Hormones and Heart Disease in Women: Hidden Risks After Menopause
Hormonal changes after menopause increase heart disease risk. Learn how estrogen, testosterone, thyroid, and cortisol affect your heart.
HEART
Dr. T.S. Didwal, M.D.(Internal Medicine)
4/9/202617 min read
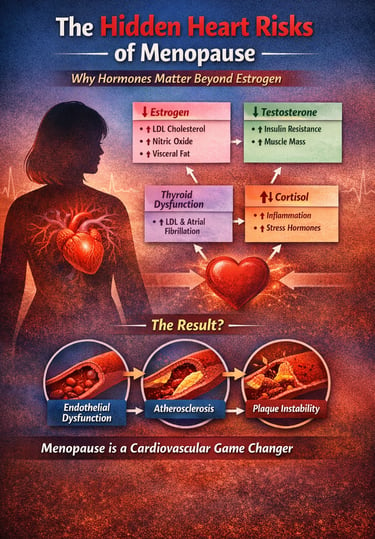
Hormonal changes after menopause do more than cause symptoms—they directly increase heart disease risk. The decline in estrogen, testosterone, and thyroid function, combined with elevated cortisol, disrupts blood vessels, cholesterol metabolism, inflammation, and fat distribution. This hormonal imbalance drives endothelial dysfunction, accelerates atherosclerosis, and increases the risk of heart attack and stroke. Understanding and addressing these hormonal shifts is essential for protecting cardiovascular health in women after menopause.
A Clinician’s Perspective on Hormones and Heart Disease in Women
Menopause is not a benign transition—it is a cardiometabolic inflection point.
In clinical practice, the sharp rise in cardiovascular events after menopause is striking. This is not merely an age-related risk; it reflects a coordinated hormonal shift that alters vascular biology, metabolism, and inflammatory tone in a clinically meaningful way.The estrogen-centric model is incomplete.
While estrogen decline impairs endothelial nitric oxide production and accelerates atherogenesis, focusing solely on estrogen risks missing the broader endocrine disruption. Testosterone, thyroid hormones, and cortisol form an interconnected network that modulates cardiovascular risk at multiple levels.Symptoms are subtle, but pathology is progressive.
Many women present with fatigue, central weight gain, poor sleep, or palpitations—symptoms often attributed to “normal menopause.” However, these may represent early manifestations of metabolic and vascular dysfunction. Delayed recognition remains a critical gap in preventive cardiology.Screening must evolve beyond traditional risk factors.
A comprehensive approach should include hormonal profiling alongside metabolic markers such as ApoB, insulin resistance indices, and inflammatory markers. This integrated model allows earlier identification of high-risk individuals who would otherwise be missed.Therapeutic strategies must be individualized and time-sensitive.
Evidence supports the concept of a “window of opportunity” for hormone replacement therapy, particularly when initiated early in menopause. Equally important are lifestyle interventions—resistance training, sleep optimization, and dietary patterns—that target the hormonal drivers of disease.The clinical imperative is clear: shift from reactive to proactive care.
Cardiovascular disease in postmenopausal women is not inevitable. With timely recognition of hormonal convergence and targeted intervention, clinicians can meaningfully alter the trajectory of disease and redefine prevention in women’s heart health.
For decades, cardiovascular disease in women was misunderstood, underdiagnosed, and frequently underestimated. Yet today, it stands as the leading cause of death among women worldwide—surpassing all cancers combined. What makes this reality even more concerning is that much of this risk accelerates silently during a period often framed as a natural life transition: menopause. Far from being merely a reproductive milestone, menopause represents a profound cardiometabolic turning point driven by complex hormonal shifts that extend well beyond estrogen alone (Guo & Lau, 2025).
