Vascular Calcification Is Not ‘Normal Aging’ — It’s Dysregulated Calcium Biology
Is vascular calcification inevitable with age? Learn how micronutrient imbalances in vitamin D, K2, and magnesium drive arterial stiffness and heart risk.
AGINGMETABOLISM
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/5/202615 min read
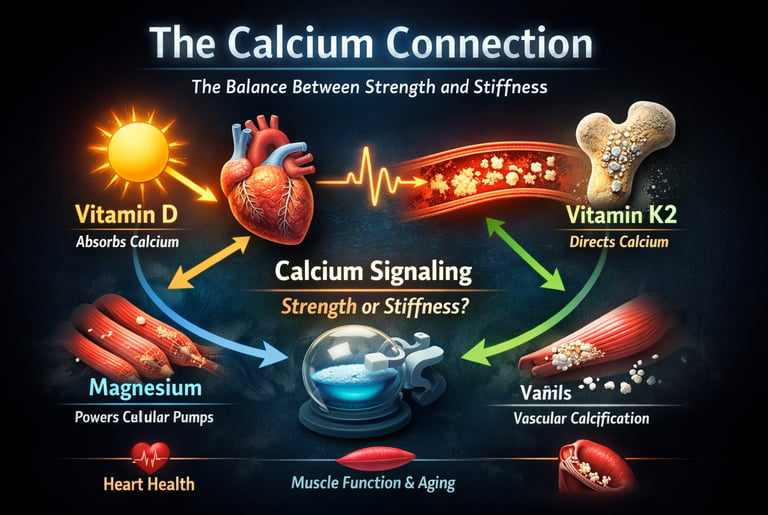
Every heartbeat begins with calcium.
Not metaphorically — literally. The contraction of cardiac muscle, the constriction and relaxation of arteries, the firing of neurons, and the strength of your skeletal muscles all depend on calcium ions moving in exquisitely timed waves across cellular membranes. A deviation of mere micromoles can mean the difference between rhythmic contraction and arrhythmia, between flexible arteries and vascular stiffness.
Yet calcium does not act independently. It is governed by a tightly coordinated micronutrient network — one that modern medicine is only beginning to fully appreciate. Vitamin D determines how much calcium enters the bloodstream by regulating intestinal transport proteins through the vitamin D receptor. Vitamin K2 activates Matrix Gla Protein (MGP), the most potent endogenous inhibitor of vascular calcification. Magnesium stabilizes ATP as Mg-ATP, the biologically active energy substrate required to power calcium pumps that reset cells after each contraction (Liu & Dudley, 2025).
When this network becomes unbalanced, the consequences are not subtle. Vitamin D supplementation without sufficient vitamin K2 may elevate circulating calcium while leaving arterial walls unprotected — a phenomenon described as the “calcium paradox” (D’Elia et al., 2026). Ageing muscle fibers lose precise calcium channel coupling, contributing to sarcopenia and reduced strength (Dong & Maturana, 2025). Meanwhile, immune cells within arterial plaques actively regulate calcium crystal deposition, revealing vascular calcification as an inflammatory, cell-mediated process rather than passive degeneration (Sheng et al., 2025).
Calcium, in other words, is not merely a structural mineral. It is a signaling language. And whether that language promotes strength or stiffness depends on how well vitamin D, K2, and magnesium are orchestrating the conversation.
Clinical Pearls
1. The Calcium Paradox & The Traffic Director
Vitamin D facilitates intestinal calcium absorption via VDR-mediated upregulation of TRPV6; however, without Vitamin K2-dependent carboxylation of Matrix Gla Protein (MGP), this systemic calcium remains "itinerant," increasing the risk of medial vascular calcification.
Think of Vitamin D as the "Delivery Truck" that brings calcium into your body, and Vitamin K2 as the "Traffic Controller." Without the controller, the calcium gets lost and ends up "parking" in your arteries (causing stiffness) instead of your bones.
