Your Cholesterol Isn’t the Whole Story: The Hormone Imbalances Driving Heart Disease
Discover how testosterone, thyroid, and cortisol silently drive heart disease risk—even when cholesterol and blood pressure are normal
HEART
Dr. T.S. Didwal, M.D.(Internal Medicine)
4/8/202617 min read
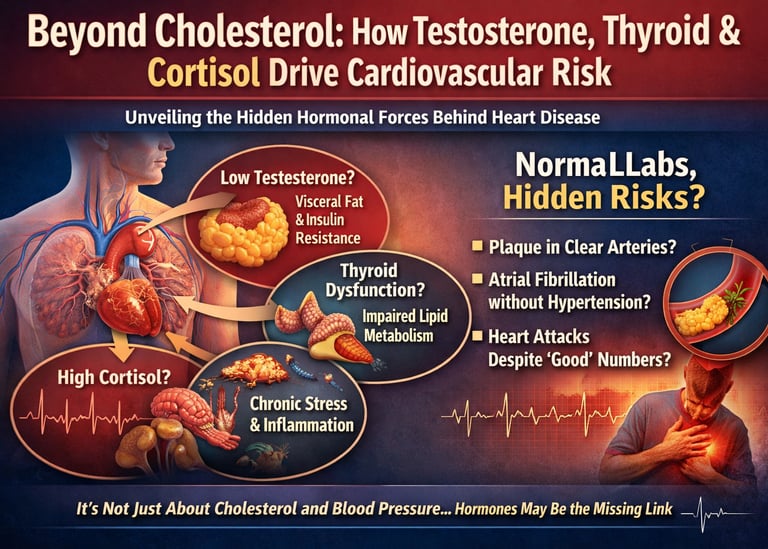
Hormones like testosterone, thyroid hormones, and cortisol play a central role in cardiovascular health by regulating metabolism, vascular function, inflammation, and autonomic balance. Even subtle hormonal imbalances—often within normal lab ranges—can increase the risk of heart disease, atrial fibrillation, and metabolic dysfunction. Emerging evidence shows that evaluating these endocrine pathways alongside cholesterol and blood pressure provides a more accurate and upstream assessment of cardiovascular risk.
Subclinical hyperthyroidism is associated with ~2–3× higher risk of atrial fibrillation (RR ≈ 2.0–3.1)
Elevated cortisol levels correlate with ~30–50% increased cardiovascular event risk across cohort studies
Low testosterone in men is linked to ~20–30% higher all-cause and cardiovascular mortality risk
A Clinician’s Perspective: hormones and heart disease
Cardiovascular medicine is due for a paradigm shift
For decades, risk assessment has focused on downstream markers like LDL cholesterol, blood pressure, and glucose. While essential, these are expressions of disease—not always its origin.Residual risk is the clinical red flag
Patients with “controlled” lipids and blood pressure still develop atherosclerosis, atrial fibrillation, and heart failure. This persistent residual risk suggests missing upstream drivers in our current models.Hormones are central—not secondary—regulators
Testosterone, thyroid hormones, and cortisol directly influence:Endothelial function
Lipid metabolism and ApoB dynamics
Insulin sensitivity
Autonomic balance
These are core cardiovascular pathways, not peripheral modifiers.
Subclinical dysfunction is clinically meaningful
Hormonal values within “normal ranges” may still fall outside physiological optimal zones, contributing to long-term risk (Iglesias & Iglesias, 2026).
This is where traditional lab interpretation often fails.Current clinical practice has a diagnostic blind spot
Common scenarios where endocrine drivers are overlooked:Unexplained hypertriglyceridemia
Resistant hypertension
Atrial fibrillation without structural disease
Central obesity with normal BMI
These patients are often treated symptomatically, without addressing root causes.
Emerging evidence supports endocrine-driven cardiovascular risk
Dysregulation of the hypothalamic–pituitary axis is linked to:Endothelial dysfunction
Adverse cardiac remodeling
Increased cardiovascular events
Clinical implication: expand, don’t replace, current models
The goal is not to abandon LDL or BP targets, but to integrate hormonal evaluation into risk stratification.Toward precision cardiology
Targeted testing of testosterone, thyroid function, and cortisol rhythm allows clinicians to:Identify upstream drivers
Personalize interventions
Potentially alter disease trajectory earlier
The future of prevention
Cardiovascular care will evolve from “How low is the LDL?” to
“What endocrine signals are driving this patient’s risk?”