Emerging evidence reveals that the decline in estrogen is only one part of a broader endocrine disruption. Testosterone levels fall, thyroid function becomes increasingly unstable, and cortisol regulation—governed by the hypothalamic-pituitary-adrenal axis—often becomes dysregulated. These hormonal changes do not occur in isolation; rather, they interact in a tightly interconnected network that directly influences vascular function, lipid metabolism, insulin sensitivity, inflammation, and fat distribution. The result is a convergence of risk pathways that collectively accelerate the development of atherosclerosis and cardiovascular disease (Gersh et al., 2024).
Critically, this transition is often clinically silent. Women may present with subtle symptoms—fatigue, weight gain, sleep disturbances, palpitations—that are frequently attributed to “normal aging” or menopause itself, delaying recognition of underlying cardiometabolic dysfunction. Meanwhile, adverse changes such as endothelial dysfunction, visceral fat accumulation, and low-grade inflammation continue to progress beneath the surface (Frank-Raue & Raue, 2023).
Understanding this hormonal convergence is not merely an academic exercise—it is a clinical imperative. By recognizing menopause as a window of both vulnerability and opportunity, clinicians and patients alike can shift from reactive treatment to proactive prevention, targeting the root hormonal drivers of cardiovascular risk before irreversible damage occurs.
The Hormonal Convergence Model of Postmenopausal Cardiovascular Risk
A systems-level framework integrating endocrine decline with cardiometabolic disease pathways:
Core Hormonal Disruptions
↓ Estrogen
↓ Endothelial nitric oxide (NO) production → impaired vasodilation
↑ LDL-C and ApoB-containing lipoproteins
↑ Visceral adiposity → pro-inflammatory adipokine profile
↓ Testosterone (in women)
↑ Insulin resistance → hyperinsulinemia
↓ Skeletal muscle mass → reduced glucose disposal
↑ Central fat accumulation → metabolic inflexibility
Thyroid Dysfunction (Hypo-/Hyperthyroidism)
Hypothyroidism → ↑ LDL, ↓ LDL receptor activity
Hyperthyroidism → ↑ risk of atrial fibrillation
Both → adverse cardiac remodeling and vascular effects
↑ Cortisol (HPA Axis Dysregulation)
↑ Chronic low-grade inflammation (↑ IL-6, TNF-α)
↑ Endothelial oxidative stress
↑ Hypertension, hyperglycemia, and visceral fat deposition
Converging Pathophysiological Pathways
Endothelial Dysfunction
Reduced NO bioavailability + increased oxidative stress
Impaired vascular reactivity and early vascular aging
Atherosclerosis Progression
Lipid abnormalities (↑ LDL, ApoB, triglycerides)
Chronic inflammation + endothelial injury → plaque formation
Plaque Instability
Inflammatory cytokines weaken fibrous cap
Increased risk of plaque rupture → myocardial infarction/stroke
Clinical Insight
These hormonal changes do not act in isolation—they amplify each other
Menopause represents a cardiometabolic inflection point, not just a reproductive transition
Early identification + targeted intervention can interrupt this convergence cascade and reduce long-term cardiovascular risk
Takeaway: Postmenopausal cardiovascular disease is best understood not as a single risk factor problem, but as a multi-hormonal systems failure driving vascular pathology.
Section 1: Testosterone in Women — Far More Than a "Male Hormone"
The Overlooked Role of Testosterone
When most people think of testosterone, they think of men. But testosterone is a vital hormone in women too — influencing muscle mass, bone density, mood, libido, and, critically, metabolic and cardiovascular health. Its role in women has been dramatically underestimated.
Vallejo and Palacios (2025) challenge the reductive framing of testosterone in women as merely a driver of sexual desire. Their research demonstrates that testosterone's physiological reach extends far beyond the bedroom — with meaningful implications for insulin sensitivity, lipid metabolism, vascular tone, and inflammatory regulation. When testosterone levels decline in perimenopause, these broader metabolic functions are compromised.
