Fatty Liver and Exercise: How Resistance Training Can Reverse MASLD in 12 Weeks
Best exercises for liver health in MASLD. Learn a safe, effective strength training plan that reduces fatty liver and improves metabolic health.
METABOLISM
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/13/202612 min read
Fatty liver disease is no longer a silent condition—and exercise may be its most powerful therapy. Metabolic dysfunction–associated steatotic liver disease (MASLD), previously known as NAFLD, now affects more than one in three adults worldwide and is tightly linked to insulin resistance, obesity, and cardiometabolic disease. Once considered a passive fat-storage disorder, MASLD is now recognized as a dynamic, reversible metabolic condition—especially when targeted with the right type of physical activity.
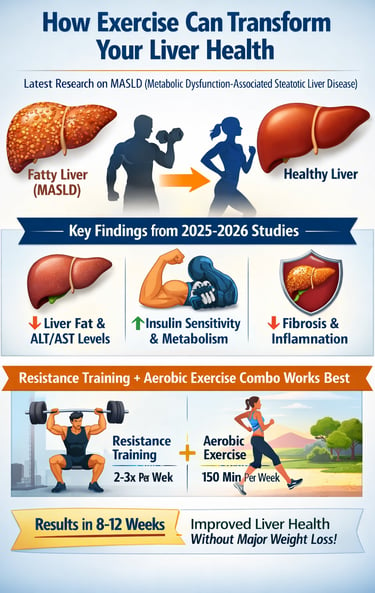
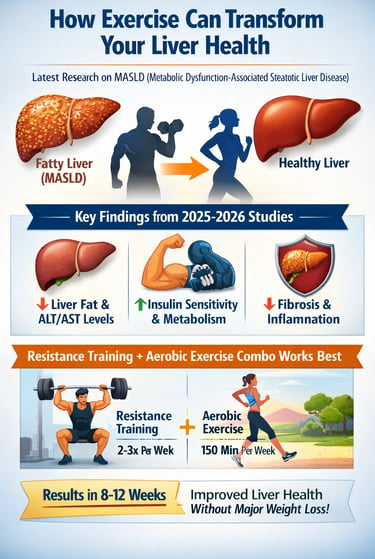
Emerging research from 2025–2026 has fundamentally reshaped how clinicians understand MASLD treatment. Large systematic reviews and meta-analyses show that structured exercise—particularly resistance training—can significantly reduce intrahepatic fat, improve liver enzymes, and slow fibrosis progression, even without major weight loss (Medeiros et al., 2025; Chen et al., 2026). These benefits extend beyond the liver, improving insulin sensitivity, mitochondrial function, bile acid metabolism, and systemic inflammation—key drivers of disease progression (Liu & Liu, 2025; Shi et al., 2025).
Importantly, resistance training appears uniquely effective in targeting hepatic steatosis through muscle-liver crosstalk and metabolic signaling, while aerobic exercise provides complementary cardiovascular and anti-inflammatory benefits. When combined, these modalities consistently outperform either approach alone (Shi et al., 2025; Rajewski et al., 2025).
For patients and clinicians alike, this evidence delivers a clear message: MASLD is not inevitably progressive. With properly designed resistance and aerobic exercise programs, meaningful improvements can occur within 8–12 weeks, making exercise not an optional lifestyle add-on, but a first-line therapeutic strategy (Chen et al., 2026).
This article synthesizes the most recent high-quality evidence to explain how and why exercise transforms liver health—and how to apply these findings in real-world MASLD management.
Clinical Insight
In clinical practice, many MASLD patients assume that significant weight loss is required before liver improvement occurs. However, several recent studies show that resistance training alone can significantly reduce hepatic fat and normalize liver enzymes, even when body weight changes are modest.
Fatty Liver & Exercise — Quick Facts
• MASLD affects ~30–38% of adults worldwide
• Resistance training 2–3× per week significantly reduces liver fat
• Benefits occur even without major weight loss
• Combined aerobic + resistance exercise produces the best results
• Measurable improvement typically occurs within 8–12 weeks
Clinical pearls
. The "Metabolic Bypass" Effect
Scientific: Resistance training (RT) improves hepatic steatosis and insulin sensitivity through non-canonical pathways, independent of significant total body mass reduction. RT upregulates glucose transporter type 4 (GLUT4) in skeletal muscle, creating a "glucose sink" that alleviates the metabolic burden on the liver.