Beyond Cholesterol: How Testosterone, Thyroid & Cortisol Drive Cardiovascular Risk
This chapter explores the three pillars of hormonal cardiovascular risk: testosterone, thyroid hormones, and cortisol. For each, we'll cover the physiology, the clinical evidence, the controversies, and — most importantly — what you and your care team can actually do about it.
Cardiovascular medicine is entering a necessary phase of recalibration. For decades, we have anchored risk assessment and treatment strategies around downstream markers—LDL cholesterol, blood pressure, and glycemic indices. These remain important, but they are not primary drivers. Increasingly, clinical experience and emerging literature suggest that we are often treating the biochemical consequences of disease rather than its upstream endocrine origins. The patient with “well-controlled” lipids who still progresses to atherosclerosis is no longer an anomaly—it is a signal that our framework is incomplete.
From a clinician’s perspective, the most striking shift is the recognition that hormonal systems are not peripheral modifiers but central regulators of cardiovascular physiology. Testosterone, thyroid hormones, and cortisol collectively govern vascular tone, lipid trafficking, insulin sensitivity, and autonomic balance. Subclinical disturbances—values that fall within laboratory reference ranges but outside physiological optimality—can meaningfully alter cardiovascular trajectory over years (Iglesias & Iglesias, 2026). Yet these markers are rarely integrated into routine cardiometabolic evaluation.
In practice, this creates a blind spot. Patients with unexplained hypertriglyceridemia, resistant hypertension, atrial fibrillation without structural disease, or central adiposity despite normal BMI are often escalated pharmacologically without a parallel endocrine assessment. The result is therapeutic inertia at the root-cause level. Evidence increasingly supports that dysregulation of the hypothalamic–pituitary axis contributes to endothelial dysfunction, adverse cardiac remodeling, and increased cardiovascular events—even when traditional risk scores appear reassuring (Tsai et al., 2024; Júnior et al., 2025).
The clinical imperative, therefore, is not to replace established risk models, but to expand them. Incorporating targeted hormonal evaluation—testosterone, thyroid function, and cortisol rhythm—into selected patients represents a shift toward precision cardiology. This approach allows clinicians to identify upstream drivers, personalize interventions, and potentially alter disease trajectory earlier. The future of cardiovascular prevention will not be defined solely by how aggressively we lower LDL, but by how intelligently we understand and correct the endocrine signals shaping the vascular environment.
Testosterone regulates visceral fat, insulin sensitivity, endothelial nitric oxide, and erythropoiesis.
The thyroid controls heart rate, LDL receptor activity, cardiac contractility, and lipid turnover.
Cortisol modulates blood pressure, glucose metabolism, visceral adiposity, and autonomic tone.
The Endocrine Architecture of the Cardiovascular System
Your heart and blood vessels are not just plumbing. They are endocrine-sensitive tissue. Three major axes govern their function: the hypothalamic–pituitary–gonadal (HPG) axis, the hypothalamic–pituitary–thyroid (HPT) axis, and the hypothalamic–pituitary–adrenal (HPA) axis. Dysregulation in any one of these does not produce a single isolated effect — it ripples across endothelial function, lipoprotein metabolism, vascular inflammation, coagulation pathways, and autonomic nervous system tone (Júnior et al., 2025)
A landmark 2026 review in the Journal of Endocrinological Investigation described this as the "hormonal crossroads of the heart," noting that the heart is simultaneously a target of endocrine signals and a source of its own hormones, including atrial natriuretic peptide (ANP) (Iglesias & Iglesias, 2026). Understanding this two-way dialogue is essential for any clinician managing patients with unexplained dyslipidemia, resistant hypertension, or central obesity despite normal BMI.
Clinical Concept
Hormonal risk layering means that a patient with borderline TSH, low-normal testosterone, and elevated evening cortisol may carry substantially more cardiovascular risk than their Framingham score suggests — even with normal LDL and blood pressure.
