Mitochondria: The Missing Link Between Fatigue, Weight Gain, and Brain Fog
Why are you always tired? Discover the cellular science behind mitochondrial dysfunction and proven strategies to restore energy.
METABOLISM
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/6/202615 min read
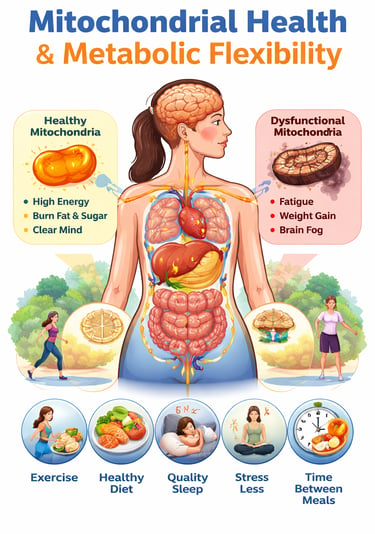
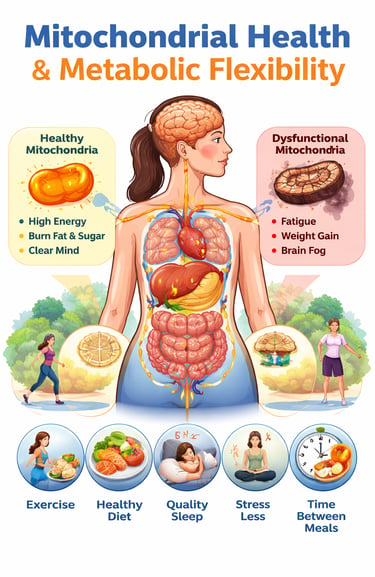
How impaired oxidative phosphorylation, reduced fatty acid oxidation, reactive oxygen species, and loss of metabolic flexibility are reshaping our understanding of chronic disease — and what you can do about it.
Inside every cell in your body are tiny structures called mitochondria. Think of them as your body’s power plants. Their job is to turn the food you eat into energy. When they work well, you feel energetic, mentally sharp, and physically strong. When they don’t, you may feel constantly tired, struggle with weight, or have difficulty controlling blood sugar.
Recent research shows that mitochondrial dysfunction plays a central role in common conditions such as obesity, type 2 diabetes, and age-related muscle loss (Yu et al., 2025). One major reason is something called metabolic inflexibility — the inability to smoothly switch between burning sugar and burning fat for fuel (Ang, 2025). A healthy body burns sugar after meals and shifts to fat between meals or during exercise. When this flexibility is lost, fat can accumulate in organs like the liver and muscles, promoting insulin resistance and inflammation (Aqeel et al., 2025).
Inefficient energy production can also increase harmful byproducts called reactive oxygen species (ROS). In small amounts, these molecules are normal and even helpful. But when they build up, they damage cells and weaken mitochondria further, creating a vicious cycle (Ansari et al., 2025). Interestingly, scientists have also found that metabolic inflexibility is sometimes temporarily protective during tissue repair, showing that the body’s energy system is complex and adaptive (Wang et al., 2024).
The encouraging news is that mitochondria respond strongly to lifestyle changes. Regular exercise, especially strength training and brisk walking, improves mitochondrial function. Time between meals can enhance fat burning. A whole-food diet, quality sleep, and stress management all support healthier energy production.
Many symptoms — fatigue, stubborn weight gain, brain fog — may reflect stressed mitochondria. The good news? Your cellular energy system can improve at any age.
Clinical pearls
1. The "Energy Brownout" Phenomenon
Scientific Context: Impaired Oxidative Phosphorylation (OXPHOS) means your cells are struggling to complete the final step of turning oxygen and nutrients into ATP. Even if your blood oxygen levels are normal, the "turbines" (ATP Synthase) aren't spinning efficiently.
Is this happening to me? If you feel "fine" at rest but hit a physical or mental wall almost immediately upon exertion, your cells are likely experiencing a localized energy shortage.
What this means for your fatigue: This isn't just "being tired." It’s a cellular brownout. Your brain and muscles are demanding high-voltage power, but your mitochondria are only providing low-voltage flickers.
The Reversibility: Yes. Zone 2 exercise (steady-state aerobic work) acts as "grid maintenance," slowly repairing these turbines and increasing their density.
