The Invisible Orchestra: How Calcium, Magnesium, and Vitamin D Work Together
Discover how calcium, magnesium, and vitamin D work together to control cellular signaling, muscle contraction, bone health, and cardiovascular protection.
NUTRITION
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/16/202617 min read
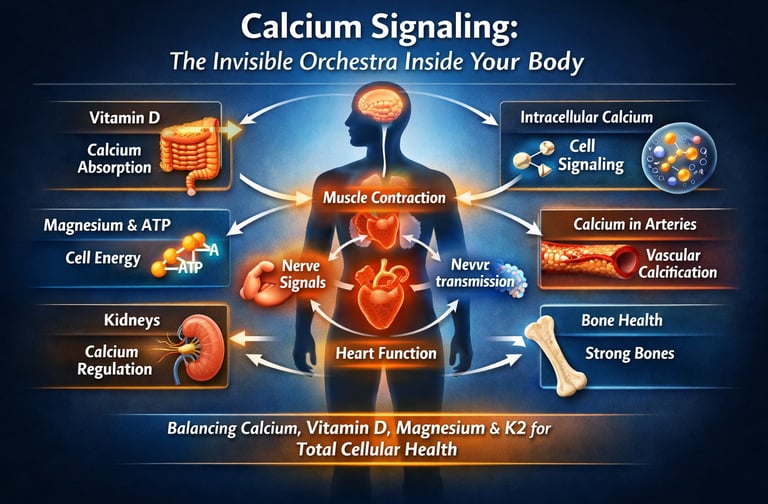
Calcium is more than a bone mineral—it is a critical cellular signaling molecule. Its function depends on a coordinated network with vitamin D and magnesium, which regulate calcium absorption, intracellular signaling, muscle contraction, and vascular health. When this micronutrient triad is balanced, it supports bone strength, metabolic function, and cardiovascular protection.
Every second inside the human body, billions of cells exchange microscopic signals that determine whether the heart beats rhythmically, muscles contract smoothly, and blood vessels remain flexible and healthy. At the center of this vast communication network lies one of the most tightly regulated minerals in biology: calcium. Although nearly 99% of the body’s calcium is stored in bones, the remaining 1% circulating in blood and inside cells acts as a powerful intracellular signaling molecule that controls nerve transmission, hormone release, muscle contraction, and metabolic regulation (Deluque et al., 2025).
But calcium does not operate alone. Its biological effects depend on a tightly coordinated system of micronutrients and regulatory hormones, including vitamin D, magnesium, phosphorus, and vitamin K2. Together, these nutrients form an integrated calcium signaling network that determines how calcium is absorbed from the intestine, recycled by the kidneys, stored in bone, or used as a cellular messenger. When this system functions properly, calcium helps maintain bone density, cardiovascular stability, neuromuscular function, and metabolic health. When it fails, the consequences can range from osteoporosis and muscle cramps to vascular calcification and heart disease (Wimalawansa, 2024).
Modern research is revealing that calcium’s role extends far beyond structural bone health. Inside cells, brief surges of intracellular calcium act as biochemical switches that activate enzymes, regulate gene expression, and trigger adaptive responses to exercise and metabolic stress. For example, calcium-dependent signaling pathways such as CaMKII activation play a crucial role in mitochondrial biogenesis, glucose uptake, and muscle adaptation during high-intensity exercise (Hoffman et al., 2025).
At the same time, disturbances in calcium regulation can have paradoxical effects. In conditions such as chronic kidney disease or metabolic dysfunction, calcium may leave bones and deposit in arterial walls, contributing to vascular calcification and cardiovascular disease (Cabrera et al., 2025).
Understanding how calcium interacts with vitamin D, magnesium, and cellular signaling pathways reveals a deeper truth: calcium homeostasis is not just about bones—it is about the health of every cell in the body.
Key Takeaways
• Calcium signaling controls muscle contraction, nerve transmission, and hormone release
• Vitamin D enables intestinal calcium absorption
• Magnesium powers ATP-dependent calcium pumps
• Dysregulated signaling can lead to vascular calcification
• Exercise activates calcium-dependent pathways that improve metabolic health
Clinical pearls
1. The "Traffic Cop" Principle (Calcium Distribution)
Scientific Perspective: Vascular calcification is an active, cell-mediated transformation where vascular smooth muscle cells (VSMCs) adopt an osteogenic (bone-building) phenotype. This is often driven by a deficiency in Matrix Gla Protein (MGP) activation, which requires Vitamin K2.
