Incretin Therapy Explained: GLP-1, Tirzepatide & Next-Gen Drugs for Diabetes, Weight Loss, Heart & Kidney Health
Discover how incretin-based therapies like Semaglutide and Tirzepatide are transforming the treatment of Type 2 Diabetes Mellitus and Obesity—with proven benefits for heart, kidney, and metabolic health.
DIABETESOBESITY
Dr. T.S. Didwal, M.D.(Internal Medicine)
4/18/202616 min read
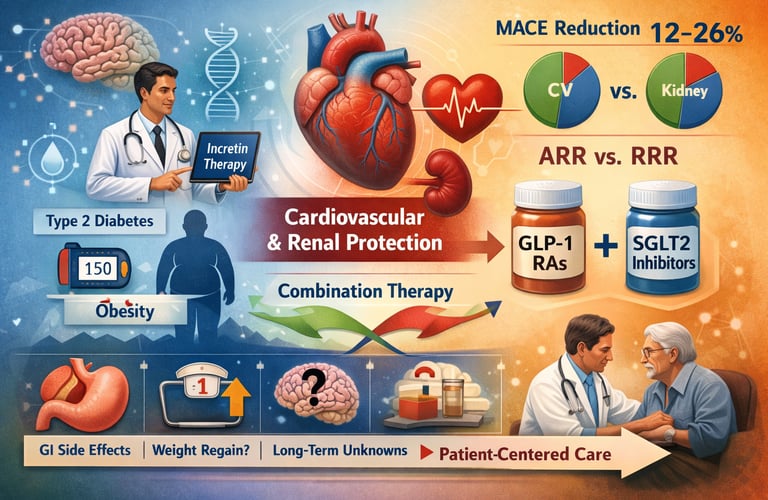
What are incretin-based therapies, and how do they benefit health?
Incretin-based therapies, including GLP-1 receptor agonists and dual agonists like tirzepatide, are medications used to treat Type 2 Diabetes Mellitus and Obesity. They work by mimicking gut hormones that enhance insulin secretion, reduce appetite, and slow gastric emptying. Beyond blood sugar control, these drugs significantly reduce the risk of heart attack, stroke, and kidney disease, with major trials showing a 12–26% reduction in major cardiovascular events. They are now considered foundational therapies for improving long-term metabolic, cardiovascular, and renal health.
LEADER trial (liraglutide): Demonstrated a 13% reduction in major adverse cardiovascular events (MACE) and a significant reduction in cardiovascular mortality in patients with type 2 diabetes at high CV risk.
SUSTAIN-6 trial (semaglutide): Showed a 26% reduction in MACE, with notable reductions in nonfatal stroke and favorable cardiovascular risk
profile.
Clinician’s Perspective: Incretin Therapy in Modern Practice
Shift from glucocentric to organ-protective care: Management of Type 2 Diabetes Mellitus now prioritises reducing cardiovascular, renal, and metabolic risk—not just HbA1c lowering. Incretin-based therapies are central to this paradigm shift.
Early use in high-risk patients: In individuals with established ASCVD, heart failure risk, or Chronic Kidney Disease, initiate GLP-1 RAs early—independent of baseline HbA1c—aligned with ADA/EASD recommendations.
Agent selection matters: Choose agents with proven CVOT benefit (e.g., semaglutide, liraglutide, dulaglutide). Consider dual agonists like tirzepatide for patients prioritizing weight reduction and deeper glycemic control.
Weight-centric approach: For patients with Obesity, these agents function as primary therapies—not adjuncts—often achieving 10–20% weight loss with downstream metabolic benefits.
Bottom Line
Incretin therapies are no longer optional add-ons—they are foundational, disease-modifying agents in cardio-renal-metabolic medicine, requiring early, individualized, and sustained use.
