Can GLP-1 Drugs Prevent Heart Attacks? New Trial Data Changes Everything
Learn how GLP-1 drugs like semaglutide may lower heart attack risk, who should consider them, and what the latest research shows.
HEART
Dr. T.S. Didwal, M.D.(Internal Medicine)
4/7/202618 min read
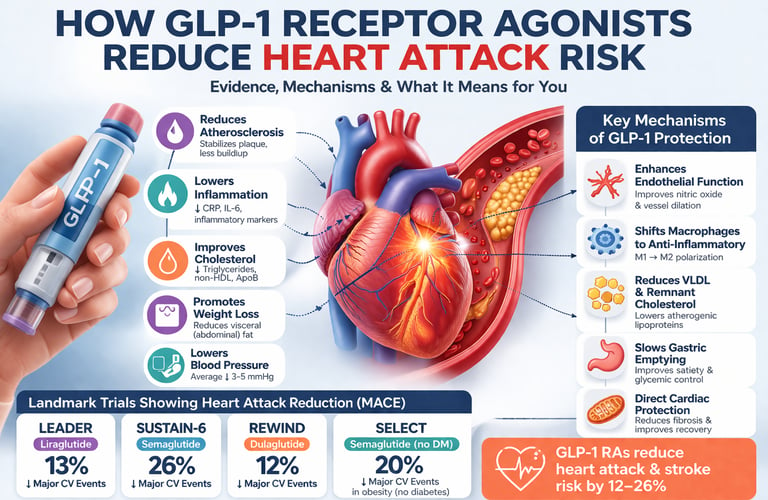
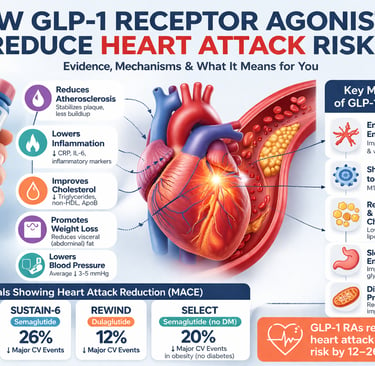
GLP-1 receptor agonists reduce heart attack risk by 12–26% in large clinical trials by targeting atherosclerosis, inflammation, and visceral fat—effects largely independent of blood sugar control.
Key Cardiovascular Outcomes at a Glance
~14% reduction in MACE
→ Observed with liraglutide in the LEADER trial~26% reduction in cardiovascular death
→ One of the most significant mortality benefits seen in LEADER~20% reduction in MACE
→ Demonstrated with semaglutide 2.4 mg in the SELECT trial (non-diabetic population)~12% reduction in MACE
→ Reported with dulaglutide in the REWIND trial
Landmark Trial Snapshot
LEADER Trial
Drug: Liraglutide
Participants: 9,340
Duration: 3.8 years
Outcome: Positive (significant MACE reduction and CV mortality benefit)
SUSTAIN-6 Trial
Drug: Semaglutide
Participants: 3,297
Duration: 2.1 years
Outcome: Positive (notable reduction in MACE, particularly stroke)
REWIND Trial
Drug: Dulaglutide
Participants: 9,901
Duration: 5.4 years
Outcome: Positive (benefit extends to primary prevention population)
SELECT Trial
Drug: Semaglutide 2.4 mg
Participants: 17,604
Duration: 3.3 years
Outcome: Positive (significant MACE reduction in obesity without diabetes)
Clinical Note
MACE (Major Adverse Cardiovascular Events) includes:
cardiovascular death, nonfatal myocardial infarction, and nonfatal strokeReported values represent approximate relative risk reductions based on published cardiovascular outcome trials
Clinician’s perspective: GLP-1 Receptor Agonists and the Redefinition of Cardiovascular Risk
We are witnessing a paradigm shift in cardiovascular prevention. For decades, risk reduction has centered on LDL cholesterol and blood pressure. Yet patients with well-controlled LDL and HbA1c continue to experience myocardial infarctions, highlighting the limitations of traditional risk models (Rosen & Ingelfinger, 2026).
GLP-1 receptor agonists challenge the glucose-centric view of diabetes care. These agents demonstrate that targeting upstream metabolic dysfunction—particularly visceral adiposity and insulin resistance—can yield cardiovascular benefits that exceed those expected from glycemic control alone (Drucker, 2025).
