Autonomic Nervous System & Vagus Nerve: Hidden Drivers of Metabolism
Learn how your **autonomic nervous system controls metabolism, blood sugar, fat burning, and heart health—and why stress can trigger metabolic disease.
METABOLISM
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/17/202618 min read
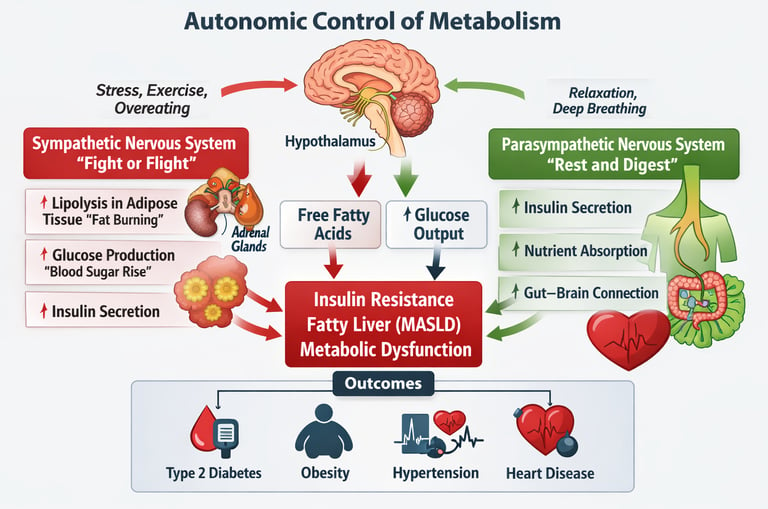
The autonomic nervous system (ANS) plays a central role in regulating metabolism. It controls how the body burns fat, releases glucose, and manages blood pressure through two branches: the sympathetic nervous system, which mobilizes energy during stress, and the parasympathetic nervous system, which promotes recovery, insulin secretion, and metabolic balance.
Your metabolism is not controlled only by calories, hormones, or exercise. Hidden behind every heartbeat, breath, and digestive signal is a powerful biological command system: the autonomic nervous system (ANS). This vast neural network—working entirely outside conscious control—acts as the body’s metabolic control center, constantly deciding whether to burn fat, release glucose, store energy, or raise blood pressure.
Every second, the brain communicates with the liver, pancreas, fat tissue, and muscles through two opposing neural forces: the sympathetic nervous system and the parasympathetic nervous system. When the sympathetic system activates—during stress, exercise, or fasting—it mobilizes energy by stimulating lipolysis in adipose tissue, increasing hepatic glucose production, and suppressing insulin secretion. These rapid metabolic shifts evolved to help humans survive danger, but when sympathetic signaling remains chronically elevated—as happens with persistent stress, poor sleep, and overeating—it can drive insulin resistance, visceral fat accumulation, and metabolic dysfunction–associated steatotic liver disease (MASLD) (Luukkonen et al., 2025; Wangler et al., 2026).
Conversely, the parasympathetic system—primarily mediated through the vagus nerve—promotes nutrient absorption, insulin release, and energy storage during the “rest-and-digest” state. This neural pathway also links the brain to the gut microbiome, forming part of the emerging gut–brain–metabolism axis, a critical regulator of inflammation, glucose control, and metabolic health (Kurhaluk et al., 2025).
Modern research increasingly shows that disturbances in autonomic balance—often reflected by reduced heart rate variability (HRV)—are closely associated with type 2 diabetes, hypertension, obesity, and cardiovascular disease (Patel et al., 2025). In other words, metabolic disease is not only a hormonal disorder; it is also a disorder of neural regulation.
Understanding how the autonomic nervous system controls metabolism provides a powerful new framework for preventing and treating cardiometabolic disease. It reveals why stress, sleep, exercise, diet, and even breathing patterns can profoundly influence fat burning, insulin sensitivity, blood pressure, and long-term cardiovascular health.
How the Nervous System Controls Metabolism
• Sympathetic system → releases glucose and burns fat
• Parasympathetic system → supports insulin and digestion
• Chronic stress → raises blood sugar and visceral fat
• High HRV → indicates metabolic resilience
Clinical pearls
1. The "Overflow" Phenomenon (Lipolysis)
Scientific Perspective: Chronic overnutrition triggers the Sympathetic Nervous System (SNS) to drive continuous, unregulated lipolysis. This ignores the body's energy needs and floods the portal circulation with free fatty acids, directly causing hepatic steatosis (fatty liver) and insulin resistance (Luukkonen et al., 2025).
