Is Your Fat Aging Faster Than You? How Adipose Tissue Controls Lifespan and Disease
Adipose tissue isn’t just fat—it may control how fast you age. Explore biomarkers, inflammation, and proven strategies for healthier aging.
AGINGMETABOLISM
Dr. T.S. Didwal, M.D.(Internal Medicine)
4/16/202616 min read
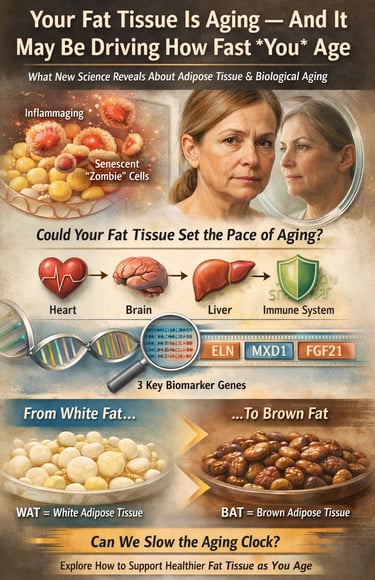

Adipose tissue ageing refers to the progressive decline in fat tissue function, characterised by increased inflammation, accumulation of senescent cells, and impaired hormone signalling. As fat ages, it releases harmful molecules through the senescence-associated secretory phenotype, contributing to insulin resistance, cardiovascular disease, and systemic aging.
Emerging research in geroscience suggests that adipose tissue may influence the overall pace of biological aging by affecting metabolic health, immune function, and organ performance. Key biomarkers such as ELN, MXD1, and FGF21 are being studied to measure fat tissue aging and guide future therapies.
In simple terms: healthy fat supports longevity, while dysfunctional fat accelerates aging and disease risk.
Clinician’s Perspective
Adipose tissue is a central metabolic organ—not a passive depot
Modern evidence firmly establishes adipose tissue as an endocrine and immunologically active organ regulating systemic metabolism, inflammation, and energy homeostasis. Its dysfunction is a primary driver, not merely a consequence, of cardiometabolic disease (Wang et al., 2025).Adipose aging is clinically visible long before disease onset
Subtle shifts—central fat redistribution, rising triglycerides, declining insulin sensitivity—often precede overt diagnoses by years. These changes reflect early adipose dysfunction and should be treated as actionable warning signals rather than benign age-related changes.Visceral fat is the most clinically relevant compartment
Expansion of visceral adipose tissue correlates strongly with insulin resistance, hepatic steatosis, and systemic inflammation. Waist-to-height ratio and imaging-based fat quantification provide more meaningful risk assessment than BMI alone.Chronic low-grade inflammation (“inflammaging”) is adipose-driven
Aging fat becomes enriched with senescent cells exhibiting the senescence-associated secretory phenotype, releasing pro-inflammatory cytokines that amplify metabolic and vascular risk (Liu et al., 2025).Biomarkers are transitioning from research to clinical relevance
Emerging markers such as FGF21, adiponectin-to-leptin ratio, and inflammatory cytokine profiles offer early insight into adipose health. While not yet routine, they are likely to become part of precision metabolic care (Xie et al., 2025).Therapeutic focus should shift from weight loss to fat quality
The goal is not simply to reduce fat mass but to improve adipose function—enhancing insulin sensitivity, reducing inflammation, and preserving metabolic flexibility.Exercise remains the most potent intervention
Combined aerobic and resistance training improves adipokine signaling, promotes mitochondrial function, and may partially reverse adverse adipose remodeling.Metabolic health must be managed proactively and longitudinally
Early control of glucose, lipids, and blood pressure slows adipose aging and reduces downstream organ damage.Future therapies are promising, but adjunctive
Targets such as FGF21 analogues and senolytics may complement—but not replace—lifestyle interventions.Clinical takeaway
Healthy aging is not defined by the absence of fat, but by functionally resilient adipose tissue—a modifiable determinant of long-term metabolic and systemic health.
Why Your Fat Tissue Deserves More Respect
What if the pace at which you age is not primarily determined by your heart, your brain, or even your DNA—but by your fat?
For decades, adipose tissue was dismissed as little more than passive energy storage. Today, that view has been decisively overturned. A growing body of research in geroscience and metabolic medicine now positions adipose tissue as a central regulator of systemic aging—an active endocrine organ capable of influencing inflammation, insulin sensitivity, immune function, and even brain health (Wang et al., 2025; Liu et al., 2025).
