Can High Iron Accelerate Aging? What Ferritin Levels Reveal About DNA Damage
Ferritin may be within the normal range yet still affect aging. New research suggests lower iron levels may reduce oxidative stress and metabolic risk
AGING
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/9/202616 min read
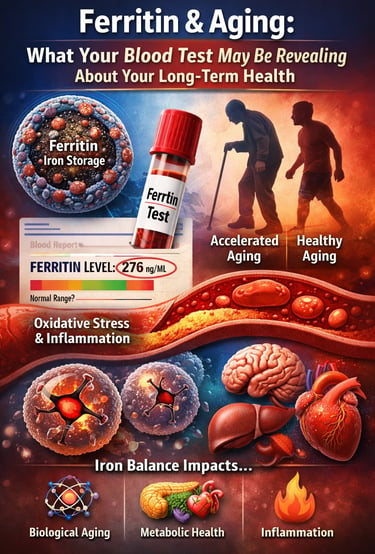
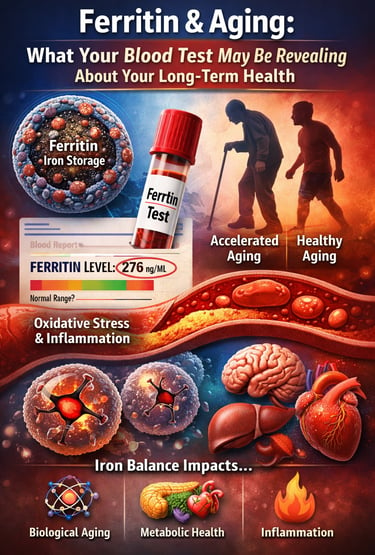
High iron levels, reflected by elevated ferritin in blood tests, may accelerate aging by increasing oxidative stress and cellular damage. Scientists now suggest that maintaining balanced iron stores may be important for protecting long-term metabolic health and healthy aging
For most people, a ferritin blood test is just another number buried in routine laboratory results. If it falls within the “normal range,” it is rarely discussed further. But emerging research suggests that ferritin levels may reveal far more about your health than simply whether you have enough iron to avoid anemia. In fact, ferritin may offer valuable insights into iron homeostasis, metabolic health, inflammation, and even the biological aging process.
Iron is an essential mineral that supports oxygen transport, mitochondrial energy production, and immune defense. Yet iron is also biologically powerful — and potentially harmful when present in excess. Excess iron may also trigger ferroptosis, a form of iron-dependent cell death increasingly linked to aging and neurodegenerative disease.
Because the human body has no efficient mechanism to actively excrete excess iron, small amounts absorbed from food accumulate gradually over decades. This means that iron balance depends almost entirely on tightly regulated absorption mechanisms and cellular storage systems (Zeidan et al., 2024).
This is where ferritin plays a central role. Ferritin is the body’s primary iron storage protein, acting as a protective container that safely stores iron and releases it when cells require it for metabolic processes. Clinically, serum ferritin levels are considered the most reliable marker of total body iron stores, making them a critical indicator of iron status in both deficiency and excess (Kotla et al., 2022).
However, ferritin is not merely a passive storage molecule. It is also closely linked to oxidative stress, inflammation, metabolic dysfunction, and age-related disease risk. Excess iron can catalyze the formation of reactive oxygen species through biochemical reactions that damage cellular structures and DNA, contributing to the gradual biological changes associated with aging (Mangan, 2021). Recent epidemiological research further suggests that higher iron stores may be associated with accelerated biological aging, as measured by epigenetic aging markers (Von Holle et al., 2025).
In other words, ferritin may function as more than a laboratory value — it may serve as a window into long-term metabolic health and the biology of aging itself. Understanding what your ferritin level truly represents could help you interpret routine blood tests in a completely new way.
Clinical pearls
1. The "Inflammation Mirror" (Ferritin + hs-CRP)
Scientific Perspective: Ferritin is an acute-phase reactant regulated by cytokines like IL-6. In the presence of systemic inflammation, serum ferritin levels rise independently of actual iron stores. To differentiate between "true iron overload" and "inflammatory signaling," clinicians must evaluate High-sensitivity C-reactive protein (hs-CRP).
