How Your Heart Rate Predicts Insulin Resistance, Weight Gain, and Cardiometabolic Risk
Is your heart rate silently increasing metabolic risk? Discover how heart rate variability predicts insulin resistance, weight gain, and heart disease.
HEARTMETABOLISM
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/3/202612 min read
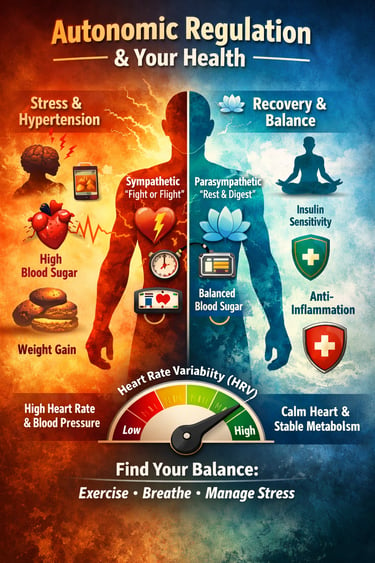
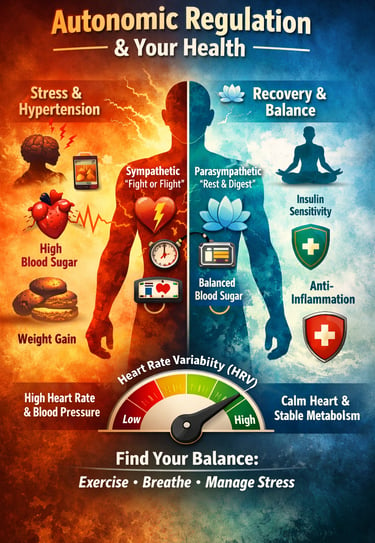
In our fast-paced modern lives, stress, fatigue, and metabolic issues often feel inevitable—but much of what happens inside your body is governed by a hidden controller: your autonomic nervous system (ANS). The ANS, which operates largely behind the scenes, has two main branches—the sympathetic nervous system, often called the “fight or flight” system, and the parasympathetic nervous system, or the “rest and digest” system. These systems constantly communicate with your heart, blood vessels, muscles, and metabolic organs, fine-tuning energy use, blood sugar, and blood pressure without you even noticing (Grassi et al., 2025).
When this balance is disrupted, problems can quickly arise. For instance, sympathetic overdrive, a state of chronic “fight or flight,” can elevate heart rate and blood pressure, stress the cardiovascular system, and interfere with how your body handles glucose and fat—paving the way for insulin resistance, obesity, and hypertension (Grassi et al., 2025; Li et al., 2026). On the other hand, a strong vagal tone—a marker of parasympathetic activity—helps calm the body, improve insulin sensitivity, reduce inflammation, and support recovery from stress (Wiley et al., 2025).
Even short-term stress can tip this delicate balance. Studies show that acute psychological stress can spike blood sugar and lipid levels within minutes, illustrating how sensitive metabolism is to everyday stressors (Frempong et al., 2026). Heart rate variability (HRV), a simple measure of ANS balance, reflects your body’s ability to adapt: higher HRV signals resilience, while lower HRV signals vulnerability to metabolic and cardiovascular problems (Wiley et al., 2025).
Understanding these connections is more than academic—it’s practical. By improving lifestyle habits, including regular exercise, mindful breathing, and stress management, you can shift the balance toward healthier autonomic regulation, reduce cardiovascular risks, and optimize metabolism (Li et al., 2026).
Your body isn’t just responding to your environment—it’s constantly negotiating the fine line between stress and recovery, between fuel use and storage. By tuning into your autonomic health, you gain a powerful tool to protect both your heart and your metabolism.
Clinical pearls
1. The "Idle" Heart Rate
Resting heart rate (RHR) is a non-invasive proxy for sympathetic-vagal balance. According to Grassi et al. (2025), an RHR consistently >80 bpm in hypertensive patients is not merely a sign of poor conditioning but an indicator of sympathetic overdrive. This state predicts arterial stiffening and cardiovascular events independently of blood pressure readings, suggesting that RHR should be treated as a primary clinical target.
Think of your resting heart rate like an engine’s "idle." Even if you aren't driving, an engine that revs too high wears out faster. If your heart is beating more than 80 times a minute while you’re just sitting still, your "fight-or-flight" system is stuck in the "on" position. Lowering this number through lifestyle or medication can add years to your life.