Mauvais-Jarvis (2025) further substantiates this in a precision medicine framework, documenting that testosterone therapy in menopausal women may improve metabolic markers including glucose tolerance and lipid profiles — key cardiovascular risk factors. Low testosterone in women has been associated with increased adiposity, insulin resistance, and dyslipidemia — the classic triad that sets the stage for cardiovascular disease.
A comprehensive review by the Pharmaceutical Journal on testosterone in menopause synthesizes clinical evidence suggesting that women with low testosterone may experience a more adverse cardiometabolic profile. Importantly, targeted testosterone therapy — when appropriately prescribed and monitored — shows promise in reversing some of these metabolic disruptions without significant adverse effects.
Clinical Pearl
Testosterone in women peaks in the mid-twenties and declines gradually, reaching its lowest levels in postmenopause. Many women are never tested for testosterone deficiency because clinicians focus solely on estrogen. This diagnostic blind spot may contribute to unrecognized cardiovascular risk.
Practical Application: Testosterone and Your Heart
Ask your healthcare provider to test total and free testosterone levels as part of a menopause hormone panel.
Lifestyle interventions — resistance training in particular — naturally support healthy testosterone levels and simultaneously benefit heart health.
Low testosterone can contribute to fatigue, weight gain, and reduced motivation for exercise — all factors that worsen cardiovascular outcome
Emerging short-term data support the use of low-dose transdermal testosterone (often with estrogen) for potential improvements in body composition and metabolic markers in carefully selected women, with reassuring short-term cardiovascular safety profiles. However, long-term cardiovascular outcome trials are still lacking, and guidelines emphasize its primary role in hypoactive sexual desire disorder. Use remains off-label in most regions and requires individualized monitoring. Emerging use but not yet universally recommended
Section 2: Thyroid Dysfunction — A Cardiovascular Threat Hidden in Plain Sight
Why Menopausal Women Face Cumulative Thyroid Risk
Thyroid disorders are significantly more common in women than in men, and their prevalence rises substantially around perimenopause and postmenopause. This timing is not coincidental — it reflects a convergence of hormonal shifts, immune system changes, and accumulated biological stressors.
Frank-Raue and Raue (2023) offer a compelling analysis of thyroid dysfunction in peri- and postmenopausal women, describing what they term cumulative risks — the compounding effect of age-related thyroid decline on top of the menopausal hormonal shift. Hypothyroidism, the most common thyroid disorder in this group, is associated with elevated LDL cholesterol, impaired cardiac contractility, diastolic dysfunction, and increased arterial stiffness. These are direct pathways to heart disease.
Hyperthyroidism, conversely, increases the risk of atrial fibrillation — an irregular heart rhythm that dramatically elevates stroke risk. Subclinical thyroid dysfunction, even when thyroid-stimulating hormone (TSH) falls within borderline ranges, can silently exert adverse cardiovascular effects. Many women in their 40s and 50s attribute thyroid symptoms — fatigue, weight gain, brain fog, palpitations — to menopause itself, delaying diagnosis by months or even years.
Did You Know?
It is estimated that up to 1 in 5 women over the age of 60 has some degree of thyroid dysfunction, yet a significant proportion remain undiagnosed. Routine TSH screening in perimenopausal women could prevent a meaningful proportion of avoidable cardiovascular events.
Practical Application: Protecting Your Heart Through Thyroid Health
• Insist on a comprehensive thyroid panel — TSH, free T3, free T4, and thyroid antibodies — at your annual wellness check, especially if you are perimenopausal.
• Persistent fatigue, cold sensitivity, hair loss, or unexplained weight gain in a menopausal woman should prompt thyroid evaluation before being attributed solely to hormonal changes.
• Treated thyroid dysfunction substantially reduces cardiovascular risk. Optimized thyroid function is a modifiable heart health lever.
• Selenium and iodine are key micronutrients for thyroid health; discuss supplementation needs with your provider.