Testosterone and Cardiovascular Health
What Testosterone Actually Does in Your Body
Testosterone is far more than a "male hormone." In men and women alike, it plays a critical role in maintaining lean muscle mass, regulating insulin sensitivity, controlling visceral fat distribution, and supporting endothelial nitric oxide (NO) production — the very molecule that keeps arteries flexible and responsive. Low testosterone accelerates visceral adiposity, which drives insulin resistance, which elevates inflammatory cytokines (IL-6, TNF-α), which damage the arterial wall. This chain reaction is the mechanistic link between hypogonadism and cardiovascular disease.
The U-Shaped Risk Curve
One of the most important — and underappreciated — concepts in endocrine cardiology is that both extremes of testosterone are dangerous. This is not a simple "more is better" relationship.
The Testosterone U-Shaped Risk Curve
Low Testosterone
↑ Visceral adiposity
↑ Insulin resistance
↑ Inflammatory cytokines
↑ CVD mortality
High / Supraphysiologic
↑ Hematocrit
↑ Hyperviscosity
↑ Thrombosis risk
Adverse lipid shifts
The pivotal TRAVERSE trial, Lincoff et al., 2023, published in the New England Journal of Medicine, finally provided high-quality RCT evidence on this question. Among middle-aged and older men with hypogonadism and elevated cardiovascular risk, testosterone-replacement therapy (TRT) did not significantly increase the rate of major adverse cardiovascular events compared with placebo. Importantly, however, there was a higher rate of pulmonary embolism and atrial fibrillation in the TRT group — consistent with the hyperviscosity concern at the high end of the curve. The message is nuanced: TRT is not categorically dangerous, but optimal management requires the "physiologic window hypothesis" — targeting restoration to normal range, not maximization.
Practical Pearl
For symptomatic hypogonadal men receiving TRT, monitor hematocrit regularly. A hematocrit above 54% is a signal to reduce the dose. Also track lipid profiles, PSA, and cardiometabolic markers every 3–6 months in the first year of treatment.
Thyroid Dysfunction: The Metabolic Metronome of the Heart
Thyroid hormones are the master regulators of cardiac pace and lipid metabolism. When these levels fluctuate, they create a ripple effect that compromises both the electrical rhythm and the structural integrity of the cardiovascular system.
The Direct and Indirect Cardiac Drivers
Biological Activity: Triiodothyronine (T3) is the active hormone that targets cardiac myocytes (heart muscle cells).
Direct Impact: It regulates heart rate, the force of myocardial contraction (inotropy), and systemic vascular resistance.
The Lipid Gatekeeper: Thyroid hormones control the number of LDL receptors on the liver.
The "Slow-Motion" Effect: Reduced thyroid activity leads to fewer receptors → slower clearance of LDL from the blood → elevated ApoB and LDL-C.
Subclinical Hypothyroidism (SCH): The Silent Threat
Definition: Characterized by an elevated TSH with a "normal" free T4 reading.
Prevalence: Affects 4–8% of the population and up to 20% of women over age 60.
Prognostic Risk: A 2025 study in Frontiers in Endocrinology found that elevated TSH significantly worsens outcomes in patients with existing coronary disease, especially when TSH exceeds 10 mIU/L.
Structural Damage: Mendelian randomization (causal evidence) links thyroid dysfunction to left ventricular hypertrophy and increased arterial stiffness (Ruan et al., 2024)
TSH Risk Stratification: Cardiovascular Impact & Clinical Action
The following points break down how Thyroid-Stimulating Hormone (TSH) levels correlate with cardiovascular health and the corresponding medical response.
Optimal Range (0.4–2.5 mIU/L)
Impact: Represents the lowest cardiovascular disease (CVD) risk zone and an optimal metabolic rate.
Action: Continue routine monitoring as part of standard wellness checks.
Borderline Range (2.5–4.5 mIU/L)
Impact: Associated with early shifts in LDL cholesterol and subtle changes in endothelial (blood vessel) function.
Action: Review clinical symptoms and order a comprehensive lipid panel to assess cumulative risk.