What Are Incretins? The Gut-Hormone You've Never Heard Of
For decades, the treatment of Type 2 Diabetes Mellitus and Obesity followed a predictable path: lower blood sugar, prescribe lifestyle change, and manage complications as they arise. But what if the same class of medications could do far more—simultaneously reducing cardiovascular risk, slowing kidney decline, and even influencing brain health? That possibility is no longer theoretical. It is the reality emerging from the rapidly evolving science of incretin-based therapies.
At the center of this shift are GLP-1 and GIP, gut-derived hormones that regulate insulin secretion, appetite, and energy balance. In healthy physiology, these hormones account for up to 70% of postprandial insulin release—a phenomenon known as the incretin effect. In Type 2 Diabetes Mellitus, however, this system is blunted, contributing to hyperglycemia and metabolic dysfunction (Grespan et al., 2022). Modern pharmacology has leveraged this insight to develop agents that restore and amplify incretin signaling with remarkable clinical impact.
Drugs such as Semaglutide, Liraglutide, and the dual agonist Tirzepatide have demonstrated not only robust glycemic control and substantial weight loss, but also significant reductions in major adverse cardiovascular events and progression of kidney disease (Nauck et al., 2026; Avogaro, 2025). These findings mark a paradigm shift: treatment is no longer confined to glucose-lowering but extends to comprehensive metabolic and organ protection.
What makes this moment particularly compelling is that we are only at the beginning. With next-generation multi-agonists and expanding evidence in neurodegenerative disease, incretin therapies are redefining the boundaries between endocrinology, cardiology, nephrology, and neurology. The question is no longer whether these drugs work—but how broadly their benefits will reshape modern medicine.
The Incretin Defect in Type 2 Diabetes: Why the System Breaks Down
In people without diabetes, the incretin effect is robust and beautifully orchestrated. But in people with type 2 diabetes mellitus (T2DM), this system is significantly impaired — and that impairment has real consequences.
A landmark systematic review and meta-analysis published in The Journal of Clinical Endocrinology & Metabolism by Grespan et al. (2022) rigorously quantified this problem. Analyzing data from multiple controlled studies, the authors demonstrated a substantial and consistent loss of the incretin effect in people with T2DM compared to matched healthy controls. This means the gut-to-pancreas signaling pathway is misfiring — the hormones are released, but the pancreas is far less responsive to them.
Why does this matter practically?
Post-meal blood sugar spikes are higher and last longer
Appetite regulation is disrupted, making it harder to feel full
Glucagon excess drives the liver to release unnecessary glucose into the bloodstream
Insulin secretion in response to meals is blunted at exactly the time it's most needed
This incretin defect is not just a symptom of diabetes — evidence suggests it may contribute to its progression. This insight fundamentally changed how scientists and physicians think about treatment: rather than simply giving more insulin from outside, why not fix the broken internal signaling system?
That question launched the incretin drug revolution.
GLP-1 Receptor Agonists: The First Wave That Changed Everything
The development of GLP-1 receptor agonists (GLP-1 RAs) — medications that mimic the action of natural GLP-1 but last far longer in the body — represents one of the most significant advances in metabolic medicine in the past two decades.
Drugs like semaglutide (Ozempic, Wegovy), liraglutide (Victoza, Saxenda), dulaglutide (Trulicity), and exenatide (Byetta, Bydureon) work by activating GLP-1 receptors throughout the body. Because they are engineered to resist the enzyme that breaks down natural GLP-1, they remain active for hours to days rather than the mere two-minute half-life of the natural hormone.
As comprehensively reviewed by Nauck et al. (2026) in The Lancet, these agents deliver a remarkable array of benefits:
Significant HbA1c reduction (glycated haemoglobin, a key measure of long-term blood sugar control)
Weight loss ranging from 5% to over 15% of body weight depending on the agent
Blood pressure reduction
Favorable effects on cholesterol profiles
Reduced risk of major adverse cardiovascular events (MACE), including heart attack and stroke
Their profile is so compelling that current international guidelines from the American Diabetes Association (ADA) and European Association for the Study of Diabetes (EASD) recommend GLP-1 RAs as preferred agents not just for blood sugar control, but specifically for patients with established or high-risk cardiovascular disease, chronic kidney disease, and obesity-driven T2DM.
