Reset Your Metabolism: How Nutrient Sensing Pathways Control Aging, Cellular Energy, and Longevity
Discover how AMPK, mTOR, NAD+, and FOXO regulate metabolism, aging, and energy—and learn science-backed strategies to improve longevity
AGINGMETABOLISM
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/4/202611 min read
The Hidden Switches That Control How You Age
What if aging is not just the passage of time — but the result of tiny molecular switches constantly responding to how you eat, move, and live?
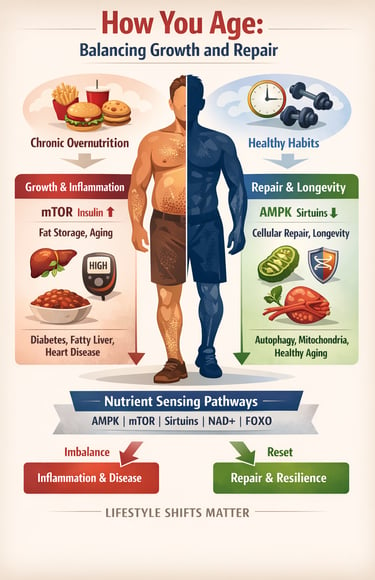
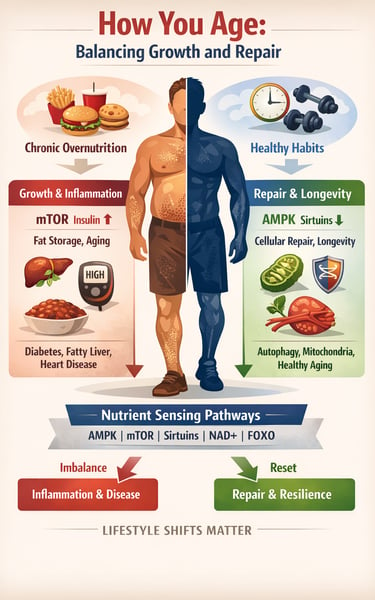
Inside every cell are nutrient-sensing pathways that decide whether your body should grow, store energy, repair damage, or conserve fuel. These pathways — including AMPK, mTOR, sirtuins, NAD+, and FOXO — function like a metabolic control panel. When balanced, they promote resilience, efficient energy use, and cellular repair. When overstimulated by chronic overnutrition and inactivity, they accelerate inflammation, insulin resistance, and biological aging (Penugurti et al., 2024; Zhang et al., 2025).
In modern life, food is constantly available. Frequent meals, high sugar intake, and excess refined carbohydrates keep insulin and mTOR persistently activated — signaling the body to remain in growth mode rather than repair mode (Zhang et al., 2025). Meanwhile, protective pathways such as AMPK and sirtuins, which are activated during fasting and exercise, become relatively suppressed (Yusri et al., 2025). Over time, this imbalance contributes to conditions like type 2 diabetes, fatty liver disease, cardiovascular disease, and sarcopenia (Wang et al., 2026).
The exciting discovery from recent research is that these systems are not fixed. They are dynamic and highly responsive to lifestyle interventions. Exercise, time-restricted eating, and calorie moderation can recalibrate these pathways, restoring metabolic flexibility and cellular repair mechanisms (Dai et al., 2025).
Clinical pearls
1. The "Metabolic Switch" (AMPK)
Scientific Perspective: AMPK activation via a 12–16 hour fast or Zone 2 exercise triggers the "fuel gauge" to shift from glucose oxidation to fatty acid oxidation and autophagy. According to Penugurti et al. (2024), this is the primary defense against cellular senescence.
"Think of your body like a hybrid car. If you’re always snacking, you’re only running on electricity (sugar). Fasting lets the engine kick in to burn the 'backup fuel' (fat) and clears out the 'exhaust' (cellular junk) that makes you feel sluggish."
2. The mTOR "Growth vs. Repair" Trade-off
Scientific Perspective: Chronic mTORC1 hyperactivation—driven by high-leucine protein and refined carbs—inhibits lysosomal function and proteostasis (Zhang et al., 2025). This accelerates tissue aging even while promoting muscle growth.
You can’t renovate a house while the party is still going on. mTOR is the 'party' (growth). You need quiet periods—like time-restricted eating—to let the 'cleaning crew' (repair) come in and fix the floorboards so the house lasts 100 years instead of 50."