Subclinical Hypothyroidism (4.5–10 mIU/L)
Impact: Increased risk of atherosclerosis (plaque buildup) and rising diastolic blood pressure.
Action: Move toward an individualized treatment decision based on the patient's existing heart disease risk factors.
Overt Hypothyroidism (>10 mIU/L)
Impact: Significant elevation in heart failure risk and overall cardiovascular mortality.
Action: Strong clinical indication to initiate thyroid hormone replacement therapy..
Hyperthyroidism: The Arrhythmia Engine
Electrical Overdrive: Excess thyroid hormone creates a state of "metabolic heat," resulting in tachycardia and increased oxygen demand.
AFib Connection: Genetically predicted hyperthyroid states are a direct driver of Atrial Fibrillation (AFib).
Subclinical Risk: Even a suppressed TSH with normal T4/T3 triples AFib risk in older adults.
The "Masking" Effect: Thyroid dysfunction often presents as a heart problem (arrhythmia or heart failure) rather than a glandular one.
The Clinical Pearl: Dyslipidemia, hypertension, and arrhythmias are often symptoms of an upstream thyroid imbalance. If you only treat the cholesterol, you are managing the shadow while ignoring the source.
Cortisol, Chronic Stress, and the Heart
The HPA Axis and Your Cardiovascular System
The hypothalamic–pituitary–adrenal (HPA) axis orchestrates the body's stress response. When functioning normally, it follows a precise circadian rhythm: cortisol peaks in the early morning (6–8 AM) to mobilize energy, then drops to a nadir by midnight. This rhythm is not cosmetic — it synchronizes cardiovascular function, metabolism, and immune activity. When chronic stress, poor sleep, or chronic disease flatten or invert this curve, the cardiovascular consequences are profound.
A 2025 review in The American Journal of Medicine on integrative HPA dysfunction management highlighted that dysregulated cortisol patterns are increasingly recognized as an independent contributor to cardiometabolic disease, and that measuring a single morning cortisol value — as is commonly done — systematically underestimates the degree of HPA disruption (An Integrative Approach, 2025)
How Cortisol Damages the Heart
Chronically elevated cortisol drives cardiovascular damage through multiple simultaneous pathways: it elevates blood glucose via gluconeogenesis, induces peripheral insulin resistance, promotes visceral fat deposition (the most metabolically dangerous fat depot), and raises blood pressure through mineralocorticoid-receptor activation. At the arterial level, cortisol promotes endothelial dysfunction, accelerates inflammatory plaque development, and destabilizes existing plaques (Knezevic et al., 2023).
Cushing's syndrome — a disease of profound chronic cortisol excess — serves as the clinical extreme, with patients developing premature atherosclerosis, metabolic syndrome, and cardiovascular events at rates far exceeding age-matched controls. But you don't need Cushing's syndrome to suffer cortisol-driven cardiovascular harm. A 2024 systematic review and meta-analysis in the International Journal of Cardiology (Tsai et al., 2024) pooled data across large cohort studies and confirmed that elevated stress hormones — including cortisol and catecholamines — are independently associated with significantly increased cardiovascular disease risk.
Autonomic Dysfunction: The Missing Link
Chronic HPA activation does not occur in isolation — it perpetuates sympathetic nervous system overdrive. The clinical signature of this is reduced heart rate variability (HRV), a marker of diminished vagal tone that predicts both arrhythmia risk and all-cause cardiovascular mortality. Psychological stress, social isolation, depression, and job strain have all been robustly linked in cohort data to incident cardiovascular events — the mechanism operating substantially through this autonomic-cortisol axis (Tsai et al., 2024). Stress-induced cardiomyopathy (Takotsubo syndrome) represents the acute extreme of this pathway.
Actionable Insight
Mindfulness-based stress reduction (MBSR), sleep optimization, and structured aerobic exercise (at moderate, not extreme, intensity) all demonstrably reduce HPA axis dysregulation. These are not soft lifestyle suggestions — they are mechanistically grounded cardiovascular interventions
The "Endocrine Triangle" of Cardiovascular Risk
The clinical danger of siloed thinking about these hormones is that they do not act independently — they form a mutually reinforcing network. Low testosterone promotes insulin resistance, which disrupts HPA regulation, which elevates cortisol, which further suppresses gonadotropin release, which further lowers testosterone. Hypothyroidism reduces metabolic clearance of cortisol, prolonging its exposure. Chronic cortisol elevation impairs thyroid hormone conversion (T4 → T3). These axes interact synergistically, not additively.