2. The "Fuel Clog" (Reduced Beta-Oxidation)
Scientific Context: When CPT-I (the gatekeeper enzyme) is sluggish, fatty acids can't enter the mitochondria to be burned. They "back up" in the cell, converting into toxic lipid intermediates like ceramides.
Why can’t I lose weight? It’s not just about calories; it’s about access. Your body may have plenty of fat to burn, but the "door" to the furnace is locked. If you can't burn fat, your body signals for more hunger (glucose) because it thinks it’s starving.
Is this happening to me? If you feel "hangry" two hours after eating or can't skip a meal without a total crash, your fat-burning machinery is likely offline.
The Reversibility: High-intensity interval training (HIIT) and time-restricted eating can "force" the CPT-1 gate to reopen by depleting glycogen and demanding fat oxidation.
3. The "Exhaust Pipe" Leak (ROS & Oxidative Stress)
Scientific Context: Inefficient electron transport leads to electron leakage, where oxygen prematurely turns into Reactive Oxygen Species (ROS). This is like an engine that isn't just failing to move the car—it's also blowing hot, corrosive smoke back into the engine block.
What this means for your fatigue: ROS causes "systemic friction." Your body has to spend precious energy on "cleanup" and repair instead of performance, leading to that heavy, "toxic" feeling of exhaustion.
Is this reversible? Absolutely. Antioxidant-rich nutrition and deep sleep act as the "coolant" and "repair crew," neutralizing these leaks before they cause permanent DNA damage.
4. The "Stuck Gear" (Metabolic Inflexibility)
Scientific Context: Metabolic flexibility is the ability to switch between glucose (high-octane, fast-burning) and fat (long-range, slow-burning). Dysfunction leaves the body "stuck in one gear"—usually the glucose gear.
Why can’t I lose weight? Because you are stuck in glucose-burning mode, your body will protect its fat stores and instead demand sugar. This creates a cycle where you are overfed but cellularly underpowered.
What this means for your fatigue: You are riding a "blood sugar rollercoaster." When the sugar runs out, you crash, because your mitochondria have forgotten how to switch to the fat tank.
The Reversibility: This is highly reversible through "metabolic cross-training"—alternating between periods of fasting and nutrient-dense feeding.
5. The "Sarcopenic Cycle" (Muscle as the Battery)
Scientific Context: Skeletal muscle is your largest reservoir of mitochondria. As we age, a downregulation of OXPHOS genes leads to Sarcopenia (muscle loss), which in turn reduces the total number of "power plants" available to the body.
Is this happening to me? If you find it harder to hold your posture, open jars, or climb stairs, your "battery capacity" is shrinking.
Why can't I lose weight? Fewer mitochondria mean a lower Basal Metabolic Rate. You are burning less energy even while sleeping.
The Reversibility: Resistance training is the primary "software update" for this. It tells the body to build more muscle fibers and, crucially, to populate them with fresh, high-performing mitochondria.
What Are Mitochondria, and Why Do They Matter?
Before diving into dysfunction, it helps to understand normal function. Mitochondria produce a molecule called ATP (adenosine triphosphate) — the universal energy currency of the cell. They do this through a tightly coordinated process called oxidative phosphorylation, which takes place along the inner mitochondrial membrane via a series of protein complexes known as the electron transport chain (ETC).
Think of the ETC as a line of molecular workers passing electrons down a relay chain. As electrons move, they pump protons across a membrane, building up potential energy that is then used to spin a molecular turbine — ATP synthase — which snaps phosphate groups onto ADP to make ATP. It is one of the most elegant engineering feats in all of biology. But when any part of this chain misfires, the whole system suffers.
Pillar 1: Impaired Oxidative Phosphorylation
Oxidative phosphorylation (OXPHOS) is the primary mechanism by which mitochondria generate ATP. When it is impaired, cells simply cannot produce enough energy to meet their metabolic demands. This is not just a problem for rare genetic diseases — it is increasingly recognized as a shared mechanism across common chronic conditions.
A landmark 2025 study published in Scientific Reports by Yu et al. used integrated transcriptomics — a powerful technique that reads the gene expression profiles of thousands of cells simultaneously — to compare the molecular fingerprints of sarcopenia and obesity. Despite being superficially different conditions (one involving muscle wasting, the other excess fat), the researchers found that OXPHOS dysfunction was a shared underlying mechanism in both. Specifically, genes encoding proteins in mitochondrial complexes I through V were consistently downregulated across both conditions, suggesting that the mitochondria in affected individuals are simply not generating energy efficiently (Yu et al., 2025).