3. The NAD+ "Gas Tank" (Sirtuins)
Scientific Perspective: Sirtuins are NAD+-dependent; they cannot repair DNA if NAD+ is depleted by chronic inflammation (which activates CD38). Yusri et al. (2025) note that restoring NAD+ isn't just about supplements; it's about lowering the "leak" caused by systemic inflammation.
"Your Sirtuins are like expert mechanics, but NAD+ is the fuel for their power tools. As we age, the fuel tank leaks. By eating anti-inflammatory foods and staying active, we plug the leaks so the mechanics can actually do their jobs."
4. The FOXO "Stress Shield"
Scientific Perspective: FOXO3 is the "longevity gene" that moves into the nucleus when insulin levels are low. It activates antioxidant enzymes like SOD2 and Catalase (Wang et al., 2026). High-insulin diets keep FOXO "locked out" of the nucleus where it can’t protect the cell.
"FOXO is your body's internal umbrella. When you keep your blood sugar stable, that umbrella stays open to protect you from the 'rain' of oxidative stress. If your insulin is always high, the umbrella stays closed, and your cells get soaked."
5. The "Inflammaging" Feedback Loop
Scientific Perspective: Overnutrition causes "nutrient-induced inflammation." This dysregulates the gut-brain axis and suppresses the production of short-chain fatty acids (SCFAs), which are essential for FOXO and SIRT1 signaling (Nanda & Patro, 2026).
"Overeating isn't just about calories; it’s about 'noise.' Too much food creates biological static that drowns out the signals your body uses to stay young. Eating fiber-rich, Mediterranean-style meals clears that static so your cells can hear the 'stay healthy' instructions again."
6. The Synergy of "The Big Two" (Fasting + Exercise)
Scientific Perspective: Exercise creates an acute energy deficit (AMPK spike), while fasting provides the prolonged low-insulin window for mTOR suppression. Dai et al. (2025) found that the combination yields a 25% better improvement in insulin sensitivity than either alone.
If you only exercise but eat all day, or only fast but never move, you’re only using half the toolkit. Combining a brisk walk with a consistent eating window is the '1-2 punch' that forces your metabolism to stay flexible and resilient."
I. AMPK: The Metabolic Master Switch for Cellular Homeostasis
From a clinical standpoint, AMPK (AMP-activated protein kinase) is the primary defender against metabolic syndrome and malignancy. It functions as a cellular "fuel gauge" that restores energy balance during metabolic stress.
Clinical Relevance: AMPK activation promotes insulin sensitivity, reduces systemic inflammation, and stimulates mitochondrial biogenesis.
Oncology Intersection: A 2024 review (Penugurti et al.) identifies AMPK as a critical "crossroads" in cancer therapy. While it suppresses early-stage tumor growth by inhibiting mTOR, it can paradoxically support survival in established tumors under nutrient stress.
Therapeutic Pearl: Pharmacological activators like metformin or natural mimetic berberine are used to "trick" the body into a fasted state, potentially reducing cancer risk by 30-50% in preclinical models.
II. mTOR: Managing the Growth-Repair Trade-off in Chronic Disease
mTOR (mechanistic target of rapamycin) is the primary anabolic driver. While essential for muscle protein synthesis (hypertrophy), chronic mTOR hyperactivation is a hallmark of "Inflammaging" and organ fibrosis.
Cardiovascular Impact: Recent data (Wang et al., 2025) highlight mTOR’s role in maladaptive cardiac remodeling. Nutrient overload hyperactivates the mTOR/insulin axis, driving hypertrophy and heart failure.
Neurological Implications: Zhang et al. (2025) suggest that mTOR overactivity inhibits autophagy, leading to the accumulation of misfolded proteins seen in neurodegenerative diseases.
Clinical Intervention: Pulsatile inhibition—rather than chronic suppression—is key. Strategies like Time-Restricted Feeding (TRF) allow for mTOR-driven repair during the day and AMPK-driven cleanup at night.
III. Sirtuins & NAD+: The Epigenetic Engine of Mitochondrial Health
Sirtuins (SIRT1-7) are NAD+-dependent enzymes responsible for DNA repair and genomic stability. Clinically, they are the "guardians" of the mitochondria.
The NAD+ Decline: Biological aging is characterized by a 50% drop in NAD+ by age 50 (Yusri et al., 2025). This depletion starves Sirtuins, leading to the mitochondrial decay seen in Type 2 Diabetes and Alzheimer’s.