Think of ferritin as a storage tank that leaks when there is a "fire" (inflammation) in the body. If your ferritin is high but your hs-CRP is also high, your high iron number might just be a sign that your body is fighting inflammation, not that you actually have too much iron. You need both tests to see the full picture.
2. The Menopausal "Metabolic Shift"
Scientific Perspective: The cessation of menses leads to a rapid loss of the primary iron excretion pathway. The resulting "ferritin surge" is strongly correlated with a decline in insulin sensitivity and an increase in LDL cholesterol. This suggests iron accumulation may be a driver, not just a bystander, of menopausal metabolic syndrome.
For women, menopause isn't just about hormones; it’s about an "iron reset." Because you stop losing iron every month, your levels can skyrocket quickly. This sudden jump in iron can "rust" your metabolism, making it harder for your body to manage blood sugar and cholesterol.
3. The Pro-Oxidant Aging Clock
Scientific Perspective: Labile plasma iron (LPI) participates in the Fenton Reaction, generating highly reactive hydroxyl radicals (cdot OH). This leads to lipid peroxidation and DNA damage, which are captured by epigenetic clocks as "accelerated biological aging."
Too much iron acts like biological "rust." It creates tiny sparks of oxidative stress that damage your DNA. This makes your cells act much older than they actually are, potentially speeding up the aging process across your entire body.
4. The "Normal" vs. "Optimal" Gap
Scientific Perspective: Standard reference ranges for ferritin are often based on population averages that include individuals with subclinical metabolic disease. Research suggests that the "all-cause mortality" curve for ferritin may begin to rise well before the upper limit of 300 ng/mL—often at levels as low as 100–150 ng/mL in certain populations.
Just because your lab result isn't "red" or "out of range" doesn't mean it’s perfect. The "normal" range is based on the average person, but the average person is often not in peak health. For longevity, "middle-of-the-pack" ferritin is usually safer than being at the very top of the range.
5. Transferrin Saturation: The "Traffic" Check
Scientific Perspective: Ferritin tells you the size of the warehouse, but Transferrin Saturation (TSAT) tells you how crowded the "transport trucks" are. A high ferritin with a low TSAT often points to "Anemia of Chronic Disease" (iron is trapped in storage), whereas high levels of both suggest true systemic iron overload.
If ferritin is your warehouse, transferrin is the fleet of trucks moving iron around. You want to know if the warehouse is full (High Ferritin) OR if the trucks are just overstuffed (High Saturation). Checking both helps your doctor figure out if iron is actually stuck where it shouldn't be.
6. Selective Absorption (The Heme vs. Non-Heme Strategy)
Scientific Perspective: Heme iron (from meat) is absorbed via a relatively unregulated pathway, whereas non-heme iron (from plants) is strictly regulated by hepcidin. In aging populations with rising iron stores, shifting the dietary ratio toward non-heme sources allows the gut to "downregulate" absorption more effectively.
Your body is much better at "saying no" to iron from beans and spinach than iron from steak. If your iron levels are creeping up as you age, eating more plant-based iron gives your body more control over how much it actually lets in, preventing accidental "over-filling."
What Is Ferritin, and Why Does It Matter?
Most people know iron as the mineral found in red meat and leafy greens, the one your doctor checks when you feel unusually tired. But iron does not simply float freely through your blood — most of it is carefully packaged inside a protein shell called ferritin.
Think of ferritin as the body's iron warehouse. When iron is needed, ferritin releases it. When there is excess, ferritin stores it safely away. A ferritin blood test, therefore, does not just tell you how much iron you have available — it tells you how much iron your body has in reserve.
But ferritin is not just a passive storage container. According to Kotla et al. (2022), ferritin performs a wide range of active biological functions, including anti-inflammatory activity, immune signaling, and iron detoxification at the cellular level. Ferritin also exists in two locations: inside cells (intracellular ferritin), where it stores iron, and in the bloodstream (serum ferritin), where elevated levels can act as a signal of inflammation, oxidative damage, or metabolic dysfunction.
This dual nature is what makes ferritin so clinically interesting — and also what makes interpreting it so nuanced.