Section 3: The Cortisol–Menopause Connection — Stress, Hormones, and the Inflamed Heart
How Menopause Resets the Stress Response
Cortisol is the body's primary stress hormone, produced by the adrenal glands in response to physical or psychological threat. In normal physiology, cortisol rises and falls in a predictable daily rhythm. In menopause, this rhythm is disrupted — often profoundly.
The relationship between estrogen and cortisol is bidirectional and complex. Estrogen modulates the hypothalamic-pituitary-adrenal (HPA) axis — the command-and-control system for cortisol release. As estrogen declines in menopause, HPA axis regulation becomes less precise. The result: many postmenopausal women experience chronically elevated or dysregulated cortisol levels, even without exceptional life stressors. This state, sometimes called functional hypercortisolism, carries significant cardiovascular consequences.
Guo and Lau (2025), writing in Current Treatment Options in Cardiovascular Medicine, identify hormonal disruption — including dysregulated cortisol — as a key mediator of the cardiovascular risk acceleration seen in menopausal women. Elevated cortisol promotes hypertension, hyperglycemia, dyslipidemia, and systemic inflammation — all established cardiovascular risk factors.
Gersh et al. (2024), publishing in Progress in Cardiovascular Diseases, extend this analysis by documenting how estrogen loss disrupts vascular endothelial function. The endothelium — the cellular lining of blood vessels — depends on estrogen for nitric oxide production, a key vasodilatory molecule. Without adequate estrogen to counterbalance cortisol's vasoconstrictive and pro-inflammatory effects, blood vessels become stiffer, less responsive, and more prone to plaque formation.
The Inflammation Connection
Chronic cortisol elevation does not just raise blood pressure — it drives low-grade systemic inflammation, a hallmark of atherosclerosis (coronary artery disease). Managing psychological stress, sleep quality, and adrenal health is therefore not a lifestyle luxury for postmenopausal women — it is a cardiovascular necessity.
Practical Application: Taming Cortisol for a Healthier Heart
• Prioritize sleep: cortisol dysregulation is strongly linked to poor sleep quality, which is itself a prevalent complaint in menopause. Eight hours of quality sleep is a genuine cardiac intervention.
• Mind-body practices — yoga, meditation, diaphragmatic breathing — have measurable cortisol-lowering effects and demonstrable cardiovascular benefits.
• Avoid prolonged high-intensity exercise if you are already under chronic stress; opt for moderate-intensity workouts that lower cortisol rather than spike it further.
• Adaptogenic herbs such as ashwagandha show preliminary evidence of HPA axis modulation, though clinical use should be discussed with a qualified provider.
• Consider saliva or urinary cortisol testing for a detailed view of your daily cortisol rhythm.
Section 4: The Visceral Fat Shift — When Hormones Reroute Body Fat to the Danger Zone
Understanding the Menopausal Fat Redistribution
One of the most clinically significant — and visually noticeable — changes of menopause is a shift in where the body stores fat. Pre-menopause, women typically store fat subcutaneously, distributed around the hips, thighs, and buttocks (gynoid or pear-shaped fat distribution). After menopause, fat redistribution occurs toward the abdomen — specifically, the deep visceral fat compartment surrounding internal organs.
This is not merely a cosmetic change. Visceral adipose tissue (VAT) is metabolically active in the most problematic sense: it secretes pro-inflammatory cytokines, free fatty acids, and adipokines that directly promote insulin resistance, dyslipidemia, hypertension, and vascular inflammation. It is now understood that visceral fat is an independent risk factor for cardiovascular disease, separate from body mass index (BMI).
Khalifey et al. (2026) in Frontiers in Reproductive Health document the cardiovascular impact of this hormonal-metabolic shift, noting that hormone replacement therapy (HRT) may partially attenuate visceral fat accumulation by restoring estrogenic metabolic signaling. Xia and Khalil (2025), writing in the International Journal of Molecular Sciences, provide molecular-level mechanistic evidence for how estrogen loss dysregulates lipid metabolism and promotes atherogenic lipid profiles — elevating LDL and triglycerides while reducing protective HDL.
