Visceral Fat Treatment: GLP-1, Semaglutide & SGLT2 Explained
Struggling with belly fat? Discover how GLP-1 drugs like Semaglutide target dangerous visceral fat and why they work better than diet alone.
OBESITY
Dr. T.S. Didwal, M.D.(Internal Medicine)
4/5/202614 min read
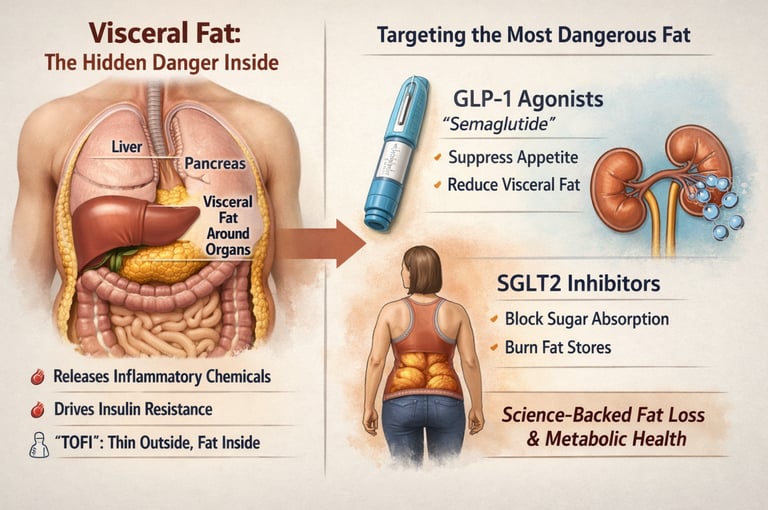
Visceral fat is the dangerous fat stored around internal organs that drives insulin resistance and heart disease. Medications like Semaglutide and Empagliflozin reduce visceral fat by suppressing appetite, improving insulin sensitivity, and increasing fat metabolism—often more effectively than lifestyle changes alone in high-risk patients
Best Medications to Reduce Visceral Fat
GLP-1 receptor agonists (e.g., Semaglutide)
SGLT2 inhibitors (e.g., Empagliflozin)
Dual incretin therapy (e.g., Tirzepatide)
Combination therapy (GLP-1 + SGLT2)
Clinical pearls
1. The "Preferential Loss" Phenomenon
Scientific Perspective: GLP-1 receptor agonists (GLP-1 RAs) do not just cause generalized weight loss; they exhibit a high affinity for reducing Visceral Adipose Tissue (VAT). Imaging substudies from the STEP trials show that while total body weight might drop by 15%, visceral fat can decrease by up to 40%. This suggests that these drugs modulate the metabolic activity of deep fat depots directly, rather than just inducing a caloric deficit.
Don’t be discouraged if the number on the scale isn't moving as fast as you hoped. You are likely losing the "dangerous" fat inside your abdomen—the kind that causes heart disease—much faster than the "pinchable" fat under your skin. Your belt loop is often a better health indicator than your scale.
2. Targeting the TOFI Phenotype
Scientific Perspective: Body Mass Index (BMI) is an insufficient metric for cardiometabolic risk. Patients with the TOFI (Thin Outside, Fat Inside) phenotype may have a normal BMI but carry high levels of ectopic fat in the liver and around organs. Clinicians should use the TG/HDL ratio and waist circumference as primary screening tools for these patients.
You can be "skinny" and still be at risk for type 2 diabetes or fatty liver disease. If you have a "pouch" or a high waist measurement despite a normal weight, your internal health might need the same medical attention as someone with clinical obesity.
3. The Power of "Metabolic Multitasking" (Combination Therapy)
Scientific Perspective: Combining GLP-1 RAs with SGLT2 inhibitors creates a dual-pathway caloric deficit. GLP-1s reduce "energy in" via central satiety, while SGLT2s increase "energy out" via glycosuria (excreting sugar in urine). This synergy not only accelerates fat loss but provides simultaneous protection for the heart and kidneys.