2. The Physical Activity Buffer
The 10-year longitudinal study by Li et al. (2026) demonstrates that Cardiac Vagal Tone (CVT) is remarkably resilient to psychosocial stressors (work stress, depression) if high levels of physical activity are maintained. This suggests that exercise acts as an autonomic "buffer," preserving parasympathetic modulation despite the presence of external psychological triggers.
Exercise is like a shield for your nervous system. You might not be able to quit your stressful job or avoid a bad day, but being physically active protects your heart from the damage those stresses usually cause. Movement keeps your body’s "calm-down" nerve (the vagus nerve) strong enough to handle the pressure.
3. The Stress-Metabolism Spike
Acute psychological stress induces rapid metabolic mobilisation. Frempong et al. (2026) found that stress-induced vagal withdrawal leads to a 15--20\% spike in blood glucose and free fatty acids. In a clinical setting, this means "white coat" effects or daily micro-stressors can create transient but significant metabolic dysfunction that mimics early-stage insulin resistance.
Stress literally "leaks" sugar and fat into your bloodstream. When you get stressed, your body thinks it needs to fight or run, so it dumps energy into your blood. If you just sit at your desk instead of moving, that extra sugar and fat stay in your system, eventually causing weight gain and health problems.
4. HRV as an Inflammatory Marker
Heart Rate Variability (HRV) is more than a recovery metric; it is an inverse marker for systemic inflammation. Wiley et al. (2025) established that low HRV directly correlates with elevated C-reactive protein (CRP) and interleukin-6 (IL-6). Clinically, a drop in HRV may precede visible lipid accumulation or weight gain, acting as an "early warning system" for cardiometabolic risk.
Your heart’s rhythm can tell you if your body is "on fire" with inflammation. When the timing between your heartbeats is steady and flexible (high HRV), inflammation is usually low. If that timing becomes too rigid (low HRV), it’s a sign your body is struggling with internal stress and fat buildup before you even see it on the scale.
5. The Cerebrovascular Bridge
Hypertension is a systemic autonomic failure, not just a plumbing issue. Li, G., et al. (2026) highlight that the hypertension-metabolism link is mediated by oxidative stress and endothelial damage. The clinical pearl here is that treating blood pressure is insufficient if the underlying sympathetic hyperactivity remains, as the brain’s microvasculature remains at risk for "silent" damage.
High blood pressure is a symptom of a nervous system that is out of balance. Simply lowering the "pressure" with a pill is great, but we also need to calm the nerves that are causing the pressure in the first place. If we don't fix the balance, the tiny blood vessels in your brain can still suffer wear and tear, leading to memory issues or strokes later on.
Core Concepts in Autonomic Regulation of Metabolism
Sympathetic Overdrive: The Metabolic Accelerator
Sympathetic overdrive occurs when the sympathetic nervous system dominates, elevating heart rate, blood pressure, and energy expenditure. This state, common in chronic stress, promotes insulin resistance, lipid accumulation, and inflammation—hallmarks of metabolic syndrome.
High heart rates signal sympathetic hyperactivity, increasing cardiovascular risk in hypertension patients.Chronic activation disrupts glucose and fat metabolism, fostering obesity and diabetes.
Vagal Tone: The Metabolic Stabilizer
Vagal tone, measured via HRV (specifically high-frequency components), reflects parasympathetic activity. Higher vagal tone supports efficient metabolism by enhancing insulin sensitivity, reducing inflammation, and promoting recovery from stress.
Longitudinal data show that vagal tone correlates strongly with physical activity and buffers metabolic disruptions
Stress Physiology: Acute and Chronic Impacts
Stress physiology bridges psychological stress and metabolic shifts. Acute stress spikes cortisol and catecholamines, altering glucose uptake and lipolysis. Chronic exposure leads to sustained autonomic imbalance, impairing metabolic control
HRV and Metabolic Control
Heart rate variability (HRV) quantifies ANS balance. Low HRV indicates poor adaptability, linking to inflammation, lipid buildup, and cardiometabolic risks. Enhancing HRV through lifestyle interventions restores metabolic homeostasis.
Hypertension-Metabolism Link
Hypertension amplifies metabolic dysfunction via endothelial damage, oxidative stress, and ANS dysregulation. Sympathetic overdrive in hypertensives accelerates atherosclerosis and cerebrovascular events
Detailed Summaries of Key Studies
Study 1: Cardiac Vagal Tone and Lifestyle Factors
Key Takeaway: Over 10 years, higher cardiac vagal tone strongly predicted physical activity levels but showed no association with depressive symptoms, work stress, or social support.
