Reverse Metabolic Aging: How Exercise Rebuilds Mitochondria and Restores Cellular Energy
Discover how exercise triggers mitochondrial biogenesis, improves cellular energy, and helps reverse metabolic aging through powerful molecular pathways.
EXERCISEAGING
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/7/202615 min read
"Exercise is the closest thing we have to a polypill — but the real story is not in how it burns calories. It is in how it reprograms your cells."
What if the most powerful therapy for metabolic disease was not a pill, but a biological signal your body has been designed to generate for millions of years?
Most people think of exercise mainly as a way to burn calories or lose weight. You go for a walk, jog, or lift some weights, and you assume the main benefit is simply using up energy. But modern research in metabolism and exercise physiology shows that something much deeper is happening inside your body.
Every time your muscles contract—even during something as simple as brisk walking, climbing stairs, or cycling—your body activates a complex network of molecular signals that influence how your cells produce energy, regulate blood sugar, and repair themselves. In fact, scientists now describe exercise as a form of molecular medicine because it can directly improve the biological processes that drive many chronic diseases (Stead et al., 2024).
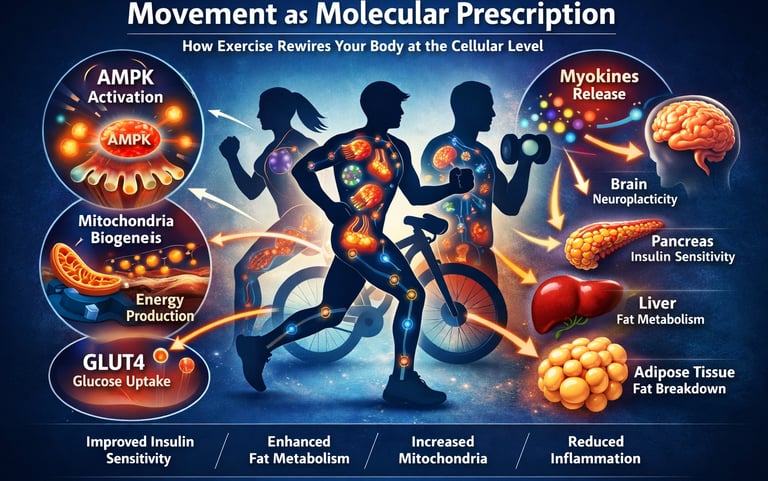
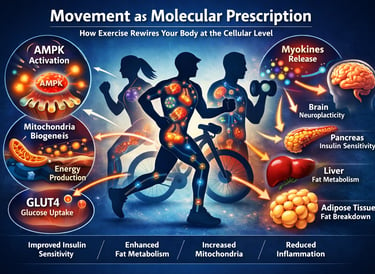
One of the most important discoveries in recent decades is that skeletal muscle is not just a structure that moves your body—it is also an endocrine organ. When muscles work, they release signaling molecules called myokines, which travel through the bloodstream and communicate with other organs such as the liver, pancreas, fat tissue, and brain. These signals help regulate insulin sensitivity, inflammation, fat metabolism, and even cognitive health (De Sousa, 2026).
Exercise also activates powerful metabolic pathways inside cells. One of the key regulators is AMP-activated protein kinase (AMPK)—often described as the cell’s energy sensor. When AMPK is activated during physical activity, it helps the body burn fat more efficiently, improve glucose uptake, and stimulate the production of new mitochondria, the tiny energy-producing structures inside cells (Carapeto et al., 2024).
These effects are particularly important because many modern health problems—including type 2 diabetes, obesity, cardiovascular disease, and metabolic aging—are closely linked to impaired mitochondrial function and poor metabolic regulation.
Clinical Pearls: Translating Molecular Exercise Science Into Practice
1. Exercise Activates the Body’s Master Energy Sensor (AMPK)
Scientific Perspective:
During physical activity, the intracellular AMP/ATP ratio increases, activating AMP-activated protein kinase (AMPK). This signalling pathway enhances glucose uptake, stimulates fatty acid oxidation, inhibits lipogenesis, and activates downstream metabolic regulators such as SIRT1 and PGC-1α, thereby promoting mitochondrial biogenesis and metabolic flexibility.