The magnitude and consistency of cardiovascular outcome trial data are difficult to ignore. Across trials such as LEADER, SUSTAIN-6, REWIND, and SELECT, reductions in major adverse cardiovascular events range from 12% to 26%, with signals extending to non-diabetic populations (Moiz et al., 2025; Jackson-Morris et al., 2026).
Mechanistically, these drugs act where traditional therapies do not. Beyond lipid lowering, GLP-1 RAs modulate endothelial function, reduce vascular inflammation, improve lipoprotein quality, and directly influence atherosclerotic plaque biology—key drivers of residual cardiovascular risk (Yang et al., 2026).
This class is particularly relevant in the modern phenotype of cardiometabolic disease. Patients with obesity, metabolic syndrome, and atherogenic dyslipidemia represent a growing proportion of cardiovascular cases—precisely the population in which GLP-1 therapies show the greatest impact.
However, clinical adoption must remain nuanced. Cost, tolerability, and long-term adherence remain real-world barriers. Furthermore, these agents complement—not replace—foundational therapies such as statins and antihypertensives.
The SELECT trial may ultimately redefine eligibility for pharmacologic prevention. Treating obesity as a primary cardiovascular risk factor—independent of diabetes—represents a conceptual shift with far-reaching implications for guidelines and public health strategy.
The challenge now is implementation. Identifying the right patient, at the right time, with the right combination of therapies will determine whether this scientific breakthrough translates into population-level impact.
A New Era in Heart Disease Prevention
For decades, the prevention of heart attacks has followed a familiar and largely effective script: lower LDL cholesterol with statins, control blood pressure, and encourage lifestyle change. Yet despite these interventions, cardiovascular disease remains the leading cause of death worldwide, exposing a critical gap in our ability to address the deeper metabolic drivers of atherosclerosis (Rosen & Ingelfinger, 2026). In particular, individuals with obesity, insulin resistance, and type 2 diabetes continue to experience disproportionately high rates of myocardial infarction—even when traditional risk factors appear well controlled. This paradox has forced clinicians and researchers to reconsider whether targeting glucose and LDL cholesterol alone is sufficient.
Enter glucagon-like peptide-1 receptor agonists (GLP-1 RAs), a class of therapies originally developed for glycemic control that has rapidly emerged as one of the most transformative advances in cardiometabolic medicine. Over the past decade, large-scale cardiovascular outcome trials have demonstrated that these agents reduce major adverse cardiovascular events—including heart attacks and strokes—by approximately 12–26%, with benefits that extend beyond glucose lowering (Moiz et al., 2025; Yang et al., 2026). Perhaps most strikingly, the recent SELECT trial showed a 20% reduction in cardiovascular events in individuals with obesity but without diabetes, fundamentally reshaping our understanding of cardiovascular prevention (Jackson-Morris et al., 2026).
These findings signal a paradigm shift: heart disease is not merely a lipid or pressure problem—it is a complex metabolic and inflammatory disorder. GLP-1 receptor agonists appear to target this root biology directly, influencing atherosclerosis, systemic inflammation, and visceral adiposity through mechanisms that are only beginning to be fully understood (Drucker, 2025)
1. What Are GLP-1 Receptor Agonists?
GLP-1 is a hormone naturally produced in the gut in response to food intake. It stimulates insulin secretion, suppresses glucagon, slows gastric emptying, and sends satiety signals to the brain. GLP-1 receptor agonists are synthetic versions of this hormone — engineered to be more potent and longer-lasting than the body's own GLP-1, which has a half-life of only a few minutes.
Agents in this class include:
Liraglutide (Victoza/Saxenda)
Semaglutide (Ozempic/Wegovy — injectable; Rybelsus — oral)
Dulaglutide (Trulicity)
Exenatide (Byetta/Bydureon)
Tirzepatide (Mounjaro/Zepbound — dual GLP-1/GIP agonist)
As Rosen & Ingelfinger (2026) outline in the New England Journal of Medicine, these agents represent a major pharmacological shift — from glucose-lowering tools to broad-spectrum cardiometabolic therapies with pleiotropic effects on the heart, vasculature, kidneys, and brain. Their expanding therapeutic footprint has prompted a complete rethinking of how we approach chronic cardiometabolic disease (Moiz et al., 2025; Drucker, 2025).