Eating too much doesn't just "add weight"; it keeps your body's "fat-burning" switch stuck in the 'on' position. This sounds good, but it actually overflows your liver with fat it can’t process, making it harder for your body to manage blood sugar.
2. Vagal Tone as a Metabolic "Shield"
Scientific Perspective: High vagal tone (Parasympathetic activity) is a prerequisite for a healthy "cephalic phase" insulin response. It coordinates the gut-brain axis, utilizing the vagus nerve to sense microbial metabolites like short-chain fatty acids to maintain glucose homeostasis (Kurhaluk et al., 2025).
Your nervous system starts managing your meal before you even take a bite. By practicing "rest-and-digest" habits—like slow breathing or mindful eating—you strengthen your vagus nerve, which helps your body prime itself to handle sugar and inflammation.
3. Stress as a Molecular Disruptor (FGF21)
Scientific Perspective: Psychosocial stress isn't just "in the head"; it manifests molecularly via the HPA axis and mitochondrial signaling. Specifically, stress alters Fibroblast Growth Factor 21 (FGF21), a liver hormone that regulates fat-burning and energy expenditure (Kurade et al., 2025).
Being stressed at work or in relationships changes your chemistry. It disrupts a specific hormone (FGF21) that usually helps you stay lean and sensitive to insulin. In this sense, "stress management" is actually a form of weight and diabetes management.
4. HRV: The Dashboard of Metabolic Health
Scientific Perspective: Heart Rate Variability (HRV) serves as a non-invasive proxy for autonomic flexibility. Low HRV is an independent predictor of cardiovascular events in metabolic syndrome patients, reflecting a dangerous dominance of the SNS (Patel et al., 2025).
Your heart shouldn't beat like a perfectly steady metronome; it should be flexible. If you use a smartwatch to track HRV, a "low" score is a signal that your nervous system is too rigid, which puts extra strain on your heart and metabolism.
5. The Hypertension-Diabetes "Shared Root"
Scientific Perspective: Hypertension and Type 2 Diabetes are not separate diseases but often the same autonomic dysfunction (sympathetic overactivation) manifesting in different organs. Treating one via the SNS/RAAS pathway naturally improves the other (Li et al., 2026).
High blood pressure and high blood sugar are often two branches of the same tree. Because they share the same "root" in your nervous system, the same lifestyle changes—like exercise and better sleep—can often fix both problems at the same time.
Quick Medical Overview: Autonomic Nervous System and Metabolism
Key Concepts Explained
• Autonomic Nervous System (ANS)
The autonomic nervous system is the brain-controlled network that automatically regulates essential body functions such as heart rate, metabolism, digestion, blood pressure, and hormone release without conscious effort.
• Sympathetic Nervous System
This branch activates the body’s “fight-or-flight” response. It increases heart rate, stimulates the liver to release glucose into the bloodstream, promotes fat breakdown (lipolysis), and raises blood pressure to prepare the body for action.
• Parasympathetic Nervous System
Often described as the “rest-and-digest” system, this branch supports recovery and metabolic balance. It stimulates insulin secretion, improves digestion, enhances nutrient absorption, and promotes energy storage after meals.
• Heart Rate Variability (HRV)
HRV measures the variation in time between heartbeats. It is an important indicator of autonomic balance. Higher HRV reflects a healthy and flexible nervous system, while low HRV is associated with metabolic syndrome, insulin resistance, and increased cardiovascular risk.
• Chronic Sympathetic Activation
When the sympathetic nervous system remains persistently active due to chronic stress, poor sleep, or excessive caloric intake, it can contribute to insulin resistance, fatty liver disease, hypertension, and obesity.
• Lifestyle Factors That Influence Autonomic Balance
Daily habits strongly affect autonomic function. Regular physical activity, quality sleep, stress management techniques, and a balanced diet can improve vagal tone, reduce sympathetic overactivation, and support healthy metabolism.