When you move your muscles, your cells switch into “energy-repair mode.” This helps your body burn sugar and fat more efficiently and improves how your metabolism works over time.
2. Muscle Contraction Lowers Blood Sugar Even Without Insulin
Scientific Perspective:
Exercise induces insulin-independent translocation of GLUT4 transporters to the skeletal muscle cell membrane, enabling glucose uptake independent of insulin receptor signaling. This mechanism is particularly beneficial in insulin resistance and type 2 diabetes.
Even if your body does not respond well to insulin, exercise still pulls sugar out of your blood and into your muscles, helping control blood glucose naturally.
3. Exercise Builds Healthier Mitochondria
Scientific Perspective:
Repeated exercise activates PGC-1α–mediated mitochondrial biogenesis, increasing mitochondrial density, oxidative phosphorylation capacity, and metabolic efficiency while reducing oxidative stress and metabolic dysfunction.
Exercise helps your cells build more and better “energy factories.” This means more stamina, better metabolism, and improved overall health.
4. Muscle Is an Endocrine Organ
Scientific Perspective:
Contracting skeletal muscle releases myokines, including irisin, IL-6, and myostatin regulators, which influence systemic metabolic pathways, modulate inflammation, and affect brain-derived neurotrophic factor (BDNF) signalling.
When you exercise, your muscles release beneficial chemical messengers that communicate with your brain, liver, and fat tissue—helping improve mood, metabolism, and overall health
5. Consistency of Exercise Sustains Metabolic Benefits
Scientific Perspective:
Exercise-induced metabolic signaling pathways—including GLUT4 expression, AMPK activation, and mitochondrial enzyme activity—remain elevated for approximately 24–48 hours, emphasizing the importance of regular activity.
The benefits of exercise fade if you stop moving for several days, so regular activity—even simple walking—helps keep your metabolism working efficiently.
Part I: The Master Energy Sensor — AMPK Activation
What Is AMPK and Why Does It Matter?
Adenosine monophosphate-activated protein kinase, or AMPK, is often called the cell's "energy gauge." When your cells use up their energy stores during exercise — when the ratio of AMP to ATP rises — AMPK flips on like a circuit breaker, triggering a cascade of energy-restoring, health-promoting responses.
Think of AMPK as a wise internal accountant. When the energy budget runs low, it immediately begins optimizing: it shuts down processes that consume too much energy, and switches on those that generate it. But crucially, many of those energy-generating responses also happen to be powerfully anti-aging and anti-diabetic.
Recent research has expanded our understanding of AMPK beyond muscle and liver. In a landmark 2024 study published in Nature Metabolism, Carapeto et al. demonstrated that exercise activates AMPK not just in skeletal muscle but in pancreatic islets — the clusters of cells responsible for producing insulin. Remarkably, this AMPK activation reduced cellular senescence (the process by which old, dysfunctional cells accumulate and drive disease) in both mouse and human pancreatic tissue (Carapeto et al., 2024). This finding reframes exercise as a therapy that directly protects the insulin-producing machinery of the pancreas — a profound implication for type 2 diabetes prevention and management.
The AMPK-SIRT1-PGC-1α Longevity Axis
AMPK does not work in isolation. It activates SIRT1, a protein deacetylase involved in stress resistance and longevity, which in turn amplifies the activity of PGC-1α — the master regulator of mitochondrial biogenesis and energy metabolism. This coordinated AMPK/SIRT1/PGC-1α axis is, in essence, the molecular heartbeat of why exercise extends healthspan.