2. Magnesium: The "Spark Plug" for Cellular Pumps
Intracellular calcium homeostasis is an energy-dependent process. The SERCA and PMCA pumps require magnesium-bound ATP (Mg-ATP) to function. Hypomagnesemia leads to "sluggish" calcium extrusion, resulting in cytosolic calcium overload and muscular/vascular hypertonicity.
Your cells have tiny "pumps" that push calcium out after every muscle contraction or heartbeat. These pumps run on a specific fuel that only works when magnesium is present. If you are low on magnesium, the pumps fail, calcium gets trapped inside, and your muscles or heart can’t fully relax—leading to cramps or high blood pressure.
3. The Aging Muscle "Leak" (RyR1 Uncoupling)
Sarcopenia is driven partly by the oxidative modification and "leakiness" of the Ryanodine Receptor (RyR1). This leads to a chronic, low-level dissipation of calcium from the sarcoplasmic reticulum, triggering calpain-mediated proteolysis and diminishing the force-generating capacity of the muscle fiber.
As we age, the "storage tanks" for calcium in our muscles can start to leak. Instead of a strong, controlled burst of power when you move, the calcium just "dribbles" out. This constant leak "rusts" the muscle from the inside, making it weaker and harder to build back up.
4. Macrophages: From Immune Cells to Bone-Makers
Vascular calcification is an active, immune-mediated transition. M1-polarized macrophages secrete pro-inflammatory cytokines (IL-6, TNF-α) that induce an osteogenic phenotype in vascular smooth muscle cells, effectively "reprogramming" soft tissue to behave like bone tissue.
Your immune cells (macrophages) are supposed to be the "cleanup crew." But when they are chronically inflamed, they accidentally give your blood vessels the wrong instructions, telling them to start acting like bone. This turns flexible arteries into rigid "pipes."
5. The Magnesium/Calcium Competition
Magnesium serves as a physiological calcium channel blocker. By competing for the same entry points in vascular smooth muscle, adequate magnesium levels prevent the excessive calcium influx that drives vasoconstriction and platelet aggregation.
Magnesium and calcium are like two people trying to get through the same narrow door. When you have enough magnesium, it blocks "too much" calcium from rushing into your blood vessel cells. This keeps your arteries relaxed and helps prevent the "clots" that can lead to heart attacks.
How Vitamin D Governs Calcium Absorption
Vitamin D, in its biologically active form as 1,25-dihydroxycholecalciferol (calcitriol), acts as a steroid hormone. It binds to the vitamin D receptor (VDR), which is expressed in virtually every tissue of the body. In the intestine, VDR activation upregulates transport proteins — including TRPV6 and calbindin-D9k — that physically move calcium from the gut lumen into the bloodstream. Without adequate vitamin D, dietary calcium absorption can fall below 15%, regardless of how much calcium you consume.
But here is the paradox that defines modern calcium physiology: if vitamin D drives calcium into the bloodstream, what prevents that calcium from depositing in arteries, kidneys, and soft tissues? The answer is vitamin K2.
Vitamin K2: The Calcium Traffic Director
Vitamin K2 — specifically its long-chain form MK-7 (menaquinone-7) — activates a family of proteins called Gla-proteins through a process known as carboxylation. Two of these proteins are central to cardiovascular health:
Matrix Gla Protein (MGP) is the most potent known inhibitor of vascular calcification in the human body. It is produced in arterial wall smooth muscle cells and acts as a molecular "stop sign" for calcium crystal formation. However, MGP is only active when it has been carboxylated by vitamin K2. Uncarboxylated MGP — found in people with K2 deficiency — is biologically inert and may actually accelerate calcification.
Osteocalcin similarly depends on K2-mediated carboxylation to bind calcium within the bone mineral matrix. Inactive osteocalcin allows calcium to "leak" from bones without being properly incorporated into the mineral structure.
D'Elia et al. (2026) detail the molecular pharmacology of this axis, noting that the D–K2 combination modulates endothelial function, inflammatory cytokine expression, and insulin sensitivity through mechanisms that extend well beyond simple mineral transport. Their analysis positions vitamin D and K2 not as passive dietary supplements but as molecules with ligand-receptor pharmacodynamics, dose-response relationships, and identifiable therapeutic windows — characteristics that define drugs, not just nutrients.