Autonomic State vs Metabolic Effect
Sympathetic dominance:Increased lipolysis, glucose release
Low vagal tone: Insulin resistance
High HRV:Metabolic resilience
Chronic stress:Visceral fat gain
Signs Your Autonomic Nervous System May Be Affecting Your Metabolism
When the autonomic nervous system becomes imbalanced, particularly when sympathetic activity remains chronically elevated and parasympathetic (vagal) activity is reduced, several metabolic and cardiovascular symptoms can appear. Many people experience these signs for years before developing conditions such as type 2 diabetes, metabolic syndrome, or hypertension.
Common warning signs include:
Persistently High Resting Heart Rate
A resting heart rate consistently above 80–85 beats per minute may indicate sympathetic dominance and reduced vagal tone.
Low Heart Rate Variability (HRV)
Wearable devices increasingly measure HRV. Low HRV reflects reduced autonomic flexibility and is associated with insulin resistance and cardiovascular risk.
Stress-Related Blood Sugar Spikes
Some individuals notice elevated glucose levels during psychological stress, even without dietary changes. This occurs because sympathetic activation stimulates hepatic glucose release.
Abdominal or Visceral Fat Accumulation
Chronic stress signaling and cortisol elevation promote fat deposition around abdominal organs, a key driver of metabolic syndrome.
Poor Sleep or Frequent Night Awakenings
Autonomic imbalance often disrupts sleep architecture, preventing the parasympathetic nervous system from restoring metabolic balance.
Digestive Disturbances
Low vagal activity may impair digestion, leading to bloating, altered gut motility, or microbiome imbalance, which can further influence metabolic regulation.
Elevated Blood Pressure
Persistent sympathetic activation increases vascular tone and renin–angiotensin signaling, contributing to hypertension.
I. Sympathetic Nervous System and Metabolism: How Stress Triggers Fat Burning and Glucose Release
The sympathetic nervous system (SNS) is a key regulator of metabolic function. Often called the body’s “fight-or-flight” system, it prepares the body to respond to physical or psychological stress. When activated by factors such as exercise, emotional stress, cold exposure, or low blood sugar, the SNS releases the neurotransmitter norepinephrine and stimulates the adrenal glands to produce epinephrine (adrenaline).
These signals rapidly alter metabolism by increasing fat breakdown, glucose production, heart rate, and blood pressure, ensuring that energy is quickly available to the brain and muscles.
Sympathetic Activation and Fat Metabolism
One of the most important metabolic actions of the SNS is the stimulation of lipolysis, the process by which stored triglycerides in adipose tissue are broken down into free fatty acids (FFAs) for energy.
This occurs through a well-defined biological pathway:
• Norepinephrine binds to β-adrenergic receptors on fat cells
• Intracellular signaling pathways activate hormone-sensitive lipase (HSL)
• Stored triglycerides are broken down into fatty acids and glycerol
These fatty acids are then transported to the liver and muscles, where they can be oxidized for energy.
However, emerging research shows that chronic sympathetic activation may disrupt metabolic balance.
A 2025 study published in the Journal of Hepatology found that persistent sympathetic stimulation caused by chronic overnutrition can drive continuous lipolysis. This results in excessive delivery of fatty acids to the liver, promoting hepatic steatosis, insulin resistance, and metabolic dysfunction (Luukkonen et al., 2025).
Key Insight:
Overeating does not simply increase calorie intake—it may also chronically stimulate sympathetic signaling, creating a metabolic cycle that promotes fatty liver disease and insulin resistance.
A later review in Nature Reviews Endocrinology confirmed that sympathetic nerve fibers directly innervate adipose tissue, making neural signaling a primary regulator of fat mobilization (Wangler et al., 2026).
How the Sympathetic Nervous System Raises Blood Sugar
In addition to regulating fat metabolism, the SNS strongly influences blood glucose regulation.
During stress or physical exertion, sympathetic signals instruct the liver to release glucose through glycogenolysis, ensuring that muscles and the brain have sufficient fuel.
At the same time, the SNS:
• Suppresses insulin secretion from the pancreas
• Stimulates glucagon release
• Increases hepatic glucose production
These effects temporarily increase blood glucose levels, which is beneficial in acute stress situations.
However, chronic sympathetic activation—often caused by long-term psychological stress—can keep blood sugar levels persistently elevated. Over time this contributes to insulin resistance and the development of type 2 diabetes.
