Scientists Are Rethinking Depression: The Hidden Role of Metabolism and Brain Energy
New studies suggest insulin resistance, obesity, and metabolic inflammation may play a major role in depression and psychiatric disorders.
METABOLISM
Dr. T.S. Didwal, M.D.(Internal Medicine)
5/15/202614 min read
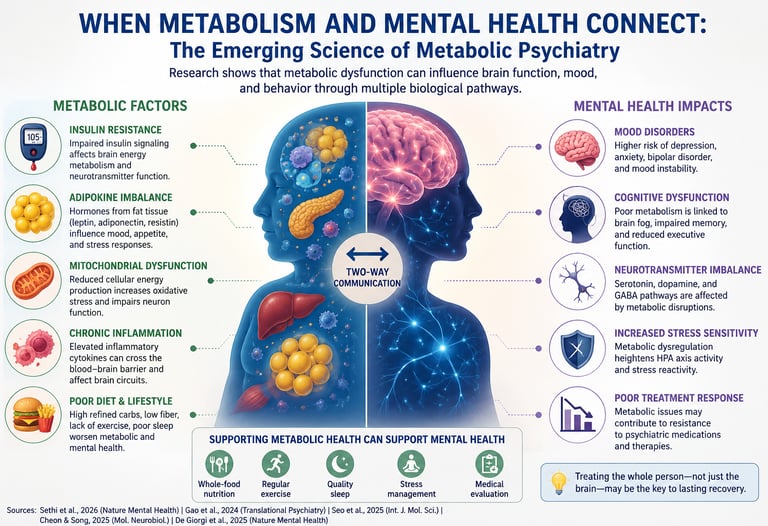
For decades, mental health and metabolic health were treated as two entirely separate worlds. Depression was considered a disorder of neurotransmitters. Obesity was viewed as a problem of weight. Type 2 diabetes was categorized as a disease of blood sugar. But modern neuroscience and metabolic medicine are now dismantling those artificial boundaries. Emerging evidence suggests that the brain and the body are engaged in a continuous biochemical dialogue — and when metabolism begins to fail, mental health may deteriorate alongside it (Sethi et al., 2026).
This shift has given rise to a rapidly evolving field known as metabolic psychiatry, which investigates how insulin resistance, mitochondrial dysfunction, chronic inflammation, impaired glucose metabolism, and metabolic syndrome influence psychiatric disorders, including depression, anxiety, bipolar disorder, and schizophrenia (Gao et al., 2024). Researchers are increasingly recognizing that the brain is not only a psychological organ — it is also one of the most metabolically demanding organs in the human body. Although the brain accounts for only about 2% of total body weight, it consumes nearly 20% of the body’s energy supply to sustain cognition, emotional regulation, memory, and neurotransmission (Seo et al., 2025).
When cellular energy systems become impaired, the consequences may extend far beyond fatigue or weight gain. Neuroinflammation, oxidative stress, impaired insulin signaling, and disrupted mitochondrial energy production are now being linked to mood disorders, cognitive dysfunction, emotional dysregulation, and treatment-resistant depression (Cheon & Song, 2025). Scientists are also exploring how gut-brain signaling, adipokines, GLP-1 pathways, and systemic inflammation may reshape brain chemistry itself (De Giorgi et al., 2025).
The implications are profound. Mental illness may no longer be understood solely as a disorder of “brain chemistry,” but increasingly as a whole-body metabolic condition involving interconnected networks of hormones, immune signaling, energy metabolism, and neural function.
What Is Metabolic Psychiatry?
Metabolic psychiatry is the study and clinical application of how disorders of metabolism — including insulin resistance, lipid dysregulation, chronic inflammation, and mitochondrial dysfunction — contribute to the onset, severity, and treatment resistance of mental health conditions.
A landmark 2026 review published in Nature Mental Health by Sethi et al. synthesized the current state of this evidence base. The researchers found that systemic metabolic abnormalities are highly prevalent in people living with psychiatric illness and may contribute directly to increased mortality, greater illness severity, and resistance to standard psychiatric treatments (Sethi et al., 2026). In other words, treating only the brain — and ignoring what is happening in the rest of the body — may explain why so many patients never fully recover.
This does not mean mental illness is "just" a metabolic disease. Rather, it means the mind and the metabolism are locked in a constant conversation, and when that conversation goes wrong, both suffer.