Next-Generation Multi-Agonist Therapies: When One Hormone Is Not Enough
If targeting one incretin receptor produces impressive results, what happens when you target two — or even three — simultaneously?
The answer, as emerging evidence shows, is transformative.
Tirzepatide (Mounjaro, Zepbound) — a dual GLP-1/GIP receptor co-agonist — was the first of a new class of multi-target incretin therapies to reach clinical practice. By activating both GLP-1 and GIP receptors simultaneously, tirzepatide achieves synergistic effects on insulin secretion, glucagon suppression, gastric emptying, and brain-mediated appetite control. Clinical trials have shown weight loss exceeding 20% in some patients and HbA1c reductions superior to semaglutide monotherapy.
But the science does not stop there. As detailed in Podder et al. (2026) in the journal Diabetology, a new generation of triple agonists is now in advanced clinical development:
Retatrutide targets GLP-1, GIP, and glucagon receptors simultaneously. Glucagon receptor activation — counterintuitive as it sounds — appears to enhance fat burning (lipolysis) in adipose tissue while the GLP-1 component prevents dangerous blood sugar rises. Early trials suggest weight loss approaching or exceeding 25%.
Cagrilintide + semaglutide (CagriSema) combines a GLP-1 RA with an amylin analog, targeting complementary appetite-regulating pathways and demonstrating superior glycemic and weight outcomes.
Other combinations targeting FGF21 (fibroblast growth factor 21) receptors alongside GLP-1 are in early trials for nonalcoholic fatty liver disease (NAFLD) and metabolic syndrome.
The biological logic of multi-agonism is elegant: metabolic disease is driven by multiple dysregulated pathways, so medicines that address several simultaneously produce effects that exceed the sum of their individual parts. As Miramontes-González et al. (2026) note in Diabetes Therapy, this multi-agonist approach is quite literally "rewriting diabetes therapy" — transforming what were once medications for blood sugar into comprehensive metabolic, cardiovascular, and organ-protective agents.
Heart Benefits: Connecting the Gut to Your Cardiovascular System
Perhaps no aspect of incretin medicine has generated more scientific excitement — or more real-world impact — than the cardiovascular benefits of these drugs.
The story begins with an unexpected trial design. Regulatory agencies, concerned after a cardiovascular safety scandal with an older diabetes drug class, began requiring cardiovascular outcomes trials (CVOTs) for all new diabetes medications in the mid-2000s. Researchers braced for drugs that might "just be safe." Instead, multiple GLP-1 RA trials came back with something remarkable: these drugs didn't just avoid harm — they actively reduced cardiovascular events.
Avogaro (2025), writing in The Lancet Diabetes & Endocrinology, provides a sophisticated mechanistic framework for understanding why:
Direct cardiac effects: GLP-1 receptors are expressed on heart muscle cells. Activation improves cardiac contractility, reduces inflammation in cardiac tissue, and protects against ischemia-reperfusion injury (the damage that occurs when blood flow is restored after a blockage).
Anti-atherosclerotic effects: GLP-1 RAs reduce the inflammatory processes that drive plaque formation in arteries, lower LDL cholesterol and triglycerides, and improve endothelial (blood vessel lining) function.
Blood pressure reduction: Multiple mechanisms contribute, including natriuresis (salt excretion through the kidneys), vasodilation, and modest weight-related reductions.
Anti-arrhythmic properties: Emerging evidence suggests GLP-1 RAs may reduce the risk of atrial fibrillation, a common and dangerous heart rhythm disorder.
For heart failure with preserved ejection fraction (HFpEF) — a form of heart failure that is increasingly prevalent and for which no medications previously showed benefit — the STEP-HFpEF trial with semaglutide showed dramatic improvements in symptoms, physical function, and quality of life. This represents a genuine paradigm shift.