Having "extra calcium" in your heart doesn't mean you have too much calcium in your body; it means the calcium is lost. Think of Vitamin K2 and Vitamin D as the "traffic cops" that direct calcium into your bones and keep it out of your arteries.
2. The Magnesium-ATP Nexus
Scientific Perspective: Biologically active ATP exists almost exclusively as a Mg -ATP complex. Magnesium is a mandatory cofactor for the Na/K ATPase pump, which maintains the electrical gradient required for cellular signaling and muscle relaxation.
Magnesium is the "spark plug" for your cellular battery. If you are low on magnesium, your body can’t use the energy it makes. This is why "unexplained" fatigue or leg cramps often persist even if your calorie intake and basic blood work look normal.
3. The Vitamin D "Floor" vs. "Ceiling"
Scientific Perspective: While rickets is prevented at low levels, genomic stability and immune modulation via the Vitamin D Receptor (VDR) show optimal signaling at serum levels above 40 ng/mL. Intestinal calcium absorption can drop by 75\% when levels fall below clinical sufficiency.
There is a difference between "not being sick" and "being optimal." Just because your lab report doesn't have an "L" for low doesn't mean you have enough Vitamin D to help your muscles recover or your immune system stay sharp.
4. The "Silent Drain" of Common Medications
Scientific Perspective: Chronic use of Proton Pump Inhibitors (PPIs) and certain diuretics alters the expression of TRPM6 and TRPM7 transporters in the kidney and intestine, leading to significant secondary magnesium and calcium malabsorption.
Medications for heartburn or blood pressure are often necessary, but they can "leak" minerals out of your system over time. If you are on these long-term, you need to be more intentional about your mineral intake to refill the bucket being drained.
5. Intensity-Dependent Muscle Signaling
Scientific Perspective: High-Intensity Interval Training (HIIT) activates CaMKII (Calcium/calmodulin-dependent protein kinase II) more robustly than steady-state exercise. This kinase is the master switch for mitochondrial biogenesis (creating new energy factories in the cell).
Not all workouts are created equal for your metabolism. Short bursts of high effort send a "loud" calcium signal to your cells that tells them to get stronger and more efficient, whereas a slow walk sends a "whisper." Both are good, but they tell your body to do different things.
6. The Paradox of Supplement "Spikes"
Scientific Perspective: Rapid spikes in serum calcium from high-dose carbonate supplements can temporarily overwhelm the kidneys' regulatory capacity, potentially promoting transient hypercalcemia and seeding mineral deposits in soft tissue.
When it comes to calcium, "slow and steady" wins. Getting your calcium from food (like yogurt or leafy greens) provides a slow trickle into the blood that your body can handle. If you must use a supplement, take smaller doses with meals rather than one giant pill.
Why Calcium Is More Than a Bone Mineral
Calcium is widely recognized for its role in bone health, but in human physiology it functions primarily as a universal cellular signaling molecule. Only about 1% of total body calcium circulates in blood and inside cells, yet this small fraction regulates critical biological processes including muscle contraction, nerve transmission, hormone secretion, vascular tone, and metabolic signaling. Calcium homeostasis depends on a tightly integrated network involving vitamin D, magnesium, parathyroid hormone, and vitamin K–dependent proteins, which collectively control calcium absorption in the intestine, recycling in the kidneys, storage in bone, and intracellular signaling pathways. Disruptions in this regulatory system can lead to a range of disorders including osteoporosis, vascular calcification, muscle dysfunction, and metabolic disease. Modern research increasingly shows that maintaining optimal calcium balance requires more than adequate calcium intake—it requires coordinated micronutrient support and proper regulation of calcium signaling across multiple organ systems.
Calcium signaling refers to the process by which changes in intracellular calcium concentration act as biochemical signals that regulate cellular activity across the nervous, muscular, cardiovascular, and endocrine systems.
Part I: Calcium Signaling: The Hidden Regulator of Cellular Health
Most people associate calcium primarily with bone health, but the most critical biological functions of calcium occur outside the skeleton. Approximately 99% of the body’s calcium is stored in bone, while the remaining 1% circulates in blood and inside cells, where it acts as a powerful intracellular signaling molecule controlling muscle contraction, nerve transmission, hormone release, and metabolic regulation (Deluque et al., 2025).