2. The Cardiovascular Outcome Trials: What the Evidence Shows
The story of GLP-1 RAs and heart disease is inseparable from a series of large, rigorous randomized controlled trials known as cardiovascular outcome trials (CVOTs). Designed primarily to prove cardiovascular safety (following regulatory mandates after concerns about earlier diabetes drugs), these trials ended up demonstrating something far more exciting: genuine cardiovascular benefit.
LEADER Trial (Liraglutide)
The LEADER trial enrolled 9,340 participants with type 2 diabetes and high cardiovascular risk over a median follow-up of 3.8 years. Liraglutide reduced the primary composite endpoint — major adverse cardiovascular events (MACE: cardiovascular death, nonfatal myocardial infarction, and nonfatal stroke) — by approximately 13% compared to placebo. Crucially, cardiovascular death was reduced by about 22%, and all-cause mortality also declined significantly.
SUSTAIN-6 Trial (Semaglutide)
The SUSTAIN-6 trial tested once-weekly injectable semaglutide in 3,297 patients with type 2 diabetes and established cardiovascular disease or high risk. MACE was reduced by approximately 26%, driven primarily by a reduction in nonfatal stroke. The trial was shorter (2.1 years) and somewhat underpowered for individual MACE components, but the signal was strong and consistent.
REWIND Trial (Dulaglutide)
REWIND distinguished itself by including a broader population — notably, a larger proportion of patients without prior cardiovascular events (primary prevention). Over 5.4 years, dulaglutide reduced MACE by approximately 12%. Importantly, this benefit extended to patients without established cardiovascular disease, suggesting a role in primary prevention.
SELECT Trial (Semaglutide in Non-Diabetics)
Perhaps the most practice-changing of all, the SELECT trial enrolled 17,604 individuals with established cardiovascular disease and obesity — but without diabetes. Semaglutide 2.4 mg reduced MACE by approximately 20%. This was a landmark result: a glucose-lowering drug reducing cardiovascular events in people who don't have diabetes. More on this in Section 6.
3. How Do GLP-1 RAs Reduce Heart Attack Risk? (The Mechanisms)
Understanding why these drugs work requires going beyond glycemic control. As Yang et al. (2026) demonstrated in a comprehensive umbrella review of meta-analyses published in JAMA Network Open, GLP-1 RAs produce a remarkable array of beneficial effects across multiple organ systems — many of which are independent of blood sugar lowering.
3.1 Anti-Atherosclerotic Effects
GLP-1 receptors are expressed directly on endothelial cells, vascular smooth muscle cells, and macrophages within arterial walls. Activation of these receptors improves nitric oxide bioavailability, reduces endothelial dysfunction, and suppresses the foam cell formation that underpins atherosclerotic plaque development.
Animal and imaging studies have shown that GLP-1 RAs reduce carotid intima-media thickness — a surrogate marker of atherosclerosis — and appear to stabilize coronary plaques by strengthening fibrous caps and reducing necrotic core size. These are not just cosmetic effects; vulnerable plaques that rupture are the direct cause of most heart attacks.
3.2 Anti-Inflammatory Actions
Chronic low-grade inflammation is now recognized as a central driver of atherosclerosis and acute cardiovascular events. GLP-1 RAs reduce circulating levels of C-reactive protein (CRP), interleukin-6 (IL-6), and other inflammatory markers. They also modulate macrophage phenotype — shifting cells from pro-inflammatory (M1) to anti-inflammatory (M2) states within plaques (Saran et al., 2026).
This anti-inflammatory signature may help explain why cardiovascular event curves in the major trials begin to separate earlier than would be expected if weight loss alone were driving the benefit.
3.3 Lipoprotein Remodeling and Triglyceride Reduction
GLP-1 RAs reduce hepatic VLDL output and increase lipoprotein lipase (LPL) activity, resulting in significant reductions in fasting and postprandial triglycerides — sometimes in the range of 20–30%. This matters because remnant cholesterol carried by triglyceride-rich lipoproteins is an underappreciated driver of residual cardiovascular risk, particularly in people with normal LDL cholesterol. Non-HDL cholesterol and ApoB-containing particles also decline with GLP-1 therapy (Ilias et al., 2025).
