Why Your Metabolism Slows Down: The Hidden Role of Mitochondrial Dysfunction in Obesity
Discover how mitochondrial dysfunction drives obesity, insulin resistance, and metabolic disease. Learn how excess nutrition damages cellular energy systems, increases oxidative stress, and disrupts metabolism—plus evidence-based strategies to restore mitochondrial health and improve long-term metabolic outcomes.
OBESITY
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/27/202617 min read
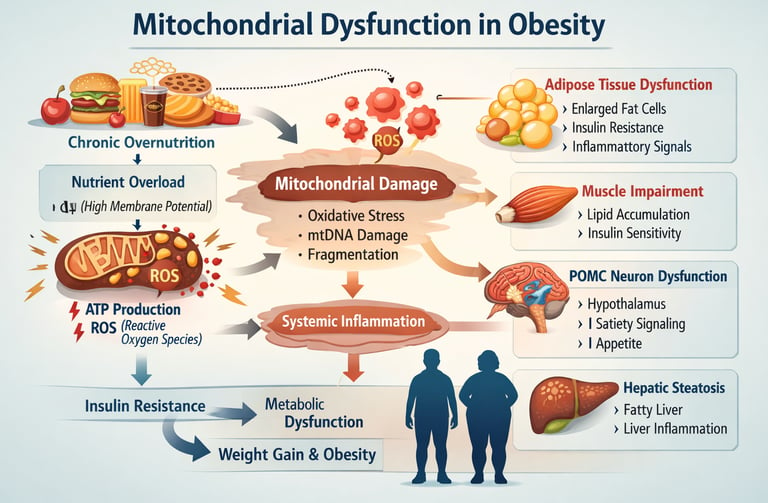
Obesity isn’t just about calories—it begins when your mitochondria lose control of energy, inflammation, and hunger signals. Mitochondrial dysfunction in obesity occurs when excess nutrients overwhelm cellular energy systems, increasing reactive oxygen species (ROS) and impairing ATP production. This leads to insulin resistance, inflammation, and disrupted appetite regulation via POMC neurons, making obesity a disease of cellular energy failure rather than just excess calorie intake.
Obesity begins at the cellular level—when mitochondria fail, metabolism follows.
Your weight is regulated not just by calories, but by how efficiently your cells produce energy.
Mitochondrial dysfunction transforms excess nutrition into inflammation and insulin resistance.
When cellular energy systems break down, fat storage and hunger signals spiral out of control.
Obesity is not just overeating—it is impaired energy metabolism driven by mitochondrial stress.
Clinical pearls.
1. The "Nutrient Flood" Paradox
Scientific Tone: Chronic overnutrition induces a state of "electron pressure" where the mitochondrial membrane potential (Delta psi) becomes excessively high, leading to mandatory electron leak and a surge in superoxide (ROS) production rather than efficient ATP synthesis.
Think of your mitochondria like a small campfire. If you throw a whole tree on it at once, you don't get a bigger, better fire; you get a massive amount of smoke (oxidative stress) and the fire actually struggles to stay lit.
2. Fragmentation vs. Efficiency (The RalA Mechanism)
Scientific Tone: Obesity-induced activation of the RalA protein in white adipose tissue triggers excessive mitochondrial fission via DRP1 recruitment, resulting in fragmented organelles with reduced oxidative capacity.
Healthy mitochondria like to stay connected in a long, powerful network. In obesity, a specific molecular "scissors" (RalA/DRP1) snips them into tiny, weak pieces. These smaller fragments are less efficient at burning fat and harder for the body to repair.
3. The Brain's Energy "Thermostat"
Scientific Tone: Mitochondrial dysfunction in POMC neurons within the hypothalamus disrupts satiety signaling, suggesting that the inability to lose weight is partially driven by an organelle-level failure in the brain's appetite-regulation circuitry.
Your brain has a built-in "fullness" sensor. When the power plants in those specific brain cells fail, the sensor stops working. This means that feeling hungry isn't always about "willpower"—it’s a signal that your brain's energy factories need support.