This is clinically important because it reframes obesity not merely as a problem of overeating, and sarcopenia not merely as a problem of under-exercising. Both may, at their root, reflect a failing cellular energy system — one that cannot adequately process the fuel it receives. For patients, this means that addressing mitochondrial health directly — not just counting calories or lifting weights in isolation — may be an essential part of long-term recovery.
Signs that OXPHOS may be impaired include:
Persistent fatigue that does not improve with rest
Brain fog or difficulty concentrating
Muscle weakness disproportionate to activity level
Poor exercise tolerance
Pillar 2: Reduced Fatty Acid Oxidation and Impaired β-Oxidation
Fats are the body's preferred long-term energy source. Under healthy conditions, fatty acids are shuttled into the mitochondria and broken down through a process called β-oxidation (beta-oxidation), which systematically strips off two-carbon units to generate acetyl-CoA — a key fuel for the Krebs cycle and, ultimately, ATP production. When this process is impaired, fats accumulate in tissues where they do not belong, triggering inflammation, insulin resistance, and cellular damage.
A comprehensive 2025 review by Aqeel et al., published in Diabetes Research and Clinical Practice, provides an in-depth look at the mechanisms behind impaired β-oxidation and its role in mitochondrial dysfunction. The authors highlight how disruptions in key enzymes — including carnitine palmitoyltransferase I (CPT-I), which acts as the gatekeeper for fatty acid entry into the mitochondria — can bottleneck the entire fat-burning process. When CPT-I activity is reduced, fatty acids accumulate in the cytoplasm, where they are converted into harmful intermediates such as ceramides and diacylglycerols, both of which interfere with insulin signaling and promote inflammatory cascades (Aqeel et al., 2025).
The authors further note that impaired β-oxidation creates a vicious cycle: the buildup of lipid intermediates damages the mitochondria themselves, further reducing their capacity to oxidize fatty acids, which in turn leads to greater lipid accumulation. In the context of type 2 diabetes, this cycle is particularly destructive — the pancreatic beta cells that produce insulin are themselves highly dependent on mitochondrial fatty acid oxidation for their function and survival.
Practical insight: This is one reason why simply reducing dietary fat intake, without addressing the underlying mitochondrial capacity to process fat, often fails to produce lasting metabolic improvements.
Pillar 3: Reactive Oxygen Species — When Byproducts Become Harmful
Every energy-producing process generates some waste. In mitochondrial energy production, the primary waste products are reactive oxygen species (ROS) — highly unstable molecules that contain oxygen and readily react with other molecules. In small amounts, ROS serve important signaling functions, helping cells respond to stress and regulate growth. But when they accumulate unchecked — a state called oxidative stress — they damage DNA, proteins, and the lipid membranes of cells, including the mitochondria themselves.
A thorough 2025 review by Ansari et al., published in Discovery Chemistry, catalogues the diverse sources of ROS generation in biological systems. The authors explain that mitochondria are both the primary producers of endogenous ROS (particularly at complexes I and III of the electron transport chain) and among the most vulnerable targets of oxidative damage. When electron transport is inefficient — as in OXPHOS dysfunction — electrons "leak" prematurely onto molecular oxygen, generating superoxide radicals. These can be dismuted into hydrogen peroxide, which, in the presence of iron, forms the highly reactive hydroxyl radical (Ansari et al., 2025).
The downstream effects are wide-ranging. ROS-induced damage to mitochondrial DNA is of particular concern because, unlike nuclear DNA, mitochondrial DNA lacks repair mechanisms and protective histone proteins. Accumulated mitochondrial DNA mutations impair the production of ETC subunits, further reducing OXPHOS efficiency and generating even more ROS — another self-reinforcing cycle.
Ansari et al. (2025) also discuss the body's antioxidant defense systems, including superoxide dismutase (SOD), catalase, and glutathione peroxidase, which under normal conditions neutralize ROS before they cause damage. In chronic disease states, the balance tips: ROS production outpaces antioxidant capacity, leading to chronic oxidative stress.
Key diseases linked to excessive ROS include:
Cardiovascular disease (oxidized LDL, endothelial damage)
Neurodegenerative diseases (Alzheimer's, Parkinson's)
Type 2 diabetes (beta-cell destruction)
Non-alcoholic fatty liver disease (NAFLD)
Cancer (DNA mutations)
Pillar 4: Loss of Metabolic Flexibility
Of all the concepts in this article, metabolic flexibility may be the most important for everyday health — and the least discussed. Metabolic flexibility refers to the body's ability to efficiently switch between different fuel sources — primarily glucose and fatty acids — depending on what is available and what is physiologically appropriate. After a carbohydrate-rich meal, a metabolically flexible person readily burns glucose. During fasting or prolonged exercise, they seamlessly transition to burning fat. A metabolically inflexible person, by contrast, is "stuck" — they struggle to efficiently use either fuel.