A 2026 review by De Sousa published in Molecular Biology Reports synthesizes how this axis connects to irisin and BDNF (brain-derived neurotrophic factor), forming an integrated signalling network that links physical activity to muscle health, brain plasticity, and systemic metabolic protection When you exercise regularly, you are not just activating this axis transiently — you are building a more sensitive, more efficient molecular signalling infrastructure over time.
Clinical Takeaway: AMPK activation during exercise is a master switch for metabolic health. It reduces pancreatic senescence, stimulates fat oxidation, improves insulin sensitivity, and triggers downstream longevity pathways. This is why exercise works across so many disease categories simultaneously.
Part II: GLUT4 — The Insulin-Independent Glucose Gateway
The Problem With Insulin Resistance
In type 2 diabetes and metabolic syndrome, the tissues of the body stop responding properly to insulin's signal to take up glucose. The pancreas tries to compensate by producing more and more insulin, but cells simply stop listening. This is insulin resistance, and it lies at the heart of most modern chronic disease.
Most therapeutic strategies target the pancreas (stimulating more insulin production) or peripheral insulin receptors. But exercise reveals a different, often overlooked pathway: the activation of GLUT4 transporters independent of insulin.
How Exercise Bypasses Insulin Resistance
GLUT4 is a protein that acts like a gate in the cell membrane, allowing glucose to enter from the bloodstream. Normally, this gate opens only when insulin sends the signal. But during muscle contraction, GLUT4 transporters migrate to the cell surface through a completely separate signalling pathway — one that works even when insulin signalling is broken.
This is clinically transformative. It means that even in someone whose cells have completely lost sensitivity to insulin, muscle contraction can still pull glucose out of the bloodstream and into the cells where it is needed.
McGee and Hargreaves (2024), writing in Free Radical Biology and Medicine, provide a comprehensive overview of how exercise performance and metabolic health are regulated through GLUT4, highlighting the downstream benefits for glucose homeostasis that occur independently of the insulin receptor pathway .This mechanism partially explains why even a single bout of moderate exercise can lower blood glucose for 24–48 hours in individuals with impaired glucose regulation.
Clinical Takeaway: Exercise activates GLUT4-mediated glucose uptake through a pathway that bypasses broken insulin signalling. This makes movement one of the most powerful tools available for managing blood glucose in insulin-resistant individuals — and it works from the very first session.
Part III: Building Better Energy Factories — Mitochondrial Biogenesis
Why Mitochondria Are Central to Metabolic Health
Mitochondria are the energy factories inside every cell. They generate the ATP that powers muscle contraction, thought, heartbeat, and every other biological process. In metabolic disease, mitochondrial function is almost universally compromised. Fewer mitochondria, less efficient mitochondria, more oxidative stress — these are hallmarks of insulin resistance, obesity, type 2 diabetes, and accelerated aging.
The good news? Exercise is the most potent known stimulus for mitochondrial biogenesis — the process of making new mitochondria and improving the quality of existing ones.
What the Latest Meta-Analysis Tells Us
A systematic review and meta-analysis published in Biomolecular Concepts, pooled data from randomized controlled trials to examine the effect of different exercise types on mitochondrial biogenesis in skeletal muscle (Abrego-Guandique et al., 2025). The findings confirm that both aerobic and resistance exercise reliably increase markers of mitochondrial biogenesis, particularly through upregulation of PGC-1α — the same transcriptional coactivator activated downstream of the AMPK/SIRT1 axis described earlier.
Both modalities increase mitochondrial density, improve oxygen-consumption efficiency, and reduce the oxidative damage that characterises metabolically unhealthy muscle tissue. Importantly, even relatively modest exercise interventions — conducted over weeks to months — produced measurable improvements in mitochondrial markers in previously sedentary individuals.
Aerobic vs. Resistance: Different Paths, Complementary Outcomes
Stead, Mackin, and Handschin (2024), in a thorough review in Free Radical Biology and Medicine, map the distinct molecular adaptations that occur with aerobic versus resistance training. Aerobic exercise predominantly drives mitochondrial biogenesis, fat oxidation capacity, and cardiovascular efficiency. Resistance training, by contrast, primarily induces protein synthesis, myofiber hypertrophy, and improvements in neuromuscular force generation (Stead et al., 2024).