Measure What Matters
BMI does not capture the cardiometabolic risk of visceral fat accumulation. Waist circumference greater than 35 inches (88 cm) in women is a more clinically relevant marker of cardiovascular risk. A DEXA scan or abdominal CT can precisely quantify visceral fat, though waist measurement remains a practical clinical tool.
Practical Application: Reducing Visceral Fat and Protecting Your Heart
• Reduce ultra-processed foods, refined carbohydrates, and sugar — the primary dietary drivers of visceral fat accumulation.
• Resistance training (2–3 sessions per week) is specifically effective at reducing visceral fat, even without overall weight loss.
• High-intensity interval training (HIIT) has been shown in multiple studies to selectively reduce abdominal and visceral fat in postmenopausal women.
• Discuss with your provider whether HRT is appropriate for you — beyond symptom management, it may also mitigate the metabolic consequences of menopausal fat redistribution.
• Monitor waist circumference monthly — more useful than body weight alone for tracking cardiovascular risk.
Hormone Replacement Therapy: A Nuanced Cardiovascular Conversation
No discussion of hormones and heart disease in women is complete without addressing hormone replacement therapy (HRT) — and the scientific nuance the topic demands. For years, HRT was broadly discouraged following a misinterpreted Women's Health Initiative study. The current evidence landscape is considerably more sophisticated.
Khalifey et al. (2026) and Xia and Khalil (2025) both emphasize what is now called the "timing hypothesis" or "window of opportunity": HRT initiated early in menopause (within ten years of final menstrual period or before age 60) is associated with reduced cardiovascular risk, improved vascular function, and more favorable metabolic outcomes. When initiated late, in women with established atherosclerosis, benefits are less certain, and risks may outweigh them.
The type of HRT matters too. Transdermal estrogen (patches, gels) carries a lower risk of blood clots compared to oral formulations. Micronised progesterone is preferred over synthetic progestins for cardiovascular safety. Individualized, precision-based HRT — tailored to a woman's specific hormonal profile, timing, risk factors, and preferences — represents the current state of the art in menopausal medicine. Recent data reinforce that transdermal estrogen (with or without progestogen) generally carries a more favorable cardiovascular profile than oral routes."
HRT is not indicated for primary CVD prevention — it's for symptom relief, with potential secondary CV benefits in the right window
Important Disclaimer
This article is for educational purposes and does not constitute medical advice. The decision to use hormone replacement therapy is a complex, individualized clinical decision that should be made in partnership with a qualified healthcare provider, taking into account your personal medical history, risk profile, and preferences.
Frequently Asked Questions
1. Does menopause increase the risk of heart disease?
Yes. The risk of heart disease in women escalates sharply after menopause due to the decline of estrogen, which previously protected the blood vessels. This hormonal shift leads to increased arterial stiffness, higher LDL cholesterol, and a redistribution of body fat to the visceral (abdominal) area, directly increasing cardiovascular risk.
2. What are the best hormone tests for women in menopause?
To accurately assess cardiovascular and hormonal health during menopause, clinicians recommend a comprehensive panel including:
Estradiol: To check estrogen levels.
Total and Free Testosterone: To assess metabolic and muscle health.
TSH, Free T3, and Free T4: To rule out thyroid-driven heart risks.
Cortisol: To evaluate HPA axis/stress response regulation.
Lipid Profile & HbA1c: To monitor cholesterol and blood sugar.
3. Is HRT safe for the heart after age 60?
According to the "Window of Opportunity" hypothesis, Hormone Replacement Therapy (HRT) is most effective and safest for the heart when initiated within 10 years of menopause onset or before age 60. Research suggests that early initiation supports vascular health, whereas starting HRT much later may not provide the same cardiac benefits and could carry higher risks for women with existing plaque buildup.