Nauck et al. (2026) synthesize the evidence across multiple CVOTs and conclude that next-generation incretin agents, particularly dual and triple agonists, are expected to show even stronger cardiovascular risk reduction as their greater metabolic potency translates to greater reductions in the inflammatory, metabolic, and hemodynamic drivers of cardiovascular disease.
Kidney Protection: The Renal Benefits You May Not Know About
The kidneys are among the most common organ systems damaged by both diabetes and obesity. Diabetic kidney disease (DKD) is the leading cause of kidney failure worldwide, and conventional treatment — while helpful — has rarely stopped progression.
Incretin-based therapies are changing this narrative too.
As highlighted across both Nauck et al. (2026) and Miramontes-González et al. (2026), GLP-1 receptor agonists exert kidney protection through multiple mechanisms:
Reduced glomerular hyperfiltration: In early diabetic kidney disease, the kidneys work at dangerous overdrive. GLP-1 RAs normalize filtration rates, reducing the pressure-driven damage to delicate glomerular (filtering) structures.
Anti-inflammatory and anti-fibrotic effects: Chronic kidney inflammation drives scar tissue (fibrosis) formation — the irreversible end-stage of kidney damage. GLP-1 RAs suppress key inflammatory pathways, including NF-κB and NLRP3 inflammasome activation, within kidney tissue.
Blood pressure and uric acid reduction: Both are major independent drivers of kidney damage that GLP-1 RAs favorably modify.
Albuminuria reduction: Protein in the urine (albuminuria) is both a marker and driver of kidney disease progression. Multiple trials show significant reductions in albuminuria with GLP-1 RA therapy.
The landmark FLOW trial of semaglutide in people with T2DM and chronic kidney disease demonstrated a 24% reduction in the risk of major kidney disease events — including kidney failure, need for dialysis, and kidney-related death. This trial was so compelling it was stopped early by its independent safety board because the evidence of benefit was overwhelming.
The mechanistic and clinical evidence now positions GLP-1 RAs — and likely next-generation multi-agonists — as foundational kidney-protective therapies, not just as blood sugar medications that happen to help kidneys on the side.
Brain Health: The Surprising New Frontier of Incretin Medicine
The most exciting and perhaps unexpected frontier in incretin research involves the brain.
GLP-1 receptors are expressed not just in the pancreas, gut, and heart — they are found extensively throughout the central nervous system, including the hypothalamus (appetite regulation), the hippocampus (memory and learning), and the brainstem. This neurological footprint is now attracting intense scientific interest in the context of cognitive impairment, Alzheimer's disease, and Parkinson's disease.
Giofrè et al. (2025), writing in Frontiers in Endocrinology, provide a comprehensive narrative review of both preclinical (animal/cell model) and clinical evidence for incretin-based therapy in cognitive impairment associated with T2DM. Key findings include:
In preclinical models:
GLP-1 receptor agonists reduce amyloid-beta plaques and tau protein tangles — the hallmark pathological features of Alzheimer's disease
They reduce neuroinflammation, oxidative stress, and synaptic dysfunction
They promote neurogenesis (growth of new nerve cells) in the hippocampus
They improve spatial memory and learning performance
In clinical studies:
Observational data show that patients with T2DM on GLP-1 RAs have significantly lower rates of dementia compared to those on other diabetes medications
A Phase 2 trial of liraglutide in Alzheimer's disease showed signals of slowed cognitive decline and reduced brain atrophy on MRI
Liraglutide and semaglutide are currently in large Phase 3 trials specifically for Alzheimer's prevention and treatment
For Parkinson's disease, a clinical trial of exenatide (a GLP-1 RA) showed that treated patients had significantly better motor function scores after a washout period, suggesting a genuine disease-modifying (neuroprotective) effect — not just symptomatic improvement.