At the cellular signalling level, these distinctions are mediated by different phosphorylation and acetylation patterns in muscle proteins — a point elegantly demonstrated in a 2025 study published in Nature Communications. Using phosphoproteomics and acetylproteomics, the researchers mapped the specific molecular modifications that occur in human skeletal muscle following both aerobic and resistance exercise sessions, revealing distinct but partially overlapping signalling landscapes (Pataky et al., 2025). This molecular atlas of exercise adaptation confirms that the two exercise modalities are complementary rather than interchangeable — a powerful argument for combining both in any therapeutic exercise prescription.
Clinical Takeaway: Exercise builds and improves mitochondria — the fundamental units of metabolic health. Aerobic training and resistance training produce different but synergistic molecular adaptations. For optimal metabolic health, both are needed. Neither alone tells the whole story.
Part IV: Myokines — Muscle's Secret Hormonal Language
The Discovery That Changed Exercise Science
Perhaps the most revolutionary insight of the past two decades in exercise biology is this: contracting skeletal muscle is an endocrine organ. When muscle fibres work hard, they release signalling proteins called myokines — cytokines produced and secreted by muscle — that travel through the bloodstream and influence other tissues.
This discovery fundamentally reframes how we understand exercise's systemic benefits. The health effects of physical activity are not merely mechanical or metabolic in the narrow sense — they are hormonal, communicating with fat tissue, liver, brain, bone, and immune cells.
Irisin: The Cross-Organ Messenger
Among the myokines, irisin has attracted enormous scientific interest. Released by exercising muscle, irisin crosses the blood-brain barrier, stimulates BDNF production in the hippocampus (supporting cognitive function and mood), promotes browning of white adipose tissue (converting calorie-storing fat into calorie-burning fat), and improves bone density.
De Sousa's (2026) analysis of the AMPK/SIRT1/PGC-1α/Irisin/BDNF axis elegantly shows how these molecules form a unified cross-organ communication system. PGC-1α activation in muscle drives irisin release; irisin then acts in the brain to stimulate BDNF, creating a molecular bridge between physical activity and cognitive resilience. This molecular pathway offers one mechanistic explanation for why regular exercise is consistently associated with reduced risk of depression, dementia, and cognitive decline
IL-6: Context Is Everything
Another important myokine is interleukin-6 (IL-6). In the context of chronic disease, IL-6 is associated with low-grade systemic inflammation and metabolic harm. But during exercise, acutely elevated IL-6 released from contracting muscle serves entirely different, anti-inflammatory functions — stimulating glucose uptake, promoting fat oxidation, and regulating energy homeostasis.
This distinction — between chronic inflammatory IL-6 and exercise-induced myokine IL-6 — is clinically important. It illustrates a broader principle: the same molecule can have diametrically opposite effects depending on context, timing, and origin. Exercise does not simply reduce inflammation by reducing body fat. It actively teaches the immune system a more adaptive response.
Clinical Takeaway: Muscle is an endocrine organ. Every time you exercise, your muscles release hormones that communicate with your brain, liver, fat cells, and immune system. This is the molecular basis for exercise's seemingly miraculous multi-system benefits — from better mood and sharper cognition to improved blood sugar and reduced inflammation.
Part V: The Phosphoproteomic Revolution — Understanding Exercise at Molecular Resolution
Science's ability to study exercise has entered a new era. Rather than measuring simple outcomes like blood glucose or oxygen consumption, researchers can now map thousands of molecular modifications in muscle tissue simultaneously — revealing the full complexity of exercise's cellular effects.
Pataky et al. (2025) conducted exactly such an analysis, profiling the phosphoproteome and acetylproteome of human skeletal muscle following aerobic and resistance exercise sessions. Their Nature Communications study identified hundreds of protein modifications specific to each exercise type, many of which had not previously been linked to exercise adaptation. This kind of molecular cartography is beginning to explain why different exercise types produce different outcomes — and may eventually allow us to design exercise prescriptions with the same precision we currently reserve for pharmaceutical dosing.
