyond Glucose Lowering
Cardiometabolic medicine is undergoing a profound transformation. For decades, the management of type 2 diabetes mellitus (T2DM) revolved around a singular objective: lowering blood glucose. Glycated haemoglobin (HbA1c) became the central metric of success, guiding therapeutic decisions and defining clinical outcomes. Yet an uncomfortable paradox persisted—patients achieved “target” glycemic control while continuing to suffer myocardial infarctions, strokes, and premature cardiovascular death. The limitation was not in the effort, but in the framework itself. Glucose control alone was never sufficient to address the complex pathophysiology linking insulin resistance, visceral adiposity, inflammation, and atherosclerosis (Rosen & Ingelfinger, 2026).
Consider a typical patient: a 58-year-old individual with T2DM, an HbA1c of 7.1%, and no overt symptoms. On paper, the disease appears controlled. Beneath the surface, however, lies a different reality—endothelial dysfunction, elevated remnant lipoproteins, chronic low-grade inflammation, and progressive plaque formation within coronary arteries. This disconnect between glycemic metrics and cardiovascular risk has driven a paradigm shift from glucocentric care to comprehensive cardiometabolic risk reduction (Nauck, 2025).
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) have emerged at the center of this shift. Initially developed as glucose-lowering agents, they are now recognized as multi-system therapies capable of modifying the underlying drivers of cardiovascular disease. Large-scale cardiovascular outcome trials and meta-analyses demonstrate consistent reductions in major adverse cardiovascular events (MACE), effects that cannot be explained by glycemic control alone (Galli et al., 2025; Kong et al., 2026). Instead, GLP-1 RAs act through a convergence of mechanisms—weight reduction, anti-inflammatory signalling, endothelial restoration, and direct anti-atherosclerotic effects—that redefines how clinicians approach both diabetes and cardiovascular prevention.
1. The Incretin System: A Quick Refresher
Glucagon-like peptide-1 (GLP-1) is a hormone secreted by L-cells in the distal small intestine and colon in response to food. Its primary role is to stimulate insulin secretion in a glucose-dependent fashion — a crucial safety feature that prevents hypoglycemia. However, native GLP-1 has a half-life of only 1–2 minutes, rapidly degraded by the enzyme dipeptidyl peptidase-4 (DPP-4).
What makes GLP-1 RAs clinically transformative is where their receptors are found. GLP-1 receptors are not just in the pancreas — they are expressed throughout the body: in the brain (appetite regulation), heart (myocardial protection), vasculature (endothelial function), and kidney (sodium handling).1 This widespread receptor distribution explains why GLP-1 RAs produce effects that cascade across multiple organ systems simultaneously.
2. Pharmacology: Not All GLP-1 RAs Are Equal
GLP-1 receptor agonists are structurally engineered versions of native GLP-1, modified to resist DPP-4 degradation. Key pharmacological distinctions include:
Short-acting agents (e.g., exenatide twice daily): Greater impact on postprandial glucose; act largely on gastric emptying.
Long-acting agents (e.g., semaglutide weekly, liraglutide daily): Continuous receptor activation; greater effects on fasting glucose, weight, and cardiovascular outcomes.
Oral semaglutide: The first oral GLP-1 RA, expanding accessibility beyond injectable formulations.
The degree of cardiovascular protection varies by agent and cannot be assumed to be a pure class effect — a nuance that Westermeier and Fisman (2025) thoroughly examine in their historical and mechanistic review of GLP-1 RA cardiometabolic protection.
3. Mechanisms of Cardiovascular Protection
The cardiovascular benefits of GLP-1 RAs are not reducible to weight loss alone. A 2025 systematic review and meta-analysis of 99,599 patients published in the Journal of the American College of Cardiology (Galli et al.) confirms multiple parallel pathways operating simultaneously.
Visceral fat reduction & adipokine remodeling: Sustained weight loss, particularly of metabolically active visceral adipose tissue, reduces pro-inflammatory adipokines, leptin excess, and adiponectin deficiency.