Clinical Targets: NAD+ precursors (NR/NMN) have shown efficacy in human trials, improving muscle NAD+ by up to 60%.
Gastrointestinal Link: New research (Sah et al., 2025) connects Sirtuin activity to gut microbiome health, where SIRT1 activation reduces gut dysbiosis and associated systemic frailty.
IV. FOXO: The Transcription Factor for Stress Resilience
The FOXO family (specifically FOXO3) is often called the "Centenarian Gene." These factors translate nutrient signals into a survival script by upregulating antioxidant defenses.
Clinical Profile: FOXO activity is inversely related to insulin levels. High-carbohydrate diets keep FOXO "locked" in the cytoplasm, preventing it from entering the nucleus to perform DNA repair (Wang et al., 2026).
Longevity Mimetics: Future therapeutics are focusing on FOXO mimetics that could theoretically extend human healthspan by 15-25% by enhancing innate cellular stress resistance.
V. Overnutrition: Clinical Manifestations of Sensor Dysregulation
Chronic nutrient surplus (the "Western Diet") flips these sensors into a disease-promoting state.
The Pathology: Chronic insulin spikes suppress FOXO and SIRT1 while locking mTOR in the "on" position.
The Result: This leads to Non-Alcoholic Fatty Liver Disease (NAFLD) and Cardiovascular Disease (CVD).
Prescription: Nanda & Patro (2026) emphasize that switching to a Mediterranean-style pattern (rich in polyphenols like resveratrol) can recalibrate these sensors and reduce metabolic syndrome risk by 40%.
VI. Clinical Protocols: Implementing Fasting and Exercise
Lifestyle interventions remain the most potent "drugs" for sensor recalibration.
Synergy in Obesity: A 2025 meta-analysis (Dai et al.) found that combining Intermittent Fasting (IF) with High-Intensity Interval Training (HIIT) resulted in 25% better insulin sensitivity and 3-5% more fat loss than either intervention alone.
Mechanism: Exercise provides the acute AMPK "spike," while fasting provides the necessary mTOR "silence" for autophagy to occur.
Why Nutrient Sensing Matters in Real Life
In clinic, aging rarely announces itself as “mTOR activation” or “NAD+ depletion.” It shows up as rising fasting glucose, increasing abdominal fat, elevated blood pressure, stubborn fatigue, or slowly declining muscle mass. What we label as type 2 diabetes, fatty liver disease, sarcopenia, or cardiovascular risk often shares a common upstream driver: dysregulated nutrient sensing pathways.
When calories are constantly abundant, insulin and amino acids chronically stimulate mTOR, pushing the body toward growth rather than repair . At the same time, low cellular energy signaling through AMPK declines, reducing autophagy and metabolic flexibility (Penugurti et al., 2024). NAD+ levels gradually fall with age, weakening sirtuin-mediated mitochondrial repair and increasing inflammatory stress. FOXO longevity pathways become suppressed under persistent insulin signaling, limiting stress resistance and cellular resilience .
Clinically, this translates into a body that stores more, repairs less, inflames easily, and ages faster.
The encouraging news? These pathways are dynamic. Interventions such as structured exercise and intermittent fasting reactivate AMPK, suppress excessive mTOR signaling, improve insulin sensitivity, and restore metabolic flexibility (Dai et al., 2025). Even modest weight loss and time-restricted eating can shift cellular signaling from “constant growth mode” to “repair and resilience mode.”
Longevity is not simply about adding years. It is about restoring balance between growth and repair — and that balance begins at the level of nutrient sensing.
Metabolic Reset Protocol
This schedule balances the traditional meal structure with the clinical goal of pulsatile mTOR activation and AMPK restoration.
Breakfast: The "Protein-First" Metabolic Prime
Time: 08:00 AM – 09:00 AM
The Plate: High protein, high healthy fats, zero refined carbs.
Example: scrambled eggs with spinach and sautéed mushrooms in olive oil, plus half an avocado.
The Science: Starting with protein and fats rather than "cereal and juice" prevents an early-morning insulin spike. This keeps FOXO active longer while providing the amino acids needed to prevent muscle wasting (sarcopenia).
Clinical Pearl: Drink your coffee or green tea black to keep Sirtuins focused on DNA repair.
Lunch: The Micronutrient Load
Time: 01:00 PM – 02:00 PM
The Plate: Large Mediterranean-style salad or bowl.