Because calcium plays such essential roles in cellular physiology, the body maintains extremely tight control over blood calcium levels. Even small fluctuations can have serious consequences. Low calcium levels may trigger muscle spasms, neuromuscular irritability, and cardiac arrhythmias, whereas excessive calcium can contribute to kidney stones, neurological symptoms, and metabolic disturbances.
Two organs form the core of this regulatory system: the small intestine and the kidneys.
The small intestine absorbs calcium from food through specialized transport proteins. Calcium enters intestinal cells through TRPV6 channels, binds to the carrier protein calbindin, and is transported across the cell before being released into the bloodstream. Meanwhile, the kidneys regulate calcium excretion, filtering calcium from the blood and selectively reabsorbing it through renal tubular transport systems, particularly TRPV5 channels (Deluque et al., 2025).
This process is tightly controlled by parathyroid hormone (PTH). When blood calcium levels fall, PTH is released from the parathyroid glands within minutes. PTH increases calcium reabsorption in the kidneys, stimulates bone to release calcium, and activates vitamin D to enhance intestinal calcium absorption. When calcium levels rise, PTH secretion declines and excess calcium is excreted in urine.
Although this system is remarkably precise, it can be disrupted by vitamin D deficiency, kidney disease, medications, or magnesium deficiency, all of which interfere with normal calcium homeostasis.
Why this matters:
If you have experienced abnormal calcium levels, kidney stones, or bone density loss, the intestine–kidney calcium regulation axis may be under strain. Adequate hydration, balanced dietary calcium, and optimal vitamin D status are essential for maintaining stable calcium metabolism.
Part II: How Vitamin D Controls Calcium Absorption
Vitamin D is widely known as the “sunshine vitamin,” but in biological terms it functions more accurately as a prohormone that regulates calcium metabolism, immune function, and gene expression.
Vitamin D exists in two primary forms:
Vitamin D₂ (ergocalciferol) from plant sources
Vitamin D₃ (cholecalciferol) produced in the skin through sunlight exposure or obtained from animal foods
Both forms are converted in the liver into 25-hydroxyvitamin D [25(OH)D], the storage form measured in blood tests. The kidneys then convert this compound into the biologically active hormone 1,25-dihydroxyvitamin D (calcitriol) (Wimalawansa, 2024).
Calcitriol regulates calcium metabolism by binding to vitamin D receptors (VDRs) within cell nuclei. This interaction activates genes that increase production of calcium-transport proteins such as TRPV6 channels and calbindin, dramatically enhancing intestinal calcium absorption.
Without adequate vitamin D, the body absorbs only a fraction of dietary calcium. Research suggests that vitamin D deficiency may reduce intestinal calcium absorption by up to 70–75%, even when dietary calcium intake is sufficient.
Beyond bone health, vitamin D plays broader roles in cardiovascular health, immune regulation, insulin sensitivity, and neuromuscular function. Low vitamin D levels have been associated with increased risks of osteoporosis, type 2 diabetes, cardiovascular disease, and autoimmune disorders (Wimalawansa, 2024).
Current research suggests that optimal blood levels of 25-hydroxyvitamin D may exceed traditional laboratory reference ranges, with many experts recommending concentrations above 40 ng/mL for optimal physiological function.
Why this matters:
Individuals with limited sunlight exposure, darker skin pigmentation, or predominantly indoor lifestyles are at increased risk of vitamin D deficiency. Maintaining adequate vitamin D status through sun exposure, diet, or supplementation (1,000–4,000 IU/day) supports healthy calcium absorption and bone metabolism.
Part III: Magnesium and Calcium Balance in Muscle and Nerve Function
Magnesium is one of the most important yet often overlooked minerals in human physiology. Nearly every reaction involving ATP—the body’s primary energy molecule—requires magnesium. In fact, ATP functions biologically as Mg-ATP, meaning magnesium must be present for cellular energy metabolism to occur.
A 2025 review by Liu and Dudley highlighted the critical role of magnesium in mineral balance, cellular signaling, and cardiovascular physiology (Liu & Dudley, 2025).
Magnesium regulates calcium signaling in several important ways:
It acts as a natural calcium channel regulator, limiting excessive calcium entry into cells.
It supports ATP-dependent calcium pumps, which remove calcium from the cytoplasm after cellular activation.