Ferritin as an Inflammatory Biomarker
Ferritin is not only an iron storage protein but also an acute-phase reactant that rises in response to inflammation. During infections, chronic inflammatory diseases, liver disorders, or metabolic conditions, immune signaling pathways stimulate ferritin production as part of the body’s defense response. As a result, elevated serum ferritin may reflect systemic inflammation rather than true iron overload, which is why clinicians often interpret ferritin alongside markers such as C-reactive protein (CRP) and transferrin saturation to determine the underlying cause.
Iron Homeostasis: The Delicate Balance Your Body Must Maintain
Your body has no meaningful way to actively excrete iron. Unlike sodium, potassium, or water, iron levels are regulated almost entirely through absorption — the gut either takes in more or less depending on what your body needs. This means that when iron accumulates over time, there is limited ability to simply "flush it out."
This biological quirk becomes particularly relevant as we age. Zeidan et al. (2024) conducted a thorough review of iron homeostasis in older adults and found that aging disrupts the finely tuned systems that normally keep iron in balance. Older adults face a double-edged challenge: they are at risk for both iron deficiency (often from poor dietary intake, reduced absorption, or chronic blood loss) and iron excess (from decades of accumulation and declining iron regulatory efficiency).
The study highlights that elevated serum ferritin in older adults is not uncommon, and rather than reassuring clinicians that iron stores are adequate, high ferritin in this population should prompt deeper investigation. Inflammation, metabolic syndrome, liver disease, and early signs of cellular iron overload can all drive ferritin upward — even in the absence of true iron excess in tissues.
For older patients and their caregivers, the key message from this research is clear: ferritin is not just a number to optimize upward. Balance — genuine homeostasis — is the goal.
Menopause and the Ferritin Surge: A Critical Window for Women
One of the most striking and practically relevant findings in recent iron research concerns what happens to ferritin during menopause. For most of their reproductive lives, women maintain lower iron stores than men because monthly menstruation provides a natural, ongoing pathway for iron loss. Premenopausal women are, in fact, at higher risk of iron deficiency than men.
But everything changes at menopause.
Kim et al. (2025) followed a large cohort of women through the menopausal transition and found that ferritin levels rise acceleratedly — not just gradually — during this period. What is particularly important about this finding is that the rate of ferritin increase, not just its final level, was associated with markers of metabolic health, including changes in BMI, blood pressure, fasting glucose, and lipid profiles.
In other words, the speed at which a woman's ferritin rises during menopause may be an early warning signal of metabolic risk. Women who experienced a steeper ferritin trajectory were more likely to show signs of metabolic deterioration, including early markers of insulin resistance and cardiovascular risk.
This finding has meaningful clinical implications. Ferritin is not routinely tracked with high frequency during the menopausal transition, yet this research suggests it could serve as an accessible, low-cost biomarker for identifying women at heightened metabolic risk during this critical life stage — before more serious conditions develop.
If you are a woman approaching or going through menopause, it may be worth having a conversation with your healthcare provider about tracking your ferritin over time, not just as a one-time snapshot.
Iron Status and Biological Aging: Are High Iron Stores Aging You Faster?
Perhaps the most provocative idea emerging from this body of research is that elevated iron — often considered a sign of good nutritional status — may actually accelerate the biological aging process.
Von Holle et al. (2025) examined the relationship between body iron status and biological age in a substantial epidemiological dataset. Rather than using chronological age (the number of candles on your birthday cake), the researchers used epigenetic clocks and other validated biological aging markers — measures of how old your cells actually behave relative to your calendar age.
Their findings were striking: higher iron stores were associated with accelerated biological aging, independent of other risk factors. Individuals with elevated ferritin showed epigenetic patterns consistent with being biologically older than their years would suggest.
The proposed mechanisms are compelling. Iron is a potent pro-oxidant — when present in excess, it participates in chemical reactions that generate free radicals and oxidative stress. This oxidative burden damages DNA, disrupts cell signaling, and triggers inflammation. Over years and decades, this cumulative oxidative stress is one of the primary drivers of cellular aging.
Additionally, excess iron can accumulate in tissues — the liver, heart, and brain — where it contributes to local damage and organ dysfunction over time. This is not only a concern for people with hereditary hemochromatosis (a genetic condition causing severe iron overload) but may also affect individuals with chronically high-normal iron stores.
The takeaway is not that you should be afraid of iron. Rather, it reinforces that more iron is not always better, and that pursuing unnecessarily high ferritin levels — whether through diet, supplementation, or not addressing accumulation — may carry real costs for how you age.