Key Insight: Supplementing vitamin D without adequate K2 may inadvertently increase cardiovascular risk by elevating serum calcium without activating the MGP-dependent protection against arterial calcification — a phenomenon sometimes called the "calcium paradox."
Clinical Implications of the D–K2 Axis
In practice, this "calcium paradox" — high dietary calcium intake combined with poor vascular outcomes — has been documented in epidemiological literature and may be explained, at least in part, by widespread K2 insufficiency in Western populations, whose fermented food intake is far below that of traditional East Asian diets rich in natto (fermented soybeans), the most concentrated dietary source of MK-7.
Calcium Handling in Skeletal Muscle — What Aging Does to Your Fibers
One of the most clinically underappreciated consequences of aging is the change in skeletal muscle calcium channel function — a process that contributes to sarcopenia (age-related muscle loss), exercise intolerance, and falls in older adults.
A 2025 study by Dong and Maturana, published in Frontiers in Molecular Biosciences, provides a molecular-level analysis of how aging alters calcium channels in skeletal muscle (Dong & Maturana, 2025).
Magnesium and ATP: The Overlooked Foundation of Energy, Muscle Function, and Heart Health
When people talk about calcium balance, they often focus on vitamin D and vitamin K2. But there’s another nutrient that quietly makes the entire system work:
Magnesium.
In fact, magnesium deficiency is extremely common. Depending on how it’s measured, research suggests that up to half — and possibly more — of adults may not get enough magnesium, especially in modern, processed-food diets.
And this matters far more than most people realize.
Why Magnesium Is Essential for Cellular Energy
Here’s a key biochemical fact that surprises even many clinicians:
ATP — the body’s main energy molecule — only works when it is bound to magnesium.
The active form isn’t just ATP.
It’s Mg-ATP (magnesium-bound ATP).
That means every energy-dependent process in your body — including:
Muscle contraction
Nerve signaling
Heart rhythm
Blood vessel relaxation
Calcium removal from cells
— depends on adequate magnesium levels.
Without magnesium, the body’s “energy currency” cannot be properly used.
Magnesium and Calcium: A Delicate Balance
Calcium helps muscles contract.
Magnesium helps them relax.
After every heartbeat or muscle movement, calcium must be pumped back out of cells so the muscle can relax. These pumps require magnesium-powered ATP to function.
When magnesium levels are low:
Calcium removal slows down
Muscles remain partially tense
Blood vessels stay more constricted
Blood pressure can rise
The heart works harder
This imbalance can contribute to:
Hypertension
Heart rhythm disturbances (like atrial fibrillation)
Insulin resistance
Type 2 diabetes
Magnesium and Cardiometabolic Health
Magnesium also acts as a natural calcium channel blocker in blood vessels. This helps:
Promote arterial relaxation
Support healthy blood pressure
Reduce abnormal clotting
Maintain normal heart rhythm
Low magnesium has been linked to metabolic syndrome, high blood pressure, and arrhythmias — all conditions tied to abnormal calcium regulation inside cells.
How Aging Disrupts the Muscle Calcium System
As we get older, the finely tuned system that controls muscle contraction doesn’t break down all at once — it gradually becomes less efficient. Think of it like an aging electrical system: the wiring still works, but the signals are weaker and slower.
1️⃣ Weaker Communication Between Nerves and Muscle
In young muscle, there is tight communication between the nerve signal and the calcium release system inside the muscle cell. With aging, that connection loosens.
The result?
When the brain sends a signal to move, less calcium is released, so the muscle contracts with less strength — even if you are trying just as hard.
This helps explain why strength and power decline faster than muscle size alone would suggest.
2️⃣ “Leaky” Calcium Channels
Over time, increased oxidative stress (a normal part of aging) can damage the calcium release channels inside muscle cells.
Instead of releasing calcium only when needed, these channels begin to leak small amounts continuously.