3.4 Blood Pressure and Weight Reduction
These agents produce significant and sustained weight loss — particularly visceral fat reduction — which in turn reduces insulin resistance, lowers systemic blood pressure (typically by 3–5 mmHg), and reduces cardiac workload. Visceral fat is a metabolically active organ that secretes adipokines, fuels systemic inflammation, and drives atherogenesis. Shrinking it is not a cosmetic benefit — it is a vascular benefit.
3.5 Direct Cardiac Effects
There is emerging evidence that GLP-1 receptors in cardiac tissue may confer direct cardioprotective effects — including reduced cardiac fibrosis and improved post-ischemic recovery. Whether this translates meaningfully to clinical outcomes in humans remains an active area of investigation (Drucker, 2025).
4. Are the Cardiovascular Benefits Independent of Blood Sugar Lowering?
This is one of the most important mechanistic questions in the field — and the evidence increasingly says yes. Several lines of evidence support independence from glycemic effects:
First, in the LEADER and SUSTAIN-6 trials, HbA1c differences between the GLP-1 and placebo groups were modest (typically 0.4–0.8%), yet the cardiovascular benefit was substantially larger than what could be attributed to glycemic improvement alone.
Second, and most compellingly, the SELECT trial demonstrated a 20% MACE reduction in people without diabetes — people for whom glycemic effects are essentially irrelevant. As Jackson-Morris et al. (2026) argue in The Lancet Global Health, this firmly repositions GLP-1 RAs as cardiometabolic drugs, not merely antidiabetic drugs.
Third, the early separation of event curves in some trials — occurring within months of drug initiation — suggests mechanisms beyond the slow, gradual changes expected from glucose control or even meaningful weight loss.
5. Who Benefits Most? Clinical Selection and Risk Stratification
Not all patients benefit equally, and understanding who gains the most from GLP-1 RA therapy is essential for precision prescribing.
The greatest benefit has been observed in patients with:
Established atherosclerotic cardiovascular disease (ASCVD): prior MI, stroke, or peripheral arterial disease
Obesity (BMI ≥ 27) with additional cardiovascular risk factors
Type 2 diabetes with high-risk features (elevated HbA1c, long disease duration, kidney disease)
Emerging benefits have been demonstrated in:
Obesity without diabetes (SELECT trial population)
Patients with heart failure with preserved ejection fraction (HFpEF) — an area of active investigation
More uncertain benefits exist in:
Primary prevention populations without ASCVD or high-risk features
Patients with BMI < 27
As Moiz et al. (2025) detail in their narrative review in eClinicalMedicine, the positioning of GLP-1 RAs in clinical guidelines is rapidly evolving — with the American Diabetes Association, European Society of Cardiology, and other bodies now recommending GLP-1 RAs as first-line agents for patients with type 2 diabetes and established cardiovascular disease, irrespective of glycemic control targets.
6. The SELECT Trial: A Paradigm Shift for Non-Diabetics
The SELECT trial (Semaglutide Effects on Cardiovascular Outcomes in People with Overweight or Obesity) deserves particular attention. It enrolled 17,604 adults aged 45 or older with a BMI ≥ 27, established cardiovascular disease, but without type 2 diabetes. After a median of 3.3 years, weekly subcutaneous semaglutide 2.4 mg reduced the risk of cardiovascular death, nonfatal MI, or nonfatal stroke by 20%.
This was not driven solely by weight loss. Statistical analyses accounting for weight change showed residual cardiovascular benefit, suggesting that anti-inflammatory and direct vascular mechanisms play a meaningful independent role (Jackson-Morris et al., 2026).
The SELECT trial has fundamentally changed the conversation: GLP-1 RAs are no longer just for people with type 2 diabetes. They represent a new class of cardiovascular prevention therapy for high-risk individuals with obesity — a population that represents hundreds of millions of people worldwide.
7. Practical Applications: What This Means in Real Life
Important Points for Patients
Ask your doctor if you qualify
If you have type 2 diabetes along with a history of heart attack, stroke, or established heart disease — or if you have obesity with additional cardiovascular risk factors — ask your doctor specifically about GLP-1 receptor agonists. Current guidelines recommend these medications for heart protection, not just blood sugar control.
Don’t stop your existing medications
GLP-1 receptor agonists work alongside, not instead of, your current treatments. Continue taking your statins, blood pressure medicines, and other prescribed heart medications. Think of GLP-1 therapy as an extra layer of protection for your heart.