A 2026 review in Nature Reviews Endocrinology emphasized that sympathetic innervation of the liver, pancreas, and skeletal muscle forms a neural network that tightly regulates glucose metabolism (Wangler et al., 2026).
II. Parasympathetic Nervous System: The Metabolic Brake
If the sympathetic system acts as the metabolic accelerator, the parasympathetic nervous system (PNS) functions as the body’s metabolic brake.
The PNS is primarily mediated by the vagus nerve, the longest cranial nerve, which connects the brainstem to the heart, lungs, pancreas, and gastrointestinal tract.
This system dominates during the “rest-and-digest” state, promoting processes that restore metabolic balance after stress or activity.
Key parasympathetic metabolic functions include:
• Stimulating insulin secretion
• Enhancing nutrient absorption
• Promoting glycogen synthesis
• Supporting fat storage and metabolic recovery
The Vagus Nerve and the Gut–Brain–Metabolism Axis
The vagus nerve is increasingly recognized as a critical regulator of the gut–brain axis, a communication network linking the nervous system, digestive system, and the gut microbiome.
A review published in the International Journal of Molecular Sciences showed that vagal signaling integrates immune, microbial, and metabolic signals that influence energy balance and systemic inflammation (Kurhaluk et al., 2025).
High vagal tone—a measure of parasympathetic activity—is associated with:
• Improved insulin sensitivity
• Reduced chronic inflammation
• A healthier and more diverse gut microbiome
In contrast, low vagal activity is linked to insulin resistance, metabolic syndrome, and gut dysbiosis.
Vagal sensory fibers detect metabolites produced by gut bacteria, such as short-chain fatty acids, and transmit these signals to the hypothalamus, a brain region that regulates hunger, satiety, and energy balance.
This neural communication explains why gut health and metabolic health are closely interconnected.
Patient-Friendly Insight:
Practices that enhance vagal tone—such as slow breathing, meditation, yoga, singing, and regular exercise—may support metabolic health through the gut-brain axis.
III. Stress Hormones and Metabolism: The Brain–Hormone Connection
The autonomic nervous system works closely with the hypothalamic-pituitary-adrenal (HPA) axis, the body’s central stress hormone system.
Together, these networks coordinate a complex physiological response that influences energy metabolism, inflammation, and glucose regulation.
FGF21: A Metabolic Hormone Linking Stress and Energy Balance
Recent research has identified fibroblast growth factor 21 (FGF21) as an important hormone connecting stress physiology with metabolic regulation.
A 2025 study published in Nature Metabolism demonstrated that FGF21 levels in humans are influenced by both mitochondrial stress and psychological stress (Kurade et al., 2025).
FGF21 plays several metabolic roles:
• Enhancing fat oxidation
• Improving insulin sensitivity
• Regulating energy expenditure
The researchers observed that individuals exposed to higher levels of psychosocial stress showed altered FGF21 signaling, particularly when mitochondrial function was impaired.
Key Insight:
Psychological stress may directly influence metabolism through mitochondrial pathways and hormone signaling, reinforcing the connection between mental health and metabolic disease.
Cortisol, Catecholamines, and the Metabolic Stress Response
Two additional stress hormones—cortisol and norepinephrine—play major roles in the metabolic stress response.
Cortisol increases:
• Gluconeogenesis (glucose production)
• Appetite for calorie-dense foods
• Visceral fat accumulation
Chronic cortisol elevation, as seen in conditions such as Cushing’s syndrome or chronic psychological stress, strongly contributes to metabolic syndrome and insulin resistance.
At the same time, norepinephrine stimulates lipolysis and hepatic glucose output, amplifying the metabolic effects of the autonomic nervous system.
Together, these hormonal and neural signals explain why chronic stress is a powerful driver of obesity, type 2 diabetes, and cardiovascular disease.
IV. Heart Rate Variability and Metabolic Health: A Window Into Autonomic Function
Heart rate variability (HRV)—the natural variation in the time between consecutive heartbeats—is one of the most practical clinical markers of autonomic nervous system balance. Contrary to what many people assume, a healthy heart does not beat with perfect regularity. Instead, it constantly adjusts its rhythm in response to signals from the sympathetic and parasympathetic nervous systems.