This is why a patient may present with what appears to be "just" atherogenic dyslipidemia — but the root cause is actually the convergence of subclinical hypothyroidism, low-testosterone-driven insulin resistance, and elevated evening cortisol from poor sleep. Statin therapy addresses the symptom. Endocrine assessment addresses the cause.
Practical Clinical Application: What to Do With This Information
When to Screen
Consider hormonal cardiovascular screening in patients with: unexplained dyslipidemia (especially high TG + low HDL), resistant hypertension, central obesity with normal BMI, low libido or fatigue alongside metabolic changes, or atrial fibrillation without a structural cause.
The Integrated Biomarker Panel
A practical first-pass hormonal cardiovascular screen includes: total and free testosterone (8 AM fasting), TSH and free T4, a timed cortisol (ideally morning + evening to assess diurnal rhythm), fasting insulin and HOMA-IR, and ApoB. This is not expensive medicine — it is precise medicine.
Step 1: Identify Atypical Patterns. Look for dyslipidemia, hypertension, or metabolic syndrome that is disproportionate to diet and activity level.
Step 2: Screen Endocrine Axis Order testosterone, TSH/free T4, and timed cortisol. Consider 24-hour urinary cortisol if clinical suspicion is high.
Step 3: Stratify Risk. Use hormonal findings alongside ApoB, hsCRP, and coronary artery calcium (CAC) scoring for integrated risk stratification.
Step 4: Target the Upstream Driver. Treat hypothyroidism, address hypogonadism if symptomatic and confirmed, and implement structured HPA rehabilitation strategies.
Step 5: Monitor Longitudinally Re-assess hormonal and cardiovascular markers at 3 and 12 months. Hormonal optimization is not a one-time intervention.
Frequently Asked Questions
To dominate Google Search snippets (and AI-powered overviews), the content must be structured for the "Position Zero" algorithm. This requires a "Definition-First" approach: a clear, 40–60 word direct answer followed by a high-value breakdown.
Is testosterone therapy safe for men with heart disease?
Direct Answer: Recent high-quality evidence, including the 2023 TRAVERSE trial, suggests that Testosterone Replacement Therapy (TRT) does not increase major adverse cardiovascular events (MACE) in symptomatic hypogonadal men. However, it may increase risks for atrial fibrillation and pulmonary embolism if hematocrit levels exceed 54%.
Optimal Window: Aim for physiologic restoration, not supraphysiologic levels.
Key Metric: Monitor hematocrit and PSA every 3–6 months.
Risk Factors: A history of blood clots or untreated sleep apnea requires caution.
How does a TSH of 5.0 affect heart disease risk?
Direct Answer: A TSH of 5.0 mIU/L indicates subclinical hypothyroidism, which is linked to an increased risk of coronary heart disease and heart failure. Even subtle thyroid deficiency reduces LDL receptor activity in the liver, leading to higher ApoB levels and increased diastolic blood pressure.
Cardiovascular Impact: Slower LDL clearance and impaired arterial flexibility.
Treatment Threshold: Most guidelines strongly recommend treatment if TSH exceeds 10 mIU/L, or earlier if high cardiovascular risk exists.
Clinical Goal: Target a TSH between 0.4 and 2.5 mIU/L for optimal heart health.
Can stress and high cortisol cause a heart attack?
Direct Answer: Yes. Chronic cortisol elevation drives atherosclerosis by promoting visceral fat deposition, insulin resistance, and systemic inflammation. Acute stress spikes can also trigger Takotsubo syndrome (stress-induced cardiomyopathy) or cause the sudden rupture of unstable arterial plaques due to sympathetic nervous system overdrive.
The Mechanism: Cortisol increases blood glucose and destabilizes existing plaque.
The Warning Sign: Reduced Heart Rate Variability (HRV) indicates autonomic strain.