The Brain Is an Energy-Hungry Organ — and It Shows
To understand why metabolism matters so profoundly to mental health, it helps to appreciate just how energetically demanding the brain is. Though it accounts for only about 2% of body weight, the human brain consumes roughly 20% of the body's total energy. Every thought, emotion, memory, and decision you have costs metabolic currency.
When the systems that produce and deliver that energy are compromised, the brain does not simply work less efficiently — it starts generating psychiatric symptoms. A 2025 review in the International Journal of Molecular Sciences by Seo et al. illuminated one critical pathway in this process: a stress-signaling molecule called Growth Differentiation Factor 15 (GDF15) (Seo et al., 2025). GDF15 is released during cellular stress and metabolic strain. The researchers found that elevated GDF15 disrupts brain energy homeostasis — essentially shorting out the brain's ability to regulate its own fuel supply. This molecular link demonstrates that what happens at the cellular level during metabolic stress directly reshapes the biology of mood and cognition.
Think of it this way: if your phone is running low on battery, it starts cutting non-essential functions. The brain, faced with an energy crisis, does something similar — and the functions it cuts first are often those governing emotional regulation, motivation, and clear thinking.
What the Genetics Tell Us: Metabolic Syndrome Is Not Just Correlated With Mental Illness — It May Cause It
For years, researchers wondered whether the high rates of metabolic dysfunction seen in people with depression, schizophrenia, and bipolar disorder were simply a side effect of psychiatric medications, poor lifestyle habits associated with mental illness, or coincidence. A 2024 Mendelian randomization study published in Translational Psychiatry by Gao et al. offered a more definitive answer (Gao et al., 2024).
Mendelian randomization is a powerful technique that uses genetic variants as natural experiments to test causation — not just correlation — between exposures and outcomes. By analyzing large genomic datasets, Gao and colleagues found genetic evidence that metabolic syndrome components causally influence the risk of several psychiatric disorders. This means the relationship is not merely coincidental or an artifact of shared lifestyle. Metabolic dysregulation appears to genuinely increase the biological risk of developing mental health conditions.
This is a paradigm shift with major clinical implications. It suggests that identifying and treating metabolic risk factors early — elevated blood sugar, high triglycerides, central obesity, high blood pressure — may represent a genuine strategy for psychiatric prevention, not just physical health management.
The Gut, the Brain, and a River of Molecules
A 2025 multi-omics study published in Frontiers in Psychiatry by Qianhao et al. took a sophisticated three-pronged approach to understanding metabolic dysregulation in depression (Qianhao et al., 2025). By combining metabolomics (the study of small molecules in the body), weighted gene co-expression network analysis (which maps how genes work together), and Mendelian randomization, the researchers identified specific metabolic pathways that appear to be causally linked to depressive illness.
What emerged was a picture of depression not as a simple chemical imbalance, but as a systems-level disruption involving amino acid metabolism, lipid processing, and inflammatory signaling — all of which are components of the broader metabolic ecosystem. Crucially, this research supports the idea that blood-based biomarkers of metabolic dysfunction could one day be used to diagnose depression subtypes and guide personalized treatment decisions. We may be approaching an era where a blood panel helps your doctor understand not just your cholesterol, but your mental health risk profile.
The Gut Microbiome: The Missing Link in Metabolic Psychiatry
One of the most exciting developments in metabolic psychiatry is the growing recognition of the microbiota-gut-brain axis — a sophisticated communication network connecting the gut microbiome with brain function and metabolism.
The gut microbiome influences mental health through multiple pathways: it produces short-chain fatty acids (SCFAs) that reduce neuroinflammation and support the blood-brain barrier, regulates tryptophan metabolism (the precursor to serotonin), modulates immune responses, and influences insulin sensitivity. When the microbiome becomes imbalanced (dysbiosis), it can trigger metabolic endotoxemia, chronic low-grade inflammation, impaired insulin signaling in the brain, and disrupted neurotransmitter balance. These changes have been linked to increased risk and severity of depression, anxiety, bipolar disorder, and even schizophrenia (Sethi et al., 2026; Qianhao et al., 2025).
Importantly, this relationship is bidirectional. Stress, poor diet, and psychiatric medications can worsen gut dysbiosis, while a healthy microbiome supports better mood regulation, cognitive function, and metabolic resilience. GLP-1 receptor agonists appear to positively influence the gut microbiome, which may partly explain their emerging benefits on both metabolic and mental health.