High HRV indicates a flexible and resilient autonomic system, reflecting strong parasympathetic (vagal) activity and effective stress adaptation. In contrast, low HRV suggests sympathetic dominance, a state associated with chronic stress, metabolic dysfunction, and increased cardiovascular risk.
Over the past decade, HRV has emerged as a valuable biomarker linking autonomic regulation with cardiometabolic health, including conditions such as metabolic syndrome, obesity, type 2 diabetes, and hypertension.
HRV as a Predictor of Cardiovascular and Metabolic Risk
Growing clinical evidence indicates that reduced HRV is not merely a marker of stress but an independent predictor of cardiovascular disease.
A 2025 study published in the Journal of Heart Valve Disease evaluated HRV as a predictor of cardiovascular outcomes in individuals with metabolic syndrome. The researchers found that low HRV independently predicted adverse cardiovascular events, highlighting the critical role of autonomic dysfunction in cardiometabolic disease (Patel et al., 2025).
Several biological mechanisms explain this association. Reduced HRV, reflecting sympathetic overactivity or reduced vagal tone, is linked to:
• Elevated blood pressure
• Insulin resistance
• Atherogenic dyslipidemia
• Chronic low-grade inflammation
• Endothelial dysfunction
Together, these abnormalities accelerate atherosclerosis and cardiovascular risk, demonstrating how autonomic imbalance contributes directly to cardiometabolic disease progression.
Can Improving Metabolism Restore HRV?
If reduced HRV reflects metabolic dysfunction, an important clinical question arises: can improving metabolic health restore autonomic balance?
Evidence suggests the answer is yes.
A 2025 systematic review and meta-analysis published in Updates in Surgery examined the effects of metabolic and bariatric surgery on autonomic function in patients with obesity-related metabolic disease. The analysis showed that procedures such as sleeve gastrectomy and gastric bypass significantly improved HRV, indicating restoration of autonomic balance (Jamialahmadi et al., 2025).
These findings suggest that reversing metabolic abnormalities—such as insulin resistance, visceral obesity, and systemic inflammation—can also repair autonomic dysfunction.
In other words, metabolic health and nervous system health are tightly interconnected.
Clinical Implications of HRV Monitoring
HRV measurement is becoming increasingly accessible due to wearable technologies and smart health devices.
Monitoring HRV may help clinicians and individuals:
• Detect early autonomic imbalance
• Assess cardiometabolic risk
• Track response to lifestyle interventions or treatment
For individuals not undergoing metabolic surgery, several lifestyle interventions are known to improve HRV and autonomic balance, including:
• Regular aerobic and resistance exercise
• Stress reduction practices such as meditation or breathing exercises
• Adequate sleep and circadian rhythm regulation
• A nutrient-dense diet supporting metabolic health
These strategies improve parasympathetic activity and reduce chronic sympathetic overactivation, promoting both cardiovascular and metabolic resilience.
V. The Hypertension–Metabolism Connection
Hypertension and metabolic disorders are deeply interconnected. Rather than existing as separate diseases, they often arise from shared biological mechanisms involving the autonomic nervous system, hormonal signaling, and metabolic regulation.
Understanding this relationship is essential for effective prevention and treatment of both cardiovascular disease and metabolic syndrome.
Shared Mechanisms Linking Hypertension and Metabolic Dysfunction
A comprehensive 2026 review in Clinical and Experimental Hypertension examined the mechanisms connecting hypertension with cardiovascular and cerebrovascular disease. The authors highlighted sympathetic nervous system overactivation as a central driver of essential hypertension (Li et al., 2026).
Excess sympathetic signaling increases blood pressure through several pathways:
• Direct vasoconstriction, increasing vascular resistance
• Activation of the renin–angiotensin–aldosterone system (RAAS)
• Increased sodium retention and fluid volume
These same pathways also contribute to metabolic dysfunction, including insulin resistance and abnormal lipid metabolism.
As a result, sympathetic overactivity represents a shared biological link between hypertension and metabolic syndrome.
Lifestyle Factors That Influence Both Blood Pressure and Metabolism
Lifestyle behaviors play a major role in regulating both autonomic function and cardiometabolic health.
A 2025 review published in Frontiers in Endocrinology emphasized that factors such as diet, physical activity, sleep quality, and stress management influence hypertension and metabolic disorders through their effects on autonomic regulation (Wang et al., 2025).