While this evidence is still maturing and not yet at the level where GLP-1 RAs can be prescribed specifically for cognitive protection, the signals are compelling enough that they are actively reshaping neuroscience research priorities. The gut-brain connection, it turns out, runs deeper than anyone imagined.
Targeting Obesity: Incretins as the Central Pillar of Weight Management
For the first time in medical history, we have pharmacological treatments for obesity that achieve weight loss comparable to bariatric surgery — without going under the knife. And they work precisely because they engage the incretin system.
Ansari et al. (2024), in a comprehensive review in Nature Reviews Endocrinology, explain why the incretin system is the ideal biological target for obesity treatment:
Central appetite suppression: GLP-1 and GIP receptors in the hypothalamus and brainstem reduce caloric intake by signaling satiety, reducing food cravings, and — fascinatingly — reducing the "reward value" of high-calorie foods in brain reward circuits.
Gastric emptying delay: Food stays in the stomach longer after meals, prolonging physical fullness.
Energy expenditure: GIP receptor activation, in particular, appears to enhance thermogenesis (heat generation) in adipose tissue, contributing to caloric burn.
Gut microbiome modulation: Emerging evidence suggests GLP-1 RAs favorably alter the gut microbiome in ways that further support metabolic health.
The clinical results speak for themselves:
Semaglutide 2.4mg (Wegovy): ~15% average body weight reduction over 68 weeks in the STEP trials
Tirzepatide 15mg (Zepbound): ~22.5% average body weight reduction in SURMOUNT-1
Retatrutide: Early trial data showing ~24% weight loss
Critically, as Ansari et al. (2024) emphasize, the benefits go far beyond aesthetics. Weight loss of this magnitude reduces joint pain, sleep apnea, fatty liver disease, hypertension, and — as we've seen — cardiovascular and kidney disease risk. The entire metabolic disease burden is shifted.
Limitations & Real-World Considerations
While incretin-based therapies are highly effective, several important limitations should be considered in clinical practice:
1. Gastrointestinal Side Effects (≈20–40%)
Nausea, vomiting, diarrhea, and early satiety are the most commonly reported adverse effects, particularly during dose escalation. These symptoms are usually transient and dose-dependent but can lead to discontinuation in a subset of patients. Gradual titration and patient counseling significantly improve tolerability.
2. Weight Regain After Discontinuation
Evidence from extension studies (e.g., STEP trials) shows that a substantial proportion of lost weight is regained after stopping therapy. This highlights that incretin-based treatments function more like chronic disease management tools rather than short-term interventions, similar to antihypertensive or lipid-lowering therapy.
3. Real-World Adherence Challenges
Despite strong efficacy in clinical trials, real-world adherence is lower due to:
Cost and limited insurance coverage
Need for long-term use
Injectable route (for most agents)
Side effects during initiation
These factors can reduce sustained effectiveness outside controlled trial settings.
4. Long-Term Safety (10+ Years)
Although medium-term data (2–5 years) demonstrate a favorable safety profile, long-term outcomes beyond a decade remain incompletely understood. Ongoing surveillance is particularly focused on:
Pancreatic safety
Gallbladder disease
Rare endocrine or gastrointestinal complications
Continued post-marketing data and longitudinal cohort studies will be essential to fully define lifetime risk-benefit balance.
Clinical Takeaway
Incretin therapies are best viewed as long-term, disease-modifying treatments that require sustained use, careful patient selection, and ongoing monitoring to maximize benefits and minimize risks.
Who Should NOT Take GLP-1 Receptor Agonists (Very Brief)
Medullary Thyroid Carcinoma or MEN2: Absolute contraindication (based on rodent tumor signal; precaution extended to humans)
Multiple Endocrine Neoplasia type 2: Avoid due to thyroid cancer risk
History of pancreatitis: Use with caution or avoid (risk remains debated but clinically relevant)
Severe Gastroparesis: May worsen symptoms due to delayed gastric emptying
Severe gastrointestinal disease: Poor tolerability (nausea, vomiting)
Pregnancy or breastfeeding: Not recommended (insufficient safety data)
Frequently Asked Questions
❓ FAQ 1: Are GLP-1 medications safe for long-term use?