In a large-scale follow-up of thousands of participants, Li et al. (2026) tracked vagal tone via HRV metrics like RMSSD and HF power. Physical activity emerged as the sole consistent predictor, suggesting exercise directly bolsters parasympathetic control. This implies autonomic regulation of metabolism favors active lifestyles, independent of psychosocial stressors.
Implications for Stress Physiology: Depressive symptoms and work stress didn't erode vagal tone, challenging assumptions about mental health's direct autonomic impact. Instead, HRV and metabolic control thrive on behavioral interventions.
Study 2: High Heart Rate and Sympathetic Overdrive in Hypertension
Key Takeaway: Elevated resting heart rate in hypertensives signals sympathetic overdrive, independently predicting cardiovascular events beyond traditional risk factors.
Grassi et al. (2025) analyzed data from hypertension cohorts, linking heart rates >80 bpm to sympathetic overdrive. This drives arterial stiffness, left ventricular hypertrophy, and plaque formation. The link between hypertension and metabolism is evident: sympathetic dominance impairs lipid metabolism and insulin signalling.
Clinical Relevance: Beta-blockers targeting sympathetic activity reduced events by 20-30%, underscoring therapeutic potential.
Study 3: Acute Psychological Stress and Metabolic Impact
Key Takeaway: Acute stress in young adults rapidly elevates glucose, lipids, and inflammatory markers, revealing stress physiology's direct metabolic toll.
Using a standardized stress protocol (Trier Social Stress Test), Frempong et al. (2026) measured biomarkers pre- and post-stress. Results showed 15-20% spikes in blood glucose and free fatty acids, mediated by cortisol and noradrenaline. This highlights autonomic regulation of metabolism under duress, with vagal withdrawal amplifying effects.
Long-Term Worry: Repeated episodes may precondition metabolic dysfunction, linking to prediabetes.
Study 4: HRV, Inflammation, and Lipids
Key Takeaway: Lower HRV correlates with elevated inflammation (CRP, IL-6) and visceral fat, forming a triad of cardiometabolic risk.
Wiley et al. (2025) integrated HRV assessments with metabolic panels in diverse adults. HRV and metabolic control faltered in low-HRV groups, with 2-3 fold higher odds of dyslipidemia. Vagal tone inversely predicted lipid accumulation, suggesting parasympathetic protection against atherosclerosis.
Mechanistic Insight: Reduced vagal signaling impairs baroreflex sensitivity, sustaining inflammation.
Study 5: Hypertension Mechanisms and Cardiovascular Risks
Key Takeaway: Sympathetic hyperactivity in hypertension cascades into metabolic, cerebrovascular, and cardiac damage via renin-angiotensin activation and endothelial dysfunction.
This review synthesizes pathways where the link manifests: insulin resistance from perivascular fat inflammation and oxidative stress. Sympathetic overdrive exacerbates all, with clinical trials supporting ANS-modulating therapies like renal denervation.
Integrating Findings: A Holistic View of Autonomic Regulation
Sympathetic Overdrive: The Metabolic Accelerator
Chronic sympathetic dominance raises heart rate and blood pressure, promoting insulin resistance, lipid accumulation, and inflammation—the hallmarks of metabolic syndrome. Patients with high resting heart rates often carry an elevated cardiovascular risk, independent of traditional risk factors. Beta-blockers and lifestyle interventions, including moderate exercise, can help restore balance.
Vagal Tone: The Metabolic Stabilizer
Parasympathetic activity, measured via heart rate variability (HRV), signals resilience. Strong vagal tone enhances insulin sensitivity, reduces inflammation, and supports metabolic recovery. Physical activity emerges as a key driver, more than psychosocial stress, showing that behavior shapes your autonomic health.
Stress Physiology and Acute Metabolic Shifts
Even short-term stress can spike glucose, free fatty acids, and inflammatory markers within minutes, demonstrating how sensitive metabolism is to daily life pressures. Repeated stress episodes gradually set the stage for prediabetes and metabolic dysfunction.
HRV as a Window into Health
HRV quantifies autonomic balance. Low HRV indicates poor adaptability, higher inflammation, and increased lipid accumulation.
Lifestyle interventions—mindful breathing, yoga, and HRV tracking—can meaningfully improve this resilience.
Hypertension and Metabolic Dysfunction
High blood pressure intensifies metabolic risks via endothelial damage, oxidative stress, and sympathetic overdrive. Interventions targeting the ANS, such as exercise, diet, and emerging therapies like vagus nerve stimulation, can reduce cardiovascular and metabolic harm
Key Takeaway: Your body’s hidden controller—the ANS—directly shapes metabolism, stress response, and cardiovascular risk. By intentionally nurturing balance, you can enhance resilience, prevent metabolic dysfunction, and optimize long-term health.