Management: Mindfulness-based stress reduction (MBSR) and sleep optimization are validated clinical interventions.
Do women need to monitor testosterone for heart health?
Direct Answer: Absolutely. Testosterone is the most abundant pro-hormone in women and is essential for maintaining endothelial nitric oxide production. After menopause, the decline in testosterone contributes to increased visceral adiposity (belly fat) and a higher risk of metabolic syndrome and cardiovascular events.
Vasculoprotective Role: Helps keep female arteries flexible and responsive.
The Menopause Shift: Lack of testosterone accelerates the transition to an "android" (apple-shaped) fat distribution.
Screening: Women with unexplained fatigue and rising lipids should assess their free testosterone levels.
What is the "Endocrine Triangle" in cardiology?
Direct Answer: The Endocrine Triangle refers to the synergistic interaction between the Thyroid (HPT), Stress (HPA), and Gonadal (HPG) axes. Dysregulation in one axis, such as high cortisol, often suppresses the others, leading to a "perfect storm" of insulin resistance, low metabolism, and vascular inflammation.
Thyroid-Cortisol Link: High cortisol impairs the conversion of T4 to active T3
Stress-Testosterone Link: Chronic stress suppresses gonadotropin release, lowering testosterone.
Clinical Importance: Addressing a single hormone in isolation often fails if the entire "triangle" is not balanced.
What lifestyle changes reduce hormonal heart risk?
Direct Answer: The most effective lifestyle interventions focus on HPA axis rehabilitation and insulin sensitivity. This includes getting 7–9 hours of quality sleep, performing moderate-intensity aerobic exercise, and ensuring adequate protein intake to maintain lean muscle mass, which supports testosterone and metabolic rate.
Exercise Dose: Avoid chronic overtraining, which can paradoxically lower T3 and raise cortisol.
Nutrition: Focus on a high-protein, anti-inflammatory diet to support the "leucine threshold" for muscle repair.
Circadian Rhythm: Morning sunlight exposure helps regulate the cortisol awakening response (CAR).
The Clinical Crossroads: Essential Pearls
1. The "Reference Range" Trap
Lab "normal" ranges for TSH and Testosterone are often population averages, not health optimizations. A TSH of 4.2 mIU/L may be "statistically normal" but is associated with a 20% increase in LDL-C and impaired brachial artery flow-mediated dilation.
Just because your lab results aren't "red" or "out of range" doesn't mean your hormones are optimal for your heart. If you are at the edge of the range and feeling symptoms, your "normal" might actually be your "sub-optimal."
2. The Diurnal Cortisol "Slope"
A single morning serum cortisol check is a poor predictor of CV risk. The diurnal slope—the drop from morning peak to evening nadir—is the true marker of HPA health. A "flat" slope (elevated evening cortisol) is more predictive of coronary artery calcification than a high morning spike.
Your body’s stress hormone should be like a slide: high in the morning to wake you up and low at night to let you heal. If your stress stays "flat" all day, your heart never gets the "rest and digest" period it needs to repair arterial walls.
3. The Female Testosterone Shield
While often ignored in women, testosterone is the most abundant pro-hormone in the female body. In post-menopausal women, low testosterone is independently associated with increased visceral adiposity and a higher risk of HCE (Hard Cardiovascular Events), likely due to the loss of its protective effect on the vascular endothelium.
Testosterone isn't just for men. For women, it’s a vital "metabolic spark plug" that helps keep belly fat down and keeps your arteries flexible. After menopause, a drop in "T" can be just as hard on your heart as a drop in estrogen.
4. The Male Estrogen Paradox
In men, some testosterone is converted to estradiol via the aromatase enzyme. Suppressing estrogen too low (often seen in aggressive TRT protocols using aromatase inhibitors) is cardio-toxic, as estradiol is required in men for lipid metabolism and preventing arterial plaque buildup.
Men, don't be afraid of a little estrogen. While too much can cause issues, having some is actually a "heart-protector." If you’re on testosterone therapy and drive your estrogen to zero, you might be accidentally increasing your risk of a heart attack.