As fat tissue ages, it undergoes profound biological changes. It becomes more inflamed, less metabolically flexible, and increasingly populated by senescent “zombie” cells that secrete harmful signaling molecules—a phenomenon known as the senescence-associated secretory phenotype. These signals do not remain confined to fat depots. They circulate throughout the body, subtly reshaping the biological environment in which every organ operates (Wang et al., 2025).
This has led to a provocative and increasingly supported hypothesis: adipose tissue may act as a “pace-maker” of biological aging, influencing how quickly—or slowly—multiple organ systems decline (Liu et al., 2025). In parallel, advances in bioinformatics and molecular profiling have begun to identify measurable signatures of this process. Recent work has highlighted key genes such as ELN, MXD1, and FGF21 as potential biomarkers that track the aging trajectory of fat tissue with remarkable precision (Xie et al., 2025).
The implication is both sobering and empowering. Aging is not merely something that happens to us over time—it is, in part, something happening within our fat tissue, shaped by metabolism, lifestyle, and molecular signaling.
Understanding this hidden biology may be one of the most important steps toward slowing the aging process itself.
What Is Adipose Tissue, and Why Does It Age Differently?
Adipose tissue comes in several forms. The type most people know — the soft tissue under the skin and around the abdomen — is called white adipose tissue (WAT). There is also brown adipose tissue (BAT), which burns energy to generate heat and is more metabolically active, and a transitional form called beige adipose tissue, which can shift between the two under certain conditions.
A landmark review published in Nature Reviews Endocrinology described how adipose tissue undergoes striking structural and functional changes as we age (Wang et al., 2025). These changes include:
A redistribution of fat from subcutaneous (under-the-skin) deposits to visceral (around-the-organs) deposits
A decline in the metabolic activity of brown and beige fat
An increase in chronic low-grade inflammation — sometimes called inflammaging
Reduced regenerative capacity of fat-resident stem cells
Impaired secretion of beneficial hormones, including adiponectin and leptin
These changes do not happen in isolation. Because adipose tissue is a major endocrine organ — meaning it releases hormones and signaling molecules into the bloodstream — its aging disrupts metabolic regulation body-wide. The consequence is a cascade that raises the risk for type 2 diabetes, cardiovascular disease, neurodegenerative conditions, and even some cancers (Wang et al., 2025).
A comprehensive review spanning 2,527 published documents over four decades found that interest in adipose tissue browning and aging has accelerated dramatically since 2015, with bioinformatics and molecular biology tools now enabling researchers to map the aging process within fat tissue at an unprecedented level of detail (Luo et al., 2026). The field has shifted from asking "does fat tissue age?" to asking "how exactly does it age — and can we measure it?"
What Are Aging Biomarkers, and Why Do We Need Them?
A biomarker is a measurable biological signal — a molecule, gene expression pattern, protein level, or cellular feature — that reliably indicates a particular biological state. In the context of aging, biomarkers help researchers and clinicians answer a deceptively complex question: How old is this tissue, really?
Your chronological age — the number of years you have been alive — tells only part of the story. Your biological age, the actual functional state of your tissues and cells, can diverge significantly from the calendar. Two people who are both 50 years old can have fat tissue that behaves as if it is 40 or 65, depending on genetics, lifestyle, metabolic health, and other factors.
Identifying reliable adipose tissue aging biomarkers is therefore crucial for several reasons. First, they allow early detection of accelerated aging processes, potentially before symptoms of metabolic disease appear. Second, they provide measurable targets for therapeutic interventions. Third, they offer a way to track whether lifestyle changes or treatments are actually slowing aging at the tissue level.
Recognizing this need, the Aging Biomarker Consortium — a multinational panel of experts — released a consensus statement in 2025 establishing a formal framework for identifying and validating biomarkers specific to adipose tissue aging (Aging Biomarker Consortium et al., 2025). This framework categorizes biomarkers across several dimensions, including cellular senescence markers, inflammatory signals, metabolic function indicators, and structural integrity measures. The consensus statement marks a turning point: the scientific community now has agreed standards for what counts as a valid adipose aging biomarker, enabling more consistent and reproducible research globally.
The Three Breakthrough Biomarker Genes: ELN, MXD1, and FGF21
Against this backdrop, a 2025 study published in Frontiers in Endocrinology made a notable contribution by using integrative bioinformatics and machine-learning approaches to sift through large genomic datasets and identify which genes are most consistently associated with adipose tissue aging (Xie et al., 2025). The study applied multiple analytical pipelines — including LASSO regression, Random Forest, and support vector machine models — and converged on three genes as the most robust aging-related biomarkers in adipose tissue: ELN, MXD1, and FGF21.