4. Muscle "Junk" and Insulin Resistance
Scientific Tone: Incomplete $\beta$-oxidation of fatty acids leads to the accumulation of intramyocellular ceramides, which inhibit the insulin signaling pathway by phosphorylating IRS-1 at inhibitory serine sites.
When muscle mitochondria can't finish "burning" the fat they take in, they leave behind toxic metabolic "trash" (ceramides). This trash acts like gum in a lock, preventing insulin from opening the door for sugar to enter your cells.
5. mtDNA as an Internal "Infection"
Scientific Tone: The leakage of mitochondrial DNA (mtDNA) into the cytosol due to failed mitophagy acts as a DAMP (Damage-Associated Molecular Pattern), activating the NLRP3 inflammasome and driving systemic metabolic inflammation.
Because mitochondria look a lot like ancient bacteria to our immune system, when they leak their DNA, the body thinks it is under attack. This triggers a "false alarm" of inflammation throughout the body that makes weight loss even harder.
6. Exercise as the Ultimate "Factory Upgrade"
Scientific Tone: Physical activity is the most potent biological trigger for PGC-1α, the master regulator of mitochondrial biogenesis, which simultaneously increases mitochondrial density and improves the quality of the existing pool through enhanced mitophagy.
Exercise is the only "medicine" that forces your body to build brand-new, high-efficiency power plants. It doesn't just burn calories while you're moving; it upgrades your cells' machinery so you burn energy more cleanly even while you're resting.
When the Powerhouse Fails: Mitochondrial Dysfunction and the Biology of Obesity
You have likely heard the phrase: mitochondria are the “powerhouses” of the cell. But in modern metabolic science, that definition is no longer sufficient. Mitochondria are not just energy generators—they are central regulators of metabolic health, integrating nutrient signals, controlling oxidative stress, and determining how efficiently the body stores or burns energy. When these cellular engines begin to fail, the consequences extend far beyond fatigue. They reshape the biology of obesity itself.
Emerging evidence from 2024–2025 reframes obesity as a condition driven, in part, by mitochondrial dysfunction rather than simple caloric excess. Chronic overnutrition floods the mitochondrial electron transport chain with substrates, elevating membrane potential and increasing the leakage of electrons that form reactive oxygen species (ROS). Over time, this oxidative stress damages mitochondrial DNA, impairs ATP production, and reduces metabolic efficiency (Tung et al., 2024; Marino et al., 2025).
Critically, this dysfunction is not uniform across the body. In adipose tissue, it promotes hypertrophy and inflammation; in skeletal muscle, it contributes to lipid accumulation and insulin resistance; and in the liver, it accelerates fat deposition and metabolic stress (Das et al., 2024; Xu et al., 2025). Perhaps most striking is the emerging role of the brain. Dysfunction within POMC neurons—key regulators of appetite and energy balance—has been shown to impair satiety signaling, biologically reinforcing weight gain (Luo et al., 2025).
This evolving framework challenges the traditional narrative of obesity as a failure of willpower. Instead, it highlights a cellular energy crisis, where excess nutrients paradoxically reduce the body’s ability to generate usable energy. Understanding this shift is essential—not only for clinicians and researchers, but for anyone seeking to address the root causes of metabolic disease.
How Mitochondria Produce Energy: The Science of OXPHOS
Mitochondria are the metabolic engines of your cells, responsible for generating adenosine triphosphate (ATP)—the primary energy currency that powers every biological process, from muscle contraction to hormone synthesis. This energy is produced through a highly efficient process called oxidative phosphorylation (OXPHOS).
During OXPHOS, electrons derived from nutrients move through the electron transport chain (ETC)—a series of protein complexes embedded in the inner mitochondrial membrane. As electrons flow through this chain, protons are pumped across the membrane, creating an electrochemical gradient known as the proton motive force (Δp). This gradient is the driving force behind ATP production.
The Formula for Cellular Energy Efficiency
\Delta p = \Delta \psi - 60,\Delta pH
Δp (Proton Motive Force): Total energy available for ATP synthesis
Δψ (Membrane Potential): Electrical gradient across the membrane
ΔpH: Chemical gradient (proton concentration difference)
This equation represents the core of human metabolism—how efficiently your body converts nutrients into usable energy.