It is required for normal parathyroid hormone (PTH) secretion, meaning severe magnesium deficiency can lead to low calcium levels despite adequate intake.
Because of these interactions, magnesium deficiency may mimic calcium deficiency symptoms, including muscle cramps, neuromuscular irritability, and cardiac rhythm disturbances.
The kidneys play a central role in magnesium balance. Approximately 70–80% of filtered magnesium is reabsorbed in the renal tubules, primarily through specialized transport proteins known as TRPM6 and TRPM7.
However, several medications—including proton pump inhibitors, diuretics, and certain antibiotics—can reduce magnesium absorption or increase renal magnesium loss.
Modern diets also contribute to magnesium deficiency. Highly processed foods often contain significantly less magnesium than whole foods, and soil depletion has reduced magnesium content in many crops.
Rich dietary sources of magnesium include:
leafy green vegetables
nuts and seeds
legumes
whole grains
dark chocolate
Why this matters:
Symptoms such as muscle cramps, fatigue, poor sleep, anxiety, or irregular heartbeat may reflect magnesium deficiency. Optimizing magnesium intake is essential for maintaining stable calcium signaling, muscle relaxation, and cardiovascular health.
Part IV: Vascular Calcification:Why Calcium Builds Up in Arteries
While calcium is essential for bone strength, problems arise when calcium accumulates in soft tissues such as blood vessels. This process, known as vascular calcification, involves the deposition of calcium phosphate crystals within arterial walls.
Contrary to common belief, vascular calcification is not simply caused by excessive calcium intake. Instead, it is a highly regulated biological process that resembles bone formation within blood vessels.
Research published in the Journal of Cardiovascular Development and Disease describes how vascular smooth muscle cells (VSMCs) can transform into bone-like cells under certain pathological conditions (Cabrera et al., 2025).
Several factors contribute to this transformation:
chronic kidney disease
oxidative stress and inflammation
elevated phosphate levels
metabolic disorders such as diabetes
deficiencies of vitamin D or vitamin K
When these factors disrupt normal signaling pathways, VSMCs begin producing matrix vesicles containing calcium and phosphate, which act as nucleation sites for mineral deposition in the arterial wall.
The clinical consequences can be severe. Vascular calcification increases arterial stiffness, blood pressure, and cardiovascular risk, significantly raising the likelihood of heart attack, stroke, and peripheral artery disease.
Researchers are currently exploring therapies that target molecular pathways involved in vascular calcification, including Wnt signaling and RANK-L pathways. Nutritional strategies are also important. Vitamin K₂ activates matrix Gla protein (MGP), a natural inhibitor of arterial calcification.
Interestingly, many individuals develop both osteoporosis and vascular calcification simultaneously, a phenomenon sometimes referred to as the calcium paradox—where calcium is lost from bone but accumulates in arteries.
Why this matters:
Maintaining balanced levels of vitamin D, magnesium, and vitamin K₂, while limiting excess dietary phosphate from processed foods, may help protect against vascular calcification and cardiovascular disease.
Part V: Exercise, CaMKII, and Metabolic Adaptation
One of the most visible roles of calcium signaling occurs in skeletal muscle contraction.
When a nerve impulse reaches a muscle cell, calcium is released from an intracellular storage structure called the sarcoplasmic reticulum. The calcium binds to the regulatory protein troponin, causing structural changes that allow myosin motors to interact with actin filaments, generating muscle contraction.
When the signal ends, calcium is pumped back into storage by ATP-dependent calcium pumps, allowing the muscle to relax.
Exercise places intense demands on this system and activates powerful cellular signaling pathways that drive long-term muscle adaptation.
A 2025 phosphoproteomics study published in Sports Medicine demonstrated that high-intensity interval training (HIIT) activates complex calcium-dependent signaling networks in skeletal muscle (Hoffman et al., 2025).
Among the most important signaling molecules activated during exercise is CaMKII (calcium/calmodulin-dependent protein kinase II). This enzyme responds to increases in intracellular calcium and regulates processes including:
mitochondrial biogenesis
glucose uptake
metabolic flexibility
gene expression related to muscle adaptation
The study also revealed that exercise intensity influences signaling patterns. Moderate exercise primarily activates metabolic pathways, whereas high-intensity exercise triggers stronger activation of calcium-dependent kinases and stress-response signaling networks.