That constant calcium leak:
Interferes with muscle recovery
Activates enzymes that slowly break down muscle proteins
Contributes to age-related muscle loss
3️⃣ Slower Muscle Relaxation and More Fatigue
After each contraction, calcium must be pumped back into storage so the muscle can relax and prepare for the next movement.
With aging, this calcium pump becomes less efficient.
This leads to:
Slower relaxation
Reduced endurance
Increased fatigue during activity
4️⃣ Reduced Calcium Refill During Exercise
During sustained activity, muscles need to refill their calcium stores.
In older adults, this refill system doesn’t work as well, which can further reduce performance during prolonged activity.
Why This Matters Clinically
These microscopic changes help explain why older adults often experience:
Slower reaction times
Reduced power (for example, climbing stairs or standing quickly)
Higher fall risk
The good news?
Resistance training helps restore calcium signaling efficiency.
Adequate magnesium and vitamin D levels also support proper muscle contraction and recovery.
Aging changes the system — but targeted lifestyle strategies can strengthen it again.ning and adequate magnesium/vitamin D status support muscle function through complementary mechanisms.
Vascular Calcification — When Calcium Ends Up in the Wrong Place
Most people have heard the phrase "hardening of the arteries." But what's actually happening inside your blood vessels — and why does it matter so much for your heart health?
When calcium builds up inside the walls of your arteries, doctors call it vascular calcification. Think of it like limescale forming inside a garden hose — the walls stiffen, the flow becomes less flexible, and over time, the whole system works less efficiently. For years, doctors assumed this was simply an unavoidable part of aging, like going grey. We now know that's not true. Vascular calcification is an active biological process — one that your immune system, your diet, and your micronutrient status all influence in meaningful ways.
A 2025 research review by Sheng and colleagues, published in Frontiers in Immunology, takes a close look at exactly how this process unfolds — and more importantly, what might be done about it (Sheng et al., 2025).
Not All Arterial Calcification Is the Same
Here's something your doctor may not have explained: there are actually two distinct types of vascular calcification, and they affect your health in different ways.
Intimal calcification happens inside fatty plaques that build up along the inner lining of arteries. This is the type most closely linked to heart attacks and coronary artery disease — the calcium essentially forms within the same deposits that can rupture and block blood flow.
Medial calcification — sometimes called Mönckeberg's sclerosis — is a different problem entirely. It occurs in the middle muscular layer of the artery wall, completely separate from any fatty plaque. Rather than causing blockages, it makes arteries rigid and stiff, like pipes made of stone instead of rubber. This drives up blood pressure, strains the heart, and is strongly linked to higher overall mortality. It's especially common in people with diabetes, chronic kidney disease, and advanced age.
Understanding which type is present matters clinically — because the mechanisms, and potentially the interventions, differ.
Your Immune System Is Involved — Here's How
One of the most important discoveries in vascular calcification research is the central role played by macrophages — white blood cells that are part of your innate immune system. Macrophages are your body's first responders. They patrol tissues, engulf debris, and orchestrate inflammation. It turns out they are also key players in whether calcium deposits form or dissolve in your arterial walls.
Macrophages can work against you here. When they become inflamed — what scientists call an M1 state — they release signalling proteins (including IL-1β, IL-6, and TNF-α) that essentially instruct the smooth muscle cells lining your arteries to start behaving like bone-forming cells. Those reprogrammed smooth muscle cells then begin depositing calcium — exactly where you don't want it. Inflamed macrophages also release enzymes that break down the elastic fibres in artery walls, releasing phosphate that acts as a seed crystal for further calcium deposits.
But macrophages also have a protective mode. When they shift into an M2 (anti-inflammatory) state, they can actually engulf and clear calcium crystals, and they secrete factors that slow down the bone-forming transformation of arterial muscle cells. The balance between these two macrophage personalities — inflammatory versus protective — appears to be one of the key factors determining whether calcification in your arteries gets worse or stabilises.
This is why chronic low-grade inflammation — driven by poor diet, sedentary behaviour, obesity, and metabolic dysfunction — accelerates arterial calcification. It tips the macrophage balance firmly toward the M1 destructive state.