This research also confirms what clinicians have long suspected: exercise is not a single intervention. It is a highly complex, multi-pathway biological stimulus that modifies gene expression, protein function, and metabolic flux in ways that no single drug could replicate. The polypharmacology of movement — its simultaneous action across dozens of pathways — is precisely what makes it so therapeutically potent.
Key Takeaways: How Exercise Reprograms Metabolism
Exercise functions as a systemic molecular therapy, not merely a calorie-burning activity.
When skeletal muscle contracts, it triggers a cascade of intracellular signaling pathways that regulate energy metabolism, inflammation, and cellular repair. This molecular response affects nearly every major organ system, including the liver, pancreas, adipose tissue, brain, and immune system. Exercise therefore acts as a whole-body metabolic regulator rather than a simple mechanical activity.AMPK activation acts as the master metabolic switch during exercise.
Physical activity increases the cellular AMP/ATP ratio, activating AMP-activated protein kinase (AMPK) — the cell’s primary energy sensor. Once activated, AMPK promotes glucose uptake, stimulates fat oxidation, suppresses energy-wasting anabolic processes, and activates longevity-associated pathways including SIRT1 and PGC-1α signaling. This axis plays a central role in improving insulin sensitivity and metabolic flexibility.Exercise enables glucose uptake even when insulin signaling is impaired.
Muscle contraction activates GLUT4 translocation through insulin-independent pathways. This allows glucose to enter skeletal muscle cells without relying on insulin receptor signaling. As a result, even individuals with insulin resistance or early type 2 diabetes can significantly improve blood glucose control through regular physical activity.Mitochondrial biogenesis is a core mechanism through which exercise restores metabolic health.
Regular aerobic and resistance exercise stimulates the production of new mitochondria and enhances the efficiency of existing ones. This process, largely driven by PGC-1α activation, improves oxidative metabolism, reduces reactive oxygen species production, and enhances cellular energy availability. Mitochondrial remodeling is a key reason exercise improves conditions such as obesity, metabolic syndrome, and age-related metabolic decline.Skeletal muscle functions as a powerful endocrine organ through myokine signaling.
During exercise, contracting muscle fibers release signaling proteins known as myokines, including irisin and IL-6. These molecules coordinate communication between muscle and distant organs, influencing fat metabolism, immune function, brain plasticity, and glucose homeostasis. This endocrine function explains why exercise has benefits far beyond the musculoskeletal system.Different exercise modalities activate complementary molecular pathways.
Aerobic exercise predominantly enhances mitochondrial density, oxidative metabolism, and cardiovascular efficiency, while resistance training stimulates muscle protein synthesis, neuromuscular strength, and glucose disposal capacity. Combining both modalities produces the most comprehensive metabolic adaptation.Consistency of movement matters more than intensity alone.
Many exercise-activated pathways — particularly GLUT4-mediated glucose uptake and AMPK signaling — remain elevated for only 24–48 hours after activity. Regular movement maintains these metabolic benefits and prevents the progressive decline in insulin sensitivity associated with sedentary lifestyles.In modern metabolic medicine, movement should be considered a foundational therapy.
Exercise simultaneously targets multiple biological drivers of chronic disease — insulin resistance, mitochondrial dysfunction, systemic inflammation, and metabolic inflexibility. No single pharmaceutical intervention currently replicates this multi-pathway therapeutic effect.
Start wherever you are. The molecular benefits of exercise begin with the first session. Even a single bout of walking activates AMPK, triggers GLUT4 translocation, and releases myokines. There is no threshold of fitness required to begin benefiting at the cellular level.
Frequently Asked Questions
Q1. Can exercise actually replace diabetes medication?