Think of it as a two-front attack: one medicine helps you feel full so you eat less, and the other helps your body flush out excess sugar through your urine. Using them together can provide a "safety net" for your heart and kidneys that one drug alone might not offer.
4. Protecting "The Engine" (Muscle Preservation)
Scientific Perspective: Rapid weight loss induced by potent incretins (like Tirzepatide) carries a risk of sarcopenia (muscle loss). To maintain a healthy Basal Metabolic Rate (BMR) and prevent weight regain, patients must consume 1.2–1.6 g/kg of protein and engage in resistance training to ensure the weight lost is predominantly fat, not lean mass.
When you're on these medications, you'll eat much less. Because you're eating less, every bite counts. You must prioritize protein (like chicken, fish, or beans) and lift weights. If you lose muscle instead of fat, your metabolism will slow down, making it much harder to keep the weight off later.
5. Chronic Management vs. Acute "Fix"
Scientific Perspective: Data from the STEP 4 trial indicate that metabolic dysregulation is a chronic condition. Discontinuing GLP-1 therapy often leads to a reversal of the metabolic benefits and weight regain. Pharmacotherapy should be viewed as a long-term "thermostat adjustment" for the metabolic system rather than a short-term intervention.
These medications are like blood pressure or cholesterol pills—they work as long as you take them. They aren't a "detox" or a quick fix to get ready for an event; they are tools to help your body's biology function correctly for the long haul.
Visceral Fat : The Hidden Cardiovascular Risk
Visceral fat is not just an aesthetic concern—it is a biologically active, disease-driving tissue that sits deep within the abdominal cavity, wrapping vital organs such as the liver, pancreas, and intestines. Unlike subcutaneous fat, which primarily serves as energy storage, visceral adipose tissue (VAT) functions as an endocrine organ, releasing free fatty acids, pro-inflammatory cytokines, and adipokines directly into the portal circulation. This unique anatomical and metabolic positioning allows visceral fat to exert a disproportionate influence on hepatic metabolism, promoting insulin resistance, dyslipidemia, and chronic low-grade inflammation—hallmarks of modern cardiometabolic disease (Drucker, 2025; Liao et al., 2023).
Clinically, this explains a critical paradox: individuals with a “normal” body mass index (BMI) may still harbor significant metabolic risk if they possess excess visceral fat—a phenotype increasingly recognized as “thin outside, fat inside” (TOFI). Traditional metrics such as body weight or BMI often fail to capture this hidden risk, whereas markers like waist circumference, triglyceride-to-HDL ratio, and insulin resistance indices provide a more accurate reflection of visceral adiposity and its downstream consequences (Rosen & Ingelfinger, 2026).
While lifestyle interventions remain foundational, they are not always sufficient—particularly in patients with established insulin resistance, type 2 diabetes, or metabolic dysfunction-associated steatotic liver disease. In this context, pharmacological therapies have emerged as powerful tools that do more than simply reduce weight—they selectively target the most dangerous fat depot. Among these, GLP-1 receptor agonists and SGLT2 inhibitors stand out for their ability to improve metabolic health while reducing visceral fat through distinct but complementary mechanisms (Morciano et al., 2024). Understanding how these therapies work—and who stands to benefit most—marks a fundamental shift from weight-centric to metabolism-centric care.
Key insight: Waist circumference, waist-to-hip ratio, and TG/HDL ratio are far better proxies for visceral fat burden than weight alone. A waist above 80 cm (women) or 94 cm (men) signals elevated visceral fat risk.
Lifestyle modification — diet and exercise — remains the foundation of any treatment plan. But for many patients, particularly those with established insulin resistance, type 2 diabetes, or significant cardiometabolic risk, lifestyle changes alone are insufficient. This is where pharmacotherapy becomes transformative.