Targeting the gut microbiome through diet (high-fiber, Mediterranean-style), fermented foods, exercise, and sleep represents one of the most accessible and promising strategies in metabolic psychiatry.
Adipokines: The Body-Fat-Brain Connection Nobody Told You About
Fat tissue — once regarded as passive storage — is now known to be a dynamic endocrine organ that secretes a family of signaling molecules called adipokines. These include leptin, adiponectin, resistin, and others that travel through the bloodstream and directly influence brain function and neurotransmitter systems.
A 2025 review in Molecular Neurobiology by Cheon and Song examined the interplay between adipokines and neurotransmission in neuropsychiatric conditions (Cheon & Song, 2025). The researchers found that adipokine imbalances — a hallmark of obesity and metabolic syndrome — interfere with dopamine and serotonin pathways, disrupt the hypothalamic-pituitary axis, and promote neuroinflammation. In people with metabolic dysfunction, leptin resistance (a condition where the brain stops responding properly to leptin signals) may contribute to depression, cognitive difficulties, and abnormal reward processing — the same symptoms that define many psychiatric conditions.
This research suggests that the expanding waistlines associated with the global obesity epidemic may also be fueling an expanding epidemic of mental ill-health — not merely through psychological stigma or social stress, but through direct molecular mechanisms.
GLP-1 Receptor Agonists: The Weight-Loss Drugs That May Also Heal the Mind
Perhaps the most exciting and hotly debated frontier in metabolic psychiatry is the potential psychiatric benefits of glucagon-like peptide-1 receptor agonists (GLP-1 RAs) — the class of medications that includes semaglutide (Ozempic, Wegovy) and liraglutide, originally developed to treat type 2 diabetes and obesity.
A comprehensive 2025 analysis published in Nature Mental Health by De Giorgi et al. reviewed the growing body of evidence on GLP-1 RAs and cognitive and mental health disorders (De Giorgi et al., 2025). The findings were striking: GLP-1 RA use was associated with reduced risk and improved symptoms across multiple cognitive and psychiatric domains, including depression and anxiety. GLP-1 receptors are found not only in the pancreas and gut but throughout the brain — including in regions governing mood, reward, and cognition.
The mechanisms proposed include reduced neuroinflammation, improved insulin signaling in the brain, protection of dopaminergic neurons, and direct modulation of reward circuitry. For patients who have both metabolic disease and treatment-resistant psychiatric conditions — a frustratingly common combination — GLP-1 RAs represent a tantalizing therapeutic bridge. It is worth noting that most evidence to date comes from observational studies and preliminary trials; larger randomized controlled trials are underway, and caution is warranted in clinical application until more definitive evidence is available.
The Big Picture: A New Clinical Roadmap
Putting this science together, what does metabolic psychiatry offer patients and clinicians in practical terms? Sethi et al. (2026) outlined a ranked framework of metabolism-based psychiatric interventions, categorized by current level of evidence:
Pharmacological approaches with emerging support include:
Metformin — the most widely used diabetes medication — which has shown benefits in reducing metabolic side effects of antipsychotic medications and may have direct mood-stabilizing properties.
GLP-1 receptor agonists — as discussed above.
Pioglitazone — an insulin sensitizer with early evidence suggesting antidepressant effects.
Lifestyle-based metabolic therapies include:
Ketogenic metabolic therapy — a high-fat, very-low-carbohydrate dietary approach that shifts the brain's primary fuel source from glucose to ketone bodies. Early clinical studies in psychiatric settings are yielding promising results, particularly for bipolar disorder and schizophrenia.
Intermittent fasting — which appears to modulate insulin sensitivity, reduce neuroinflammation, and support mitochondrial health.
Structured aerobic and resistance exercise — with robust evidence for mood improvement, cognitive enhancement, and metabolic benefit.
These approaches are not replacements for conventional psychiatric care — they are intended to complement it. The message from the emerging research is that psychiatric care and metabolic care should no longer exist in separate silos.