Iron: The Underrated Factor in Aging
Perhaps the clearest call to take iron seriously as an aging variable came from Mangan (2021), who made the case that iron has been systematically underappreciated in aging research and clinical medicine. Despite decades of evidence linking iron accumulation to oxidative stress, inflammation, and age-related disease, iron remains largely absent from mainstream conversations about healthy aging.
Mangan (2021) argues that the progressive accumulation of iron in human tissues with advancing age — a well-documented phenomenon — contributes meaningfully to the increased oxidative burden seen in older adults. Iron accumulates in the brain, liver, and cardiovascular system over a lifetime, and this accumulation correlates with functional decline and disease susceptibility.
The paper also highlights an interesting evolutionary perspective: many long-lived species maintain lower tissue iron levels, and interventions that reduce iron stores in animal models have, in some studies, extended healthy lifespan. While human evidence for iron-lowering interventions as anti-aging strategies remains limited and preliminary, the biological plausibility is well-established.
This does not mean that older adults should avoid iron-rich foods or take iron-reducing medications without medical guidance. It does mean that physicians, patients, and researchers should pay closer attention to iron status as a modifiable risk factor in aging — one that may sit alongside more commonly discussed variables like blood pressure, cholesterol, and blood glucose.
Mechanistic Link: How Excess Iron Accelerates Cellular Aging
At the cellular level, excess iron can accelerate aging through several interconnected biological mechanisms. One of the most important is the Fenton reaction, a chemical process in which free iron catalyzes the formation of highly reactive hydroxyl radicals. These radicals damage DNA, proteins, and cellular membranes, contributing to oxidative stress — one of the fundamental drivers of aging. Iron also interacts closely with mitochondria, the energy-producing structures within cells. Elevated iron levels can disrupt mitochondrial function, impair energy production, and increase the generation of reactive oxygen species. In addition, the hormone hepcidin, which regulates iron absorption and distribution, often becomes dysregulated with aging and chronic inflammation, allowing iron to accumulate gradually in tissues such as the liver, heart, and brain. Over decades, this accumulation may contribute to the biological aging patterns observed in epidemiological studies.
What Does This Mean for You? Practical Takeaways
Understanding the science is one thing; knowing what to do with it is another. Here is how to translate these research findings into practical action.
Get your ferritin tested — and track it over time. A single ferritin measurement is helpful but limited. The trajectory matters. Ask your doctor to include ferritin in your routine blood work, and keep a record of your results over the years so patterns become visible.
Context matters. A high ferritin level does not automatically mean you have dangerous iron excess. It can reflect inflammation, infection, liver disease, or other conditions. Similarly, a low ferritin number in an older adult may warrant further investigation rather than immediate supplementation. Always interpret ferritin alongside other markers — iron saturation, haemoglobin, C-reactive protein, and clinical context.
Women approaching menopause should pay particular attention. The accelerated rise in ferritin during the menopausal transition is a metabolic signal, not just a hormonal footnote. This is a meaningful window for proactive health monitoring.
Be cautious with iron supplements unless a deficiency is confirmed. Supplementing iron without an established deficiency can push ferritin higher than necessary and may contribute to the oxidative burden associated with excess iron stores.
Diet remains your best first-line tool. A balanced diet rich in vegetables, legumes, and lean proteins provides iron in a form (non-heme iron) that the body absorbs more selectively than the heme iron found in red meat. This natural selectivity helps prevent overconsumption.
If you have risk factors for iron overload — a family history of hemochromatosis, longstanding high ferritin, liver disease, or metabolic syndrome — genetic testing and specialist review are worth discussing with your doctor.
Ferritin Reference Ranges: What Is "Normal"?
Standard laboratory reference ranges for serum ferritin vary by sex and age, but common benchmarks are:
Women (premenopausal): 12–150 ng/mL
Women (postmenopausal): 12–263 ng/mL
Men: 12–300 ng/mL
However, as the research discussed above makes clear, being within the "normal" range does not always mean optimal. Some studies suggest that ferritin levels consistently above 200 ng/mL in women or above 300 ng/mL in men — even within the upper bounds of "normal" — may carry increased oxidative and metabolic risk. Always discuss your specific values with a qualified healthcare provider who knows your full health picture.