The scale isn't the boss. You can significantly "de-fat" your liver and improve your health markers even if your body weight stays the same. Building muscle creates a sponge that soaks up extra blood sugar before it can be turned into liver fat.
2. The Synergy of Modalities
Scientific: While RT specifically targets intrahepatic lipid content and ALT/AST normalization, aerobic exercise (AE) remains the gold standard for improving cardiorespiratory fitness (VO 2max) and reducing systemic inflammatory cytokines like TNF-α. A concurrent training model (RT + AE) yields the most robust reduction in the Hepatic Steatosis Index (HSI).
Think of resistance training as the "liver fixer" and aerobic exercise (like walking or cycling) as the "heart and lung protector." Doing both gives you a total body shield against the complications of MASLD.
3. Bile Acid Reshaping
Scientific: Recent evidence (Shi et al., 2025) suggests that exercise modulates the farnesoid X receptor (FXR) signaling pathway by altering the circulating bile acid profile. This reduces lipogenesis (fat creation) and promotes fatty acid oxidation within the hepatocytes.
Exercise is a chemical messenger. When you move, your body changes the "chemistry soup" (bile acids) it uses for digestion. This new mixture sends a signal to your liver to stop storing fat and start burning it for fuel.
4. The 8-to-12 Week "Threshold"
Scientific: Meta-analyses indicate that while cellular adaptations in mitochondrial efficiency occur within days, statistically significant architectural changes in hepatic tissue and stabilization of liver enzymes typically require a minimum dose-response period of 8–12 weeks of consistent intervention.
Consistency beats intensity. You don’t need to be an athlete on Day 1, but you do need to stay the course. Your liver cells need about three months of "re-training" before the results show up clearly on your blood tests and scans.
5. Myokine-Mediated Protection
Scientific: Contracting skeletal muscle acts as an endocrine organ, secreting "myokines" (such as Irisin and IL-6) that facilitate cross-talk with the liver. These myokines have been shown to inhibit the activation of hepatic stellate cells, which are the primary drivers of liver fibrosis (scarring).
Your muscles talk to your liver. Every time you lift weights or push your muscles, they release "healing signals" that travel through your blood to your liver. These signals tell the liver to stop forming scar tissue and keep its texture soft and healthy.
Understanding MASLD: Why Exercise Matters
Metabolic dysfunction-associated steatotic liver disease is characterized by excessive fat accumulation in liver cells, occurring in individuals with metabolic dysfunction markers like obesity, insulin resistance, and dyslipidemia. Unlike traditional approaches focusing on medication alone, emerging evidence emphasizes the critical role of lifestyle modifications, particularly structured exercise programs.
The liver is your body's metabolic powerhouse. When fat infiltrates hepatic tissue, it impairs insulin sensitivity, triggers inflammation, and disrupts bile acid metabolism. The exciting part? Physical exercise directly addresses these mechanisms at their source.
Key Biological Mechanisms: How Exercise Improves MASLD
1. Resistance Training – The “Glucose Sink” Effect
Resistance training increases GLUT4 expression in skeletal muscle.
GLUT4 allows muscle cells to absorb more glucose from the bloodstream.
This reduces excess glucose available for hepatic fat synthesis.
Result: Lower liver fat accumulation and improved insulin sensitivity.
2. Aerobic Exercise – Mitochondrial Fat Burning
Aerobic activity enhances mitochondrial oxidative capacity in muscle and liver.
Improved mitochondrial function increases fatty acid oxidation.
This helps the body burn circulating fatty acids rather than storing them in the liver.
Result: Reduced intrahepatic fat and improved metabolic efficiency.
3. Muscle Contraction – Myokine Signaling
Contracting skeletal muscle releases signaling molecules called myokines.
Important myokines include irisin and IL-6.
These molecules communicate with the liver and other organs.
They help reduce inflammation and inhibit activation of hepatic stellate cells, which drive fibrosis.
Result: Protection against liver scarring and disease progression.
4. Exercise-Induced Metabolism – Bile Acid and FXR Pathway
Physical activity alters bile acid composition in circulation.
These changes activate the farnesoid X receptor (FXR) signaling pathway.
FXR regulates genes involved in lipid metabolism and glucose control.
Activation of this pathway suppresses hepatic lipogenesis (fat production).
Result: Reduced liver fat formation and improved metabolic regulation.
.
Study 1: Resistance Training's Systemic Impact on MASLD
Medeiros et al. (2025) conducted a systematic review examining resistance training effects in MASLD patients. This foundational research synthesizes multiple clinical trials to provide robust evidence for strength training interventions.