Anti-atherosclerotic effects: GLP-1 RAs reduce macrophage infiltration into atherosclerotic plaques, decrease foam cell formation, and promote plaque stabilization — directly reducing rupture risk.4
Endothelial function: Improved nitric oxide bioavailability and reduced oxidative stress restore endothelial health — the vascular lining that regulates tone, inflammation, and thrombosis.
Anti-inflammatory actions: Reductions in CRP, IL-6, and TNF-α attenuate chronic low-grade inflammation, a root driver of both atherosclerosis and insulin resistance.
Lipid metabolism: Significant triglyceride reduction and improvements in remnant cholesterol; modest LDL changes but improved particle quality and reduced postprandial lipemia.
Blood pressure & natriuresis: Mild but consistent reductions in systolic blood pressure (2–3 mmHg), partly via renal sodium handling — additive to lifestyle modification.1
Direct myocardial effects: Improved myocardial glucose uptake and reduced ischemia-reperfusion injury signal a direct cardioprotective role independent of systemic metabolic effects.5
5 . GLP-1 Cardiovascular Outcome Trials (CVOTs): Evidence That Changed Clinical Practice
Since the 2008 FDA mandate for cardiovascular outcome trials (CVOTs), GLP-1 receptor agonists have built one of the most robust evidence bases in modern cardiometabolic medicine. These large-scale trials were designed not just to prove safety, but to determine whether glucose-lowering therapies could reduce cardiovascular events—and GLP-1 RAs have consistently delivered.
A 2026 umbrella review in Nature Communications (Kong et al.) confirmed a class-wide reduction in major adverse cardiovascular events (MACE), reinforcing their role as cardioprotective agents beyond glycemic control.
Key GLP-1 CVOTs and Their Clinical Impact
LEADER (Liraglutide)
Demonstrated a 13% reduction in MACE along with a significant reduction in cardiovascular mortality, establishing GLP-1 RAs as effective therapies in high-risk T2DM patients.SUSTAIN-6 (Semaglutide)
Showed a 26% reduction in MACE, with particularly strong effects on stroke reduction and nonfatal myocardial infarction.REWIND (Dulaglutide)
A landmark trial extending benefits to primary prevention populations, including patients without established cardiovascular disease, highlights broader clinical applicability.EXSCEL (Exenatide Extended-Release)
Demonstrated cardiovascular safety (non-inferiority) but not superiority, suggesting that cardiovascular benefits may vary between agents.
What Do Meta-Analyses Show?
Large meta-analyses, including Galli et al. (2025), confirm that GLP-1 receptor agonists primarily reduce:
Atherosclerotic events (myocardial infarction and stroke)
Major adverse cardiovascular events (MACE)
However, their effect on:
Heart failure hospitalization is modest
Compared to SGLT2 inhibitors, which remain superior for HF and renal outcomes
Clinical Insight: Why REWIND Matters
The REWIND trial is particularly important for clinicians because it included patients with lower baseline cardiovascular risk. This suggests that GLP-1 receptor agonists are not limited to secondary prevention—they may also play a role in early cardiovascular risk reduction, even before overt disease develops.
The "Power Couple" of Cardiometabolic Health
While both drug classes are used to manage Type 2 Diabetes, they protect the body through entirely different—and complementary—biological pathways.
GLP-1 Receptor Agonists (The "Vascular Shield")
Primary Benefit: Targets Atherosclerotic Cardiovascular Disease (ASCVD).
Heart Protection: Reduces Major Adverse Cardiovascular Events (MACE), specifically heart attack and stroke.
Core Mechanism: Acts as an anti-inflammatory and anti-atherosclerotic agent; it stabilizes arterial plaques and improves the health of the blood vessel lining.
Weight Impact: High potency for weight loss (averaging 5–15%+), targeting metabolic visceral fat.
Blood Pressure: Provides a mild reduction, typically 2–3 mmHg.
Renal Status: Emerging evidence shows significant protection against protein in the urine (proteinuria).
SGLT2 Inhibitors (The "Fluid & Filter Guard")
Primary Benefit: Targets Heart Failure (HF) and Chronic Kidney Disease (CKD).
Heart Protection: Dramatically reduces hospitalizations for heart failure and improves outcomes for patients with reduced or preserved ejection fraction.