Yes, based on current evidence. Multiple large clinical trials with follow-up periods of 2–5 years have demonstrated an excellent safety profile for GLP-1 receptor agonists. Common side effects include nausea, vomiting, and diarrhea — particularly when starting or increasing doses — but serious adverse events are rare. Concerns about pancreatitis and thyroid cancer, raised in early animal studies, have not been borne out in large human trials. As Nauck et al. (2026) note, the benefit-risk profile of these agents is clearly favorable across their approved indications. Ongoing long-term registry studies continue to monitor safety in real-world populations.
❓ FAQ 2: What is the difference between semaglutide and tirzepatide?
Semaglutide (Ozempic for diabetes, Wegovy for obesity) is a GLP-1 receptor agonist — it targets one receptor. Tirzepatide (Mounjaro for diabetes, Zepbound for obesity) is a dual GLP-1/GIP receptor co-agonist — it targets two incretin receptors simultaneously. Head-to-head trials (SURPASS-6) have shown tirzepatide achieves greater HbA1c reduction and greater weight loss than semaglutide. Both are excellent medications; the choice depends on individual patient factors, tolerability, cost, and treatment goals.
❓ FAQ 3: Can these medications really protect my kidneys?
Yes — and this is one of the most important recent discoveries. The FLOW trial of semaglutide demonstrated a 24% reduction in major kidney disease events in people with T2DM and chronic kidney disease (Nauck et al., 2026; Miramontes-González et al., 2026). GLP-1 RAs reduce kidney-damaging inflammation, lower blood pressure, decrease albuminuria, and normalize hyperfiltration — multiple protective mechanisms working together. They are now recommended alongside SGLT-2 inhibitors as preferred agents in diabetic kidney disease.
❓ FAQ 4: I've heard these drugs can help with dementia. Is that true?
The evidence is exciting but still developing. Preclinical studies are very promising, showing reductions in Alzheimer's-related proteins, reduced neuroinflammation, and improved memory in animal models (Giofrè et al., 2025). In humans, observational studies show lower dementia rates in GLP-1 RA users, and Phase 3 clinical trials specifically targeting Alzheimer's disease are currently underway. It is not yet established practice to prescribe these drugs specifically for dementia prevention, but the signals are compelling enough to factor into treatment decisions for patients with both T2DM and dementia risk.
❓ FAQ 5: I don't have diabetes — can I still take these medications for weight loss?
Yes, if you meet the criteria. Semaglutide 2.4mg (Wegovy) and tirzepatide 15mg (Zepbound) are FDA-approved for chronic weight management in adults with a BMI of ≥30 kg/m², or ≥27 kg/m² with at least one weight-related condition (such as hypertension, sleep apnea, or high cholesterol) — regardless of diabetes status. As Ansari et al. (2024) outline in Nature Reviews Endocrinology, the incretin system is the central biological target for obesity pharmacotherapy, and these medications produce clinically meaningful, sustained weight loss that improves multiple aspects of health.
❓ FAQ 6: What does "loss of incretin effect" mean for someone with diabetes?
It means a key natural blood sugar control system is impaired. In healthy individuals, gut hormones (GLP-1 and GIP) released after meals account for 50–70% of insulin secretion. In people with T2DM, this gut-to-pancreas signaling is substantially blunted — the pancreas is far less responsive to these hormones than it should be (Grespan et al., 2022). This contributes to higher post-meal blood sugar spikes, excess glucagon, and progressive metabolic deterioration. GLP-1 receptor agonists bypass this defect by providing pharmacological doses of GLP-1 activity that overcome the pancreatic resistance.
❓ FAQ 7: Are there newer medications coming that are even more powerful?