GLP-1 Receptor Agonists and Visceral Fat
GLP-1 receptor agonists (GLP-1 RAs) have emerged as the most powerful pharmacological tools we currently have for reducing visceral fat. Originally developed for type 2 diabetes, they have since proven effective across a broader spectrum of metabolic disease.
Key agents
Semaglutide
Weekly injection or daily tablet. Highest efficacy for weight and visceral fat reduction in class.
Liraglutide
Daily injection. Established long-term cardiovascular safety data (LEADER trial).
Dulaglutide
Weekly injection. Good tolerability profile; widely used in type 2 diabetes management.
How they reduce visceral fat — the mechanisms
GLP-1 agonists work through multiple converging pathways. Understanding the mechanism helps clarify why they are so effective:
Central appetite suppression: GLP-1 receptors in the hypothalamus — particularly in POMC neurons — signal satiety and reduce hunger. Patients eat less without feeling deprived.
Delayed gastric emptying: Food leaves the stomach more slowly, extending fullness and blunting post-meal glucose spikes.
Improved insulin sensitivity: By reducing hyperinsulinaemia, these drugs decrease the liver's drive to synthesise new fat (de novo lipogenesis), directly reducing hepatic and visceral fat accumulation.
Preferential visceral fat reduction: MRI-based imaging studies consistently show that GLP-1 RAs reduce visceral fat disproportionately — more than their effect on total body weight or subcutaneous fat would predict. Evidence also suggests GLP-1 receptors exist directly on adipocytes, allowing local fat-remodelling effects (Drucker, 2025).
What the clinical evidence shows
A 2023 systematic review and meta-analysis by Liao et al., pooling data from multiple randomised trials, confirmed that GLP-1 receptor agonists produce statistically significant reductions in visceral fat area and liver fat across populations both with and without type 2 diabetes and NAFLD. Crucially, these reductions were independent of — and greater than — what weight loss alone would predict, indicating a direct metabolic effect on fat depots.
The landmark STEP trials of semaglutide 2.4 mg weekly demonstrated up to 15% total body weight reduction. Imaging substudies showed visceral fat reductions of 25–40%. Alongside this came measurable improvements in insulin resistance (HOMA-IR), high-sensitivity CRP (a marker of systemic inflammation), liver enzymes, and lipid profiles.
15% Body weight loss with semaglutide 2.4 mg (STEP trials)
25–40% Visceral fat reduction on MRI imaging substudies
↓ MACE Major adverse cardiovascular events reduced (SELECT trial)
A comprehensive review published in the New England Journal of Medicine (Rosen & Ingelfinger, 2026) reinforces these findings, noting that GLP-1 RAs reduce cardiovascular events, improve hepatic steatosis, and lower markers of systemic inflammation — effects that extend well beyond glycaemic control.
Bottom line: For patients with significant visceral adiposity, GLP-1 receptor agonists produce meaningful, clinically significant reductions in the most dangerous fat depot — with cardiovascular and hepatic benefits that make a compelling case for early use.
SGLT2 Inhibitors and Visceral Fat
SGLT2 inhibitors work by a completely different mechanism: they block glucose reabsorption in the kidney, causing excess sugar to be excreted in urine. This elegant approach generates a genuine caloric deficit without relying on appetite regulation, making it a complementary tool in visceral fat management.
Key agents
Empagliflozin
Strong cardiovascular and renal outcome data (EMPA-REG, EMPEROR trials). First choice in heart failure.
Dapagliflozin
Broad indication, including chronic kidney disease independent of diabetes (DAPA-CKD trial).
Canagliflozin
Additional bone and cardiovascular data. Also approved for diabetic kidney disease.
How they target visceral fat
Glycosuria-driven caloric deficit: SGLT2 inhibitors cause the kidney to excrete roughly 60–80 g of glucose per day, equivalent to approximately 200–300 calories. This creates a sustained, automatic energy deficit without dietary restriction.
Insulin-lowering: Because glucose is excreted rather than retained, circulating insulin levels fall. Lower insulin shifts the body toward fat oxidation and ketogenesis — a metabolic state that preferentially burns fat stores, including visceral fat.