Key Findings:
Resistance training significantly improves liver function markers,
including ALT and AST levels
Muscle building exercises enhance insulin sensitivity and glucose metabolism
Consistent resistance exercise programs reduce hepatic fat content
Effects are independent of significant weight loss, suggesting direct metabolic benefits
Key Takeaway: You don't need to dramatically lose weight to improve liver health. Engaging in resistance training triggers metabolic improvements at the cellular level, making this an accessible intervention even for those with modest weight changes.
This research supports integrating strength training 2-3 times weekly as a primary therapeutic tool, not just a supplement to other treatments.
Study 2: Aerobic vs. Resistance Exercise—Which Works Best?
Shi et al. (2025) directly compares aerobic exercise and resistance exercise in non-alcoholic fatty liver disease (NAFLD) patients, examining a fascinating aspect: bile acid profiles.
Key Findings:
Resistance exercise produces superior improvements in liver enzyme levels and hepatic steatosis
Aerobic training excels at improving cardiovascular health and systemic inflammation
Bile acid metabolism improves significantly with both modalities, but through different pathways
Combined aerobic and resistance training yields superior outcomes compared to either alone
Key Takeaway: This is the head-to-head comparison you've been waiting for. Resistance training appears particularly effective for direct liver
improvement, while aerobic exercise provides crucial metabolic and cardiovascular benefits. The ideal approach combines both.
A well-rounded program featuring 2-3 days of resistance training plus 150 minutes weekly of moderate-intensity aerobic activity represents the evidence-based gold standard.
Study 3: The Meta-Analysis Evidence
Chen et al. (2026) published a rigorous systematic review and meta-analysis examining resistance training specifically for metabolic dysfunction-associated steatotic liver disease. Meta-analyses represent the highest level of evidence, pooling data across numerous studies.
Key Findings:
Resistance training consistently reduces intrahepatic fat content across diverse populations
Effect sizes remain robust even when accounting for body weight changes
Progressive resistance training programs show dose-dependent improvements
Benefits emerge within 8-12 weeks of consistent training
Key Takeaway: This meta-analysis eliminates doubt about resistance training's efficacy. Multiple studies consistently show that strength training programs produce measurable improvements in liver health markers and hepatic steatosis reduction.
You can expect meaningful improvements within 2-3 months of initiating a proper resistance training program, providing concrete motivation for consistency.
Study 4: Exercise's Multifaceted Effects
Liu & Liu (2025 examined exercise interventions in subjects with metabolic dysfunction-associated steatotic liver disease, providing a holistic view of how physical activity influences multiple disease mechanisms.
Key Findings:
Physical exercise improves insulin sensitivity and glucose homeostasis
Exercise-induced adaptations enhance mitochondrial function in liver cells
Inflammatory markers (IL-6, TNF-α) decrease with consistent physical activity
Hepatic fibrosis progression slows significantly with exercise intervention
Key Takeaway: Exercise doesn't just reduce fat—it fundamentally restores your liver's cellular function and prevents disease progression toward cirrhosis. The benefits extend to improved insulin handling and reduced systemic inflammation.
Regular exercise essentially applies the brakes on disease progression, making it perhaps the most important preventive intervention available outside of medication.
Study 5: Dietary Synergy and Long-Term Prevention
Rajewski et al. (2025) synthesized evidence on how dietary interventions and physical activity work synergistically in MASLD prevention and treatment.
Key Findings:
Combined dietary modification and structured physical activity produce superior outcomes versus either alone
Resistance training enhances the benefits of calorie restriction without requiring extreme dieting
Lifestyle interventions can reverse early-stage fibrosis and prevent progression
Long-term adherence to combined exercise and nutrition programs provides sustained benefits
Key Takeaway: The magic happens at the intersection of exercise and nutrition. Physical activity allows more flexibility with dietary approaches while still achieving substantial liver health improvements.
You don't need perfection in either domain—consistent, moderate exercise combined with reasonable nutritional choices creates powerful, sustainable results.
Synthesizing the Evidence: What These Five Studies Tell Us
All five studies converge on several critical insights about exercise therapy for MASLD:
1. Resistance Training is Specifically Effective. Strength training and resistance exercise demonstrate particular efficacy for hepatic fat reduction and liver enzyme improvement. This isn't coincidental—resistance training triggers metabolic adaptations specifically addressing MASLD pathophysiology.
2. Timeline Matters Research shows meaningful improvements within 8-12 weeks, with continued benefits accumulating over months. This realistic timeline helps maintain motivation.