Let us explore each one.
ELN (Elastin)
Elastin is a structural protein responsible for the elasticity and resilience of many tissues. In adipose tissue, elastin forms part of the extracellular matrix — the scaffolding that holds cells in their proper architecture. As adipose tissue ages, ELN expression changes in ways that impair the structural integrity of the tissue. Reduced elastin production means the connective framework of fat depots becomes stiffer and less capable of responding to metabolic demands. In simple terms, aging fat tissue loses some of its physical "bounce" — and that has real consequences for how fat cells communicate and store energy (Xie et al., 2025).
ELN is also relevant beyond the fat depot itself. Elastin degradation products — fragments released when the protein breaks down — can trigger inflammatory signaling, creating a feedback loop that accelerates the aging process. Identifying ELN as a key biomarker connects structural biology to metabolic aging in a clinically meaningful way.
MXD1 (MAX Dimerization Protein 1)
MXD1 is a transcription factor — a protein that controls the expression of other genes. It functions as an antagonist to MYC, a well-known regulator of cell growth and proliferation. In the context of aging, MXD1 plays a role in balancing cell turnover versus cell senescence (the state where cells stop dividing but remain metabolically active in a damaging way).
In aging adipose tissue, dysregulation of MXD1 appears to shift this balance toward increased cellular senescence and reduced regenerative capacity (Xie et al., 2025). Senescent fat cells — sometimes called "zombie cells" — accumulate with age and secrete a cocktail of inflammatory molecules known as the senescence-associated secretory phenotype (SASP). This SASP state contributes significantly to the chronic inflammation associated with metabolic disease and accelerated systemic aging.
The discovery of MXD1 as a key adipose aging biomarker opens a potential therapeutic window: if MXD1 activity could be modulated, it might be possible to slow the accumulation of senescent fat cells, with downstream benefits for metabolic and systemic health.
FGF21 (Fibroblast Growth Factor 21)
Of the three biomarkers, FGF21 may be the most familiar to clinicians. It is a metabolic hormone produced primarily in the liver and adipose tissue that plays diverse roles in energy balance, glucose metabolism, and lipid handling. FGF21 levels tend to rise in response to fasting, exercise, and metabolic stress.
In the context of aging, FGF21 has a complex and somewhat paradoxical role. It is simultaneously a beneficial metabolic signal — improving insulin sensitivity and promoting fat oxidation — and a potential marker of metabolic stress when chronically elevated. The bioinformatics study identified FGF21 as a key discriminator of adipose tissue aging state, suggesting that changes in its expression pattern in fat tissue track meaningfully with aging-related functional decline (Xie et al., 2025).
Importantly, FGF21 has also emerged as a promising therapeutic target. Several pharmaceutical companies are developing FGF21 analogs for treating metabolic syndrome, fatty liver disease, and obesity. The identification of FGF21 as an aging biomarker strengthens the scientific rationale for these therapeutic approaches and suggests their potential benefits may extend to slowing adipose tissue aging.
How Does Adipose Tissue "Talk" to the Rest of Your Body?
One of the most important concepts in understanding adipose aging is that fat tissue does not age in silence. It communicates constantly with the rest of the body through adipokines — hormone-like molecules secreted by fat cells — and through circulating metabolites, extracellular vesicles, and even neuronal signals.
As adipose tissue ages, the quality and composition of these communications change. Production of beneficial adipokines like adiponectin (which has anti-inflammatory and insulin-sensitizing effects) tends to decline, while pro-inflammatory signals rise. The review in Nature Reviews Endocrinology described this shift as a transition from a "youthful secretome" to an "aged secretome" — a fundamentally different biochemical environment that the rest of the body must navigate (Wang et al., 2025).
This helps explain a striking observation: when researchers transplant young adipose tissue into aged animals, the recipients show improvements in metabolic function and even lifespan. The fat tissue, it seems, can rejuvenate its host — a finding that has electrified the field and underscored just how central adipose health is to systemic aging.
The review by Corvera et al. (2026), published in Endocrine Reviews, provided a comprehensive update on adipose tissue biology advances, emphasizing the growing understanding of how fat tissue interacts with the nervous system, the immune system, and distant organs, including the brain and heart. This "adipose-brain axis," the "adipose-cardiovascular axis," and similar interdependencies mean that keeping your fat tissue healthy as you age is not merely an aesthetic concern — it is a whole-body health imperative.