The Obesity Connection: When Energy Production Backfires
In states of chronic overnutrition, mitochondria are flooded with excess fuel. This drives Δψ (membrane potential) to abnormally high levels, overwhelming the ETC. As a result, electrons “leak” and react with oxygen to form reactive oxygen species (ROS)—a key driver of oxidative stress and metabolic damage.
This phenomenon explains a critical paradox:
More calories do not equal more energy—instead, they reduce metabolic efficiency.
What Is Mitochondrial Dysfunction? (A 3-Pillar Framework)
Mitochondrial dysfunction is not a single defect—it is a multidimensional breakdown of cellular energy systems. Understanding these three pillars is essential for reversing metabolic disease:
1. Impaired Biogenesis (Reduced Mitochondrial Production)
Your body fails to produce enough new mitochondria due to suppression of PGC-1α, leading to reduced energy capacity and metabolic slowdown.
2. Impaired Mitophagy (Defective Recycling System)
Damaged mitochondria are not cleared efficiently. Dysfunction in the PINK1–Parkin pathway leads to accumulation of defective organelles, triggering inflammation via the NLRP3 inflammasome.
3. Altered Dynamics (Mitochondrial Fragmentation)
Instead of forming efficient networks, mitochondria undergo excessive fission, becoming fragmented and less functional—often driven by RalA-mediated pathways.
Why This Matters for Weight Loss and Metabolic Health
When mitochondria are dysfunctional, your body loses metabolic flexibility—the ability to switch between burning carbohydrates and fat. This leads to fat accumulation, insulin resistance, and reduced energy output.
Fixing metabolism is not just about eating less—it is about restoring mitochondrial function. Evidence-based strategies such as Zone 2 exercise, time-restricted eating, and thermal stress (cold/heat exposure) directly target mitochondrial biogenesis, efficiency, and repair.
How Chronic Overnutrition Damages Mitochondria and Slows Metabolism
The Nutrient Overload Problem: When Cells Receive Too Much Fuel
Chronic overnutrition—defined as sustained intake of excess calories—creates a metabolic bottleneck at the mitochondrial level. Instead of efficiently converting nutrients into energy, mitochondria become overloaded with glucose and fatty acids. These substrates flood the electron transport chain (ETC), driving the mitochondrial membrane potential (Δψ) to abnormally high levels.
Once this threshold is exceeded, electrons begin to escape the ETC prematurely. Rather than completing oxidative phosphorylation, they react with oxygen to form reactive oxygen species (ROS)—particularly superoxide radicals, which are potent drivers of oxidative stress.
While ROS play essential roles in normal cellular signaling, chronic overproduction in obesity leads to cumulative damage. This includes:
Mitochondrial DNA (mtDNA) mutations
Lipid peroxidation of mitochondrial membranes
Dysfunction of ETC protein complexes
Over time, this creates a state where the energy-producing machinery damages itself under metabolic pressure.
The Overload Paradox: Why More Calories Reduce Energy Efficiency
A key concept in modern metabolic science is the “overload paradox.”
Excess fuel does not increase energy production—instead, it reduces mitochondrial efficiency while accelerating cellular damage.
This paradox explains why individuals with obesity often experience low energy levels despite high caloric intake, reflecting impaired ATP generation rather than nutrient deficiency.
Mitochondrial Fragmentation: Structural Breakdown in Obesity
Mitochondria are not static structures—they continuously undergo fusion (joining) and fission (splitting) to maintain quality control and adapt to energy demands. In metabolically healthy states, fusion predominates, forming elongated and efficient mitochondrial networks.
In obesity, this balance shifts toward excessive fission, leading to fragmented, dysfunctional mitochondria. A pivotal 2024 study demonstrated that activation of the protein RalA in adipose tissue drives this process by stimulating DRP1, the key regulator of mitochondrial division. The result is a fragmented mitochondrial population that is:
Less efficient at ATP production
More prone to ROS generation
Harder to clear via mitophagy
The Collapse of Mitochondrial Biogenesis
Mitochondrial renewal depends on PGC-1α, the master regulator of mitochondrial biogenesis. This protein coordinates the expression of genes required to build new mitochondria and maintain cellular energy capacity.