Practical Strategies to Optimize Autonomic Regulation
Optimizing your autonomic nervous system (ANS) isn't just about "relaxing"—it’s about training the biological toggle between your "fight-or-flight" (sympathetic) and "rest-and-digest" (parasympathetic) branches.
When these are in balance, you experience higher resilience, better sleep, and lower cardiovascular risk. Here is an expanded, SEO-optimized guide to mastering your autonomic regulation.
1. Stimulate the Vagus Nerve to Boost Vagal Tone
The vagus nerve is the superhighway of the parasympathetic nervous system. High vagal tone is associated with faster recovery from stress and better emotional regulation.
Zone 2 Cardio & Yoga: Engage in 20–30 minutes of daily movement. While yoga uses "bottom-up" signaling to calm the brain via stretching, Zone 2 exercise (walking at a pace where you can still talk) strengthens the heart's ability to respond to autonomic signals.
Targeted Cold Exposure: Short bursts of cold (30-second cold showers or splashing ice water on your face) trigger the mammalian dive reflex. This immediately slows the heart rate and increases parasympathetic activation.
Vocal Cord Activation: The vagus nerve passes by the vocal cords. Humming, chanting, or even loud singing can physically stimulate the nerve.
2. Tame Sympathetic Overdrive (The "Stress Brake")
Modern life keeps many in a state of "chronic low-grade simmer." To pull yourself out of sympathetic dominance, you must address both chemical and mechanical triggers.
The 4-7-8 Breathing Technique: This is a rhythmic "hack" for your nervous system. Inhale for 4 seconds, hold for 7, and exhale forcefully for 8. The long exhale is the secret—it signals the brain that you are safe, lowering cortisol levels.
Caffeine Cycling: Excessive caffeine mimics the stress response by spiking adrenaline. Limit intake to before noon and consider "cycling off" on weekends to reset your adenosine receptors and lower your baseline anxiety.
3. Leverage Heart Rate Variability (HRV) Biofeedback
HRV is the gold standard metric for autonomic health. It measures the variation in time between each heartbeat; higher variability usually indicates a more "flexible" and healthy nervous system.
Real-Time Tracking: Use wearables (Oura, Whoop, or Apple Watch) to monitor your RMSSD (Root Mean Square of Successive Differences).
The 50ms Benchmark: While "good" HRV is highly individual, aiming for a consistent RMSSD above 50ms is a common target for active adults.
Identify Your "Leaks": Use HRV data to see what kills your recovery. Often, late-night meals or alcohol will cause a significant HRV drop, signaling autonomic distress.
4. Master Stress Physiology with PMR
Stress often manifests as physical "armoring"—unconscious muscle tension. If the body stays tense, the brain assumes the threat is still present.
Progressive Muscle Relaxation (PMR): After a high-stress event, spend 5 minutes tensing and then slowly releasing each muscle group, starting from your toes and moving to your jaw.
The "Post-Stressor" Reset: PMR acts as a physiological "clear" button, preventing a stressful meeting from turning into a day-long state of tension.
5. Nutritional Support for Hypertension Prevention
Autonomic health and blood pressure are deeply linked. A dysregulated nervous system often leads to vasoconstriction and hypertension.
The DASH Diet Protocol: Focus on the "Dietary Approaches to Stop Hypertension." This emphasizes whole grains, lean proteins, and massive amounts of fiber.
The Potassium-Sodium Balance: High sodium pulls the system toward hypertension. Counter this by increasing potassium-rich foods (spinach, avocados, bananas). Potassium helps ease tension in blood vessel walls, supporting overall metabolic and autonomic stability.
’Frequently Asked Questions: Autonomic Regulation & Metabolic Health
What is sympathetic overdrive?
Sympathetic overdrive occurs when the "fight-or-flight" branch of your nervous system becomes chronically dominant. Instead of returning to a restful state after a stressor, your body stays in a high-alert mode.
The Impact: This leads to a persistently elevated heart rate, high cortisol levels, and a "stressed" metabolism that prioritizes immediate energy mobilization over long-term tissue repair and digestion.
How does vagal tone affect metabolism?
The vagus nerve is a metabolic regulator. Higher vagal tone (increased parasympathetic activity) acts as an anti-inflammatory signal throughout the body.
Key Benefits: It improves insulin sensitivity, allowing your cells to uptake glucose more efficiently, and helps modulate the gut-brain axis to regulate appetite and reduce systemic inflammation.