The Browning Connection: A Metabolic Lifeline Against Aging
One of the most exciting research areas at the intersection of fat biology and aging is adipose tissue browning — the process by which white fat cells acquire characteristics of metabolically active brown fat cells. Brown and beige fat cells contain more mitochondria, burn more energy, and produce less inflammation than white fat cells.
The extensive bibliometric and bioinformatics review by Luo et al. (2026), covering four decades of literature on this topic, confirmed that browning capacity declines with age and that this decline is associated with worsening metabolic health. However, the review also highlighted numerous interventions — including exercise, cold exposure, certain dietary components, and pharmacological agents — that can partially restore browning capacity even in aged adipose tissue.
This is genuinely good news. It means adipose aging is not a one-way street. The plasticity of fat tissue — its ability to adapt and, to some extent, rejuvenate — offers meaningful opportunities for intervention.
Practical Applications: What You Can Do Right Now
The science of adipose tissue aging, while still evolving, already points toward several evidence-informed lifestyle practices that support healthier fat tissue as you grow older.
1. Prioritize Regular Aerobic and Resistance Exercise Exercise is the most robustly supported intervention for improving adipose tissue function. Aerobic exercise promotes fat browning, reduces visceral fat accumulation, and improves adipokine profiles. Resistance training preserves muscle-fat balance and improves insulin sensitivity. Aim for at least 150 minutes of moderate aerobic activity and two strength-training sessions per week.
2. Manage Chronic Inflammation Through Diet The Mediterranean diet pattern — rich in vegetables, legumes, whole grains, healthy fats (particularly olive oil and omega-3s), and lean protein — has been consistently associated with reduced adipose inflammation and better metabolic aging outcomes. Minimize ultra-processed foods, refined sugars, and excess saturated fats, which accelerate the pro-inflammatory shift in aging fat tissue.
3. Protect Your Sleep. Sleep disruption impairs adipose tissue function, raises cortisol, increases visceral fat deposition, and blunts FGF21 signaling. Adults should aim for 7–9 hours of quality sleep per night as a non-negotiable metabolic health practice.
4. Avoid Prolonged Sedentary Periods. Even in individuals who exercise regularly, prolonged unbroken sitting is independently associated with worse adipose tissue health. Break up sedentary time every 60–90 minutes with brief movement.
5. Manage Metabolic Risk Factors Conditions like insulin resistance, type 2 diabetes, and chronic high blood glucose accelerate adipose tissue aging through several mechanisms, including increased advanced glycation end-products (AGEs) and oxidative stress. Work with your healthcare provider to keep blood glucose, blood pressure, and lipid levels within healthy ranges.
6. Avoid Tobacco and Excess Alcohol. Both tobacco smoke and heavy alcohol consumption damage adipose tissue directly and impair its secretory function, accelerating the transition to an aged secretome.
7. Stay Connected to Emerging Therapeutics As FGF21 analogs and other adipose-targeted therapies progress through clinical trials, staying informed through your physician may become increasingly relevant — particularly for individuals with metabolic syndrome or obesity.
Frequently Asked Questions (FAQs)
Q1: What exactly does "adipose tissue aging" mean in practical terms?
Adipose tissue aging refers to the accumulation of functional changes in fat cells and the tissue environment over time — including shifts in fat distribution toward more harmful visceral depots, increased inflammation, reduced metabolic flexibility, and altered hormone secretion. These changes contribute to metabolic disease risk and accelerated systemic aging.
Q2: Can I measure my adipose tissue's biological age?
Not yet at the consumer level — but research in this area is advancing rapidly. The Aging Biomarker Consortium's 2025 framework (Aging Biomarker Consortium et al., 2025) is establishing the scientific foundation for validated clinical tools. Blood-based markers like FGF21 levels, adiponectin-to-leptin ratios, and inflammatory markers offer partial windows into adipose tissue health.
Q3: Are ELN, MXD1, and FGF21 the only important adipose aging biomarkers?
No — they are among the most robust candidates identified using advanced machine-learning methods in one important 2025 study (Xie et al., 2025). The broader field has identified dozens of candidate biomarkers. What distinguishes ELN, MXD1, and FGF21 is the convergence of multiple analytical approaches pointing to their significance.
Q4: Does losing weight improve adipose tissue aging?
Intentional, sustained weight loss — particularly reduction of visceral fat — generally improves adipose tissue function, reduces inflammatory markers, and shifts adipokine profiles in a favorable direction. However, the quality of weight loss matters: rapid weight loss through extreme caloric restriction can impair adipose tissue health in other ways. Gradual fat loss through lifestyle modification is preferred.