A 2025 review by Ang, published in Cell Reports Medicine, provides an authoritative analysis of metabolic flexibility across the whole body and individual tissues, with a focus on its implications in cardiometabolic disease. Ang (2025) distinguishes between whole-body metabolic flexibility — typically measured by the respiratory exchange ratio (RER) in response to feeding and fasting — and tissue-specific flexibility, which can differ markedly between, say, skeletal muscle, the liver, and adipose tissue. Insulin-resistant skeletal muscle, for example, is impaired in its ability to upregulate glucose oxidation after a meal, while also struggling to increase fat oxidation during fasting — the hallmark of metabolic inflexibility.
Ang (2025) further highlights that metabolic inflexibility is not merely a consequence of obesity or insulin resistance — it may actively drive disease progression. When tissues cannot effectively switch fuels, substrate competition leads to the accumulation of partially oxidized metabolites, increased ceramide synthesis, and ectopic lipid deposition in the liver and muscle — all of which independently worsen insulin resistance and cardiometabolic risk..
How These Four Pillars Interconnect
It would be misleading to treat these four mechanisms in isolation. They are deeply interdependent:
Impaired OXPHOS reduces the mitochondria's capacity to oxidize either glucose or fatty acids efficiently, contributing to both ROS generation and metabolic inflexibility.
Reduced β-oxidation means fatty acids cannot be efficiently burned, which forces greater reliance on (often impaired) glucose metabolism and accelerates lipid accumulation and inflammatory signaling.
Elevated ROS damages mitochondrial membranes, proteins, and DNA, further impairing both OXPHOS and fatty acid oxidation — and activating inflammatory pathways that worsen insulin resistance.
Metabolic inflexibility is both a consequence and a driver of the above: when cells cannot switch fuels cleanly, substrate overflow amplifies ROS production and lipotoxicity, while reduced energy availability impairs the very mitochondrial biogenesis needed to repair the system.
Together, these four mechanisms form a self-reinforcing web that, left unaddressed, can progress from subclinical dysfunction to frank metabolic disease.
Mitochondrial Health & Metabolic Flexibility Checklist
Assess your cellular energy efficiency across five key domains.
1. The Fatigue Profile (OXPHOS Efficiency)
These markers indicate how well your "molecular turbines" are spinning.
Do you feel "fine" at rest, but hit a sudden physical or mental wall shortly after starting a task?
Does your fatigue persist even after a full 8 hours of quality sleep?
Do you experience "post-exertional malaise" (feeling significantly worse the day after a workout)?
Do you struggle with "brain fog" that makes it hard to focus on complex mental tasks?
2. The Metabolic Flexibility Test (Fuel Switching)
These markers indicate if you are "stuck" in glucose-burning mode.
Do you experience a "mid-afternoon crash" or feel shaky if a meal is delayed?
Do you find it impossible to exercise in a fasted state (e.g., before breakfast)?
Do you experience "hanger" (irritability paired with hunger) frequently?
Do you have intense cravings for sugar or refined carbohydrates, especially under stress?
3. The "Fuel Clog" Assessment (Fatty Acid Oxidation)
These markers suggest fatty acids are building up in the cell rather than being burned.
Is your weight loss "stalled" despite a significant calorie deficit?
Have you been told you have "ectopic fat" (Fatty Liver/NAFLD)?
Do you have high fasting triglycerides (typically >100 mg/dL)?
Do you notice a "heavy" feeling in your muscles even with light activity?
4. The Recovery & Friction Check (ROS/Oxidative Stress)
These markers suggest your "exhaust pipe" is leaking, causing cellular damage.
Do you have persistent, low-grade joint or muscle aches with no clear injury?
Do you feel "puffy" or inflamed, particularly in your face or hands, in the mornings?
Is your skin losing its resilience or showing signs of "premature aging"?
Do you have a slow recovery time from common colds or minor injuries?
5. The "Battery Capacity" Indicator (Sarcopenia & Muscle)
These markers assess the size and health of your mitochondrial reservoir.