In obesity, PGC-1α signaling is suppressed due to chronic inflammation and oxidative stress. This leads to:
Reduced mitochondrial number
Declining ATP production
Loss of metabolic flexibility
Importantly, human population studies confirm that reduced mitochondrial DNA copy number—a direct marker of impaired biogenesis—is strongly associated with increased cardiometabolic risk.
Why This Matters for Metabolic Health
The combined effects of nutrient overload, oxidative stress, mitochondrial fragmentation, and impaired biogenesis create a self-reinforcing cycle of metabolic dysfunction. This cycle underlies key features of obesity, including insulin resistance, fatigue, and progressive weight gain.
Chronic overnutrition does not simply add excess energy—it reprograms mitochondrial function, reducing efficiency and increasing cellular damage. Breaking this cycle requires targeted strategies that restore mitochondrial balance, not just calorie restriction.
It Is Not the Same Everywhere: Tissue-Specific Breakdown
One of the most important and underappreciated aspects of mitochondrial dysfunction in obesity is that it does not affect all tissues equally. Different organs face different challenges — and understanding these distinctions is key to understanding why obesity is such a complex, multisystem disease.
White Adipose Tissue
Mitochondrial failure leads to fat cell swelling, local oxygen shortage (hypoxia), and loss of beneficial "beige" fat cells that burn calories as heat.
Skeletal Muscle
Incomplete fatty acid burning creates toxic lipid intermediates (ceramides, DAGs) that directly block insulin signaling pathways.
The Liver
Initial mitochondrial adaptation eventually fails, driving the progression from fatty liver to NAFLD/NASH and increasing systemic lipid overflow.
The Brain (POMC Neurons)
Hypothalamic neurons that suppress appetite lose mitochondrial function, undermining the body's most fundamental hunger-regulation system.
White Adipose Tissue: Where Fat Storage Goes Wrong
Fat cells — adipocytes — are not passive storage units. They are metabolically active, hormone-secreting cells whose proper function depends critically on healthy mitochondria. Research by Xu and colleagues (2025) in Acta Biochimica et Biophysica Sinica demonstrates that mitochondrial dysfunction during adipocyte differentiation actively disrupts the maturation of healthy fat cells, pushing the system toward adipocyte hypertrophy (abnormal enlargement) and inflammatory signaling.
A particularly important consequence is the loss of "beige" adipocytes — a specialized subtype of fat cell that can generate heat by uncoupling the proton gradient from ATP synthesis (a process called thermogenesis). In obesity, the conditions needed to maintain or recruit beige adipocytes are systematically dismantled by mitochondrial fragmentation and ROS, eliminating a meaningful calorie-burning mechanism.
Skeletal Muscle: The Insulin Resistance Factory
Skeletal muscle is responsible for the majority of glucose uptake in the body after a meal. When mitochondria in muscle cells cannot fully oxidize fatty acids, the incomplete breakdown products — ceramides and diacylglycerols (DAGs) — accumulate inside muscle fibers. These lipid intermediates are directly toxic to insulin signaling, phosphorylating IRS-1 at inhibitory sites and blocking the glucose uptake cascade. This is one of the most well-characterised cellular mechanisms linking mitochondrial dysfunction to systemic insulin resistance.
The Brain's Role: A Surprise Contributor
Perhaps the most provocative recent finding comes from the brain. Luo and colleagues (2025) published research in Biochimica et Biophysica Acta (Molecular Basis of Disease) demonstrating that POMC neurons — specialized hypothalamic neurons that are the brain's primary appetite-suppression circuit — are exquisitely sensitive to mitochondrial dysfunction. When these neurons lose mitochondrial integrity, they fail to fire appropriately in response to satiety signals. The result is not just a matter of willpower or habit — it is a hardwired change in the brain's energy-sensing circuitry that actively promotes continued weight gain. This finding fundamentally challenges the narrative that obesity is simply a behavioral problem.