Core Mechanism: Acts as a "smart diuretic" (natriuresis); it reduces the heart's workload (preload/afterload) by removing excess salt and water through the kidneys.
Weight Impact: Modest weight loss (averaging 2–3%) primarily through calorie loss in the urine.
Blood Pressure: Significant moderate reduction due to decreased fluid volume.
Renal Status: Currently the "gold standard" for slowing the progression of kidney disease (established for eGFR ≥ 20).
Why "Both" is Often the Best Answer
The most recent clinical guidelines (2025-2026) emphasize that these medications are complementary, not competitive.
Dual-Pathway Protection: Using both agents provides a "belt and braces" approach—one protects the "pipes" (arteries) from clogging while the other protects the "pump" (heart) and "filters" (kidneys).
Additive Benefits: Combining them leads to superior blood pressure control, greater weight loss, and more robust protection against the "cardio-renal-metabolic" syndrome.
Standard of Care: For high-risk patients with both heart disease and kidney concerns, the combination is now strongly recommended as the foundational therapy regardless of the patient's starting A1c.Nauck (2025) in The Lancet provides a framework for next-generation incretin therapy that includes dual agonism — using the complementary mechanisms of these drug classes to achieve comprehensive cardiometabolic risk reduction.2
6. Beyond Diabetes: Expanding Indications
Perhaps the most exciting development is the recognition that GLP-1 RAs benefit patients regardless of diabetes status. Rosen and Ingelfinger's 2026 review in the New England Journal of Medicine outlines the expanding therapeutic frontier.1
6.1 Obesity Without Diabetes
The SELECT trial demonstrated that semaglutide 2.4 mg reduced MACE by 20% in non-diabetic obese individuals with established cardiovascular disease — a landmark finding that repositions GLP-1 RAs as primary cardiometabolic agents divorced from glycemic indication.
6.2 Non-Alcoholic Fatty Liver Disease (NAFLD)
GLP-1 RAs reduce hepatic fat content and liver inflammation, with semaglutide showing histological improvement in early trials for NASH (non-alcoholic steatohepatitis). The liver is emerging as a key therapeutic target.
6.3 Chronic Kidney Disease
Emerging renal outcome data suggest GLP-1 RAs may attenuate proteinuria and preserve eGFR decline, complementing SGLT2 inhibitors in the diabetic kidney disease population.3
6.4 Dual and Triple Agonists: The Next Frontier
Tirzepatide (GLP-1/GIP dual agonist) has demonstrated superior weight loss and glycemic control compared to semaglutide alone. GLP-1/glucagon co-agonists are in development for NASH and metabolic syndrome. The era of precision cardiometabolic polypharmacy is beginning.2
7. Practical Clinical Applications: Turning Evidence into Real-World Results
Translating the latest evidence on atherogenic dyslipidemia into everyday practice requires a structured, patient-centered framework. GLP-1 receptor agonists (GLP-1 RAs) stand out as a powerful tool because they simultaneously improve glycemic control, promote weight loss, reduce insulin resistance, and favorably impact the metabolic triad (high triglycerides, low HDL, and small dense LDL particles).
Ideal Patient Profile for GLP-1 RA Therapy
Consider GLP-1 RAs in patients with type 2 diabetes (T2DM) who also have:
Established atherosclerotic cardiovascular disease (**ASCVD**) or high CV risk
BMI >27 kg/m²
Features of atherogenic dyslipidemia (elevated triglycerides, low HDL-C, or high remnant cholesterol)
The REWIND trial with dulaglutide supports this approach even in primary prevention, showing cardiovascular benefit in a broad population with T2DM and CV risk factors (many without prior events).
First-Line or Add-On Strategy
In patients with T2DM + ASCVD: Use a GLP-1 RA with proven CV benefit independent of HbA1c. It can be added to metformin or used as monotherapy in metformin-intolerant individuals.
GLP-1 RAs help address the root drivers of atherogenic dyslipidemia by lowering triglycerides, improving the TG/HDL ratio, and reducing remnant cholesterol through weight loss and enhanced insulin sensitivity.