5. The "Goldilocks" Exercise Zone
While moderate aerobic exercise improves the HPT (Thyroid) axis and insulin sensitivity, chronic "extreme" volume (e.g., ultra-endurance) can induce Exercise-Induced Overstress (EIO), characterized by suppressed T3 levels and chronically elevated nocturnal cortisol, which paradoxically increases atrial fibrillation risk.
When it comes to your heart, you can actually have too much of a good thing. Intense, non-stop training without recovery can "burn out" your thyroid and keep your stress hormones high. Think of exercise as a medicine: the right dose heals, but an overdose can stress the heart.
6. The Sleep-Lipid Link
Sleep fragmentation (less than 6 hours) triggers a compensatory rise in evening cortisol and a suppression of growth hormone. This hormonal shift inhibits the activity of Lipoprotein Lipase (LPL), the enzyme responsible for clearing triglycerides from the blood, leading to post-prandial hyperlipidemia.
You can't out-diet a bad night's sleep. If you aren't sleeping, your hormones stay in "emergency mode," which physically prevents your body from clearing fat out of your bloodstream after you eat. Sleep is quite literally a "car wash" for your arteries
Female-Specific Cardiovascular Endocrinology
Testosterone in Women
Testosterone is essential in women for lean mass, insulin sensitivity, endothelial function, and libido.
Both low and high levels are cardiometabolically relevant:
Low testosterone → ↑ visceral fat, ↓ muscle mass, ↑ insulin resistance
High testosterone (e.g., PCOS) → ↑ ApoB, ↑ triglycerides, ↑ cardiometabolic risk
Emerging data suggest a U-shaped relationship, similar to men, though less well defined.
Clinical takeaway: Evaluate testosterone in women with unexplained metabolic dysfunction, PCOS, or fatigue syndromes.
Thyroid Dysfunction in Women
Thyroid disorders are 5–8× more common in women, especially with aging and autoimmunity.
Subclinical hypothyroidism is particularly prevalent in:
Postmenopausal women
Women with autoimmune disease (e.g., Hashimoto’s)
Cardiovascular implications:
↑ LDL-C and ApoB
↑ diastolic hypertension
↑ atherosclerosis progression
Clinical takeaway: A “normal” TSH in women—especially >2.5–3.0—may still carry cardiometabolic implications in the right context.
Cortisol and Menopause Interaction
Menopause is associated with:
Relative hypercortisolemia
Altered diurnal cortisol rhythm
↑ central adiposity and insulin resistance
Estrogen decline amplifies:
HPA axis reactivity
Sympathetic activation
This contributes to the postmenopausal surge in cardiovascular risk
Clinical takeaway: Address sleep, stress, and visceral fat aggressively in peri/postmenopausal women.
Controversy Section
Testosterone Replacement Therapy (TRT) Debate
Past concern: Increased CV events based on observational data
Current evidence (e.g., TRAVERSE RCT):
No significant increase in major adverse cardiovascular events
BUT ↑ risk of atrial fibrillation and thromboembolism
Key issue: dose and target range
Clinical consensus:Safe when used in true hypogonadism
Avoid supraphysiologic dosing
Monitor hematocrit, lipids, and CV status
Treating Subclinical Hypothyroidism (SCH)
Major debate: When to treat TSH 4.5–10 mIU/L
Arguments for treatment:
↑ LDL-C and ApoB
↑ atherosclerosis risk
Symptom burden (fatigue, weight gain)
Arguments against treatment:
Inconsistent RCT benefit on hard outcomes
Risk of overtreatment → atrial fibrillation, bone loss
Strong agreement only when:
TSH >10 mIU/L
Clinical approach:
Individualize based on:
Symptoms
Lipids/ApoB
Age and CV risk profile
Author’s Note
This chapter was written with a deliberate intention to challenge a deeply ingrained paradigm in cardiovascular medicine—the tendency to focus predominantly on downstream biomarkers while overlooking upstream biological drivers. In daily clinical practice, it is increasingly apparent that a subset of patients do not fit neatly into conventional risk models. They present with “acceptable” cholesterol, controlled blood pressure, and yet continue to progress toward cardiovascular disease. These are not outliers; they are signals that our current framework is incomplete.