Magnesium plays a critical role in this process by enabling ATP production, while vitamin D receptors present in muscle tissue contribute to muscle protein synthesis and functional performance.
Why this matters:
The ability of muscles to adapt to exercise depends on efficient calcium signaling, adequate magnesium for ATP production, and optimal vitamin D status. If exercise progress stalls, evaluating micronutrient status may reveal an overlooked factor influencing performance and recovery.
Key Mechanisms of the Calcium–Magnesium–Vitamin D–K2 Network
Understanding calcium biology requires looking beyond a single nutrient. Several micronutrients work together to regulate calcium signaling, cellular metabolism, vascular protection, and musculoskeletal health. The following mechanisms summarize how this integrated micronutrient network functions at the cellular level.
Vitamin D – The Absorption Regulator
Key Mechanism: Upregulates intestinal calcium transport proteins such as TRPV6 channels and calbindin.
Cellular Effect: Enhances active calcium transport across intestinal epithelial cells, increasing calcium availability in the bloodstream.
Clinical Outcome: Supports bone mineralization, skeletal strength, and prevention of osteomalacia or osteoporosis.
Magnesium – The Energy Enabler
Key Mechanism: Forms the Mg-ATP complex, the biologically active form of ATP required for enzymatic reactions.
Cellular Effect: Powers ATP-dependent calcium pumps (such as SERCA and Ca-ATPase) that regulate intracellular calcium levels.
Clinical Outcome: Promotes muscle relaxation, neuromuscular stability, and prevention of cramps or arrhythmias.
Vitamin K2 – The Calcium Traffic Director
Key Mechanism: Activates matrix Gla protein (MGP) and other vitamin K–dependent proteins that inhibit soft-tissue mineralization.
Cellular Effect: Prevents calcium deposition in vascular smooth muscle and arterial walls.
Clinical Outcome: Contributes to reduced vascular calcification and improved cardiovascular protection.
Calcium – The Cellular Messenger
Key Mechanism: Functions as a universal intracellular signaling ion, triggering activation of calcium-dependent enzymes such as CaMKII.
Cellular Effect: Initiates muscle contraction, metabolic signaling, and gene expression pathways in response to physiological stimuli.
Clinical Outcome: Enables muscle contraction, exercise adaptation, and normal neuromuscular function.
Takeaway: These micronutrients operate as a coordinated biochemical network, ensuring that calcium is absorbed efficiently, directed to bone, kept out of arteries, and used effectively as a cellular signaling molecule.
Putting It All Together: The Integrated Picture
Calcium is not merely a structural mineral; it is the body’s universal signaling molecule.
While most people associate calcium with bone density, modern physiology shows that its most critical function lies in intracellular signaling. Tiny fluctuations in calcium concentration act as biochemical switches that regulate nerve transmission, hormone secretion, enzyme activation, and gene expression. Because of this role, calcium signaling influences nearly every organ system, from the brain to the cardiovascular system (Deluque et al., 2025).Human health depends on a finely tuned calcium regulatory network rather than calcium alone.
Calcium metabolism is governed by a coordinated system involving vitamin D, parathyroid hormone, magnesium, phosphate balance, and vitamin K–dependent proteins. Vitamin D increases intestinal calcium absorption, magnesium enables ATP-dependent calcium pumps, and vitamin K–dependent proteins help prevent calcium from depositing in soft tissues such as arteries (Wimalawansa, 2024). Disruption of any component of this network can destabilize calcium homeostasis.Metabolic diseases increasingly appear to involve calcium signaling dysfunction.
Emerging research suggests that disturbances in cellular calcium regulation contribute to conditions such as insulin resistance, vascular calcification, hypertension, and chronic kidney disease. In these states, calcium signaling pathways may become dysregulated, causing calcium to leave bone while accumulating in arterial walls—a paradoxical process sometimes described as the “bone–vascular axis” (Cabrera et al., 2025).Exercise represents one of the most powerful natural regulators of calcium signaling.
During muscle contraction, transient surges in intracellular calcium activate signaling proteins such as calcium/calmodulin-dependent protein kinase II (CaMKII). These molecular signals stimulate mitochondrial biogenesis, glucose uptake, and long-term improvements in metabolic health, illustrating how physical activity transforms calcium from a structural mineral into a dynamic metabolic messenger (Hoffman et al., 2025).The emerging lesson for modern medicine is integration.