Practical Applications: What You Can Do Today
You do not need to wait for clinical guidelines to catch up with the science. Here are evidence-informed steps you can take now, ideally in conversation with your healthcare provider:
1. Request a Metabolic Screen. Ask your doctor for a comprehensive metabolic panel: fasting blood glucose, HbA1c, lipid panel, fasting insulin (HOMA-IR), C-reactive protein (inflammation marker), and, if possible, vitamin D levels. These tests can reveal metabolic dysfunction that may be influencing your mental health.
2. Prioritize Sleep Quality. Disrupted circadian rhythms worsen both metabolic function and psychiatric symptoms through overlapping biological pathways. Aim for 7–9 hours of consistent, good-quality sleep. Even shifting your bedtime earlier by 30 minutes can meaningfully improve insulin sensitivity.
3. Move Every Day — Even Briefly Exercise is the most robustly evidence-based lifestyle intervention for both metabolic health and mental health simultaneously. A 20–30-minute brisk walk after meals can significantly reduce postprandial blood glucose spikes — and research consistently links regular physical activity to lower rates of depression and anxiety.
4. Reduce Ultra-Processed Food Intake. Diets high in refined carbohydrates and seed oils promote insulin resistance, inflammation, and dyslipidemia — all of which the research reviewed here links to psychiatric risk. Prioritizing whole foods, adequate protein, healthy fats, and fiber supports both metabolic and mental resilience.
5. Discuss Medication Metabolic Effects With Your Psychiatrist. Many commonly prescribed antipsychotic and mood-stabilising medications carry significant metabolic side effects, including weight gain, insulin resistance, and lipid elevation. This does not mean you should stop them — but it does mean metabolic monitoring and proactive mitigation strategies should be part of your care plan.
6. Consider Stress Management as Metabolic Medicine. Chronic psychological stress drives cortisol elevation, which in turn worsens insulin resistance and promotes visceral fat accumulation. Mind-body practices, including mindfulness-based stress reduction, yoga, and breathwork, have demonstrated measurable improvements in metabolic biomarkers.
7. Advocate for Integrated Care. Ask your GP, psychiatrist, or mental health care team whether metabolic screening is part of your routine care. If it is not, advocate for it. The science clearly supports treating the whole person — not just the diagnosis.
Frequently Asked Questions (FAQs)
Q1: Does metabolic psychiatry mean my mental illness is caused by my diet? Not exactly. Your diet is one of several factors that shape your metabolic health, which in turn influences your mental health. Metabolic psychiatry identifies a range of contributors — genetics, inflammation, mitochondrial function, stress hormones, gut health — that converge to affect both systems. Diet matters, but it is one variable among many.
Q2: Can treating my blood sugar or cholesterol actually improve my mental health symptoms? Early evidence suggests yes, particularly for people whose psychiatric condition has a strong metabolic component. Research by Watson et al. (cited in Sethi et al., 2026) found that insulin resistance is associated with greater depression severity and poorer remission rates. Treating that insulin resistance may improve treatment outcomes, though this remains an active area of investigation.
Q3: Should I ask my doctor about GLP-1 medications like Ozempic for my mental health? This is a conversation worth having, particularly if you also have obesity, type 2 diabetes, or metabolic syndrome alongside your mental health condition. However, GLP-1 RAs are not yet approved specifically for psychiatric indications, and the evidence base, while exciting, is still developing (De Giorgi et al., 2025). Discuss the full risk-benefit picture with your prescribing physician.
Q4: Are the metabolic effects of psychiatric medications something I should be worried about? Yes — and your care team should be proactively monitoring for them. Certain antipsychotics, mood stabilizers (including valproate and lithium), and some antidepressants can worsen insulin resistance, cause weight gain, and elevate cholesterol. These effects can be managed with appropriate lifestyle strategies and sometimes with adjunctive medications like metformin. Do not adjust or stop medications without medical guidance.
Q5: Is the ketogenic diet safe for people with mental illness? Emerging evidence is promising, but ketogenic metabolic therapy should be undertaken with clinical supervision, particularly in people taking psychiatric medications. Some anticonvulsant medications used as mood stabilizers interact with ketosis. There are clinical programs in several countries specifically designed to deliver ketogenic therapy safely in psychiatric populations (Sethi et al., 2026).
Q6: How does stress cause metabolic problems that affect mental health? Chronic stress activates the hypothalamic-pituitary-adrenal (HPA) axis, releasing cortisol. Prolonged cortisol elevation promotes insulin resistance, visceral fat deposition, and systemic inflammation — all metabolic changes that in turn further dysregulate the stress response and impair neurotransmitter function (Cheon & Song, 2025). This creates a vicious cycle that can entrench both metabolic and psychiatric illness.