Set realistic expectations about side effects
Nausea, reduced appetite, and mild stomach discomfort are common, especially during the first few weeks when the dose is being increased. Starting with a low dose and increasing slowly helps most people tolerate it better. These symptoms usually improve within 4–8 weeks.
Guidance for Clinicians
Reframe the conversation — it’s not just a diabetes drug
For patients with established heart disease (ASCVD) and obesity — even without diabetes — GLP-1 receptor agonists (especially semaglutide 2.4 mg) now have strong Class I evidence for reducing cardiovascular risk. Use data from the SELECT trial to guide discussions with suitable patients
.
Individualize therapy: GLP-1 vs SGLT2 based on patient profile
Prefer GLP-1 receptor agonists for patients where obesity and atherosclerotic cardiovascular risk are the main concerns.
Prefer SGLT2 inhibitors for patients with heart failure with reduced ejection fraction (HFrEF) or chronic kidney disease (CKD).
Combination therapy (GLP-1 + SGLT2) may provide additional benefits in selected high-risk patients.
8. Safety Considerations and Honest Risk-Benefit Framing
No medication is without risk, and GLP-1 RAs are no exception. The most common adverse effects are gastrointestinal: nausea, vomiting, diarrhea, and constipation. These are generally dose-dependent, most prominent during initial dose escalation, and improve significantly over time for most patients.
More serious but rare concerns include:
Pancreatitis: While earlier observational data raised concerns, large meta-analyses have not confirmed a significantly elevated risk of acute pancreatitis. Nonetheless, GLP-1 RAs are generally avoided in patients with a history of pancreatitis or severe hypertriglyceridemia.
Gallbladder disease: Rapid weight loss — from any cause — increases gallstone risk. GLP-1 RAs are associated with a modest increase in cholecystitis and cholelithiasis events across trials.
Thyroid C-cell concerns: In rodent models, GLP-1 RAs stimulate thyroid C-cell proliferation and increase medullary thyroid carcinoma risk. Human data have not replicated this finding, and the absolute risk (if any) appears extremely low. However, GLP-1 RAs are contraindicated in patients with a personal or family history of medullary thyroid carcinoma or MEN2.
As Ilias et al. (2025) frame it in Frontiers in Clinical Diabetes and Healthcare, GLP-1 RAs represent a double-edged sword of innovation and risk — extraordinary efficacy coupled with a side effect profile that requires thoughtful dose titration and patient education. For most high-risk cardiovascular patients, the benefit-risk balance is strongly favorable.
Health Economics: "Value" vs. "Cost"
The "Financial Toxicity" of GLP-1 drugs remains the primary barrier to population-level impact.
The Price Gap: Generic statins cost pennies a day, whereas GLP-1 RAs are premium-priced therapies.
Systemic Value: While these drugs could save $15–20 billion annually in heart attack and stroke care, the current cost-effectiveness ratio often exceeds $100,000 per Quality-Adjusted Life Year (QALY).
The Future: True cost-effectiveness will likely arrive only as biosimilars enter the market or as healthcare systems negotiate "value-based" pricing models.
9. The Future: Dual and Triple Agonists
The next frontier in this space involves agents that target multiple incretin receptors simultaneously. Tirzepatide — a dual GLP-1/GIP agonist — produces greater weight loss than any prior GLP-1 RA and has demonstrated impressive glycemic control in the SURPASS trials. Its cardiovascular outcome trial (SURPASS-CVOT) is ongoing. Early data and mechanistic reasoning suggest its cardiovascular benefits will likely exceed those of GLP-1 RAs alone.
Beyond tirzepatide, retatrutide — a triple agonist targeting GLP-1, GIP, and glucagon receptors — is producing weight losses approaching 25% of body weight in early trials. As Saran et al. (2026) note in Molecular Diversity, these next-generation agents represent the frontier of cardiometabolic pharmacology, with the potential to make obesity a genuinely treatable chronic disease at scale.
Tirzepatide (SURPASS-CVOT) vs. Semaglutide
The 2026 data from the SURPASS-CVOT trial has finally settled the head-to-head debate between "Dual" and "Single" agonists.
Metabolic Superiority: Tirzepatide (GLP-1/GIP) is the clear winner for weight loss and HbA1c reduction.
Cardiovascular Parity: Despite the extra metabolic "punch," Tirzepatide demonstrated non-inferiority but not clear superiority over standard GLP-1s for heart protection.