"Mitochondrial dysfunction in POMC neurons contributes to the regulation of obesity, suggesting that the brain's energy-sensing machinery is itself a target of metabolic disease — not merely a bystander."Luo et al., 2025
The Vicious Cycle: Inflammation, Damaged DNA, and Cellular Chaos
Mitochondrial dysfunction does not stay confined to energy metabolism. It triggers a cascade of inflammatory events that makes the entire metabolic situation progressively worse — creating a self-reinforcing vicious cycle that is difficult to interrupt without deliberate intervention.
When the Cleanup System Fails: Mitophagy
Cells have a dedicated quality-control process for damaged mitochondria called mitophagy — a selective form of autophagy where the cell identifies, tags, and recycles dysfunctional mitochondria. The primary pathway runs through two proteins: PINK1 and Parkin. In obesity, this system becomes overwhelmed and impaired, leading to an accumulation of damaged mitochondria that continue to generate ROS and leak their contents into the cell.
Mitochondrial DNA as a Danger Signal
One of the most alarming consequences of mitophagy failure is the release of mitochondrial DNA (mtDNA) into the cytoplasm and eventually the bloodstream. Because mtDNA has bacterial evolutionary origins, the immune system recognizes it as a danger-associated molecular pattern (DAMP) — effectively treating it like a sign of bacterial invasion. This triggers activation of the NLRP3 inflammasome, one of the most potent drivers of chronic low-grade inflammation associated with obesity and metabolic syndrome.
The comprehensive review by Marino and colleagues (2025) in Biomolecules elegantly maps this progression — from initial mitochondrial impairment in peripheral tissues through to neuroinflammation in the central nervous system, demonstrating that the inflammatory consequences of mitochondrial dysfunction are not local but systemic.
Depot Matters: Not All Fat Is Created Equal
An increasingly important refinement in this field is the recognition that mitochondrial dysfunction does not manifest uniformly across different fat depots. Subcutaneous fat (under the skin), visceral fat (around organs), and brown adipose tissue all have distinct mitochondrial profiles — and their metabolic consequences differ substantially.
Das and colleagues (2024), writing in the International Journal of Molecular Sciences, provide a detailed comparison of adipocyte mitochondrial function across fat depots in both obesity and type 2 diabetes. Their analysis highlights that visceral adipose tissue — the depot most strongly associated with cardiometabolic risk — shows the most pronounced mitochondrial dysfunction, including the greatest reductions in oxidative capacity and the highest rates of ROS production. This depot-specific understanding is beginning to shape how researchers think about therapeutically targeting fat tissue mitochondria.
How to Fix Mitochondrial Dysfunction: Practical Applications
The good news? Research from 2024 and 2025 shows that mitochondrial health is remarkably plastic. You can "reboot" your cellular energy production. Here are the most effective, science-backed levers to restore your metabolism.
1. Prioritize Exercise for Mitochondrial Biogenesis
Physical activity is the most powerful "drug" for your cells. It triggers PGC-1α, the master switch that tells your body to build brand-new mitochondria.
The Protocol: Aim for 150 minutes of moderate-intensity aerobic exercise per week combined with two days of resistance training.
The Benefit: Increases mitochondrial density and clears out cellular "junk" (mitophagy).
2. Implement Time-Restricted Eating (TRE)
Chronic overnutrition overwhelms your mitochondria. Giving your system a break allows your cells to switch from "storage mode" to "repair mode."
The Protocol: Maintain a consistent 10-to-12-hour eating window (e.g., 8:00 AM to 6:00 PM).
The Science: Fasting activates AMPK and Sirtuins, enzymes that repair damaged DNA and improve insulin sensitivity.
3. Adopt an Anti-Inflammatory, Pro-Mitochondrial Diet
What you eat provides the building blocks for mitochondrial membranes.
Focus On: Omega-3 fatty acids (fatty fish), polyphenols (berries, green tea, dark chocolate), and magnesium (leafy greens).