Smart Combination Therapy
Combine therapies based on dominant risks:
High-risk heart failure (HF) or chronic kidney disease (CKD): Prioritise or add an SGLT2 inhibitor (strong cardiorenal protection).
All patients: Optimise statin therapy and follow the ABCS framework — A1C control, Blood pressure management, cholesterol optimisation (including ApoB/non-HDL/remnant-C), and Smoking cessation
Emerging data suggest GLP-1 RA + SGLT2i combination may offer additive benefits for reducing major adverse cardiovascular events in high-risk T2DM patients.
Recommended Monitoring Protocol
HbA1c: Every 3 months initially, then every 6 months once stable.
Weight: At every visit (expect meaningful loss that further improves the lipid triad).
Renal function: eGFR and urine albumin-to-creatinine ratio (ACR) every 6–12 months.
Lipids: Full panel (including calculated remnant cholesterol and non-HDL-C) every 6–12 months to track improvements in triglycerides and atherogenic particles.
Managing GI Side Effects
Start at the lowest dose and titrate slowly.
Advise taking with food.
Nausea is common but usually subsides within 4–8 weeks.
For persistent vomiting, reduce the dose rather than discontinue — most patients tolerate therapy long-term.
Addressing Access and Cost Barriers
Cost and prior authorization remain real-world challenges. Document ASCVD or high CV risk clearly to support approval. Manufacturer patient assistance programs can improve access for eligible individuals. In many cases, the long-term reduction in cardiovascular events and need for other therapies justifies the investment.
Clinical Pearl
In patients with "normal" LDL-C but persistent atherogenic dyslipidemia and insulin resistance, GLP-1 RAs offer benefits that extend far beyond glucose lowering — they directly target the metabolic drivers of residual heart disease risk.
This framework integrates seamlessly with lifestyle interventions (low-refined-carb diet, post-meal walks, omega-3 intake) discussed earlier, creating a comprehensive approach to reversing the metabolic triad and lowering long-term cardiovascular risk.
8. Adverse Effects and Important Cautions
No drug is without risk. Clinicians and patients should be aware of the following:
Gastrointestinal (most common): Nausea, vomiting, diarrhea, and constipation affect up to 30–40% of patients. Usually dose-dependent and transient. Slow titration is the most effective mitigation strategy.
Pancreatitis (rare, controversial): Current evidence does not establish causality; risk appears low. Monitor for abdominal pain and avoid use in patients with a history of pancreatitis.
Gallbladder disease: Rapid weight loss increases bile lithogenicity. Gallstones and cholecystitis are more frequent — screen with imaging if symptoms arise.
Delayed gastric emptying: Clinically relevant when patients undergo general anaesthesia. Advise extended pre-procedural fasting and inform the anaesthetic team.
Thyroid C-cell tumors: Rodent data; no confirmed human signal. Contraindicated in personal/family history of medullary thyroid carcinoma or MEN2.
Faqs
1. Can I use GLP-1 medications if I don’t have diabetes?
Yes. While originally developed for Type 2 Diabetes, several GLP-1 receptor agonists (such as semaglutide 2.4 mg) are FDA-approved specifically for chronic weight management and cardiovascular risk reduction in adults with obesity or overweight. Clinical trials like SELECT have shown these medications reduce the risk of heart attack and stroke even in patients with normal blood sugar levels.
2. How do GLP-1 drugs protect the heart beyond lowering blood sugar?
GLP-1 RAs protect the heart through multifactorial mechanisms including:
Anti-inflammatory Action: Reducing systemic inflammation (CRP and IL-6) that leads to plaque buildup.
Plaque Stabilization: Preventing existing arterial plaques from rupturing.
Endothelial Health: Improving the flexibility and function of blood vessel linings.
Weight & Pressure: Reducing visceral fat and lowering systolic blood pressure.
3. Is semaglutide better than liraglutide for heart protection?
Current evidence suggests that long-acting GLP-1 RAs (like weekly semaglutide or dulaglutide) generally show more robust cardiovascular benefits than shorter-acting versions. In the SUSTAIN-6 trial, semaglutide showed a 26% reduction in major adverse cardiovascular events (MACE), whereas the LEADER trial showed a 13% reduction for liraglutide. However, the "best" choice depends on your specific risk profile and medication tolerance.