Have you noticed a decrease in your grip strength or difficulty opening jars?
Do you find it harder to maintain your posture throughout a long day?
Has your "walking speed" naturally slowed down over the last few years?
Are you losing muscle mass despite eating adequate protein?
Interpreting Your Results
0–3 Checks: Your mitochondria are likely resilient. Focus on "maintenance" via Zone 2 cardio and nutrient-dense eating.
4–8 Checks: You may be experiencing "Metabolic Inflexibility." Prioritize time-restricted eating and HIIT to force fuel-switching.
9+ Checks: Significant "Mitochondrial Dysfunction" is likely present. Consider a clinical workup focusing on HOMA-IR, Triglyceride/HDL ratios, and a deep dive into sleep and micronutrient cofactors (CoQ10, Magnesium, B-Vitamins).
Practical Applications: Supporting Your Mitochondria
The good news is that mitochondria are remarkably responsive to lifestyle interventions. Here is what the evidence supports:
1. Exercise — Especially Resistance and High-Intensity Interval Training (HIIT)
Exercise is the most potent known stimulus for mitochondrial biogenesis — the creation of new mitochondria. Both endurance and resistance training upregulate PGC-1α, the master regulator of mitochondrial biogenesis, while also improving OXPHOS efficiency and β-oxidation capacity. Even moderate, consistent activity — 30 minutes of brisk walking five days per week — meaningfully improves metabolic flexibility over time.
2. Time-Restricted Eating and Intermittent Fasting
Periods of fasting give mitochondria a metabolic "workout" by forcing the transition from glucose to fatty acid oxidation. This trains the machinery of β-oxidation and improves whole-body metabolic flexibility, as outlined by Ang (2025). Fasting also stimulates mitophagy — the selective removal of damaged mitochondria — clearing the way for healthier replacements.
3. A Nutrient-Dense, Anti-Inflammatory Diet
Reducing ultra-processed foods and refined carbohydrates lowers the chronic glucose and insulin spikes that suppress fat oxidation and promote metabolic inflexibility. Foods rich in polyphenols — berries, olive oil, green tea, dark chocolate — support antioxidant defenses and reduce ROS-mediated damage, as highlighted by Ansari et al. (2025).
4. Targeted Nutritional Support
Coenzyme Q10 (CoQ10): A critical component of the electron transport chain; supplementation has shown promise in conditions involving OXPHOS impairment.
Magnesium: Required as a cofactor for over 300 enzymatic reactions, including many in mitochondrial energy metabolism.
B vitamins (especially B1, B2, B3, B5): Essential cofactors for the Krebs cycle and β-oxidation.
Alpha-lipoic acid: A potent mitochondria-targeted antioxidant.
5. Sleep Optimization
Poor sleep acutely impairs mitochondrial function and increases ROS production. Prioritizing 7–9 hours of quality sleep per night is one of the most underrated mitochondrial interventions available.
6. Stress Reduction
Chronic psychological stress elevates cortisol, which promotes glucose mobilization, suppresses fatty acid oxidation, and increases mitochondrial ROS production. Mind-body practices such as yoga, meditation, and controlled breathing can meaningfully reduce oxidative stress burden.
Frequently Asked Questions (FAQs)
1. Why do I feel tired all the time even after sleeping well?
Feeling constantly tired—even after a full night’s sleep—can happen when your body’s energy-producing systems are not working efficiently. Inside your cells are tiny structures called mitochondria, often called the power plants of the cell. Their job is to convert food and oxygen into energy.
When mitochondria become less efficient—because of poor diet, inactivity, insulin resistance, chronic stress, or inflammation—cells produce less energy. As a result, your brain, muscles, and organs may feel like they are running on a low battery. This can cause persistent fatigue, brain fog, and reduced stamina even when sleep seems adequate.
2. What is mitochondrial dysfunction in simple terms?
Mitochondrial dysfunction simply means that your cells are not producing energy as efficiently as they should.
Normally, mitochondria convert nutrients like glucose and fats into a molecule called ATP, which powers every process in the body—from muscle movement to brain function. When mitochondria are damaged or overloaded, this energy production slows down.
Think of it like a power station that cannot generate enough electricity for the city. The result can be fatigue, slower metabolism, muscle weakness, and reduced physical performance.