Absolutely — and they are arriving soon. Retatrutide (a triple GLP-1/GIP/glucagon receptor agonist) has shown weight loss approaching 25% in Phase 2 trials and is in advanced Phase 3 development. CagriSema (cagrilintide + semaglutide) combines complementary appetite pathways for superior outcomes. Orforglipron — a once-daily oral (pill) GLP-1 RA — could eliminate the need for injections. As Podder et al. (2026) detail, the pipeline of incretin-based multi-agonist therapies is extraordinarily rich, and the next 5 years will likely see multiple new agents reach patients with even greater efficacy and more convenient delivery.
Clinical pearls
The "Incretin Defect" Correction
Scientific Perspective: In T2DM, the "incretin effect" (the insulin response to oral vs. IV glucose) is severely blunted. GLP-1 RAs do not just "add" a hormone; they restore a signaling pathway that accounts for up to 70% of postprandial insulin secretion.
Think of your gut and pancreas like a radio and a receiver. In diabetes, the volume is turned so low they can’t hear each other. This medicine doesn't just lower sugar; it "fixes the volume" so your body can communicate properly again.
2. Weight Loss vs. Muscle Preservation
Scientific Perspective: High-potency incretins (like tirzepatide and retatrutide) can cause rapid weight loss, where 20-40% of the weight lost can be lean muscle mass if not managed. This is critical for metabolic rate and "geroscience" outcomes.
Losing weight is great, but losing muscle is not. To get the best results, you must pair these medications with resistance training and high protein intake to ensure you are losing fat, not the "engine" (muscle) that keeps you strong.
3. Beyond the A1c: Organ Protection
Scientific Perspective: GLP-1 RAs provide cardiovascular (CV) and renal benefits that are often independent of glucose lowering. Receptors in the heart and kidneys reduce systemic inflammation and oxidative stress directly.
This isn't just a "sugar pill." Even if your blood sugar numbers look perfect, the medicine is working "behind the scenes" like a shield, protecting your heart from inflammation and your kidneys from scarring.
4. Managing the "Appetite Window"
Scientific Perspective: These medications shift the "hedonic" value of food. By modulating the brain's reward centres (the mesolimbic system), they reduce "food noise"—the intrusive, obsessive thoughts about eating.
One of the biggest changes you'll feel is that the "voice" in your head constantly telling you to snack goes quiet. Use this quiet period to build a healthy relationship with food, rather than just eating less of the same processed items.
5. Gastrointestinal Titration & Tolerance
Scientific Perspective: GLP-1-induced
nausea is primarily due to a transient delay in gastric emptying and central nervous system signaling. Slow upward titration (the "Start Low, Go Slow" approach) allows the brainstem receptors to desensitize.
If you feel nauseous early on, it’s usually because your stomach is moving slower than your brain is used to. Eating smaller, more frequent meals and avoiding high-fat foods during the first few weeks can help your body "calibrate" to the new hormone levels.
Comparison of Incretin Generations
First Generation
Agents: Liraglutide, Exenatide
Key Benefit: Effective blood sugar control with modest weight loss
Second Generation
Agent: Semaglutide (Ozempic/Wegovy)
Key Benefit: Significant cardiovascular protection with ~15% weight loss
Third Generation
Agents: Tirzepatide, Retatrutide
Key Benefit: Dual/triple receptor action, >20% weight loss, and potential improvement or reversal of Nonalcoholic Fatty Liver Disease
Takeaway: Each generation represents a step forward—from glucose control → cardiovascular protection → comprehensive metabolic disease modification.
Author’s Note
The field of incretin-based therapy is evolving at an extraordinary pace, reshaping how we understand and manage Type 2 Diabetes Mellitus, Obesity, and their complications. What was once considered a niche hormonal pathway has now emerged as a central therapeutic axis influencing the heart, kidneys, and potentially even the brain.