4. Can GLP-1 RAs and SGLT2 inhibitors be taken together?
Yes. Recent clinical guidelines often recommend the combination of GLP-1 RAs and SGLT2 inhibitors for high-risk patients. They offer synergistic protection: GLP-1s focus on preventing atherosclerosis (clogged arteries), while SGLT2s focus on preventing heart failure and protecting kidney function. This "dual-pathway" approach provides the most comprehensive cardiometabolic defense available.
5. What are the most common side effects of GLP-1 medications?
The most common side effects are gastrointestinal, including nausea, vomiting, diarrhea, and constipation. These symptoms occur in approximately 30–40% of patients but are usually temporary and dose-dependent. To minimize these effects, doctors typically use a "start low, go slow" titration schedule, gradually increasing the dose over several months.
6. What is the difference between Tirzepatide and Semaglutide?
While semaglutide (Ozempic/Wegovy) is a single agonist targeting the GLP-1 receptor, tirzepatide (Mounjaro/Zepbound) is a dual agonist (GIP and GLP-1). By targeting two metabolic pathways instead of one, tirzepatide generally produces greater weight loss and blood sugar reduction, though both are highly effective for cardiovascular protection.
7. Does insurance cover GLP-1 drugs for heart health?
Coverage varies significantly by provider and plan. Most insurance companies cover GLP-1 RAs for Type 2 Diabetes. Coverage for obesity or cardiovascular prevention is expanding but often requires "prior authorization" from a doctor to prove the patient meets specific criteria, such as a certain BMI or a history of heart disease.
Clinical pearls
1. The "Plaque Stabilizer" Effect
Scientific Perspective: GLP-1 RAs are essentially anti-atherosclerotic agents. They reduce macrophage accumulation and the expression of pro-inflammatory cytokines (IL-6, TNF-alpha) within the vascular wall. This stabilizes the fibrous cap of atherosclerotic plaques, making them less likely to rupture and cause a myocardial infarction.
Think of these drugs like "internal armor" for your blood vessels. Beyond just lowering sugar, they help calm down inflammation in your arteries, making it less likely that existing blockages will break off and cause a heart attack or stroke.
2. Glucose-Dependent Safety
Scientific Perspective: The insulinotropic effect of GLP-1 RAs is glucose-dependent. This means they only stimulate the pancreas to release insulin when blood glucose levels are elevated. As glucose levels normalize, the insulin secretion stimulus diminishes, resulting in a negligible risk of therapy-induced hypoglycemia.
One of the biggest fears with diabetes medicine is "going too low" (hypoglycemia). Because this medication only works when your sugar is actually high, it’s much safer than older treatments and won't cause your blood sugar to crash unexpectedly.
3. The "Twinning" Strategy (Synergy with SGLT2i)
Scientific Perspective: Combining GLP-1 RAs with SGLT2 inhibitors provides dual-pathway protection. GLP-1 RAs target Atherosclerotic Cardiovascular Disease (ASCVD) via anti-inflammatory pathways, while SGLT2 inhibitors target Heart Failure (HF) and Chronic Kidney Disease (CKD) via natriuresis and osmotic diuresis.
If your doctor prescribes two different types of "diabetes" drugs, they aren't just doubling up. One protects the "pipes" (your arteries) from clogging, while the other protects the "pump" (your heart) and the "filters" (your kidneys).
4. Brain-Gut Reset (Not Just "Fullness")
Scientific Perspective: GLP-1 receptors in the hypothalamus and hindbrain modulate reward-based eating and homeostatic hunger. These agents decrease "food noise"—the intrusive, obsessive thoughts about eating—by slowing gastric emptying and signaling the brain's satiety centers.
This isn't just "willpower in a pen." It actually changes how your brain communicates with your stomach. Many people find that the "chatter" in their head about food finally goes quiet, making it easier to make healthy choices without feeling like they are constantly fighting an urge.