The Clinical Choice: For a patient focused on pure heart protection, semaglutide remains the gold standard. For a patient requiring maximal weight loss alongside heart protection, Tirzepatide is the preferred tool.
Precision medicine approaches — using genetic markers, metabolic phenotyping, and AI-driven risk clustering — may eventually allow clinicians to predict which patients will derive the greatest cardiovascular benefit from specific agents, further optimizing outcomes (Jackson-Morris et al., 2026).
10. What the Guidelines Currently Recommend
Current guidance from major cardiology and endocrinology bodies (including the American Diabetes Association, American College of Cardiology, and European Society of Cardiology) reflects the accumulating trial evidence:
GLP-1 RAs with proven cardiovascular benefit are recommended as first-line add-on therapy for patients with type 2 diabetes and established ASCVD, independent of HbA1c targets.
Semaglutide 2.4 mg (Wegovy) is now approved specifically for cardiovascular risk reduction in adults with obesity and established cardiovascular disease.
Guidelines are expected to continue evolving rapidly as data from ongoing trials mature.
Key Takeaway Summary Box
GLP-1 receptor agonists reduce heart attack risk through multiple complementary mechanisms — including atherosclerosis reduction, anti-inflammatory effects, lipoprotein remodeling, blood pressure lowering, and visceral fat loss — that are substantially independent of their glucose-lowering properties. Multiple large cardiovascular outcome trials confirm clinically meaningful MACE reductions of 12–26%, now extending to people without diabetes.
Clinical Insight: Why ARR and NNT Matter
While relative risk reductions (e.g., 12–26% reduction in MACE) are commonly reported in cardiovascular outcome trials, they can overestimate the perceived magnitude of benefit if not interpreted in context. Absolute Risk Reduction (ARR) provides a more clinically meaningful measure by reflecting the actual difference in event rates between treatment and placebo groups.
From ARR, we derive the Number Needed to Treat (NNT)—the number of patients who must be treated over a specific time period to prevent one adverse event. This metric translates trial data into real-world clinical decision-making.
For example, in the LEADER trial:
ARR ≈ 1.9%
NNT ≈ 53 over 3.8 years
This means that treating 53 high-risk patients with liraglutide for nearly 4 years would prevent one major cardiovascular event.
In practice, lower NNT values indicate greater clinical impact, particularly in high-risk populations such as patients with established atherosclerotic cardiovascular disease. Incorporating ARR and NNT alongside relative risk provides a more balanced, evidence-based perspective—essential for both clinician judgment and patient counseling.
Absolute Risk: The NNT Reality Check
Statins in Primary Prevention (Moderate Risk): * Absolute Risk Reduction (ARR): ~1.1% over 5 years.
NNT: 90. You must treat 90 people for 5 years to prevent one heart attack or stroke.
Statins in Secondary Prevention (Post-MI): * ARR: ~3.5% over 5 years.
NNT: 28. In patients with established disease, statins are highly efficient "heavy lifters."
Semaglutide in Obesity without Diabetes (SELECT Trial): * ARR: ~1.5% over 3.3 years.
NNT: 67. For high-risk patients with obesity, the benefit is significant and additive to existing statin therapy.
Liraglutide in Type 2 Diabetes (LEADER Trial): * ARR: ~1.9% over 3.8 years.
NNT: 53. In diabetic populations with high CV risk, the impact is even more pronounced.
FAQs
Do GLP-1 drugs prevent heart attacks in non-diabetics?
Yes. Clinical evidence, specifically from the SELECT trial, shows that GLP-1 receptor agonists like semaglutide (Wegovy) can reduce the risk of major adverse cardiovascular events (MACE) by 20% in adults with obesity and established heart disease, even if they do not have diabetes.
How quickly do GLP-1 medications provide heart protection?
Cardiovascular benefits can begin to emerge within the first few months of treatment. While weight loss takes time, the drug’s anti-inflammatory effects and improvements in vascular function (endothelial health) provide early protective signals, as seen in the rapid separation of event curves in clinical trials.
GLP-1 RAs vs. SGLT2 Inhibitors: Which is better for the heart?
The choice depends on the specific cardiovascular risk:
GLP-1 RAs: Best for preventing atherosclerotic events like heart attacks and strokes.
SGLT2 Inhibitors: Best for preventing heart failure hospitalizations and slowing kidney disease.