The Vitamin K2 Link: Your Arteries Need It Too
Here is where the vitamin K2 story becomes particularly compelling — and personal.
You may already know that vitamin K2 activates a protein called Matrix Gla Protein (MGP), which acts as a natural brake on arterial calcification. What Sheng et al. (2025) highlight is that it's not just the smooth muscle cells in your artery walls that produce MGP — your macrophages produce it too.
When you have adequate vitamin K2, the MGP released by macrophages is fully activated (carboxylated) and contributes to keeping calcium out of the arterial wall. When K2 is low — as it commonly is in people eating a typical Western diet — that macrophage-derived MGP is inactive and useless. The result is a double failure: both the structural protection and the immune protection against calcification are compromised at the same time.
This is a critical reason why vitamin K2 is increasingly being studied as a genuine cardiovascular intervention, not just a bone health supplement.
The Bottom Line for Patients
Vascular calcification is not inevitable, and it is not passive. It is driven by inflammation, immune cell behaviour, micronutrient status — particularly vitamin K2 — and metabolic health. Keeping your macrophages in a calmer, more protective state through anti-inflammatory lifestyle choices, optimising your K2 intake, and managing blood sugar and blood pressure are among the most evidence-supported strategies for keeping your arteries pliable and calcium-free for longer.
Key Takeaways
Calcium is a signaling ion, not just a structural mineral.
Every heartbeat, muscle contraction, neuronal impulse, and vascular tone adjustment depends on tightly regulated intracellular calcium flux. Dysregulation manifests as arrhythmia, arterial stiffness, sarcopenia, or metabolic dysfunction — not merely bone disease.Vitamin D determines calcium entry into systemic circulation.
Through activation of the vitamin D receptor (VDR), calcitriol upregulates intestinal transport proteins (TRPV6, calbindin), increasing calcium absorption. However, elevated serum calcium without adequate regulatory mechanisms may increase ectopic deposition risk.Vitamin K2 directs calcium to the correct anatomical destination.
K2-dependent carboxylation activates Matrix Gla Protein (MGP) and osteocalcin. Active MGP inhibits vascular calcification, while osteocalcin ensures proper skeletal mineral incorporation. K2 deficiency renders these proteins biologically inert.Magnesium stabilizes cellular energy required for calcium control.
ATP is biologically active only as Mg-ATP. Calcium pumps (SERCA, PMCA) require magnesium-bound ATP to restore intracellular calcium balance after contraction. Magnesium deficiency impairs relaxation, increases vascular tone, and promotes arrhythmogenic instability.The “calcium paradox” reflects network imbalance.
High calcium intake or vitamin D supplementation without adequate K2 and magnesium may increase circulating calcium while reducing protection against arterial deposition.Aging disrupts calcium channel precision.
Oxidative modification of ryanodine receptors (RyR1), DHPR uncoupling, and reduced SERCA activity contribute to sarcopenia, slower contraction-relaxation cycles, and increased fall risk.Vascular calcification is immune-regulated, not passive.
Macrophage polarization influences smooth muscle osteogenic transformation. Inflammatory cytokines accelerate calcification, while adequate K2-dependent MGP provides local defense.Cardiometabolic disease reflects calcium mismanagement at scale.
Hypertension, insulin resistance, arrhythmias, and arterial stiffness share a common denominator: disrupted intracellular calcium handling.Micronutrient sufficiency is systems maintenance.
Vitamin D, K2, and magnesium operate as an integrated regulatory axis — not isolated supplements.Calcium physiology is precision engineering.
Protecting cardiovascular and muscular health requires maintaining the full regulatory network, not simply increasing calcium intake.
Frequently Asked Questions
1️⃣ What is vascular calcification?
Vascular calcification means calcium is being deposited in your blood vessel walls instead of staying in your bones where it belongs.
Over time, this can make arteries stiff and less flexible, increasing the risk of:
High blood pressure
Heart disease
Stroke
It is not just “getting older.” It’s a biological process influenced by inflammation, metabolism, and nutrient balance.
2️⃣ Is vascular calcification a normal part of aging?
It becomes more common with age, but it is not inevitable.