Key Takeaways: Ferritin, Iron, and the Biology of Aging
1. Ferritin Is More Than a Routine Lab Marker
Ferritin is widely used to assess iron stores, but its clinical significance extends far beyond diagnosing iron deficiency. As the body’s primary iron storage protein, ferritin reflects the long-term balance between iron intake, storage, and utilization. Because iron influences oxygen transport, mitochondrial energy production, and immune function, ferritin can provide a deeper window into overall metabolic health (Kotla et al., 2022).
2. Iron Balance Is a Lifelong Biological Challenge
Unlike many nutrients, the human body has no efficient mechanism to actively eliminate excess iron. Iron regulation depends primarily on intestinal absorption and cellular storage systems. Over decades, even small daily surpluses may gradually accumulate, making iron homeostasis increasingly difficult to maintain with aging (Zeidan et al., 2024).
3. Aging and Iron Accumulation Are Closely Linked
Research increasingly suggests that iron levels tend to rise gradually with age, particularly in tissues such as the liver, brain, and cardiovascular system. This accumulation contributes to oxidative stress and cellular damage, processes that are central to biological aging and the development of chronic disease (Mangan, 2021).
4. Ferritin May Reflect Metabolic Health—Especially in Women
Recent studies indicate that ferritin levels often increase rapidly during the menopausal transition due to the cessation of menstrual iron loss. Importantly, the rate of increase in ferritin has been linked to metabolic markers such as insulin resistance, blood pressure changes, and lipid alterations, suggesting ferritin may serve as an early metabolic warning signal (Kim et al., 2025).
5. Excess Iron May Accelerate Biological Aging
Iron is a powerful pro-oxidant capable of generating reactive oxygen species that damage DNA and cellular structures. Epidemiological research now suggests that higher iron stores may correlate with accelerated biological aging, as measured by epigenetic aging markers and other molecular indicators (Von Holle et al., 2025).
6. The Goal Is Balance, Not Maximization
Iron remains an essential nutrient, and deficiency carries serious health risks. However, modern research increasingly emphasizes that optimal aging depends on maintaining iron balance rather than simply maximizing iron stores.
7. Ferritin Deserves a Place in Preventive Health Discussions
Tracking ferritin over time—alongside other markers such as hemoglobin, transferrin saturation, and inflammatory markers—may provide valuable insights into long-term metabolic health and aging trajectories.
Frequently Asked Questions (FAQs)
1. What is ferritin, and how is it different from iron?
Iron is the mineral itself — found in food and used by your body for countless functions, including making red blood cells. Ferritin is the protein that stores iron inside your cells. When doctors measure serum ferritin, they are measuring how much iron your body has in reserve. It is the most reliable single marker of your body's total iron stores.
2. Can ferritin be too high even if I feel fine?
Yes, absolutely. High ferritin often produces no noticeable symptoms in the early stages. This is part of what makes it potentially problematic — iron can accumulate quietly in tissues and contribute to oxidative stress and metabolic changes long before symptoms appear. This is why regular blood testing is valuable even when you feel well.
3. Is high ferritin always a sign of iron overload?
Not necessarily. Ferritin is also an acute-phase reactant, meaning it rises in response to inflammation, infection, liver disease, and certain cancers. A high ferritin reading should always be interpreted in context. Your doctor will likely check additional markers — such as transferrin saturation and a complete blood count — to determine whether the elevation reflects true iron excess or another underlying condition.
4. Why do ferritin levels rise during menopause?
Before menopause, monthly menstruation provides a natural pathway for iron loss, keeping ferritin relatively low in most women. Once menstruation ceases, this iron-loss mechanism disappears, and ferritin levels tend to rise. Research by Kim et al. (2025) found that this rise is often more accelerated than previously recognized and may serve as an early marker of metabolic changes during the menopausal transition.
5. Can high iron levels really speed up biological aging?
The evidence suggests it can. Von Holle et al. (2025) found that higher iron stores are associated with accelerated biological aging as measured by epigenetic clocks. The likely mechanism involves iron's role as a pro-oxidant — excess iron promotes the formation of free radicals, which damage DNA and accelerate cellular aging processes over time.