For example:
• Exercise increases vagal tone and improves insulin sensitivity
• Healthy dietary patterns influence gut–brain signaling and inflammation
• Stress management reduces sympathetic nervous system activation
These lifestyle interventions therefore improve both blood pressure control and metabolic health simultaneously.
Evidence From Experimental Studies
Mechanistic insights also come from experimental research.
A 2026 study in the Journal of Nutrition and Metabolism investigated how diet interacts with hypertension in an animal model. The researchers found that hypertensive animals showed significant alterations in lipid metabolism, oxidative stress markers, and inflammatory signaling compared with normotensive controls (Smoradkova et al., 2026).
Importantly, dietary modifications were able to partially reverse these metabolic abnormalities.
Although animal studies cannot fully replicate human physiology, these findings support the concept that hypertension is not merely a vascular disorder but a systemic metabolic condition.
Clinical Implication: Treat the System, Not Just the Blood Pressure
These findings highlight an important clinical principle: treating hypertension should not focus solely on lowering blood pressure numbers.
Lifestyle interventions that address autonomic balance, metabolic health, and systemic inflammation can simultaneously improve:
• Blood pressure control
• Insulin sensitivity
• Lipid metabolism
• Cardiovascular risk
For this reason, integrated lifestyle strategies—including dietary improvements, regular exercise, adequate sleep, and stress management—are often more effective long-term than relying on pharmacotherapy alone.
Bringing It All Together: The Autonomic-Metabolic Web
• Your nervous system constantly regulates metabolism
The autonomic nervous system (ANS) works automatically to control heart rate, digestion, hormone release, and energy balance. It has two main branches: the sympathetic system, which activates during stress or physical activity, and the parasympathetic system, which supports recovery and energy storage.
• Stress signals can raise blood sugar and fat levels
When the sympathetic nervous system is activated, the body releases stress hormones like adrenaline and norepinephrine. These signals stimulate the liver to release glucose and trigger fat cells to break down stored fat for energy. In short bursts this response is healthy, but chronic stress can keep these pathways switched on, contributing to insulin resistance and metabolic dysfunction.
• The vagus nerve helps restore metabolic balance
The parasympathetic system—largely controlled by the vagus nerve—supports digestion, insulin secretion, and anti-inflammatory signaling. Strong vagal activity is associated with better glucose control, improved gut health, and lower cardiovascular risk.
• Heart rate variability reveals nervous system health
Doctors increasingly use heart rate variability (HRV) as a window into autonomic balance. Higher HRV reflects a flexible and resilient nervous system, while low HRV is linked with metabolic syndrome, hypertension, and cardiovascular disease.
• Lifestyle habits directly influence this neural-metabolic system
Regular exercise, good sleep, stress reduction, and a nutrient-rich diet can improve autonomic balance. These habits enhance vagal tone, reduce sympathetic overactivation, and support healthy metabolism and cardiovascular function.
The bottom line: Metabolism is not just about what you eat—it is also about how your brain and nervous system regulate energy, hormones, and organ function. By supporting this neural network through healthy lifestyle choices, you can protect both your metabolic and cardiovascular health for years to come.
Key Lifestyle Habits That Strengthen Autonomic–Metabolic Health
• Regular Aerobic and Resistance Exercise
Consistent physical activity improves insulin sensitivity, heart rate variability (HRV), and vagal tone. Both aerobic exercise and resistance training enhance metabolic flexibility, reduce visceral fat, and support healthy glucose regulation.
• Stress Management Practices
Chronic psychological stress activates the sympathetic nervous system, increasing cortisol levels and metabolic strain. Techniques such as mindfulness meditation, breathing exercises, and yoga help restore autonomic balance by reducing sympathetic dominance and enhancing parasympathetic activity.
• High-Quality Sleep
Adequate sleep is essential for maintaining healthy autonomic regulation. Sleep deprivation reduces HRV, increases sympathetic activity, and worsens insulin resistance, increasing the risk of metabolic syndrome and cardiometabolic disease.
• A Fiber-Rich, Microbiome-Supporting Diet
A diverse diet rich in fiber, plant-based foods, and fermented products supports the gut microbiome. The gut communicates with the brain through the gut–brain axis and vagus nerve, influencing autonomic signaling, inflammation, and metabolic regulation.