Ectopic fat reduction: Morciano et al. (2024) comprehensively reviewed SGLT2 inhibitor effects on adipose tissue metabolism, demonstrating measurable reductions in visceral adipose tissue and liver fat (hepatic steatosis) on imaging, alongside improvements in adipokine profiles — including increased adiponectin, the anti-inflammatory fat hormone.
Unique cardiorenal advantages
What sets SGLT2 inhibitors apart is their profound cardiovascular and kidney-protective effects, independent of glucose lowering. They reduce hospitalisations for heart failure, slow chronic kidney disease progression, and lower blood pressure — benefits that make them indispensable for specific patient profiles regardless of visceral fat goals.
3 kg: Average weight loss (modest, but disproportionately visceral)
↓ HF: Heart failure hospitalisations significantly reduced
↓ ALT: Liver fat and ALT reduction in NAFLD patients
Combining GLP-1 and SGLT2: A Synergistic Strategy
Used together, GLP-1 receptor agonists and SGLT2 inhibitors offer complementary and additive benefits. GLP-1 RAs suppress appetite and reduce caloric intake; SGLT2 inhibitors create a parallel caloric deficit through glucose excretion and shift metabolic fuel preference toward fat oxidation.
Clinical studies confirm that combination therapy produces greater reductions in HbA1c, body weight, visceral fat, and blood pressure than either agent alone, with additive cardiovascular risk reduction. For patients with severe insulin resistance, high visceral fat burden, or established cardiometabolic disease, early combination therapy is increasingly considered standard of care rather than a last resort.
Important: Both drug classes require individualised prescribing. GLP-1 agonists carry the risk of gastrointestinal side effects; SGLT2 inhibitors require adequate renal function and carry a small risk of genital infections. Always discuss risks and benefits with your treating physician.
Emerging Therapies: The Next Generation
The pharmacological landscape for visceral fat and obesity is evolving rapidly. Several agents are already transforming practice, while others are in advanced development.
Tirzepatide — dual GIP/GLP-1 agonism
Tirzepatide activates both GLP-1 and GIP (glucose-dependent insulinotropic peptide) receptors simultaneously. The synergy between these two incretin pathways produces weight loss and visceral fat reduction exceeding anything achieved by GLP-1 agonism alone. The SURMOUNT-1 trial demonstrated weight loss up to 22.5% at the highest dose — a magnitude previously achievable only with bariatric surgery.
Tirzepatide
GLP-1 + GIP dual agonist. Up to 22% weight loss. Now approved for obesity (Zepbound) and T2D (Mounjaro).
Retatrutide
Triple agonist (GLP-1 + GIP + glucagon). Phase 3 trials ongoing. May further enhance energy expenditure.
Amylin analogues
Cagrilintide (combined with semaglutide as CagriSema). Phase 3 showing additive fat-loss effects.
As Drucker (2025) outlined in a comprehensive review in Nature Reviews Drug Discovery, the therapeutic pipeline for GLP-1-based therapies now extends far beyond diabetes and obesity, with emerging evidence for benefit in cardiovascular disease, kidney disease, neurodegeneration, and metabolic dysfunction-associated steatohepatitis (MASH).
Comparative Effectiveness at a Glance
1. GLP-1 Receptor Agonists (e.g., Semaglutide)
Weight loss: High (≈10–15%)
Visceral fat reduction: High (≈25–40%)
Insulin sensitivity: Significant improvement
Cardiovascular benefit: Strong (reduction in major adverse cardiovascular events)
2. SGLT2 Inhibitors (e.g., Empagliflozin)
Weight loss: Moderate (≈2–4 kg)
Visceral fat reduction: Moderate
Insulin sensitivity: Moderate improvement
Cardiovascular benefit: Strong (especially for heart failure and chronic kidney disease)
3. Combination Therapy (GLP-1 + SGLT2)
Weight loss: High (additive effect)
Visceral fat reduction: High (additive effect)
Insulin sensitivity: Significant improvement
Cardiovascular benefit: Very strong (combined metabolic + cardiorenal protection)
4. Dual Incretin Therapy (e.g., Tirzepatide)
Weight loss: Very high (≈20–22%)
Visceral fat reduction: Very high
Insulin sensitivity: Marked improvement
Cardiovascular benefit: Emerging evidence (ongoing SURPASS-CVOT)
Who Should Consider These Medications?