Example: Grilled chicken or chickpeas, diverse colorful veggies (peppers, cucumbers, radishes), walnuts, and an olive oil/lemon dressing.
The Science: Diverse fiber and polyphenols support gut health and produce short-chain fatty acids (SCFAs). As noted by Nanda & Patro (2026), these SCFAs are essential signaling molecules that help activate SIRT1 and FOXO.
Activity: Take a 10-minute walk immediately after eating to activate AMPK and clear glucose from the bloodstream.
Dinner: The Autophagy Bridge
Time: 06:00 PM – 07:00 PM
The Plate: Moderate protein with complex, slow-burning carbohydrates.
Example: Baked cod or tofu with steamed asparagus and a small serving of quinoa or sweet potato.
The Science: Consuming your last calories by 7:00 PM creates a "fasting runway." By keeping dinner light and fibre-forward, you allow your insulin levels to drop quickly before sleep, which is the "on-switch" for nocturnal autophagy and growth hormone release.
The Overnight Repair Phase
Time: 07:00 PM – 08:00 AM (13-Hour Window)
Action: No snacking. Herbal tea or water only.
The Science: Even a 13-hour "circadian fast" is enough to silence mTOR and allow Sirtuins to focus on mitochondrial maintenance. This is the natural "Reset" button for your metabolic dashboard.
Frequently Asked Questions: The Science of Metabolic Longevity
1. What are nutrient sensing pathways and why do they matter for aging?
Nutrient sensing pathways are the body’s "biochemical compasses" that detect levels of glucose, amino acids, and ATP. The primary players include AMPK (energy sensor), mTOR (growth regulator), Sirtuins (DNA repair), and FOXO (stress resistance). When these pathways are balanced, they dictate whether your cells focus on growth and reproduction or repair and maintenance. Dysregulation in these systems is a primary driver of the nine hallmarks of aging.
2. How does fasting activate the AMPK energy sensor for autophagy?
Fasting triggers a rise in the AMP/ATP ratio, signaling to the cell that energy is scarce. This activates AMPK (AMP-activated protein kinase), which serves as a metabolic "master switch." According to Penugurti et al. (2024), activated AMPK shuts down energy-expensive protein synthesis and initiates autophagy—the process where cells recycle damaged components—and fat oxidation to create fresh energy.
3. Can mTOR inhibitors like Rapamycin actually extend human lifespan?
While human trials are ongoing, scientific consensus shows that inhibiting mTOR (mechanistic target of rapamycin) mimics the longevity benefits of caloric restriction. Zhang et al. (2025) highlight that reducing mTOR activity limits "cellular overgrowth," a key contributor to cancer and neurodegeneration. In animal models, mTOR inhibitors have extended lifespan by 10–20%, making them a primary focus for pharmacological anti-aging research.
4. What is the most effective way to boost NAD+ levels for Sirtuin activation?
The most potent strategy is a "dual-action" approach combining NAD+ precursors (like NR or NMN) with physical activity. Research by Yusri et al. (2025) demonstrates that this combination can increase muscle NAD+ levels by 50–60%. Sirtuins (SIRT1-7) are NAD+-dependent enzymes that stabilize the genome and optimize mitochondrial function, making NAD+ availability a bottleneck for cellular youth.
5. How does overnutrition and high insulin dysregulate the FOXO longevity pathway?
In a state of chronic nutrient surplus, high levels of insulin and IGF-1 keep the PI3K/Akt pathway hyperactive. This leads to the phosphorylation of FOXO transcription factors, effectively "locking" them out of the cell nucleus. Wang et al. (2026) note that when FOXO is inactive, the cell loses its ability to express protective genes for antioxidant defense and DNA repair, accelerating the onset of age-related diseases.
6. Is exercise more effective than intermittent fasting for metabolic health?
The latest meta-analyses suggest they are synergistic rather than competitive. Dai et al. (2025) found that combining exercise (HIIT or resistance training) with intermittent fasting led to a 3–5% greater fat loss and a 25% higher improvement in insulin sensitivity compared to doing either alone. Exercise spikes AMPK acutely, while fasting provides the sustained low-insulin window needed for deep cellular repair.