Many high-risk patients are now prescribed both to provide comprehensive "dual-organ" protection.
Is GLP-1 therapy a lifelong commitment for heart health?
Current clinical data suggests that the heart-protective benefits are maintained through long-term use. Because obesity and cardiovascular disease are chronic conditions, stopping the medication often leads to weight regain and a potential loss of the metabolic and inflammatory improvements provided by the drug.
Can I stop taking my statin if I start a GLP-1 drug?
No. GLP-1 receptor agonists and statins are complementary, not interchangeable. Statins lower LDL cholesterol (the "building blocks" of plaque), while GLP-1s reduce inflammation and stabilize existing plaques. Using them together provides the most significant reduction in heart attack risk.
What are the most common side effects of GLP-1 heart drugs?
The most frequent side effects are gastrointestinal, including:
Nausea
Vomiting
Diarrhea
Constipation
These symptoms are usually temporary and can be managed by slowly titrating the dose and eating smaller, low-fat meals.
Who benefits most from GLP-1 cardiovascular therapy?
According to current medical guidelines, the greatest benefit is seen in:
Patients with established ASCVD (prior heart attack or stroke).
Individuals with Type 2 Diabetes and high cardiovascular risk.
Adults with obesity (BMI ≥27) and pre-existing heart disease.
Clinical pearls
1. The "Beyond the Sugar" Effect
Scientific Tone: The cardiovascular benefits of GLP-1 RAs are largely pleiotropic and independent of glycemic control. Trials like SELECT demonstrate that MACE reduction occurs via anti-inflammatory pathways and plaque stabilization rather than just $HbA1c$ lowering.
Think of these meds as a "multitasker." They don't just lower your blood sugar; they act like a cooling system for your arteries, reducing the inflammation that causes "rust" (plaque) to build up in your heart.
2. Atherosclerosis vs. Heart Failure
Scientific Tone: GLP-1 RAs are primarily anti-atherosclerotic (reducing MI and stroke), whereas SGLT2 inhibitors are primarily hemodynamic (reducing heart failure hospitalizations). For patients with "cold" plumbing issues (clogs), GLP-1s are the superior choice.
Patient-Friendly Tone: There are two ways the heart fails: "clogs" and "pumps." These drugs are experts at preventing the clogs that lead to heart attacks. If your main risk is a blockage, this is often the preferred tool in the toolkit.
3. The "Slow and Low" Titration Rule
Scientific Tone: Gastrointestinal (GI) tolerability is the primary barrier to persistence. Dose titration should follow a four-week minimum interval to allow for the down-regulation of GLP-1 receptors in the area postrema of the brain and the gut, minimizing nausea.
Patient-Friendly Tone: Your body needs time to "make friends" with this medicine. If you rush the dose, your stomach will rebel. Starting with a tiny amount and increasing it slowly every month helps your brain and gut adjust without making you feel sick.
4. Protecting the "Engine" (Muscle Mass)
Scientific Tone: Rapid weight loss induced by GLP-1 RAs can lead to sarcopenic obesity if lean muscle mass is not preserved. Clinicians should mandate a minimum protein intake of $1.2\text{--}1.5\text{ g/kg}$ and resistance training to maintain the basal metabolic rate.
Patient-Friendly Tone: When you lose weight fast, your body might accidentally burn muscle instead of just fat. To keep your heart (which is a muscle!) and your strength healthy, you must eat plenty of protein and do some strength exercises, like lifting light weights or using resistance bands.
5. The "Fullness" Feedback Loop
Scientific Tone: GLP-1 RAs delay gastric emptying and enhance postprandial satiety. Patients should be counseled to "eat to the signal," as overriding the satiety cue can lead to acute gastric distension and subsequent vomiting.
Patient-Friendly Tone: This drug gives your brain a "stop eating" signal much earlier than usual. Listen to it! If you try to finish a large plate out of habit even when you feel full, you’re likely to end up feeling nauseated or reaching for the Tums later.
6. Not a Statin Replacement
Scientific Tone: GLP-1 RAs do not address LDL-cholesterol through the HMG-CoA reductase pathway. They are complementary to, not a substitute for, statin therapy. Synergy between lipid-lowering and GLP-1-mediated plaque stabilization offers the maximal RRR (Relative Risk Reduction).
Patient-Friendly Tone: This is an "and," not an "instead of." While this drug helps with weight and inflammation, it doesn't scrub the cholesterol out of your blood the way a statin does. Using both together is like having a security guard and a sprinkler system for your heart.