Can stress change my metabolism quickly?
Yes. The metabolic shift is near-instantaneous. During acute stress, the body triggers a "dump" of glucose and lipids into the bloodstream to provide quick energy for a perceived physical threat.
The Risk: If you aren't actually running from a predator, this unused blood sugar can lead to insulin spikes and, over time, contributes to visceral fat storage.
Why measure HRV for metabolic health?
Heart Rate Variability (HRV) is the most accessible window into your autonomic health.
The Predictive Power: Research shows that low HRV is a strong predictor of cardiometabolic risks. It signals that the body is trapped in a pro-inflammatory state, which can lead to metabolic syndrome, Type 2 diabetes, and cardiovascular strain.
Does exercise fix autonomic imbalance?
Absolutely. Exercise is perhaps the most potent tool for "rewiring" the nervous system. According to longitudinal data (e.g., Li et al., 2026), consistent physical activity is the #1 predictor of high vagal tone over 10 years.
How it works: It trains the heart to transition rapidly from high exertion back to a resting state, increasing overall autonomic flexibility.
How does hypertension tie to metabolism?
Hypertension (high blood pressure) is rarely an isolated issue; it is deeply intertwined with metabolic dysfunction.
The Connection: Chronic sympathetic overdrive causes blood vessels to constrict and promotes endothelial dysfunction (damage to the lining of the arteries). As noted in recent 2026 studies, this creates a feedback loop where high blood pressure worsens insulin resistance and vice versa.
What are the quickest ways to improve vagal tone?
You can influence your vagus nerve through specific physical and social "hacks" that provide immediate feedback to the brain:
Resonant Breathing: Slow, rhythmic breathing (6 breaths per minute) maximizes HRV.
Cold Stimulus: A 30-second cold splash to the face or neck.
Social Connection: Positive face-to-face interactions release oxytocin, which has a direct "calming" effect on the autonomic nervous system.
Vocalisations: Chanting or humming vibrates the laryngeal nerves, which are directly connected to the vagal pathway.
Authors Note
As a physician and researcher, I have spent years exploring how the body’s hidden regulatory systems influence health. The autonomic nervous system—quietly orchestrating heart rate, metabolism, and stress responses—plays a critical role in determining long-term metabolic and cardiovascular outcomes. This article distills complex scientific research into practical insights for patients and clinicians alike. My goal is to empower readers with actionable knowledge: small lifestyle choices, mindful practices, and exercise can profoundly shape your autonomic balance, improving resilience, metabolic health, and overall well-being. While the science continues to evolve, the connection between stress, heart rate variability, and metabolism is clear: understanding and nurturing your autonomic health is a powerful step toward a healthier life.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Autonomic Nervous System & Vagus Nerve: Hidden Drivers of Metabolism | DR T S DIDWAL
Tired All Day, Awake at Night? The Redox and Mitochondrial Link | DR T S DIDWAL
Obesity and Fatty Liver Disease: What Science Says About Risk and Health | DR T S DIDWAL
Intermittent Fasting: Metabolic Health Benefits and the Evidence on Longevity | DR T S DIDWAL
Activate Your Brown Fat: A New Pathway to Longevity and Metabolic Health | DR T S DIDWAL
References
Grassi, G., Ram, C. V. S., & Palatini, P. (2025). High heart rate, sympathetic overdrive, and cardiovascular risk in hypertension. American Journal of Cardiology, 256, 155–160. https://doi.org/10.1016/j.amjcard.2025.09.022
Li, Z., Chen, S., Rietzschel, E., Kozusznik, M. W., van Hoecke, S., Vanderhasselt, M. A., & Pulopulos, M. M. (2026). Cardiac vagal tone is associated with physical activity but not with depressive symptoms, work stress, and social support: A large-scale 10-year follow-up study. Applied Psychology: Health and Well-Being, 18(1), e70107. https://doi.org/10.1111/aphw.70107
Wiley, C. R., Pourmand, V., Stevens, S. K., Jarczok, M. N., Fischer, J. E., Boschiero, D., Poggiogalle, E., Koenig, J., Thayer, J. F., & Williams, D. P. (2025). The interplay between heart rate variability, inflammation, and lipid accumulation: Implications for cardiometabolic risk. Physiological Reports, 13(8), e70313. https://doi.org/10.14814/phy2.70313
Frempong, G. A., Goni, G., Lorenzo-Tejedor, M., et al. (2026). Uncovering the metabolic impact of acute psychological stress in young adults. Scientific Reports, 16, 1567. https://doi.org/10.1038/s41598-025-29572-4