4. Why do women gain belly fat after menopause?
Postmenopausal weight gain in the midsection is driven by the loss of estrogen signaling, which causes the body to shift fat storage from the hips (subcutaneous) to the deep abdominal cavity (visceral). Visceral fat is an active endocrine tissue that releases inflammatory chemicals (cytokines), raising the risk of hypertension, insulin resistance, and heart disease.
5. How does low testosterone affect women's heart health?
While often viewed as a male hormone, testosterone in women is vital for maintaining insulin sensitivity and vascular tone. Low testosterone levels in postmenopausal women are associated with a loss of lean muscle mass, increased fat accumulation, and a more adverse "atherogenic" lipid profile, all of which contribute to a higher risk of coronary artery disease.
6. What is the link between thyroid health and heart disease in menopause?
Menopausal women face a "cumulative risk" where age-related thyroid decline overlaps with hormonal shifts. Hypothyroidism can lead to elevated LDL cholesterol and impaired heart pumping, while hyperthyroidism increases the risk of atrial fibrillation (AFib) and stroke. Because symptoms like fatigue and weight gain overlap with menopause, routine TSH screening is essential for cardiac prevention.
7. Should all postmenopausal women have their hormones tested?
Routine hormone screening is not universally recommended for all postmenopausal women, but it is highly advisable for those with cardiovascular risk factors, unexplained symptoms, or a family history of heart disease. A comprehensive panel including estradiol, testosterone (total and free), TSH, free T3, T4, DHEA-S, and cortisol provides a meaningful picture of hormonal health. Discuss your individual situation with your provider.
8 Is hormone replacement therapy safe for the heart?
The answer depends critically on timing, type, and individual risk profile. Current evidence from Khalifey et al. (2026) and Xia and Khalil (2025) supports that HRT initiated within the "window of opportunity" (early menopause, typically before age 60 or within 10 years of final period) is associated with cardiovascular benefit rather than harm. Transdermal estrogen and micronised progesterone are the preferred formulations for cardiovascular safety.Evidence is strongest for vasomotor symptom management in younger postmenopausal women, with CV effects being a potential bonus rather than the primary goal.
9. Why does fat shift to the abdomen after menopause, and why is that dangerous?
Estrogen plays a key role in directing fat to the hip and thigh region (subcutaneous fat), which is metabolically relatively inert. As estrogen declines in menopause, fat redistribution occurs toward the visceral (deep abdominal) compartment. Visceral fat is metabolically active — it secretes inflammatory molecules, disrupts insulin signaling, raises blood pressure, and promotes arterial plaque formation. This is why waist circumference is a better predictor of cardiovascular risk in postmenopausal women than body weight alone.
10. What lifestyle steps have the strongest evidence for protecting the heart after menopause?
The evidence is consistent across multiple studies: (1) Regular aerobic and resistance exercise reduces visceral fat, supports testosterone levels, and improves vascular function. (2) Mediterranean-style eating patterns reduce inflammation and support hormonal balance. (3) Prioritizing 7–9 hours of quality sleep lowers cortisol and supports cardiovascular recovery. (4) Stress management through mind-body practices reduces HPA axis dysregulation. (5) Avoiding smoking and limiting alcohol are non-negotiable. Taken together, these lifestyle pillars synergise with — and, in some cases, substitute for — pharmacological intervention.
Practical Applications at a Glance
The following evidence-based steps consolidate the practical guidance from each section of this article into an actionable framework for postmenopausal women:
GET TESTED
Request a comprehensive hormonal and metabolic panel including estradiol, total and free testosterone, TSH, free T3/T4, cortisol (ideally salivary or 24-hour urine), fasting glucose, HbA1c, lipid panel, and hs-CRP (high-sensitivity C-reactive protein, a marker of inflammation). These tests together paint a complete cardiovascular-hormonal picture.
MOVE YOUR BODY
Aim for at least 150 minutes of moderate aerobic activity per week, plus 2–3 sessions of resistance/strength training. This combination simultaneously targets visceral fat, supports testosterone, improves insulin sensitivity, and strengthens the heart.