• Avoiding Chronic Overnutrition
Consistently consuming excess calories—especially from highly processed foods—can trigger sympathetic overactivation, promoting visceral fat accumulation, fatty liver disease, and insulin resistance.
Clinical Insight
Together, these lifestyle interventions help restore autonomic balance, improve metabolic efficiency, and reduce cardiometabolic risk. Research summarized by Wang and colleagues (2025) in Frontiers in Endocrinology emphasizes that lifestyle-based strategies remain one of the most powerful tools for preventing metabolic disease and maintaining long-term metabolic health.
Frequently Asked Questions
Q1. Can stress actually cause type 2 diabetes?
Chronic psychological stress doesn't directly cause type 2 diabetes on its own, but it is a significant contributing factor. Sustained sympathetic activation raises blood glucose, promotes visceral fat accumulation, and drives insulin resistance. A 2025 study (Kurade et al.) showed that psychosocial stress disrupts FGF21, a hormone critical for insulin sensitivity, through mitochondrial pathways. Combined with genetic predisposition and poor lifestyle habits, chronic stress meaningfully elevates type 2 diabetes risk.
Q2. What is vagal tone and how can I improve it?
Vagal tone refers to the activity level of the vagus nerve — the main parasympathetic nerve controlling heart, lungs, and digestive organs. High vagal tone is associated with better metabolic health, lower inflammation, and reduced cardiovascular risk. Evidence-backed strategies to improve vagal tone include: slow, diaphragmatic breathing (e.g., 4-7-8 breathing); regular aerobic exercise; cold water facial immersion; meditation and mindfulness; singing or humming; and maintaining a healthy, diverse gut microbiome through dietary fiber and fermented foods.
Q3. What does heart rate variability (HRV) tell me about my metabolism?
HRV is the variation in time between consecutive heartbeats. High HRV indicates good autonomic flexibility (healthy balance of sympathetic and parasympathetic activity), while low HRV signals autonomic rigidity, often reflecting sympathetic dominance. Research published in 2025 (Patel et al.) showed that low HRV independently predicts cardiovascular events in people with metabolic syndrome. A separate meta-analysis (Jamialahmadi et al., 2025) confirmed that treating metabolic disease with bariatric surgery improves HRV — showing that autonomic and metabolic health rise and fall together.
Q4. Is lipolysis good or bad for health?
Lipolysis — the breakdown of fat for energy — is essential and beneficial in healthy amounts, especially during exercise or fasting. The problem arises with chronic overactivation. A 2025 study (Luukkonen et al.) demonstrated that persistent sympathetic nervous system activation due to overnutrition drives uncontrolled lipolysis, flooding the liver with fatty acids and triggering liver fat accumulation (steatosis), insulin resistance, and metabolic dysfunction. In other words, the goal is regulated lipolysis — not too little, not too much.
Q5. How are high blood pressure and diabetes connected through the nervous system?
Both hypertension and type 2 diabetes share a common autonomic root: chronic sympathetic nervous system overactivation. Elevated sympathetic tone raises blood pressure by increasing vascular resistance and activating the renin-angiotensin-aldosterone system. Simultaneously, it promotes hepatic glucose output and drives insulin resistance. The 2026 reviews by Li et al. and Wang et al. both emphasized this shared pathway — which is why lifestyle interventions like exercise and stress reduction improve both conditions simultaneously.
Q6. Can bariatric surgery improve my nervous system function?
Yes — significantly. A 2025 systematic review and meta-analysis (Jamialahmadi et al.) found that metabolic and bariatric surgeries, such as sleeve gastrectomy and gastric bypass, substantially improved heart rate variability (HRV) in patients with obesity-related metabolic disease. This suggests that by correcting the metabolic environment — reducing insulin resistance, inflammation, and visceral fat — bariatric surgery reverses the autonomic dysfunction that characterizes metabolic syndrome, restoring healthier sympathetic-parasympathetic balance.
Author’s Note
This article explores the growing body of scientific research showing that metabolism is not regulated only by calories, hormones, or diet. Increasing evidence from neuroscience, endocrinology, and cardiometabolic medicine demonstrates that the autonomic nervous system (ANS) plays a central role in controlling how the body burns fat, regulates blood sugar, manages blood pressure, and responds to stress.