Calcium cannot be understood in isolation. Its physiological effects depend on a micronutrient ecosystem, involving vitamin D, magnesium, phosphorus, and vitamin K2, alongside lifestyle factors such as exercise and diet quality. Protecting this integrated network may be one of the most effective strategies for preserving skeletal strength, cardiovascular resilience, and cellular metabolic health across the lifespan.
Frequently Asked Questions About Calcium, Magnesium, and Vitamin D
1. What does calcium signaling do in the body?
Calcium signaling is the process by which small changes in intracellular calcium concentration regulate cellular activities such as muscle contraction, nerve transmission, hormone secretion, and gene expression. These calcium signals act as biochemical switches that coordinate physiological responses across multiple organ systems.
2. Why do calcium, magnesium, and vitamin D work together?
Calcium metabolism depends on a coordinated micronutrient system. Vitamin D increases intestinal calcium absorption, magnesium enables ATP-dependent calcium transport pumps, and vitamin K–dependent proteins help prevent calcium from depositing in soft tissues such as arteries.
3. Can too much calcium cause heart problems?
Excess calcium intake—particularly from high-dose supplements—may lead to temporary spikes in blood calcium levels. In susceptible individuals, this may contribute to vascular calcification, especially when vitamin K2 and magnesium status are inadequate.
4. What foods support calcium signaling?
Foods that support calcium signaling include:
• dairy products and fortified plant milks (calcium)
• fatty fish such as salmon and sardines (vitamin D)
• leafy greens and legumes (magnesium)
• fermented foods such as natto (vitamin K2)
5. How much calcium do I actually need each day, and should I get it from food or supplements?
Most adults need between 1,000 and 1,200 mg of calcium per day, depending on age and sex. The evidence strongly favors getting this from food — dairy products, leafy greens, tofu, fortified plant milks, and canned fish with bones — rather than high-dose supplements. Calcium supplements, particularly calcium carbonate in doses above 500 mg at a time, have been associated with increased cardiovascular risk in some studies, possibly because they flood the blood with calcium quickly rather than allowing the gradual absorption that food provides. If you need to supplement, calcium citrate in smaller divided doses (250–500 mg) is generally better tolerated and absorbed.
6. Can I have low calcium even if I eat plenty of it?
Yes, absolutely. Calcium absorption depends heavily on vitamin D status, stomach acid levels, and magnesium adequacy. If you are vitamin D deficient, your intestines may absorb as little as 10–15% of the calcium you consume. Medications that reduce stomach acid (like proton pump inhibitors) impair calcium absorption. And as noted, severe magnesium deficiency can suppress PTH, causing calcium to remain low despite adequate intake. Always address vitamin D and magnesium alongside calcium.
7. I have been told I have calcium deposits in my arteries. Should I stop taking calcium supplements?
This is an important question to discuss with your doctor, but the short answer is: vascular calcification is generally not caused by dietary calcium or calcium supplements directly. It is driven by metabolic dysregulation — including chronic kidney disease, inflammation, high phosphate levels, and deficiencies in vitamin K2 and vitamin D. That said, reviewing supplement doses and ensuring you are not getting far more calcium than needed is reasonable. Ensuring adequate vitamin K2 (which directs calcium toward bone and away from arteries) and vitamin D may be more impactful than simply stopping calcium.
8. What is the relationship between magnesium and muscle cramps?
Muscle cramps can have multiple causes, but magnesium deficiency is a common and often overlooked one. Magnesium plays a critical role in allowing muscles to relax after contraction by supporting calcium reuptake into the sarcoplasmic reticulum. When magnesium is low, this process is impaired, and muscles may remain in a contracted or hyperexcitable state, leading to cramps and spasms. Athletes and people who sweat heavily are particularly at risk for magnesium depletion.
9. Does vitamin D deficiency really affect muscle function, not just bone health?
Yes. Vitamin D receptors are found in muscle tissue, and vitamin D plays a direct role in muscle protein synthesis and muscle fiber composition. Deficiency has been associated with muscle weakness, reduced athletic performance, increased fall risk in older adults, and slower recovery from exercise. The 2025 phosphoproteomics study by Hoffman and colleagues reinforces that the molecular signaling networks in exercising muscle — many of which are calcium-driven — are influenced by the broader hormonal environment, including vitamin D.