Q7: My mental health diagnosis came first — could metabolic issues still be involved? Absolutely. Metabolic abnormalities can precede psychiatric diagnoses, occur concurrently, or develop as a consequence of the illness or its treatment. The genetic research by Gao et al. (2024) demonstrates that metabolic and psychiatric vulnerabilities can share biological roots. Regardless of which came first, addressing metabolic health is relevant at any stage of a psychiatric illness.
Clinical Pearls
1. The "Power Plant" Problem (Mitochondrial Function)
Scientific Perspective: The brain accounts for 20% of total body oxygen consumption. In psychiatric disorders, mitochondrial dysfunction leads to impaired ATP production and increased oxidative stress. When neurons lack sufficient cellular energy, they cannot maintain membrane potential or facilitate proper neurotransmission.
Think of your brain like a high-end smartphone. Even if the software (your thoughts) is perfect, if the battery is damaged or old, the phone will lag, crash, or shut down features to save power. Fixing the "battery" (your cellular energy) is often necessary for the "software" to run smoothly.
2. Insulin Resistance as "Brain Fog"
Scientific Perspective: Insulin is a neuroprotective hormone that crosses the blood-brain barrier to regulate glucose uptake and synaptic plasticity. Cerebral insulin resistance impairs the brain's ability to utilize glucose, leading to cognitive deficits and mood dysregulation, often referred to as "Type 3 Diabetes" in some research circles.
Most people know insulin affects blood sugar, but it also tells your brain how to use fuel. If your brain becomes "deaf" to insulin, it’s like being in a room full of food with your mouth taped shut—the energy is there, but your brain can’t get to it, leaving you feeling exhausted and moody.
3. Adipokines: Fat as an Endocrine Organ
Scientific Perspective: Visceral adipose tissue is not inert; it secretes pro-inflammatory cytokines and adipokines (like leptin and adiponectin). These molecules can cross the blood-brain barrier, triggering neuroinflammation and disrupting the dopamine reward pathways often implicated in depression and addiction.
Body fat isn't just "stored weight"; it’s actually a busy factory sending chemical signals to your brain. If that factory is overactive, it sends "alarm" signals that cause inflammation in the brain, making it harder to feel pleasure or stay motivated.
4. The GLP-1 "Dual-Action" Breakthrough
Scientific Perspective: GLP-1 receptors are highly expressed in the ventral tegmental area (VTA) and the hippocampus. Beyond weight loss, GLP-1 agonists appear to modulate reward-seeking behavior and provide neuroprotective effects by reducing neuroinflammation and stabilizing brain energy homeostasis.
New medications like semaglutide (Ozempic) do more than just help with weight. They actually talk to the parts of your brain that control cravings and mood. It’s like a "reset button" for both your metabolism and your brain's reward system.
5. Metabolic Side Effects are Not "Secondary"
Scientific Perspective: Many second-generation antipsychotics (SGAs) and mood stabilizers are highly orexigenic (appetite-stimulating) and can cause rapid weight gain and dyslipidemia. These are not merely "side effects"—they are metabolic insults that can fundamentally counteract the primary psychiatric treatment.
If a medication helps your mood but causes you to gain 20 pounds or develop high blood sugar, it might eventually make your mental health worse in the long run. Metabolic health and mental health are two sides of the same coin; you shouldn't have to sacrifice one to fix the other.
6. The "Postprandial" Mood Connection
Scientific Perspective: Postprandial (after-meal) hyperglycemia causes transient systemic inflammation and oxidative stress. Frequent glucose spikes and crashes lead to fluctuations in glutamate and GABA, the brain’s primary "on" and "off" switches, contributing to acute anxiety and irritability.
Have you ever felt "hangry" or had a "sugar crash"? That’s your brain reacting to a roller coaster of blood sugar. By keeping your blood sugar stable (eating more fiber, protein, and healthy fats), you give your brain a steady, reliable stream of energy, which helps keep your mood stable, too.