Not in all cases, and medical supervision is essential before making any changes to prescribed treatment. However, exercise addresses insulin resistance at its molecular root in ways that most medications do not. Regular resistance and aerobic training can significantly reduce HbA1c, lower fasting insulin, and, in some cases of early-stage type 2 diabetes, may reduce medication requirements under a doctor's guidance. The GLUT4 mechanism means exercise lowers blood glucose even when insulin signalling is broken — a capability no current oral medication fully replicates.
Q2. How long before I see metabolic changes from starting exercise?
Cellular changes begin with the very first session. AMPK activates within minutes of exercise onset, GLUT4 translocation occurs during the session itself, and myokine release is immediate. Measurable improvements in fasting glucose and insulin sensitivity typically appear within 2–4 weeks of regular exercise. Mitochondrial biogenesis — the rebuilding of cellular energy factories — is detectable after 4–6 weeks of regular training.
Q3. Is walking enough, or do I need to do structured exercise?
Walking is genuinely therapeutic and should never be underestimated. It activates GLUT4, stimulates AMPK, and releases myokines. For individuals who are sedentary, moving to daily brisk walking produces substantial metabolic benefit. That said, resistance training is uniquely powerful for building muscle mass, which is the primary driver of long-term insulin sensitivity. Ideally, both should be included as fitness allows.
Q4. Why does exercise seem to help with depression and brain fog?
The irisin-BDNF connection is the key molecular answer. Irisin, released by working muscle, crosses the blood-brain barrier and stimulates BDNF production in the hippocampus — a region critical for mood regulation, memory formation, and stress resilience. Regular exercise is one of the most evidence-based interventions for mild-to-moderate depression precisely because this molecular pathway creates measurable neurological changes over time.
Q5. Does the type of exercise matter for mitochondrial health?
Both aerobic and resistance exercise improve mitochondrial function, but through partially different mechanisms. Aerobic exercise is particularly potent for increasing mitochondrial volume density and oxidative capacity. Resistance training improves mitochondrial quality and reduces mitochondrial oxidative stress in muscle. The 2025 meta-analysis by Abrego-Guandique et al. confirms that both are effective, and combining them produces the most comprehensive mitochondrial adaptation.
Q6. I have been told I am insulin-resistant but not yet diabetic. Is exercise still important?
This is actually the most important moment to intervene with exercise. Insulin resistance precedes type 2 diabetes by years, often decades, and is almost completely reversible with lifestyle change — including regular exercise — at this stage. The molecular mechanisms discussed in this article (AMPK activation, GLUT4 upregulation, mitochondrial biogenesis, myokine release) are all highly responsive to exercise in the prediabetic state. Early intervention has a disproportionately large impact on long-term outcomes.
Q7. Is there an optimal time of day to exercise for blood sugar control?
Emerging evidence suggests that post-meal exercise — even a 10–15 minute walk after eating — is particularly effective for blunting post-meal glucose spikes because the GLUT4 translocation effect is most relevant when circulating glucose is high. Morning exercise may enhance the AMPK/fat oxidation axis. For most people, however, the best time to exercise is whenever it can be done consistently. Consistency across weeks and months produces far greater metabolic benefit than optimising the timing of individual sessions.
Q8 How does exercise improve metabolic health?
Exercise functions as a systemic molecular therapy by activating cellular sensors like AMPK and PGC-1α. These pathways increase insulin sensitivity, reduce systemic inflammation, and trigger the release of myokines—hormones from skeletal muscle that communicate with the brain, liver, and pancreas to regulate energy balance and glucose homeostasis.
Q9 How does exercise increase mitochondrial biogenesis?
Exercise induces mitochondrial biogenesis primarily through the activation of the PGC-1α transcriptional coactivator. Triggered by muscle contraction and energy depletion (AMPKactivation), this process stimulates the production of new mitochondria and enhances the efficiency of existing ones, improving oxidative capacity and reducing metabolic oxidative stress.
Q10 Can exercise lower blood sugar without insulin?