Q5: Is adipose tissue aging reversible?
Partially and conditionally, yes. Research into adipose tissue browning, cellular senescence clearance (senolytics), and metabolic reprogramming suggests meaningful reversibility of certain aging features. Exercise, dietary intervention, and emerging pharmacological strategies can all shift adipose tissue toward a more youthful functional state (Luo et al., 2026; Corvera et al., 2026).
Q6: How does adipose tissue aging relate to Alzheimer's disease and brain health?
The adipose-brain axis is an active research area. Adipokines and inflammatory signals from aged fat tissue cross the blood-brain barrier and influence neuroinflammation, synaptic function, and amyloid metabolism. Maintaining metabolic and adipose health throughout life is increasingly recognized as a modifiable factor in neurodegenerative disease risk.
Q7: Should I be concerned about having too little body fat as I age?
Yes — this is an underappreciated issue. Both excess and insufficient body fat are associated with accelerated adipose tissue aging and poorer outcomes. Subcutaneous fat, especially in the legs and hips, has protective metabolic properties. Age-related fat loss (lipoatrophy) in certain depots is associated with worse insulin sensitivity and metabolic dysfunction. Healthy aging is about quality and distribution of fat tissue, not just quantity.
Clinical Pearls: The Aging Adipose Organ
1. The "Adipose-Brain Axis" Connection
Aging adipose tissue undergoes a "secretome shift," increasing the systemic release of pro-inflammatory cytokines (SASP) that can breach the blood-brain barrier. This contributes to neuroinflammation and may accelerate the progression of neurodegenerative pathologies such as Alzheimer’s disease.
Your body fat acts like a broadcasting station. As it ages, it stops sending "healthy" signals and starts sending "inflammatory" signals that can cloud your brain, making fat health a key pillar of long-term mental sharpness.
2. The Lipoatrophy Paradox
Clinical focus should remain on the quality and distribution of adipose tissue rather than just mass. Age-related subcutaneous lipoatrophy—particularly in the gluteofemoral region—leads to ectopic lipid deposition in the liver and muscle, significantly worsening insulin resistance.
Not all fat is bad. Losing the "soft" fat under your skin (especially on your legs and hips) as you age can actually be risky, as it forces fat to store itself inside your organs instead, which disrupts your metabolism.
3. Structural Integrity and the ELN Biomarker
The downregulation of the ELN (Elastin) gene leads to fibrosis and stiffening of the extracellular matrix (ECM) within fat depots. This mechanical rigidity limits the healthy expansion of adipocytes, triggering cellular stress and early senescence.
Think of your fat tissue like a sponge. To work well, it needs to be flexible. As we age, a loss of "elastin" makes the tissue stiff and brittle, preventing it from storing energy properly and causing it to "leak" inflammation into the body.
4. Therapeutic Potential of Adipose "Browning"
Adipose tissue retains significant plasticity throughout the lifespan. Interventions such as cold thermogenesis and aerobic exercise can induce "browning" of white adipose tissue (WAT), enhancing mitochondrial biogenesis and increasing the systemic metabolic rate via UCP1-mediated thermogenesis.
You can actually "re-train" your fat cells. By staying active and even exposing yourself to cooler temperatures, you can turn sluggish "white fat" into "beige fat," which acts like a furnace to burn calories and generate healthy energy.
5. FGF21: The Metabolic Stress Signal
While FGF21 is a potent insulin sensitizer, its chronic elevation in aging populations often indicates "FGF21 resistance." Clinical monitoring of this biomarker can serve as an early warning system for metabolic dysfunction before the onset of overt Type 2 Diabetes.
Think of FGF21 as a metabolic repairman. When your body is under stress, it sends out more repairmen. If your levels stay high for too long, it’s a sign that your "repair crew" is overwhelmed and your metabolism needs extra support through diet and lifestyle changes.