Patient selection matters. These medications are not one-size-fits-all, and appropriate prescribing requires consideration of each person's metabolic phenotype, comorbidities, and treatment goals.
Ideal for GLP-1 RA
BMI ≥30, or ≥27 with comorbidity
High visceral fat with low BMI (TOFI)
Type 2 diabetes + central obesity
Elevated TG/HDL ratio
High HOMA-IR (insulin resistance)
NAFLD / hepatic steatosis
Ideal for SGLT2 Inhibitor
Type 2 diabetes + established CVD
Heart failure (HFrEF or HFpEF)
Chronic kidney disease (eGFR ≥20)
Visceral adiposity + hypertension
Hyperinsulinaemia without obesity
Combination therapy candidate
Practical Clinical Algorithm for Visceral Fat Management
1. Establish Baseline & Confirm Visceral Fat Burden
Measure:
Waist circumference
TG/HDL ratio
HOMA-IR
Fasting glucose
Liver enzymes (ALT/AST)
Consider advanced assessment:
DEXA scan
Abdominal imaging (CT/MRI) if available
2. Optimise Lifestyle (Mandatory Foundation)
Nutrition: Protein-adequate, reduced refined carbohydrates
Exercise: ≥150 minutes/week (aerobic + resistance training)
Sleep: Optimise duration and quality
Stress management: Address chronic cortisol elevation
3. Initiate Phenotype-Based Pharmacotherapy
Obesity predominant:
→ Start GLP-1 receptor agonist (e.g., Semaglutide)Type 2 diabetes + CVD / heart failure / CKD:
→ Start SGLT2 inhibitor (e.g., Empagliflozin)Mixed phenotype (obesity + cardiometabolic disease):
→ Consider early combination therapy (GLP-1 + SGLT2)
4. Reassess at 12–16 Weeks
Evaluate response:
≥5% weight reduction
Decrease in waist circumference
Improvement in TG/HDL ratio
If inadequate response:
Escalate dose
Add complementary pharmacotherapy
5. Escalate Therapy if Needed
Consider dual incretin therapy:
Tirzepatide
Emerging options:
Triple agonists (GLP-1 + GIP + glucagon)
Refer to:
Obesity medicine specialist for complex or refractory cases
Key Clinical Principle
Treat visceral fat as a chronic metabolic disease requiring:
Long-term therapy
Continuous monitoring
Integrated lifestyle + pharmacological approach
If you want, I can convert this into:
A flowchart diagram (ideal for your website UX)
Or a clinical decision tree infographic (high engagement + shareability)
Practical Applications: What This Means for You
Understanding the science is valuable. Translating it into your daily life and conversations with your doctor is what matters most. Here are practical takeaways:
Get the right tests. Ask your doctor for a fasting insulin level, TG/HDL ratio, and HOMA-IR calculation — not just a fasting glucose. These reveal insulin resistance and visceral fat risk that BMI misses entirely.
Combine medication with protein-adequate eating. GLP-1 agonists reduce appetite significantly. Without adequate protein intake (1.2–1.6 g/kg body weight), you risk losing lean muscle mass alongside fat. Prioritise protein at every meal.
Exercise is non-negotiable — even on medication. Resistance training preserves muscle and amplifies visceral fat reduction during GLP-1 therapy. Research consistently shows that combining exercise with pharmacotherapy produces better body composition outcomes than either approach alone (Popoviciu et al., 2023).