The Goal: These nutrients reduce ROS (Reactive Oxygen Species) and prevent the "leakage" of mitochondrial DNA that causes systemic inflammation.
4. Optimize Sleep for Cellular Cleanup
Sleep isn't just for your brain; it’s for your bioenergetics. During deep sleep, your body performs mitophagy—the process of recycling broken mitochondria.
The Protocol: Prioritize 7–9 hours of quality sleep.
The Risk: Chronic sleep deprivation raises oxidative stress and mimics the mitochondrial profile of a high-fat diet.
5. Leverage Thermal Stress (Sauna and Cold Plunge)
Extreme temperatures act as a "hormetic stressor," making your mitochondria more resilient.
Heat (Sauna): Activates heat shock proteins that protect mitochondrial folding.
Cold Exposure: Promotes the recruitment of beige fat, a specialized type of adipose tissue that burns calories specifically to generate heat.
6. Use Targeted Supplementation (Consult a Physician)
While lifestyle is the foundation, specific "mitoceuticals" can support the Electron Transport Chain (ETC).
Top Candidates: CoQ10 (a vital electron carrier), NAD+ precursors (like NMN or NR), and MitoQ.
Note: Always check with a healthcare provider to ensure these don't interfere with existing medications.
7. Monitor Key Metabolic Biomarkers
You cannot manage what you do not measure. Use these clinical markers as a "proxy" for your mitochondrial health:
Fasting Insulin & Glucose: Markers of metabolic flexibility.
Triglycerides: High levels often indicate impaired fat oxidation.
Waist Circumference: Visceral fat is the most significant indicator of mitochondrial fragmentation.
The Bottom Line
If you choose only one intervention, make it consistent movement. Exercise remains the "gold standard" for reversing mitochondrial dysfunction and reclaiming your metabolic health.
Editorial: Mitochondrial Dysfunction and the Biology of Obesity
Obesity is no longer a disease of excess calories alone—it is a disease of cellular energy failure. At the center of this paradigm shift lies the mitochondrion, whose dysfunction reframes obesity as a disorder of impaired energy processing rather than simple energy imbalance.
Chronic overnutrition creates a bioenergetic bottleneck. Excess substrates flood the electron transport chain, elevating membrane potential (Δψ) beyond physiological thresholds, leading to electron leak and overproduction of reactive oxygen species (ROS). This marks the transition from efficient ATP generation to oxidative stress–driven damage.
The “overload paradox” defines modern metabolic disease. More fuel does not translate into more usable energy; instead, it degrades mitochondrial efficiency, damages mtDNA, and impairs oxidative phosphorylation—creating a state of cellular energy insufficiency despite caloric excess.
Mitochondrial dysfunction is tissue-specific and systemically integrated. In adipose tissue, it drives hypertrophy and inflammation; in skeletal muscle, it promotes lipid-induced insulin resistance; in the liver, it accelerates steatosis; and in the hypothalamus—particularly within POMC neurons—it disrupts satiety signaling, biologically reinforcing weight gain.
Failure of mitochondrial quality control amplifies disease progression. Impaired mitophagy (via PINK1–Parkin pathways) leads to accumulation of damaged mitochondria, while released mitochondrial DNA activates the NLRP3 inflammasome, linking bioenergetic failure to chronic low-grade inflammation.
At the molecular level, suppression of PGC-1α represents a critical inflection point. Reduced mitochondrial biogenesis prevents recovery, locking cells into a state of declining metabolic flexibility.
Therapeutically, mitochondria are not passive victims—they are modifiable targets. Exercise remains the most potent activator of mitochondrial biogenesis and oxidative capacity, while fasting-related pathways (AMPK, SIRT1) restore quality control mechanisms.
The clinical implication is profound: obesity must be approached as a systems-level mitochondrial disease. Effective management requires restoring energy flux, not merely restricting caloric intake.
Future medicine will likely target mitochondrial networks directly, integrating pharmacology, lifestyle, and precision biomarkers to reverse metabolic dysfunction at its cellular roots.