Take the Next Step: From Reading to Action
✅ Right Now (Today)
Share this article with a friend, family member, or healthcare provider who cares about healthy aging
Write down one lifestyle habit from the Practical Applications section you will commit to this week
✅ This Week
Speak with your doctor about your visceral fat levels — a DEXA scan or waist-to-height ratio can offer a useful starting point
Ask whether an FGF21 blood test is appropriate for your health context
Consider scheduling a full metabolic panel if you have not had one recently
✅ This Month
Start or upgrade an exercise routine that includes both aerobic and resistance components
Begin a 4-week Mediterranean diet trial and track your energy, sleep quality, and waist measurement
Explore whether your healthcare provider is aware of senolytics or FGF21 therapeutics in ongoing clinical trials relevant to your situation
Author’s Note
This article was written with a deliberate goal: to bridge the gap between rapidly evolving molecular science and real-world clinical understanding. The biology of adipose tissue has advanced dramatically in recent years, moving from a simplistic “energy storage” model to a complex, systems-level view of fat as a dynamic regulator of aging, metabolism, and disease risk.
In preparing this piece, the focus was on synthesizing high-quality, peer-reviewed evidence—particularly recent work in geroscience, endocrinology, and metabolic research—into a format that remains both scientifically rigorous and clinically meaningful. Special care was taken to present emerging concepts, such as adipose-driven aging and biomarker discovery, with appropriate nuance. While findings around genes like ELN, MXD1, and FGF21 are promising, they represent a rapidly developing field rather than settled clinical doctrine.
For clinicians, the intention is to encourage earlier recognition of adipose dysfunction as a precursor to disease. For general readers, the aim is to reframe fat not as an adversary, but as a biologically active partner in health—one that responds to lifestyle, environment, and time.
Importantly, this is not a call to overmedicalize aging, but to better understand it. Many of the most powerful interventions remain foundational: physical activity, nutrition, sleep, and metabolic risk management. Future therapies may refine our approach, but they will build upon—not replace—these pillars.
Science is still unfolding in this space. As with all areas of medicine, today’s insights will evolve with tomorrow’s evidence.📢 Call to Action (CTA) + User Engagement Strategy
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Your Body Fat Is an Endocrine Organ—And Its Hormones Shape Your Heart Health | DR T S DIDWAL
Tired All Day, Awake at Night? The Redox and Mitochondrial Link | DR T S DIDWAL
Obesity and Fatty Liver Disease: What Science Says About Risk and Health | DR T S DIDWAL
Intermittent Fasting: Metabolic Health Benefits and the Evidence on Longevity | DR T S DIDWAL
Activate Your Brown Fat: A New Pathway to Longevity and Metabolic Health | DR T S DIDWAL
References
Aging Biomarker Consortium, Yu, J., Zhang, Y., Zhang, T., Bi, Y., Chen, Y., Chen, Z., Dai, Z., Guo, F., Guo, L., Hu, C., Kong, X., Li, J., Liu, P., Liu, Y., Qu, J., Tang, Q., Wang, C., Wang, L., Wang, J., … & Ma, X. (2025). A framework of biomarkers for adipose tissue aging: a consensus statement by the Aging Biomarker Consortium. Life Medicine, 4(5), lnaf027. https://doi.org/10.1093/lifemedi/lnaf027
Corvera, S., Rajan, A., Townsend, K. L., Shamsi, F., Wu, J., Svensson, K. J., Zeltser, L. M., Collins, S., Reis, T., Tseng, Y. H., & Goodyear, L. J. (2026). Advances in adipose tissue biology. Endocrine Reviews, 47(1), 75–92. https://doi.org/10.1210/endrev/bnaf032
Liu, J., Huang, Q., & Liu, F. (2025). Fat talks first: how adipose tissue sets the pace of aging? Life Medicine, 4(5), lnaf028. https://doi.org/10.1093/lifemedi/lnaf028
Luo, Y., Song, Y., Zeng, M., Li, B., Li, Y., & Dong, Z. (2026). The past, present, and future of adipose tissue browning and aging: A review combined with bibliometrics and bioinformatics of 2527 documents published over the past four decades. Ageing Research Reviews, 114(Suppl C), Article 102959. https://doi.org/10.1016/j.arr.2025.102959
Wang, G., Song, A., & Wang, Q. A. (2025). Adipose tissue ageing: implications for metabolic health and lifespan. Nature Reviews Endocrinology, 21, 623–637. https://doi.org/10.1038/s41574-025-01142-8
Xie, X., Wang, H., Xiao, F., Jiang, X., Chen, Y., Huang, C., Tang, D., Wang, Y., Rui, S., Cheng, X., Deng, B., Yang, G., & Deng, W. (2025). Identifying aging-related biomarkers in adipose tissue using integrative bioinformatics and machine-learning approaches: discovery of ELN, MXD1, and FGF21 as key genes. Frontiers in Endocrinology, 16, 1638343. https://doi.org/10.3389/fendo.2025.1638343