3. Multifaceted Benefits Exercise interventions improve not just liver markers but also insulin sensitivity, inflammatory profiles, and cardiovascular health—addressing multiple metabolic dysfunction components simultaneously.
4. Combination Approach Wins. The convergent evidence strongly supports combining resistance training (2-3×/week) with aerobic activity (150 min/week) alongside reasonable dietary modifications for optimal outcomes.
5. Disease Reversal is Possible. These studies demonstrate that MASLD isn't necessarily progressive. Active intervention through structured exercise programs can reverse disease processes and prevent fibrosis progression.
Implementing an Exercise Program for MASLD: Practical Guidance
Resistance Training Protocol
Frequency: 2-3 sessions weekly with recovery days between Duration: 30-45 minutes per session Intensity: Moderate to vigorous (maintaining ability to speak but not sing) Components:
Major compound movements (squats, deadlifts, chest press, rows)
Progressive overload (gradually increasing weight/resistance)
Full-body engagement across sessions
Aerobic Exercise Protocol
Frequency: 5 days weekly or 150 minutes moderate-intensity weekly Options: Brisk walking, cycling, swimming, elliptical training Intensity: 50-70% maximum heart rate or perceived exertion of 5-6/10
Getting Started Safely
Before initiating any exercise program, consult your healthcare provider, particularly if you have advanced liver fibrosis or cirrhosis. Start conservatively and progress gradually over 4-6 weeks. Consistency matters far more than intensity.
Frequently Asked Questions (FAQs)
Q: Can exercise actually reverse MASLD?
A: Yes, according to multiple 2025-2026 studies, structured exercise programs can reduce hepatic steatosis and normalize liver function markers. Early-stage disease shows particularly good reversibility. However, advanced fibrosis or cirrhosis may require additional interventions alongside physical activity.
Q: How much weight do I need to lose to see liver improvement?
A: This is excellent news: resistance training improves liver function even without substantial weight loss. Chen et al. (2026) specifically noted that benefits persist independent of major weight changes, suggesting metabolic improvements at the cellular level exceed what weight loss alone would predict.
Q: What if I've never done resistance training before?
A: Medeiros et al. (2025) found benefits across fitness levels. Start with bodyweight exercises, resistance bands, or light weights. Consider working with a trainer for proper form initially. Progressive adaptation occurs within weeks.
Q: Is aerobic or resistance training better?
A: Shi et al. (2025) found that resistance training specifically improves liver markers while aerobic exercise excels at cardiovascular benefits. The strongest evidence supports combining both approaches.
Q: How long before I see results?
A: Liu & Liu (2025) document meaningful improvements within 8-12 weeks. However, some metabolic improvements at the cellular level occur within 2-3 weeks, even before laboratory markers shift.
Q: Can I still exercise if I have advanced liver disease?
A: This requires individualized medical guidance. While exercise is generally beneficial, advanced cirrhosis may necessitate modifications. Always consult your hepatologist before starting new exercise programs.
Q: Do I need to change my diet while exercising?
A: Rajewski et al. (2025) show that exercise combined with modest dietary modifications outperforms either alone. You don't need extreme restriction—moderate, sustainable nutritional changes paired with regular exercise create optimal results.
Q: What if I don't have time for both aerobic and resistance training?
A: While combined training is optimal, resistance training specifically shows robust liver-protective effects. If time-limited, prioritizing resistance training 2-3×/week still provides substantial benefits based on Chen et al. (2026).
Q: Are there medications that should replace exercise?
A: Current MASLD management emphasizes lifestyle interventions as first-line. While some medications may complement exercise programs, none yet match the multifaceted benefits of structured physical activity. Exercise is not optional—it's foundational.
Q: Can exercise prevent MASLD if I'm at risk?
A: Yes. Rajewski et al. (2025) specifically address MASLD prevention. Regular physical activity combined with reasonable dietary choices prevents disease development in at-risk populations, making it valuable both therapeutically and preventively.
Call to Action: Transform Your Liver Health Today
You now understand what research from 2025-2026 reveals: metabolic dysfunction-associated steatotic liver disease is not a life sentence. You have agency. The evidence is clear, the timeline is achievable, and the benefits extend far beyond liver markers.
Your Next Steps:
Schedule a consultation with your healthcare provider to discuss starting an exercise program appropriate for your current health status.
Start small and sustainable: Choose one form of resistance training and one aerobic activity you actually enjoy. Sustainability beats perfection.