Yes. Muscle contraction triggers the insulin-independent translocation of GLUT4 transporters to the cell membrane. This allows skeletal muscle to "vacuum" glucose directly from the bloodstream even in individuals with severe insulin resistance or Type 2 Diabetes, providing a potent "back door" for blood sugar control.
Q11 What is the "Endocrine Muscle" and what are Myokines?
The "endocrine muscle" refers to skeletal muscle's ability to secrete signaling proteins called myokines (such as Irisin and IL-6) during exercise. These myokines travel through the bloodstream to cross-organ targets, promoting fat oxidation in the liver, stimulating brain-derived neurotrophic factor (BDNF) for cognitive health, and reducing inflammation system-wide.
Author’s Note
This article synthesizes findings from recent advances in exercise physiology, molecular metabolism, and cellular signaling research to explain how physical activity functions as a powerful biological regulator of metabolic health. The scientific studies referenced represent a growing body of evidence showing that exercise influences multiple molecular pathways—including AMPK activation, mitochondrial biogenesis, insulin-independent glucose transport, and myokine signaling—that collectively protect against metabolic diseases such as type 2 diabetes, obesity, cardiovascular disease, and age-related metabolic decline.
While the underlying science can appear complex, the clinical message is remarkably simple: regular physical activity remains one of the most effective interventions available for maintaining metabolic resilience and healthy aging. The goal of this article is to translate current scientific knowledge into clear, practical insights that help readers better understand why movement is fundamental to long-term health.
Readers should consult qualified healthcare professionals before making significant changes to exercise or medical treatment plans.
Medical Disclaimer
The information in this article, including the research findings, is for educational purposes only and does not constitute medical advice, diagnosis, or treatment. Before starting an exercise program, you must consult with a qualified healthcare professional, especially if you have existing health conditions (such as cardiovascular disease, uncontrolled hypertension, or advanced metabolic disease). Exercise carries inherent risks, and you assume full responsibility for your actions. This article does not establish a doctor-patient relationship.
Related Articles
Can High Iron Accelerate Aging? What Ferritin Levels Reveal About DNA Damage | DR T S DIDWAL
No Time to Train? Science-Backed Workouts That Deliver | DR T S DIDWAL
References
Abrego-Guandique, D. M., Aguilera Rojas, N. M., Chiari, A., Luciani, F., Cione, E., & Cannataro, R. (2025). The impact of exercise on mitochondrial biogenesis in skeletal muscle: A systematic review and meta-analysis of randomized trials. Biomolecular Concepts, 16(1), Article bmc-2025-0055. https://doi.org/10.1515/bmc-2025-0055
Carapeto, P., Iwasaki, K., Hela, F., et al. (2024). Exercise activates AMPK in mouse and human pancreatic islets to decrease senescence. Nature Metabolism, 6, 1976–1990. https://doi.org/10.1038/s42255-024-01130-8
De Sousa, R. (2026). Molecular crosstalk for longevity: Exercise and the AMPK/SIRT1/PGC-1α/Irisin/BDNF axis. Molecular Biology Reports, 53, 142. https://doi.org/10.1007/s11033-025-11315-3
McGee, S. L., & Hargreaves, M. (2024). Exercise performance and health: Role of GLUT4. Free Radical Biology and Medicine, 224, 479–483. https://doi.org/10.1016/j.freeradbiomed.2024.09.004
Pataky, M. W., Heppelmann, C. J., Sevits, K. J., et al. (2025). Aerobic and resistance exercise-regulated phosphoproteome and acetylproteome modifications in human skeletal muscle. Nature Communications, 16, 5700. https://doi.org/10.1038/s41467-025-60049-0
Stead, C. A., Mackin, S. T., & Handschin, C. (2024). Exercise-specific adaptations in human skeletal muscle: Molecular mechanisms of making muscles fit and mighty. Free Radical Biology and Medicine, 223, 341–356. https://doi.org/10.1016/j.freeradbiomed.2024.08.010