EAT FOR YOUR HORMONES
A Mediterranean-style diet — rich in vegetables, legumes, whole grains, oily fish, olive oil, and low in ultra-processed food — supports estrogen metabolism, reduces cortisol-driven inflammation, and maintains healthy cholesterol and blood sugar levels.
SLEEP AS MEDICINE
Menopausal sleep disruption elevates cortisol, worsens insulin resistance, and accelerates visceral fat accumulation. Treating sleep disturbances — whether through HRT, cognitive behavioral therapy for insomnia (CBT-I), or environmental modification — is a genuine cardiovascular intervention.
HAVE THE HRT CONVERSATION
If you are within 10 years of menopause onset and experiencing symptoms, discuss HRT proactively with your provider. Do not wait for cardiovascular symptoms to develop. Present the evidence regarding the timing hypothesis and ask about transdermal estrogen and micronized progesterone options.
MONITOR YOUR WAIST
Measure your waist circumference monthly. Keep it below 35 inches (88 cm). This one simple metric tracks visceral fat change more meaningfully than the scale — and is among the most powerful predictors of cardiovascular risk in postmenopausal women.
Clinical pearls
1. The Testosterone Metabolic Nexus
In postmenopausal women, testosterone is not merely an androgenic precursor but a critical regulator of the GLUT4 glucose transporter and lipoprotein lipase activity. Low levels correlate with increased insulin resistance and a shift toward an atherogenic lipid profile.
Think of testosterone as a "metabolic spark plug." Even in women, it helps your body burn sugar and manage fat. When it drops during menopause, it becomes much easier to gain weight and harder for your body to process the foods you eat.
2. The "Window of Opportunity" (Timing Hypothesis)
The cardioprotective efficacy of Hormone Replacement Therapy (HRT) is dependent on the state of the vascular endothelium. Initiation within 10 years of menopause (or before age 60) preserves endothelial nitric oxide production, whereas late initiation may destabilize existing atherosclerotic plaques.
Timing is everything. Starting hormone therapy early in menopause acts like a shield for your blood vessels. However, if you wait too long—usually more than 10 years after your periods stop—the "shield" isn't as effective because the blood vessels have already undergone structural changes.
3. The Cortisol-Estrogen Seesaw
Estrogen provides a "buffer" for the Hypothalamic-Pituitary-Adrenal (HPA) axis. Its decline leads to a higher baseline of serum cortisol and an exaggerated response to stressors, promoting systemic inflammation (high-sensitivity C-reactive protein) and arterial stiffness.
Estrogen is like a volume knob for your stress response. When estrogen levels drop, the body’s "stress volume" gets stuck on high. This extra stress hormone doesn't just make you feel anxious; it actually makes your arteries stiffer and more prone to damage.
4. Visceral Fat as an Endocrine Organ
Menopausal fat redistribution from subcutaneous to visceral depots signifies a transition to a pro-inflammatory state. Visceral adipose tissue (VAT) functions as an active endocrine organ, secreting IL-6 and TNF-alpha, which directly contribute to endothelial dysfunction.
The weight shift to your midsection during menopause isn't just a cosmetic change; that "belly fat" is actually a factory for inflammation. It pumps out chemicals that irritate your heart and blood vessels, which is why your waist measurement is a more important health number than your weight on the scale.
5. Subclinical Thyroid Vigilance
Subclinical hypothyroidism (elevated TSH with normal FT4) is a significant, independent risk factor for diastolic heart failure and hypercholesterolemia in the aging female. The overlap between hypothyroid and menopausal symptoms necessitates a full thyroid panel (TSH, Free T3, Free T4, and TPO antibodies) to avoid diagnostic overshadowing.
Don't assume every "menopause symptom" is just menopause. A sluggish thyroid can mimic the fatigue, weight gain, and brain fog of menopause while quietly raising your cholesterol. A simple blood test can ensure your "engine" is still tuned correctly to protect your heart.