3. What symptoms can mitochondrial dysfunction cause?
When cellular energy production declines, the body may show several symptoms, including:
Persistent fatigue
Brain fog or difficulty concentrating
Muscle weakness or reduced endurance
Exercise intolerance
Slow recovery after physical activity
Weight gain or metabolic problems
Increased sensitivity to stress
These symptoms occur because tissues with high energy needs—such as the brain, heart, and muscles—are the first to feel the effects of reduced cellular energy.
4. Can mitochondrial dysfunction be reversed?
In many cases, mitochondrial function can improve significantly with lifestyle changes. Research shows that mitochondria respond strongly to healthy habits.
Strategies that help support mitochondrial health include:
Regular physical activity, especially resistance and aerobic exercise
Balanced nutrition rich in whole foods and adequate protein
Good sleep habits
Stress reduction
Maintaining healthy blood sugar levels
Exercise is particularly powerful because it stimulates the body to create new mitochondria and improve their efficiency.
5. How long does it take to improve mitochondrial health?
Improvements can begin within weeks of consistent lifestyle changes. Studies show that regular exercise can start improving mitochondrial function within 4–8 weeks, while metabolic health often continues to improve over several months.
The key is consistency. Mitochondria are dynamic—they constantly adapt to how you eat, move, and live.
Author’s Note
As a physician in internal medicine, I have seen countless patients struggle with fatigue, weight gain, insulin resistance, and “unexplained” metabolic decline — often despite normal basic lab results. Increasingly, research is showing that many of these issues share a common root: impaired cellular energy production.
Mitochondrial dysfunction and metabolic inflexibility are not fringe concepts. They are emerging as central mechanisms linking obesity, type 2 diabetes, cardiovascular disease, and age-related muscle loss. What is particularly encouraging, however, is that mitochondrial biology is dynamic. These cellular power plants are highly responsive to exercise, nutrition, sleep, and stress patterns.
The goal of this article is not to oversimplify complex biology — nor to promote quick fixes — but to help you understand the deeper physiology behind common symptoms. When patients understand why something is happening, lifestyle changes become purposeful rather than frustrating.
It is also important to recognize that while lifestyle strategies are powerful, they are not substitutes for individualized medical care. If you are experiencing persistent fatigue, blood sugar instability, or unexplained metabolic changes, consult a qualified healthcare professional for evaluation.
Your body is not failing you. Often, it is signaling that its energy systems need support.
Understanding those signals is the first step toward restoring metabolic resilience.
Your body’s energy problem may not be about willpower — it may be about mitochondria. These tiny structures inside your cells convert food into usable energy. When they function poorly, you may experience persistent fatigue, brain fog, poor exercise tolerance, difficulty losing weight, or unstable blood sugar — even when routine lab tests look “normal.”
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Mitochondria and Metabolic Flexibility: The Real Secret to Fat Loss and Longevity
Muscle Insulin Resistance: The Hidden Signaling Failure Behind Metabolic Disease | DR T S DIDWAL
Obesity and Fatty Liver Disease: What Science Says About Risk and Health | DR T S DIDWAL
Intermittent Fasting: Metabolic Health Benefits and the Evidence on Longevity | DR T S DIDWAL
Activate Your Brown Fat: A New Pathway to Longevity and Metabolic Health | DR T S DIDWAL
References
Ang, J. H. C. (2025). Perspectives on whole body and tissue-specific metabolic flexibility and implications in cardiometabolic diseases. Cell Reports Medicine, 6, 102354. https://doi.org/10.1016/j.xcrm.2025.102354
Ansari, W. A., Srivastava, K., Nasibullah, M., et al. (2025). Reactive oxygen species (ROS): Sources, generation, disease pathophysiology, and antioxidants. Discovery Chemistry, 2, 191. https://doi.org/10.1007/s44371-025-00275-z
Aqeel, A., Akram, A., Ali, M., Iqbal, M., Aslam, M., Rukhma, & Shah, F. I. (2025). Mechanistic insights into impaired β-oxidation and its role in mitochondrial dysfunction: A comprehensive review. Diabetes Research and Clinical Practice, 223, Article 112129. https://doi.org/10.1016/j.diabres.2025.112129
Wang, X., et al. (2024). Metabolic inflexibility promotes mitochondrial health during liver regeneration. Science, 384, eadj4301. https://doi.org/10.1126/science.adj4301
Yu, R., Miao, L., et al. (2025). Integrated transcriptomics unveils mitochondrial oxidative phosphorylation dysfunction as a shared mechanism in sarcopenia and obesity. Scientific Reports, 15, 34810. https://doi.org/10.1038/s41598-025-18824-y