Author’s Note: A Clinician’s Perspective
As a physician practising internal medicine, I have increasingly observed that the boundaries separating metabolic disease and mental illness are far less distinct than traditional medical models once suggested. Many patients living with depression, anxiety, chronic fatigue, cognitive dysfunction, obesity, insulin resistance, or type 2 diabetes are not experiencing isolated disorders affecting separate organs — they are often experiencing interconnected manifestations of systemic metabolic dysregulation.
In clinical practice, it is common to see patients whose psychiatric symptoms worsen alongside weight gain, poor sleep, chronic inflammation, sedentary behavior, or deteriorating glycemic control. Likewise, individuals with severe metabolic syndrome frequently describe brain fog, emotional exhaustion, low motivation, impaired concentration, and reduced psychological resilience. Modern research in metabolic psychiatry is beginning to provide a biological explanation for these observations through mechanisms involving neuroinflammation, mitochondrial dysfunction, impaired insulin signaling, oxidative stress, gut-brain interactions, and disrupted neurotransmitter regulation (Sethi et al., 2026; Seo et al., 2025).
Importantly, this emerging science should not be interpreted as reducing mental illness to a simple “lifestyle problem” or implying that psychiatric disorders are caused solely by diet or body weight. Mental health conditions are profoundly complex and shaped by genetics, trauma, environment, social factors, neurobiology, and metabolism simultaneously. However, ignoring metabolic health may mean overlooking one of the most modifiable contributors to psychiatric vulnerability and treatment resistance.
From a clinician’s perspective, the future of mental healthcare will likely become increasingly integrative — combining evidence-based psychiatry with metabolic screening, exercise medicine, nutritional strategies, sleep optimization, inflammation reduction, and personalized lifestyle interventions alongside conventional therapies. The goal is not to replace psychiatric care, but to strengthen it by treating the whole person rather than isolating the brain from the body.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
PCOS Is Being Renamed in 2026: What the New Science Means for Women
How Exercise Reverses Leptin Resistance and Restores Metabolic Health | DR T S DIDWAL
Your Body Fat Is an Endocrine Organ—And Its Hormones Shape Your Heart Health | DR T S DIDWAL
hsCRP Explained: What Inflammation Means for Your Heart | DR T S DIDWAL
Low-Fat vs. Low-Carb: Which Diet is Best for Weight Loss? | DR T S DIDWAL
The Role of Cholesterol in Health and Disease: Beyond the "Bad" Label | DR T S DIDWAL
The Best Dietary Fat Balance for Insulin Sensitivity, Inflammation, and Longevity | DR T S DIDWAL
References
Cheon, S. Y., & Song, J. (2025). Neuropsychiatric abnormalities in metabolic disturbances: Interplay of adipokines and neurotransmission. Molecular Neurobiology, 62, 8824–8842. https://doi.org/10.1007/s12035-025-04797-6
De Giorgi, R., Ghenciulescu, A., Dziwisz, O., et al. (2025). An analysis on the role of glucagon-like peptide-1 receptor agonists in cognitive and mental health disorders. Nature Mental Health, 3, 354–373. https://doi.org/10.1038/s44220-025-00390-x
Gao, X., Qin, Y., Jiao, S., Hao, J., Zhao, J., Wang, J., Wen, Y., & Wang, T. (2024). Genetic evidence for the causal relations between metabolic syndrome and psychiatric disorders: A Mendelian randomization study. Translational Psychiatry, 14(1), 46. https://doi.org/10.1038/s41398-024-02759-5
Qianhao, W., Jinwen, Z., Jingjie, M., Xiaoyu, C., Yangfei, Z., Wenxiu, Y., Xu, J., Xiaojun, W., Peipei, H., & Qi, G. (2025). Multi-omics investigation of metabolic dysregulation in depression: Integrating metabolomics, weighted gene co-expression network analysis, and Mendelian randomization. Frontiers in Psychiatry, 16, 1627020. https://doi.org/10.3389/fpsyt.2025.1627020
Seo, M., Pyeon, S. Y., & Kim, M. S. (2025). Molecular links between metabolism and mental health: Integrative pathways from GDF15-mediated stress signaling to brain energy homeostasis. International Journal of Molecular Sciences, 26(15), 7611. https://doi.org/10.3390/ijms26157611
Sethi, S., Berk, M., Andreazza, A. C., et al. (2026). Metabolic psychiatry targeting metabolic dysregulation in mental health. Nature Mental Health, 4, 508–520. https://doi.org/10.1038/s44220-026-00609-5