Frequently asked questions regarding mitochondrial dysfunction in obesity
1. Is mitochondrial dysfunction a primary driver or a secondary consequence of adipose tissue expansion?
The relationship is bidirectional. While chronic overnutrition (nutrient surplus) initially drives mitochondrial stress through electron transport chain (ETC) overload and ROS production, recent evidence—specifically the 2025 findings regarding POMC neurons—suggests that pre-existing or early-stage mitochondrial impairment in the hypothalamus can compromise satiety signaling, thereby accelerating the behavioral drivers of obesity.
2. How does the "proton motive force" (Delta p) contribute to cellular damage during chronic overfeeding?
In a state of nutrient excess, the delivery of NADH and FADH2 to the ETC exceeds the cell's ATP demand. This leads to an abnormally high membrane potential (Delta psi). When Delta psi exceeds a critical threshold, the movement of electrons through the complexes slows down, increasing the half-life of reduced intermediates (like ubisemiquinone). This facilitates the direct one-electron reduction of oxygen to superoxide initiating a cascade of oxidative damage.
3. What is the role of the RalA protein in obesity-related mitochondrial morphology?
Research (Xia et al., 2024) has identified RalA as a key molecular switch activated by obesity in white adipose tissue. Once active, RalA recruits the fission protein DRP1 to the outer mitochondrial membrane. This shifts the dynamic equilibrium away from fusion and toward fragmentation, resulting in smaller, spherical mitochondria that exhibit reduced respiratory capacity and increased susceptibility to apoptosis.
4. Why does mitochondrial dysfunction in skeletal muscle specifically trigger insulin resistance?
When mitochondria fail to achieve complete beta-oxidation of long-chain fatty acids, the process "stalls," leading to the accumulation of intramyocellular acylcarnitines, diacylglycerols (DAGs), and ceramides. These lipid intermediates activate stress-sensitive kinases (e.g., PKC theta, JNK), which catalyze the inhibitory serine phosphorylation of Insulin Receptor Substrate-1 (IRS-1), effectively blunting the GLUT4 translocation response to insulin.
5. How does the release of mitochondrial DNA (mtDNA) exacerbate metabolic inflammation?
Mitochondria are evolutionary endosymbionts with circular DNA containing unmethylated CpG motifs, similar to bacterial DNA. When mitophagy (autophagic clearance) is impaired, damaged mitochondria may rupture, releasing mtDNA into the cytosol. This mtDNA is recognized by cGAS-STING and NLRP3 inflammasome pathways as a Damage-Associated Molecular Pattern (DAMP), triggering the secretion of pro-inflammatory cytokines like IL-1beta
6. Can "metabolic flexibility" be restored through PGC-1α activation?
Yes. PGC-1α is the master coactivator for mitochondrial biogenesis and fat oxidation genes. By activating PGC-1α (via AMPK or SIRT1 pathways), the cell increases its mitochondrial mass and improves the efficiency of the TCA cycle. This restores the cell's ability to switch rapidly between glucose and lipid oxidation—a hallmark of metabolic health that is typically lost in the obese phenotype.
7. What is the clinical significance of the "mitochondrial DNA copy number" in population studies?
The mtDNA copy number (the ratio of mitochondrial DNA to nuclear DNA) serves as a surrogate biomarker for mitochondrial density and biogenesis. Population-level reviews (Tung et al., 2024) consistently show that a lower mtDNA copy number in peripheral blood or adipose tissue is strongly correlated with increased visceral adiposity, higher HOMA-IR scores, and a greater risk for progression to Type 2 Diabetes.
Author’s Note (Clinician’s Perspective)
As a clinician in internal medicine, one of the most important shifts I have witnessed over the past decade is the transition from viewing obesity as a lifestyle issue to recognizing it as a complex, systems-level metabolic disease rooted in cellular dysfunction. Among the many mechanisms involved, mitochondrial health has emerged as a central and unifying theme.