Author’s Note
This article was written from a clinician’s perspective with the goal of bridging the gap between rapidly evolving scientific evidence and real-world patient care. In everyday practice, it is increasingly clear that cardiovascular disease in women—particularly after menopause—is not adequately explained by traditional risk factors alone. Hormonal dynamics play a central, often underrecognized role, and this piece aims to bring that dimension into sharper clinical focus.
The concepts presented here are grounded in current peer-reviewed research and reflect an integrative understanding of endocrine and cardiovascular physiology. However, medicine is continually evolving, and recommendations—especially regarding hormone testing and hormone replacement therapy—must always be individualized. What is appropriate for one patient may not be suitable for another, depending on medical history, risk profile, and personal preferences.
Importantly, this article is not intended to replace clinical judgment or personalized medical advice. Instead, it is designed to empower both clinicians and patients with a deeper understanding of the hormonal drivers of cardiovascular risk, encouraging more informed discussions and proactive approaches to prevention.
If this work prompts you to ask better questions, seek more comprehensive evaluation, or reconsider how menopause is approached in cardiovascular care, then it has served its purpose.
TAKE ACTION NOW
Share this article with your doctor. Take your heart health into your own hands. Your heart has been working for you every moment of your life — now it is time to work for it.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Your Cholesterol Isn’t the Whole Story: The Hormone Imbalances Driving Heart Disease
GLP-1 Receptor Agonists and Cardiovascular Protection: Evidence from CVOT Trials Explained
ApoB vs LDL Cholesterol: Which Is the Most Accurate Predictor of Heart Disease?
Atherogenic Dyslipidemia Explained: The Role of ApoB in Hidden Cardiovascular Risk
Visceral Fat and Cardiovascular Risk: The Hidden Driver of Atherosclerosis
References
Frank-Raue, K., & Raue, F. (2023). Thyroid dysfunction in peri- and postmenopausal women — cumulative risks. Deutsches Arzteblatt International, 120(18), 311–316. https://doi.org/10.3238/arztebl.m2023.0069
Gersh, F., O'Keefe, J. H., Elagizi, A., Lavie, C. J., & Laukkanen, J. A. (2024). Estrogen and cardiovascular disease. Progress in Cardiovascular Diseases, 84(Suppl. C), 60–67. https://doi.org/10.1016/j.pcad.2024.01.003
Guo, R., & Lau, E. S. (2025). Matters of the heart: Menopause, hormones, and cardiovascular disease. Current Treatment Options in Cardiovascular Medicine, 27, 74. https://doi.org/10.1007/s11936-025-01131-0
Khalifey, H. T., Mahereen, R., Adwan, R., Chahine, R., Kaidali, M., Mirza, S. F., Tullah, S. N., Shaikh, S., Hammad, S., & Sukkarieh, H. H. (2026). The impact of hormone replacement therapy on cardiovascular health in postmenopausal women: A narrative review. Frontiers in Reproductive Health, 8, 1745210. https://doi.org/10.3389/frph.2026.1745210
Mauvais-Jarvis, F. (2025). Testosterone therapy and metabolic health in menopausal women. In F. Mauvais-Jarvis (Ed.), Principles of precision hormone therapy. Springer. https://doi.org/10.1007/978-3-031-89650-7_12
Pharmaceutical Journal. (n.d.). Testosterone in menopause: A review of the evidence and prescribing practice. https://pharmaceutical-journal.com/article/research/testosterone-in-menopause-a-review-of-the-evidence-and-prescribing-practice
Vallejo, M. S., & Palacios, S. (2025). Testosterone in women: Beyond hypoactive sexual desire. Gynecological Endocrinology, 41(1). https://doi.org/10.1080/09513590.2025.2592402
Xia, W., & Khalil, R. A. (2025). Hormone replacement therapy and cardiovascular health in postmenopausal women. International Journal of Molecular Sciences, 26(11), 5078. https://doi.org/10.3390/ijms26115078