Healthy arteries are flexible at any age. Calcification happens when:
Inflammation is present
Blood sugar is poorly controlled
Kidney function declines
Key nutrients are imbalanced
Aging increases risk — but lifestyle and metabolic health matter greatly.
3️⃣ What is the “calcium paradox”?
The “calcium paradox” refers to this situation:
Bones lose calcium → osteoporosis
Arteries gain calcium → stiffening
This happens when the body’s calcium regulation system is not functioning properly.
4️⃣ If calcium is important, why is it a problem in arteries?
Calcium is essential for:
Muscle contraction
Nerve signals
Heart rhythm
Bone strength
The problem is location, not calcium itself.
When regulation fails, calcium deposits in soft tissues instead of bone.
5️⃣ Does taking calcium supplements cause heart disease?
Not necessarily — but context matters.
Calcium supplements without proper balance of:
Vitamin D
Vitamin K2
Magnesium
may increase the chance that calcium is not directed properly.
It’s best to take calcium only if needed and under medical guidance.
6️⃣ Why is vitamin D important?
Vitamin D helps your intestines absorb calcium.
Low vitamin D can lead to:
Weak bones
Muscle weakness
Increased fracture risk
But very high doses without balance may not be helpful either. Testing is important.
7️⃣ What does vitamin K2 do?
Vitamin K2 helps activate proteins that:
Direct calcium into bones
Prevent calcium from depositing in arteries
Think of K2 as a “traffic controller” for calcium.
8️⃣ Why is magnesium important?
Magnesium helps:
Regulate muscle relaxation
Control blood pressure
Support heart rhythm
Stabilize calcium balance
Many people are mildly deficient due to modern diets.
9️⃣ Should I take vitamin D, K2, and magnesium together?
Sometimes they are taken together because they work in related pathways.
But:
Not everyone needs supplementation.
Doses should be individualized.
Kidney disease changes recommendations.
Always discuss with your physician.
🔟 Can vascular calcification be reversed?
Early changes may improve with:
Better blood sugar control
Blood pressure management
Reduced inflammation
Exercise
Smoking cessation
Advanced calcification is harder to reverse — prevention is key.
1️⃣1️⃣ How do I know if I have calcification?
Tests may include:
Coronary artery calcium (CAC) score (CT scan)
Ultrasound for carotid arteries
Kidney function tests
Blood markers of inflammation
Your doctor decides based on risk factors.
1️⃣2️⃣ What lifestyle changes help most?
The strongest evidence supports:
✔ Regular resistance + aerobic exercise
✔ Blood sugar control
✔ Blood pressure control
✔ Whole-food diet
✔ Adequate protein intake
✔ Avoid smoking
✔ Maintain healthy weight
Exercise improves both bone and vascular health.
1️⃣3️⃣ Is this related to diabetes?
Yes. In type 2 diabetes:
Chronic high blood sugar
Insulin resistance
Inflammation
all increase calcification risk.
Good metabolic control significantly reduces progression.
1️⃣4️⃣ Is this connected to muscle weakness?
Yes.
Vitamin D deficiency, magnesium imbalance, and metabolic disease can:
Reduce muscle strength
Increase fall risk
Increase fracture risk
Muscle health and vascular health are closely connected.
1️⃣5️⃣ What is the most important takeaway?
Vascular calcification is not simply “calcium buildup.”
It reflects:
Metabolic health
Inflammation levels
Nutrient balance
Kidney function
Lifestyle patterns
The goal is not to fear calcium —
It is to restore proper biological regulation.
Prevention and early action make the biggest difference.
Here are the specific tests and questions to consider for a systemic evaluation:
1. Assessing Vitamin D
Test: 25-Hydroxy Vitamin D [25(OH)D]. This is the standard marker for storage.
2. Assessing Magnesium
Test: Magnesium RBC (Red Blood Cell). Standard serum magnesium tests are often misleading because only 1% of the body's magnesium is in the blood. RBC testing provides a better look at cellular status.