In daily clinical practice, patients rarely present with “mitochondrial dysfunction” as a diagnosis. Instead, they come with type 2 diabetes, fatty liver disease, central obesity, dyslipidemia, or unexplained fatigue. Yet, when we look deeper, these conditions often share a common biological thread: impaired mitochondrial energy handling, increased oxidative stress, and reduced metabolic flexibility.
What is particularly important—and often misunderstood—is that these changes are not merely consequences of obesity, but active drivers of disease progression. Dysfunctional mitochondria in skeletal muscle contribute to insulin resistance; in adipose tissue, they promote inflammation and unhealthy fat expansion; and in the brain, particularly within POMC neurons, they alter appetite regulation in ways that make sustained weight loss biologically difficult.
From a treatment standpoint, this perspective changes everything. It shifts our focus away from short-term calorie restriction toward long-term restoration of metabolic function. Interventions such as structured exercise, sleep optimization, and targeted nutritional strategies are not simply “lifestyle advice”—they are mitochondrial therapies with measurable biochemical effects. Even pharmacological agents increasingly show benefits that intersect with mitochondrial pathways.
Equally important is patient communication. Understanding that their condition is rooted in biological adaptation rather than personal failure often reduces stigma and improves adherence to treatment plans.
Ultimately, addressing obesity requires more than managing weight—it requires restoring the body’s ability to efficiently produce, utilize, and regulate energy at the cellular level. That is where meaningful, lasting clinical outcomes begin. With nutrition guidance, and appropriate monitoring of the biomarkers that matter.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Managing Diabesity: A Complete Guide to Weight Loss and Blood Sugar Control | DR T S DIDWAL
Weight Loss as Immunotherapy: The Science Behind Reversing Chronic Inflammation | DR T S DIDWAL
Your Body Fat Is an Endocrine Organ—And Its Hormones Shape Your Heart Health | DR T S DIDWAL
References
Das, S., Mukhuty, A., Mullen, G. P., & Rudolph, M. C. (2024). Adipocyte mitochondria: Deciphering energetic functions across fat depots in obesity and type 2 diabetes. International Journal of Molecular Sciences, 25(12), 6681. https://doi.org/10.3390/ijms25126681
Luo, X.-d., Tang, S., Luo, X.-y., Luosang, Q., Xia, R.-h., & Wang, X.-W. (2025). Mitochondrial regulation of obesity by POMC neurons. Biochimica et Biophysica Acta (BBA) — Molecular Basis of Disease, 1871(3), 167682. https://doi.org/10.1016/j.bbadis.2025.167682
Marino, F., Petrella, L., Cimmino, F., Pizzella, A., Monda, A., Allocca, S., Rotondo, R., D'Angelo, M., Musco, N., Iommelli, P., Catapano, A., Bagnato, C., Paolini, B., & Cavaliere, G. (2025). From obesity to mitochondrial dysfunction in peripheral tissues and in the central nervous system. Biomolecules, 15(5), 638. https://doi.org/10.3390/biom15050638
Tung, P. W., Thaker, V. V., Gallagher, D., et al. (2024). Mitochondrial health markers and obesity-related health in human population studies: A narrative review of recent literature. Current Obesity Reports, 13, 724–738. https://doi.org/10.1007/s13679-024-00588-7
Xia, W., Veeragandham, P., Cao, Y., Xu, Y., Rhyne, T. E., Qian, J., Hung, C. W., Zhao, P., Jones, Y., Gao, H., Liddle, C., Yu, R. T., Downes, M., Evans, R. M., Rydén, M., Wabitsch, M., Wang, Z., Hakozaki, H., Schöneberg, J., Reilly, S. M., … Saltiel, A. R. (2024). Obesity causes mitochondrial fragmentation and dysfunction in white adipocytes due to RalA activation. Nature Metabolism, 6(2), 273–289. https://doi.org/10.1038/s42255-024-00978-0
Xu, Y., Xie, S., Han, L., Xu, L., & Zhu, Y. (2025). Mitochondrial dysfunction in adipocyte differentiation: Implications for obesity and metabolic syndrome intervention. Acta Biochimica et Biophysica Sinica. Advance online publication. https://doi.org/10.3724/abbs.2025153