This article reflects a synthesis of the most current evidence available at the time of writing (2026), including large cardiovascular outcomes trials, renal studies, and emerging data on multi-agonist therapies. However, medicine is dynamic. Ongoing trials and real-world data will continue to refine our understanding of long-term safety, optimal patient selection, and broader applications—particularly in neurodegenerative disease and precision metabolic care.
For clinicians, the challenge is no longer whether to use incretin-based therapies, but how to integrate them thoughtfully into individualized treatment plans—balancing efficacy, safety, cost, and patient preference. For patients, these therapies represent an opportunity not just for better numbers, but for meaningful reductions in long-term health risks.
Ultimately, the goal remains unchanged: to translate cutting-edge science into practical, patient-centred care that improves both lifespan and patient-centred healthspan. The science of incretins is moving at an extraordinary speed. What began as the discovery of a gut hormone in the 1980s has become the most transformative therapeutic revolution in metabolic medicine in a generation. You deserve to understand it — and to benefit from it
Disclaimer: This article is for educational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider before starting, stopping, or adjusting any medication. Individual risk profiles and treatment decisions must be individualised.
Related Articles:
Do You Need to Go Low-Carb for Diabetes? Mediterranean vs Low-Carb Explained (Doctor’s Guide)
Can You Reverse Type 2 Diabetes with Diet? Science-Based Guide 2026
Your Cholesterol Isn’t the Whole Story: The Hormone Imbalances Driving Heart Disease
Can GLP-1 Drugs Prevent Heart Attacks? New Trial Data Changes Everything
ApoB vs LDL Cholesterol: Which Is the Most Accurate Predictor of Heart Disease?
Remnant Cholesterol: The Hidden Link Between Insulin Resistance, Type 2 Diabetes, and Heart Disease
References
Ansari, S., Khoo, B., & Tan, T. (2024). Targeting the incretin system in obesity and type 2 diabetes mellitus. Nature Reviews Endocrinology, 20, 447–459. https://doi.org/10.1038/s41574-024-00979-9
Avogaro, A. (2025). Incretins and the cardiovascular system: Bridging digestion with metabolism. The Lancet Diabetes & Endocrinology, 13(9), 790–802. https://doi.org/10.1016/S2213-8587(25)00166-4
Giofrè, F., Zaffina, I., Pelle, M. C., & Arturi, F. (2025). Recent advances in incretin-based therapy for the treatment of cognitive impairment associated to the type 2 diabetes mellitus: Preclinical and clinical studies — a narrative review. Frontiers in Endocrinology, 16, 1696419. https://doi.org/10.3389/fendo.2025.1696419
Grespan, E., Guolo, A., Muscelli, E., Ferrannini, E., & Mari, A. (2022). Loss of the incretin effect in type 2 diabetes: A systematic review and meta-analysis. The Journal of Clinical Endocrinology & Metabolism, 107(7), 2092–2100. https://doi.org/10.1210/clinem/dgac213
Miramontes-González, J. P., Rodrigo-Alaíz, Á., Gabella-Martín, M., et al. (2026). Rewriting diabetes therapy: How incretin modulation is transforming cardiovascular and renal outcomes. Diabetes Therapy, 17, 317–330. https://doi.org/10.1007/s13300-025-01829-1
Nauck, M. A., Tuttle, K. R., Tschöp, M. H., & Blüher, M. (2026). GLP-1 receptor agonists and next-generation incretin-based medications: Metabolic, cardiovascular, and renal benefits. The Lancet, 407(10531), 892–90C8. https://doi.org/10.1016/S0140-6736(25)02105-1
Podder, D., Stala, O., Miah, A., Agyapong, A., Moore, M. E., Hirani, R., Diegisser, D., Garcia, V., & Etienne, M. (2026). Incretin-based multi-agonist therapies for type 2 diabetes mellitus and obesity: Mechanisms, clinical efficacy, and future directions. Diabetology, 7(3), 46. https://doi.org/10.3390/diabetology7030046