10. Is high-intensity interval training (HIIT) really better than moderate exercise for calcium and metabolic signaling?
Not necessarily better — but different. HIIT generates stronger calcium signals in muscle cells and activates a broader range of kinase pathways, including calcium-sensitive enzymes like CaMKII. This leads to adaptations like increased mitochondrial density and insulin sensitivity. However, moderate-intensity exercise has its own important signaling profile and is safer and more sustainable for many people. The ideal is probably a combination: regular moderate activity with periodic higher-intensity efforts, supported by adequate nutritional status.
11. What dietary pattern best supports calcium signaling and micronutrient integration overall?
A whole-food, varied diet is the most reliable foundation. Prioritize fatty fish (salmon, sardines, mackerel) for vitamin D and omega-3s; leafy green vegetables and legumes for magnesium and calcium; fermented dairy or fermented foods like natto for vitamin K2; nuts and seeds for magnesium and healthy fats; and limited processed foods, particularly those high in phosphate additives (found in many fast foods and sodas), which can drive vascular calcification. If you live in a region with limited sun exposure, vitamin D3 supplementation (1,000–4,000 IU daily) is widely recommended, and magnesium glycinate or citrate at 200–400 mg per day is safe for most adults.
Author’s Note
Calcium is often discussed in medicine as a nutrient associated primarily with bone health, yet modern physiology tells a far more complex story. Calcium functions as one of the body’s most important intracellular signaling molecules, helping coordinate processes as diverse as muscle contraction, nerve transmission, hormone release, and cellular metabolism. Writing this article was motivated by a desire to bridge the gap between traditional nutritional advice and the rapidly evolving science of calcium signaling and micronutrient integration.
Over the past decade, research has increasingly shown that calcium biology cannot be understood in isolation. Its regulation depends on a tightly interconnected system involving vitamin D, magnesium, phosphorus, and vitamin K–dependent proteins. Disruption of this network may contribute not only to osteoporosis but also to cardiovascular disease, metabolic dysfunction, and vascular calcification. As clinicians and researchers, it is becoming clear that maintaining calcium balance requires attention to the broader metabolic environment rather than focusing on a single nutrient.
In this article, I have attempted to synthesize insights from recent peer-reviewed studies to explain how calcium moves through the intestine, kidneys, blood vessels, and muscle cells, and how these processes interact with exercise and metabolic health. My goal is to translate complex physiological mechanisms into language that is understandable for patients while still preserving the scientific rigor necessary for evidence-based medicine.
Ultimately, understanding calcium signaling highlights an important principle of human biology: health is maintained not by individual nutrients or isolated organs, but by integrated physiological systems working in harmony. I hope this discussion helps readers appreciate how small molecular signals inside our cells influence the health of the entire body.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Can Omega-3 Boost Muscle Protein Synthesis? What the Latest Research Reveals | DR T S DIDWAL
Beyond the Low-Fat Myth: 6 New Studies Redefining Dietary Fat and Heart Health | DR T S DIDWAL
Can Plant-Based Polyphenols Lower Biological Age? | DR T S DIDWAL
References
Deluque, A. L., Dimke, H., & Alexander, R. T. (2025). Biology of calcium homeostasis regulation in intestine and kidney. Nephrology Dialysis Transplantation, 40(3), 435–445. https://doi.org/10.1093/ndt/gfae204
Wimalawansa, S. J. (2024). Physiology of Vitamin D — Focusing on Disease Prevention. Nutrients, 16(11), 1666. https://doi.org/10.3390/nu16111666
Liu, M., & Dudley, S. C., Jr. (2025). Magnesium Homeostasis and Magnesium Transporters in Human Health. Nutrients, 17(5), 920. https://doi.org/10.3390/nu17050920
Valentin Cabrera, A. M., Ashbrook, S. K., & Hutcheson, J. D. (2025). Considerations on the Development of Therapeutics in Vascular Calcification. Journal of Cardiovascular Development and Disease, 12(6), 206. https://doi.org/10.3390/jcdd12060206
Hoffman, N. J., Whitfield, J., Xiao, D., et al. (2025). Phosphoproteomics Uncovers Exercise Intensity-Specific Skeletal Muscle Signaling Networks Underlying High-Intensity Interval Training in Healthy Male Participants. Sports Medicine, 55, 1983–2004. https://doi.org/10.1007/s40279-025-02217-2