7. What is the best diet for balancing nutrient sensing pathways?
A Mediterranean-style dietary pattern is the gold standard for pathway harmony. High-fiber foods and polyphenols (like resveratrol and quercetin) activate AMPK and Sirtuins, while a moderate intake of plant-based proteins prevents chronic mTOR overstimulation. Nanda & Patro (2026) indicate that this balance reduces the risk of metabolic syndrome by 40% by aligning your biological sensors with evolutionary "thrifty" signals.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Author’s Note
As a clinician in internal medicine, I rarely see “aging” written on a prescription chart. What I see instead are its downstream expressions — rising fasting glucose, expanding waistlines, creeping blood pressure, declining muscle strength, fatty liver changes, subtle cognitive slowing. We name these type 2 diabetes, metabolic syndrome, sarcopenia, cardiovascular disease, or neurodegeneration. But increasingly, research shows they share a common upstream driver: dysregulated nutrient-sensing biology.
Pathways such as AMPK, mTOR, sirtuins, NAD+, and FOXO are not abstract molecular curiosities confined to laboratories. They are living switches inside every cell, responding continuously to how often we eat, how much we move, how we sleep, and how we manage stress. When chronically overstimulated by modern patterns of constant feeding and physical inactivity, these pathways shift the body toward growth without repair, storage without flexibility, and inflammation without resolution. When recalibrated through structured exercise, time-restricted eating, metabolic moderation, and anti-inflammatory nutrition, they restore resilience.
This article was written to bridge molecular science and practical medicine — to translate complex signaling networks into concepts that matter at the bedside and in everyday life. The goal is not to promote extreme biohacking or pharmacologic shortcuts, but to highlight a powerful truth: lifestyle remains the most potent modulator of these longevity pathways.
Aging is inevitable. Accelerated aging is often a biological miscommunication.
Related Articles
The Cardiometabolic Cascade: How Disease Starts—and How to Reverse It (2026 Science) | DR T S DIDWAL
Why Fat Tissue Is an Endocrine Organ — And What It Means for Your Health | DR T S DIDWAL
Tired All Day, Awake at Night? The Redox and Mitochondrial Link | DR T S DIDWAL
Obesity and Fatty Liver Disease: What Science Says About Risk and Health | DR T S DIDWAL
Intermittent Fasting: Metabolic Health Benefits and the Evidence on Longevity | DR T S DIDWAL
Activate Your Brown Fat: A New Pathway to Longevity and Metabolic Health | DR T S DIDWAL
References
Dai, Z. H., Wan, K. W., Wong, P. S., Huang, W. Y., Yu, A. P., & Wong, S. H. (2025). Additional effect of exercise to intermittent fasting on body composition and cardiometabolic health in adults with overweight/obesity: A systematic review and meta-analysis. Current Obesity Reports, 14(1), 54. https://doi.org/10.1007/s13679-025-00645-9
Nanda, A., & Patro, S. M. (2026). Dietary patterns and their impact on metabolic disorders: A comprehensive review. International Journal of Advanced Biochemistry Research, 10(2S), 751–758. https://doi.org/10.33545/26174693.2026.v10.i2Sj.7712
Penugurti, V., Manne, R. K., Bai, L., Kant, R., & Lin, H.-K. (2024). AMPK: The energy sensor at the crossroads of aging and cancer. Seminars in Cancer Biology, 106–107, 15–27. https://doi.org/10.1016/j.semcancer.2024.08.002
Sah, P., Rai, A. K., & Syiem, D. (2025). Sirtuin activators as an anti-aging intervention for longevity. Exploration of Digestive System. https://doi.org/10.37349/eds.2025.100881
Wang, J., Huang, Y., Wang, Z., Liu, J., Liu, Z., Yang, J., & He, Z. (2025). The mTOR signaling pathway: Key regulator and therapeutic target for heart disease. Biomedicines, 13(2), 397. https://doi.org/10.3390/biomedicines13020397
Wang, W., Zhu, H., Jiang, Q., Shi, Y., & Wang, X. (2026). FOXO: A key target in regulating aging and age-related diseases. Biogerontology, 27(1), 38. https://doi.org/10.1007/s10522-025-10380-2
Yusri, K., Jose, S., Vermeulen, K. S. et al. (2025). The role of NAD+ metabolism and its modulation of mitochondria in aging and disease. npj Metabolic Health and Disease, 3, 26. https://doi.org/10.1038/s44324-025-00067-0
Zhang, H., Xiao, X., Pan, Z. et al. (2025). mTOR signaling networks: Mechanistic insights and translational frontiers in disease therapeutics. Signal Transduction and Targeted Therapy, 10, 428. https://doi.org/10.1038/s41392-025-02493-4