Track progress: Monitor how you feel, energy levels, and—after 8-12 weeks—request updated liver function tests to document improvements.
Build community: Consider finding an exercise buddy or joining a fitness community for accountability and motivation.
Make it a priority: Schedule exercise like medical appointments. Consistency over 12 weeks creates metabolic changes worth celebrating.
Combine with nutrition: Work with a dietitian to implement modest, sustainable dietary modifications that complement your exercise program.
Your liver has remarkable regenerative capacity. By implementing the evidence-based strategies outlined in these five landmark studies, you're not just treating a disease—you're reclaiming your metabolic health and long-term wellbeing.
Author’s Note
Metabolic dysfunction–associated steatotic liver disease (MASLD) represents one of the fastest-growing chronic liver conditions worldwide, yet it remains widely misunderstood and often underestimated in clinical practice. For many years, management strategies focused almost exclusively on weight loss and pharmacologic approaches, despite inconsistent long-term success. The recent shift in terminology from NAFLD to MASLD reflects a deeper scientific understanding: this is fundamentally a metabolic disease, not merely a hepatic one.
The purpose of this article is to translate the most current 2025–2026 scientific evidence into clear, practical insights for clinicians, researchers, and health-conscious readers. The studies cited here—including systematic reviews and meta-analyses—demonstrate that structured exercise, particularly resistance training, directly targets the biological mechanisms driving hepatic steatosis, insulin resistance, inflammation, and fibrosis progression. Importantly, these benefits occur even in the absence of substantial weight loss, challenging long-held assumptions about treatment priorities.
As a physician, I wrote this piece to emphasize that exercise is not an optional lifestyle recommendation, but a foundational therapeutic intervention in MASLD management. When combined with reasonable, sustainable dietary modifications, physical activity offers a powerful, low-cost strategy capable of halting—and in many cases reversing—early disease progression.
This article is intended for educational purposes and does not replace individualized medical care. Patients with advanced liver disease should always consult their healthcare provider before initiating new exercise programs. I hope that this evidence-based synthesis empowers readers to view movement not simply as fitness, but as medicine—one of the most effective tools we currently have to restore metabolic and liver health.
Medical Disclaimer
The information in this article, including the research findings, is for educational purposes only and does not constitute medical advice, diagnosis, or treatment. Before starting a resistance exercise program, you must consult with a qualified healthcare professional, especially if you have existing health conditions (such as cardiovascular disease, uncontrolled hypertension, or advanced metabolic disease). Exercise carries inherent risks, and you assume full responsibility for your actions. This article does not establish a doctor-patient relationship.
Related Articles
Metabolic Plasticity: Epigenetic Adaptations to Calorie Restriction | DR T S DIDWAL
How to Build a Disease-Proof Body: Master Calories, Exercise & Longevity | DR T S DIDWAL
References
Chen, Y., Qiao, X., Zou, X., Xu, K., Gu, Z., Chen, G., & Dai, J. (2026). Resistance training for metabolic dysfunction-associated steatotic liver disease: A systematic review and meta-analysis. Frontiers in Physiology, 16, 1679094. https://doi.org/10.3389/fphys.2025.1679094
Liu, C., & Liu, C. J. (2025). Effects of exercise intervention in subjects with metabolic dysfunction-associated steatotic liver disease. Journal of Obesity & Metabolic Syndrome, 34(3), 239–252. https://doi.org/10.7570/jomes25028
Medeiros, D. G., Ferreira, L. F., Lamp, J. D. S., & Telles da Rosa, L. H. (2025). The impact of resistance training in patients diagnosed with metabolic dysfunction-associated steatotic liver disease: A systematic review. European Journal of Gastroenterology & Hepatology, 37(2), 129–136. https://doi.org/10.1097/MEG.0000000000002887
Rajewski, P., Cieściński, J., Rajewski, P., Suwała, S., Rajewska, A., & Potasz, M. (2025). Dietary interventions and physical activity as crucial factors in the prevention and treatment of metabolic dysfunction-associated steatotic liver disease. Biomedicines, 13(1), 217. https://doi.org/10.3390/biomedicines13010217
Shi, J., Cui, J., Zheng, T., Han, X., Wang, B., Wang, W., Zhu, C., Fang, C., Zhou, X., Cong, N., & Yin, X. (2025). Comparative effects of aerobic and resistance exercise on bile acid profiles and liver function in patients with non-alcoholic fatty liver disease. BMC Gastroenterology, 25, Article 239. https://doi.org/10.1186/s12876-025-03826-x