Monitor the right outcomes. The number on the scale is one data point. Track waist circumference, energy levels, liver enzyme normalisation, and glycaemic markers. These tell a more complete metabolic story.
Long-term commitment matters. Visceral fat accumulated over years cannot be dissolved in weeks. Sustainable medication adherence, combined with lifestyle modification, is the key to durable results. Discuss long-term prescribing plans with your clinician from the outset.
Why This Article Matters — A Clinician’s Perspective
Shift from weight-centric to fat-distribution medicine:
Modern endocrinology is moving beyond BMI toward visceral adiposity as the primary therapeutic target, given its direct role in insulin resistance, atherogenic dyslipidemia, and systemic inflammation (Drucker, 2025; Rosen & Ingelfinger, 2026).Not all weight loss is metabolically equal:
Emerging imaging data show that therapies such as Semaglutide reduce visceral fat disproportionately compared to total body weight, suggesting mechanisms beyond caloric restriction alone (Liao et al., 2023).Controversy clinicians should understand:
While some argue GLP-1–induced fat loss is purely driven by reduced energy intake, MRI-based studies demonstrate preferential VAT reduction, indicating direct or indirect adipose tissue remodeling effects.Case-based insight (real-world relevance):
A 42-year-old male with BMI 24 (TOFI phenotype), elevated TG/HDL ratio, and fatty liver showed significant waist reduction, improved ALT, and reduced HOMA-IR after 24 weeks of GLP-1 therapy—highlighting the limitation of BMI-based risk assessment.Comparative clinical positioning:
GLP-1 RAs → most potent for visceral fat reduction
SGLT2 inhibitors → cardiorenal protection + metabolic shift toward fat oxidation
Combination → additive and phenotype-driven precision therapy
Lean mass preservation — a critical gap:
Rapid weight loss without adequate protein intake or resistance training risks sarcopenia, particularly in older adults. Clinical protocols should target 1.2–1.6 g/kg protein + structured resistance training.Precision prescribing is the future:
Treatment decisions should integrate:Waist circumference
TG/HDL ratio
HOMA-IR
Hepatic biomarkers
rather than relying solely on BMI thresholds.
Key takeaway for clinicians and patients:
You don’t just lose weight with modern therapies—you strategically target the most dangerous fat depot, fundamentally altering long-term cardiometabolic risk.
Frequently Asked Questions
Do GLP-1 drugs reduce visceral fat without dieting?
Yes, GLP-1 receptor agonists like Semaglutide reduce visceral fat by suppressing appetite and lowering caloric intake—even without a formal diet. However, combining medication with a protein-adequate, lower-carbohydrate diet produces significantly better fat loss, preserves muscle mass, and improves long-term metabolic outcomes.
Is semaglutide effective for belly (visceral) fat?
Yes—semaglutide is one of the most effective medications for reducing visceral fat. Clinical trials (STEP program) show 25–40% reduction in visceral fat, which exceeds overall weight loss (~15%), indicating a direct metabolic effect on harmful fat depots, not just calorie restriction.
Which is better for visceral fat: GLP-1 or SGLT2 inhibitors?
It depends on the clinical goal. GLP-1 receptor agonists (e.g., Semaglutide) are more effective for weight and visceral fat loss, while SGLT2 inhibitors (e.g., Empagliflozin) provide moderate fat reduction with strong heart and kidney protection. Combined therapy offers the best overall metabolic benefit.
How long does it take to lose visceral fat on these medications?
Early metabolic improvements appear within 8–12 weeks, but meaningful visceral fat reduction typically requires 3–6 months. Maximum effects with semaglutide are seen around 12–18 months, making this a long-term treatment strategy.
Can non-diabetic patients use GLP-1 drugs for visceral fat?
Yes. Semaglutide (Wegovy) and Liraglutide (Saxenda) are approved for obesity in non-diabetic individuals with BMI ≥30, or ≥27 with comorbidities. For patients with normal BMI but high visceral fat (TOFI phenotype), specialist evaluation is recommended.