What You Can Do Right Now
Save this article and bring it to your next cardiology or primary care appointment.
Ask your doctor: "Given my cardiovascular risk profile, do I meet criteria for a GLP-1 receptor agonist?"
If cost or access is a concern, ask about manufacturer savings programs (several are available for Ozempic and Wegovy in many countries) or whether biosimilar options exist in your region.
Author’s Note
This article reflects a synthesis of emerging evidence at the intersection of cardiology, endocrinology, and metabolic medicine—fields that are increasingly converging as our understanding of cardiovascular disease evolves. The data presented, particularly from large cardiovascular outcome trials of GLP-1 receptor agonists, signal a meaningful shift in how we conceptualize and manage cardiometabolic risk.
However, it is important to emphasize that medicine is dynamic. While the benefits of GLP-1–based therapies are compelling, ongoing trials, longer-term safety data, and real-world evidence will continue to refine their role in clinical practice. Therapeutic decisions must remain individualized, taking into account patient-specific factors including cardiovascular risk profile, comorbidities, tolerability, accessibility, and cost.
For clinicians, the challenge lies not only in understanding the science but in translating it into thoughtful, patient-centered care. For patients and readers, this article is intended to provide clarity—not to replace medical advice, but to empower informed discussions with healthcare providers.
As research continues to unfold, one principle remains clear: cardiovascular disease is not merely a disorder of cholesterol or glucose, but a complex metabolic and inflammatory condition. Therapies that address these root mechanisms may ultimately define the next era of prevention and treatment.
Share this article with someone living with heart disease and obesity who may not know these options exist. The gap between evidence and awareness in this space remains enormous.
Bookmark and return — this field is moving exceptionally fast. Guidelines, new trial readouts, and newly approved agents are expected through 2026 and beyond.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
GLP-1 Receptor Agonists and Cardiovascular Protection: Evidence from CVOT Trials Explained
ApoB vs LDL Cholesterol: Which Is the Most Accurate Predictor of Heart Disease?
Atherogenic Dyslipidemia Explained: The Role of ApoB in Hidden Cardiovascular Risk
Visceral Fat and Cardiovascular Risk: The Hidden Driver of Atherosclerosis
Remnant Cholesterol: The Hidden Link Between Insulin Resistance, Type 2 Diabetes, and Heart Disease
Visceral Fat and Cardiovascular Risk: The Hidden Driver of Atherosclerosis
References
Drucker, D. J. (2025). Expanding applications of therapies based on GLP1. Nature Reviews Endocrinology, 21, 65–66. https://doi.org/10.1038/s41574-024-01066-9
Ilias, I., Zabuliene, L., & Rizzo, M. (2025). GLP-1 receptor agonists in diabetes and weight loss: The double-edged sword of innovation and risks. Frontiers in Clinical Diabetes and Healthcare, 5, 1530811. https://doi.org/10.3389/fcdhc.2024.1530811
Jackson-Morris, A., et al. (2026). Therapies based on GLP-1 receptor agonists: Significance, challenges, and opportunities. The Lancet Global Health. Advance online publication. https://doi.org/10.1016/S2214-109X(26)00009-4
Moiz, A., Filion, K. B., Tsoukas, M. A., Yu, O. H. Y., Peters, T. M., & Eisenberg, M. J. (2025). The expanding role of GLP-1 receptor agonists: A narrative review of current evidence and future directions. eClinicalMedicine, 86, 103363. https://doi.org/10.1016/j.eclinm.2025.103363
Rosen, C. J., & Ingelfinger, J. R. (2026). Glucagon-like peptide-1 receptor agonists. The New England Journal of Medicine, 394. https://doi.org/10.1056/NEJMra2500106
Saran, A., Raisinghani, R., Paliwal, S., & Sharma, S. (2026). GLP-1R agonists: Recent advances, current gaps, and future challenges. Molecular Diversity, 30(1), 101–112. https://doi.org/10.1007/s11030-025-11195-6
Yang, K., Liu, C., Guo, Q., & Li, Y. (2026). GLP-1 receptor agonists and noncardiometabolic outcomes: An umbrella review of meta-analyses. JAMA Network Open, 9(3), e264722. https://doi.org/10.1001/jamanetworkopen.2026.4722