5. The "Start Low, Go Slow" Mandate
Scientific Perspective: Gastrointestinal side effects (nausea/vomiting) are the primary reason for discontinuation. These are often transient and related to the rate of dose titration. Initiating at the sub-therapeutic dose (e.g., 0.25 mg semaglutide) allows the enteric nervous system to adapt to the delay in gastric emptying.
You might feel some nausea or a "heavy" stomach for the first few weeks, but this is usually temporary. The secret to success is patience—starting at a very low dose and increasing it slowly gives your body time to adjust, ensuring you get the heart-saving benefits without the side effects.
Author’s Note
This article reflects a fundamental shift in how we understand and treat cardiometabolic disease. For decades, clinical practice—my own included—was anchored in a glucose-centric model, where achieving target HbA1c was considered synonymous with effective diabetes management. Yet, over time, it became increasingly clear that many patients who appeared “controlled” on paper continued to experience cardiovascular events. This disconnect prompted a deeper re-evaluation of the underlying biology of type 2 diabetes and its intimate relationship with atherosclerosis, inflammation, and metabolic dysfunction.
The emergence of GLP-1 receptor agonists has been one of the most important therapeutic developments in this space. What makes these agents remarkable is not merely their ability to lower glucose, but their capacity to influence multiple interconnected pathways that drive cardiovascular disease. In many ways, they exemplify the transition from treating isolated biomarkers to addressing systemic disease processes.
This piece was written with both clinicians and informed patients in mind. For clinicians, the goal is to provide a mechanistic and evidence-based framework that supports more nuanced, risk-driven prescribing decisions. For patients, it aims to clarify why modern therapies may be recommended even when traditional markers like HbA1c appear well controlled.
Medicine is evolving toward precision and integration. As we move forward, the challenge will not only be understanding these therapies, but implementing them thoughtfully—balancing efficacy, safety, accessibility, and individual patient context.
Disclaimer: This article is for educational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider before starting, stopping, or adjusting any medication. Individual risk profiles and treatment decisions must be individualized.
Explore related reads on this site:
Your Cholesterol Isn’t the Whole Story: The Hormone Imbalances Driving Heart Disease
Can GLP-1 Drugs Prevent Heart Attacks? New Trial Data Changes Everything
ApoB vs LDL Cholesterol: Which Is the Most Accurate Predictor of Heart Disease?
Remnant Cholesterol: The Hidden Link Between Insulin Resistance, Type 2 Diabetes, and Heart Disease
Visceral Fat and Cardiovascular Risk: The Hidden Driver of Atherosclerosis
The #1 Diet Strategy to Reduce Visceral Fat According to Latest Research
Why Belly Fat Causes Insulin Resistance: Portal Theory Explained Simply
References
Rosen, C. J., & Ingelfinger, J. R. (2026). GLP-1 receptor agonists. The New England Journal of Medicine, 394(13), 1313–1324. https://doi.org/10.1056/NEJMra2500106
Nauck, M. A. (2025). Glucagon-like peptide-1 receptor agonists and next-generation incretin-based therapies. The Lancet. Advance online publication. https://doi.org/10.1016/S0140-6736(25)02105-1
Kong, F., Zhao, Y., Zhang, W., et al. (2026). Comprehensive evaluation of GLP-1 receptor agonists: An umbrella review of clinical outcomes across multiple diseases. Nature Communications, 17, 972. https://doi.org/10.1038/s41467-025-67701-9
Galli, M., Benenati, S., Laudani, C., Simeone, B., Sarto, G., Ortega-Paz, L., Rocco, E., Bernardi, M., Spadafora, L., D'Amario, D., Greco, E., Frati, G., Federici, M., Mehran, R., Crea, F., Angiolillo, D. J., & Sciarretta, S. (2025). Cardiovascular effects and tolerability of GLP-1 receptor agonists: A systematic review and meta-analysis of 99,599 patients. Journal of the American College of Cardiology. Advance online publication. https://doi.org/10.1016/j.jacc.2025.08.027
Westermeier, F., & Fisman, E. Z. (2025). Glucagon-like peptide-1 receptor agonists (GLP-1RAs) and cardiometabolic protection: Historical development and future challenges. Cardiovascular Diabetology, 24, 44. https://doi.org/10.1186/s12933-025-02608-9