6. Should older adults take iron supplements?
Only if a genuine deficiency has been confirmed through blood testing. Older adults are at risk for both iron deficiency and iron excess, and supplementing without clear evidence of need may be counterproductive. Zeidan et al. (2024) specifically caution against reflexive iron supplementation in older populations without thorough evaluation. Always seek guidance from your healthcare provider before starting iron supplements.
7. What is the best way to maintain healthy iron levels as I age?
A balanced, whole-food diet is the foundation. Prioritize plant-based iron sources (legumes, leafy greens, fortified cereals), which are absorbed more selectively than heme iron from red meat. Regular blood monitoring of ferritin, hemoglobin, and iron saturation allows you and your doctor to catch imbalances early. Avoid unnecessary iron supplementation, manage chronic inflammation (which elevates ferritin artificially), and stay physically active — moderate exercise supports healthy iron regulation and reduces oxidative stress.
Author’s Note
Iron metabolism is one of the most tightly regulated systems in human biology, yet it often receives far less attention in routine health discussions than glucose, cholesterol, or blood pressure. Our ferritin level may be just one number on a routine blood test, but growing scientific evidence suggests it could reveal far more about your metabolic health and how your body ages over time
The goal of this article is to highlight why ferritin—commonly reported in standard blood tests—deserves closer consideration as part of a broader conversation about metabolic health and healthy aging.
The insights presented here are based on emerging research examining the relationships between iron homeostasis, inflammation, oxidative stress, menopause, and biological aging. While the science continues to evolve, a consistent message is becoming clear: both iron deficiency and iron excess can have meaningful health consequences, and maintaining balance is critical for long-term well-being.
It is also important to emphasize that ferritin is a complex biomarker. Elevated levels do not always indicate excess iron; they may also reflect inflammation, infection, liver disease, or metabolic conditions. For this reason, ferritin should always be interpreted in context with other laboratory markers and an individual’s overall clinical picture.
My intention in writing this article is not to encourage self-diagnosis or unsupervised supplementation, but rather to help readers better understand the biological significance of a laboratory value that is often overlooked. By becoming more informed about markers like ferritin, patients can have more meaningful conversations with their healthcare providers and take a more active role in preventive health.
As always, medical decisions should be made in consultation with a qualified healthcare professional who can interpret laboratory findings within the context of your personal medical history.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
The Vitamin D–K2 Axis: A Precision Strategy for Bone, Muscle, and Vascular Integrity | DR T S DIDWAL
Living to 100: Is it Genetics or Lifestyle? What the New Science Says | DR T S DIDWAL
Time-Restricted Eating: Metabolic Advantage or Just Fewer Calories? | DR T S DIDWAL
Can You Revitalize Your Immune System? 7 Science-Backed Longevity Strategies | DR T S DIDWAL
Exercise and Longevity: The Science of Protecting Brain and Heart Health as You Age | DR T S DIDWAL
References
Kotla, N. K., Dutta, P., Parimi, S., & Das, N. K. (2022). The role of ferritin in health and disease: Recent advances and understandings. Metabolites, 12(7), 609. https://doi.org/10.3390/metabo12070609
Mangan, D. (2021). Iron: An underrated factor in aging. Aging, 13(19), 23407–23415. https://doi.org/10.18632/aging.203612
Kim, M., Chang, Y., Cho, Y., et al. (2025). Accelerated increase in ferritin levels during menopausal transition as a marker of metabolic health. Scientific Reports, 15, 29311. https://doi.org/10.1038/s41598-025-14295-3
Von Holle, A., Ramamurthy, S., Díaz Santana, M. V., Kresovich, J. K., Taylor, J. A., Xu, Z., O'Brien, K. M., Sandler, D. P., & Weinberg, C. R. (2025). Association between body iron status and biological aging. Nutrients, 17(9), 1409. https://doi.org/10.3390/nu17091409
Zeidan, R. S., Martenson, M., Tamargo, J. A., McLaren, C., Ezzati, A., Lin, Y., Yang, J. J., Yoon, H.-S., McElroy, T., Collins, J. F., Leeuwenburgh, C., Mankowski, R. T., & Anton, S. (2024). Iron homeostasis in older adults: Balancing nutritional requirements and health risks. The Journal of Nutrition, Health & Aging, 28(5), 100212. https://doi.org/10.1016/j.jnha.2024.100212