3. Assessing the Vitamin K2
Note: Direct K2 testing is rare in standard labs. Instead, clinicians look for the result of K2 deficiency.
Test: Coronary Artery Calcium (CAC) Score. This CT scan physically "sees" if calcium has deposited in your heart’s arteries.
Advanced Test: dp-ucMGP (desphospho-uncarboxylated Matrix Gla Protein). This measures how much "inactive" MGP is in your system due to K2 deficiency.
4. Metabolic & Inflammatory Context
Test: hs-CRP (High-sensitivity C-reactive protein) and HbA1c. These identify the "fire" (inflammation) that tells macrophages to turn your arteries into bone.
Author’s Note
As a physician trained in internal medicine, I have spent decades treating the downstream consequences of metabolic and cardiovascular disease — hypertension, arrhythmias, arterial stiffness, sarcopenia, and insulin resistance. What has become increasingly clear is that many of these seemingly separate conditions share a common physiological thread: disrupted calcium regulation.
This article was written to reframe calcium not as a static mineral associated only with bone density, but as a dynamic signaling molecule governed by a tightly integrated micronutrient network. Vitamin D influences how much calcium enters circulation. Vitamin K2 determines where that calcium is deposited. Magnesium provides the biochemical stability required for calcium pumps and cellular energy systems to function. When one component is neglected, the system compensates — often imperfectly.
The goal of this piece is not to promote indiscriminate supplementation. It is to encourage systems-level thinking. Nutrients operate within networks, not in isolation. Clinical outcomes reflect physiology, and physiology reflects balance.
The research emerging in 2025 and 2026 has strengthened our understanding of these interactions at the molecular level — particularly in vascular biology, skeletal muscle aging, and cardiometabolic risk. Translating that science into accessible, clinically meaningful insight is both a responsibility and a privilege.
Readers should view this article as an educational resource, not individualized medical advice. Nutrient requirements vary depending on age, renal function, medications, and underlying disease states. Thoughtful assessment, laboratory evaluation when appropriate, and professional guidance remain essential.
Calcium is not merely consumed. It is directed, buffered, signaled, and contained. Preserving health requires respecting that complexity.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
The Vitamin D–K2 Axis: A Precision Strategy for Bone, Muscle, and Vascular Integrity | DR T S DIDWAL
Living to 100: Is it Genetics or Lifestyle? What the New Science Says | DR T S DIDWAL
Time-Restricted Eating: Metabolic Advantage or Just Fewer Calories? | DR T S DIDWAL
Can You Revitalize Your Immune System? 7 Science-Backed Longevity Strategies | DR T S DIDWAL
Exercise and Longevity: The Science of Protecting Brain and Heart Health as You Age | DR T S DIDWAL
Light and Longevity: Can Sunlight Slow Cellular Aging? | DR T S DIDWAL
References
D'Elia, S., Bottino, R., Carbone, A., Formisano, T., Orlandi, M., Sperlongano, S., Castaldo, P., Molinari, D., Palladino, A., Morello, M., Titolo, G., Loffredo, F. S., Natale, F., Cirillo, P., & Cimmino, G. (2026). Modulation of cardiometabolic risk by vitamin D and K2: Simple supplementation or real drug? Uncovering the pharmacological properties. International Journal of Molecular Sciences, 27(1), 298. https://doi.org/10.3390/ijms27010298
Dong, M., & Maturana, A. D. (2025). Effects of aging on calcium channels in skeletal muscle. Frontiers in Molecular Biosciences, 12, 1558456. https://doi.org/10.3389/fmolb.2025.1558456
Liu, M., & Dudley, S. C., Jr. (2025). Magnesium homeostasis and magnesium transporters in human health. Nutrients, 17(5), 920. https://doi.org/10.3390/nu17050920
Sheng, Y., Qiu, Y., Wang, X., Shi, J., Yin, Z., Zhang, Z., Jiang, S., Zhang, J., Hu, X., & Hong, W. (2025). The role of macrophages in vascular calcification: Strategies for diagnosis and treatment. Frontiers in Immunology, 16, 1724464. https://doi.org/10.3389/fimmu.2025.1724464