In recent years, advances in fields such as neuroendocrinology, metabolic physiology, and the gut–brain axis have revealed that the brain communicates continuously with organs such as the liver, pancreas, adipose tissue, and heart through complex neural networks. These pathways influence key metabolic processes including lipolysis, glucose production, insulin secretion, and inflammation. When this regulatory system becomes chronically imbalanced—often due to persistent stress, poor sleep, sedentary behavior, or excessive caloric intake—it can contribute to metabolic disorders such as type 2 diabetes, obesity, fatty liver disease, and hypertension.
The goal of this review is to translate emerging scientific insights into a format that is understandable and useful for both clinicians and informed readers. While the concepts discussed are grounded in peer-reviewed medical literature, the article is designed to help readers appreciate how everyday lifestyle factors—such as physical activity, nutrition, sleep, and stress management—interact with the nervous system to shape long-term metabolic health.
Medical science increasingly recognizes that cardiometabolic disease is a systems problem, involving interactions between the brain, nervous system, hormones, immune signaling, and metabolism. A deeper understanding of this interconnected physiology can help guide more effective prevention strategies and more holistic approaches to treatment.
Readers should remember that the information provided here is intended for educational purposes and should not replace personalized medical advice from a qualified healthcare professional
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Tired All Day, Awake at Night? The Redox and Mitochondrial Link | DR T S DIDWAL
Obesity and Fatty Liver Disease: What Science Says About Risk and Health | DR T S DIDWAL
Intermittent Fasting: Metabolic Health Benefits and the Evidence on Longevity | DR T S DIDWAL
Activate Your Brown Fat: A New Pathway to Longevity and Metabolic Health | DR T S DIDWAL
References
Jamialahmadi, T., Abdalla, M.A., Mirhadi, E. et al. (2025). Impact of metabolic and bariatric surgery on heart rate variability: a systematic review and meta-analysis. Updates in Surgery. https://doi.org/10.1007/s13304-025-02444-2
Kurade, M., Bobba-Alves, N., Kelly, C. et al. (2025). Mitochondrial and psychosocial stress-related regulation of FGF21 in humans. Nature Metabolism, 7, 2212–2220. https://doi.org/10.1038/s42255-025-01388-6
Kurhaluk, N., Kołodziejska, R., Kamiński, P., & Tkaczenko, H. (2025). Integrative Neuroimmune Role of the Parasympathetic Nervous System, Vagus Nerve and Gut Microbiota in Stress Modulation: A Narrative Review. International Journal of Molecular Sciences, 26(23), 11706. https://doi.org/10.3390/ijms262311706
Li, G., Zhang, J. J., Mou, X., Zhuo, X., Li, W., Zhang, S., & Li, X. (2026). Mechanisms linking hypertension to cardiovascular and cerebrovascular diseases and their clinical implications: A comprehensive review. Clinical and Experimental Hypertension, 48(1). https://doi.org/10.1080/10641963.2026.2631606
Luukkonen, P. K., Donnelly, K. L., Smith, C. I. E., & Yki-Järvinen, H. (2025). Sympathetic activation of adipose tissue lipolysis underlies overnutrition-induced metabolic dysfunction. Journal of Hepatology. https://doi.org/10.1016/S0168-8278(25)02252-4
Patel, T. J., Patel, K. B., & Acharya, S. K. (2025). Assessment of Heart Rate Variability as a Predictor of Cardiovascular Events in Patients with Metabolic Syndrome. Journal of Heart Valve Disease, 30(4), 117–120.
Smoradkova, K., Szudzik, M., Maksymiuk, K., Samborowska, E., Drapala, A., Ufnal, M., & Tomasova, L. (2026). Metabolic Alterations Associated With Diet and Hypertension in Rats. Journal of Nutrition and Metabolism, 2026, 6248625. https://doi.org/10.1155/jnme/6248625
Wang, Q., Wu, D., & Huang, Y. (2025). The integral role of lifestyle in the prevention and management of hypertension and associated cardiometabolic and cognitive disorders: a review. Frontiers in Endocrinology, 16, 1682814. https://doi.org/10.3389/fendo.2025.1682814
Wangler, S., Jarczok, M. N., Ennis, M. et al. (2026). The autonomic nervous system in the regulation of glucose and lipid metabolism. Nature Reviews Endocrinology. https://doi.org/10.1038/s41574-025-01221-w