The intersection between endocrinology and cardiology is not new, but it remains under-integrated in routine care. Hormones such as testosterone, thyroid hormones, and cortisol exert continuous, system-wide effects on vascular biology, metabolism, and autonomic regulation. What is often underappreciated is that even mild o subclinical disturbances—frequently dismissed as clinically insignificant—can, over time, reshape cardiovascular risk in meaningful ways. This chapter aims to bring that hidden layer into clearer focus.
It is important to emphasize that the goal is not to replace established tools such as lipid profiling or blood pressure management. These remain foundational. Rather, the objective is to expand the clinician’s lens—to encourage a more upstream, physiology-driven approach that integrates endocrine evaluation where appropriate. In doing so, we move closer to precision medicine: identifying not just what is abnormal, but why it is abnormal.
Finally, this work reflects an evolving body of evidence. Endocrine cardiology is still a developing field, and many questions remain unanswered. Clinicians are encouraged to apply these insights judiciously, in the context of individual patient profiles, while staying aligned with emerging research and guidelines.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Hormones and Heart Disease in Women: Hidden Risks After Menopause
GLP-1 Receptor Agonists and Cardiovascular Protection: Evidence from CVOT Trials Explained
ApoB vs LDL Cholesterol: Which Is the Most Accurate Predictor of Heart Disease?
Atherogenic Dyslipidemia Explained: The Role of ApoB in Hidden Cardiovascular Risk
Visceral Fat and Cardiovascular Risk: The Hidden Driver of Atherosclerosis
References
Júnior, R. F. R., Sawaki, D., Chan, L. F., & Czibik, G. (2025). Editorial: Endocrine insights into heart disease. Frontiers in Endocrinology, 16, 1664575. https://doi.org/10.3389/fendo.2025.1664575
Iglesias, P., & Iglesias, I. (2026). Hormonal crossroads of the heart: From classic endocrine regulation to cardiac hormone secretion: An updated review. Journal of Endocrinological Investigation, 49, 747–761. https://doi.org/10.1007/s40618-025-02767-2
Júnior, R. F. R., Sawaki, D., Chan, L. F., & Czibik, G. (2025). Editorial: Endocrine insights into heart disease. Frontiers in Endocrinology, 16, 1664575. https://doi.org/10.3389/fendo.2025.1664575
Tsai, S.-Y., Hsu, J.-Y., Lin, C.-H., Kuo, Y.-C., Chen, C.-H., Chen, H.-Y., Liu, S.-J., & Chien, K.-L. (2024). Association of stress hormones and the risk of cardiovascular diseases: Systematic review and meta-analysis. International Journal of Cardiology Cardiovascular Risk and Prevention, 23, Article 200305. https://doi.org/10.1016/j.ijcrp.2024.200305
Lincoff, A. M., Bhasin, S., Flevaris, P., Mitchell, L. M., Basaria, S., Boden, W. E., … & TRAVERSE Study Investigators. (2023). Cardiovascular safety of testosterone-replacement therapy. New England Journal of Medicine, 389(2), 107–117. https://doi.org/10.1056/NEJMoa2215025
Ding, N., Hua, R., Guo, H., Xu, Y., Yuan, Z., Wu, Y., & Li, T. (2025). Effect of thyroid stimulating hormone on the prognosis of coronary heart disease. Frontiers in Endocrinology, 16, 1433106. https://doi.org/10.3389/fendo.2025.1433106
An integrative approach to HPA axis dysfunction: From recognition to recovery. (2025). The American Journal of Medicine, 138(10), 1451–1463. https://doi.org/10.1016/j.amjmed.2025.05.044
Ruan, W., Zhou, X., Li, J., Wang, T., Liu, H., Zhang, G., & Lin, K. (2024). Thyroid function effect on cardiac structure, cardiac function, and disease risk: Evidence of causal associations in European ancestry. Heart Rhythm, 21(11), 2272–2281. https://doi.org/10.1016/j.hrthm.2024.07.015
Knezevic, E., Nenic, K., Milanovic, V., & Knezevic, N. N. (2023). The role of cortisol in chronic stress, neurodegenerative diseases, and psychological disorders. Cells, 12(23), 2726. https://doi.org/10.3390/cells12232726