What are the side effects of GLP-1 and SGLT2 medications?
GLP-1 drugs commonly cause nausea, vomiting, and diarrhoea, especially early in treatment. SGLT2 inhibitors may cause genital infections and increased urination. Rare risks include gallstones (GLP-1) and euglycaemic ketoacidosis (SGLT2). Proper dose titration and monitoring reduce these risks.
Will visceral fat return after stopping medication?
Yes. Studies show that stopping Semaglutide leads to weight and visceral fat regain, indicating these therapies manage—but do not cure—underlying metabolic dysfunction. Long-term treatment, combined with lifestyle changes, is often necessary for sustained results.
Author’s Note
As a physician, one of the most important shifts I have witnessed over the past decade is the move from weight-focused medicine to metabolic-focused care. In daily practice, I routinely see patients with a “normal” body weight who carry a high burden of visceral fat—and conversely, individuals with higher BMI but relatively preserved metabolic health. This disconnect reinforces a critical point: it is not how much fat a patient carries, but where that fat is distributed, that determines risk.
The emergence of therapies such as Semaglutide and Empagliflozin has fundamentally changed how we approach this problem. These medications are not simply “weight-loss drugs.” They are metabolic modulators that improve insulin sensitivity, reduce ectopic fat, and, importantly, target visceral adiposity—the most pathogenic fat depot. In carefully selected patients, the clinical impact can be transformative, extending beyond glycaemic control to improvements in liver health, cardiovascular risk, and overall metabolic resilience.
However, pharmacotherapy is not a substitute for physiology. The most successful outcomes I see occur when medication is integrated with structured lifestyle interventions—particularly resistance training and adequate protein intake to preserve lean mass during fat loss. Equally important is individualised prescribing, guided by metabolic phenotype, comorbidities, and patient-specific goals rather than a one-size-fits-all algorithm.
There is also a need for caution. These therapies require long-term commitment, monitoring, and patient education. Expectations must be realistic, and treatment plans should be framed as part of a sustained strategy rather than a short-term intervention.
Ultimately, the goal is not simply to reduce body weight, but to redefine metabolic health at its core—targeting the drivers of disease before clinical complications emerge
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Visceral Fat and Cardiovascular Risk: The Hidden Driver of Atherosclerosis
The #1 Diet Strategy to Reduce Visceral Fat According to Latest Research
Why Belly Fat Causes Insulin Resistance: Portal Theory Explained Simply
Obesity and Fatty Liver Disease: What Science Says About Risk and Health | DR T S DIDWAL
References
Drucker, D. J. (2025). GLP-1-based therapies for diabetes, obesity and beyond. Nature Reviews Drug Discovery, 24, 631–650. https://doi.org/10.1038/s41573-025-01183-8
Liao, C., Liang, X., Zhang, X., & Li, Y. (2023). The effects of GLP-1 receptor agonists on visceral fat and liver ectopic fat in an adult population with or without diabetes and nonalcoholic fatty liver disease: A systematic review and meta-analysis. PLOS ONE, 18(8), Article e0289616. https://doi.org/10.1371/journal.pone.0289616
Morciano, C., Gugliandolo, S., Capece, U., et al. (2024). SGLT2 inhibition and adipose tissue metabolism: Current outlook and perspectives. Cardiovascular Diabetology, 23, 449. https://doi.org/10.1186/s12933-024-02539-x
Popoviciu, M. S., Păduraru, L., Yahya, G., Metwally, K., & Cavalu, S. (2023). Emerging role of GLP-1 agonists in obesity: A comprehensive review of randomised controlled trials. International Journal of Molecular Sciences, 24(13), 10449. https://doi.org/10.3390/ijms241310449
Rosen, C. J., & Ingelfinger, J. R. (2026). GLP-1 receptor agonists. New England Journal of Medicine, 394(13), 1313–1324. https://doi.org/10.1056/NEJMra2500106