Skeletal Muscle: The Missing Link in Blood Sugar Control and Metabolic Health
Discover how skeletal muscle controls blood sugar after meals and why strength, fitness, and movement are key to preventing insulin resistance
EXERCISEDIABETES
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/5/202618 min read
The role of skeletal muscle in blood sugar regulation
our muscles are not just for movement — they are your body's largest glucose-disposal organ, and understanding how they work could change the way you manage your metabolic health."
Every time you eat, your body begins a carefully coordinated process to manage the rise in blood sugar that follows. As carbohydrates are digested, glucose enters your bloodstream, and within minutes, your system must decide where that fuel should go. Most people assume the pancreas does most of this work. While insulin is certainly essential, it does not act alone.
The real metabolic workhorse is your skeletal muscle.
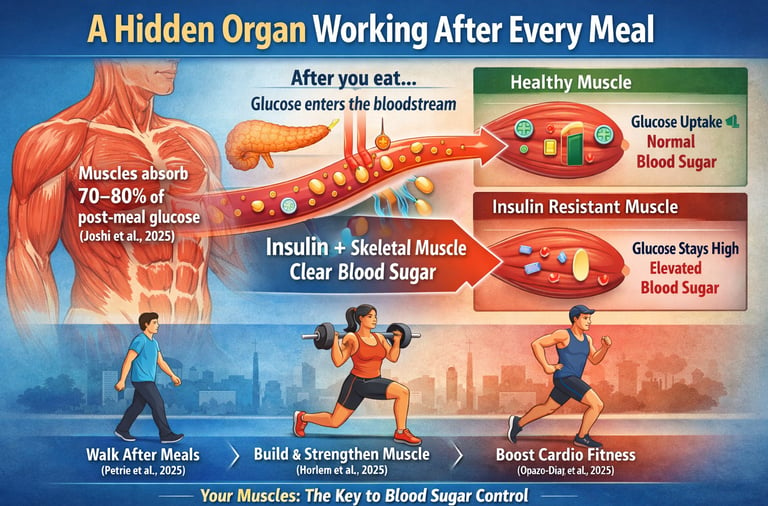
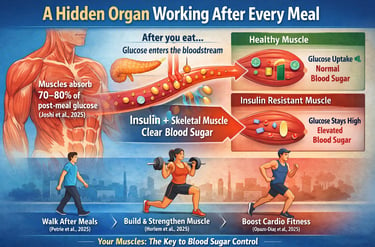
Research shows that in metabolically healthy individuals, skeletal muscle is responsible for clearing approximately 70–80% of postprandial (after-meal) glucose from the bloodstream (Joshi et al., 2025). In other words, the majority of blood sugar disposal after you eat depends not just on insulin secretion, but on how effectively your muscles respond to that signal.
When muscle cells are sensitive to insulin, glucose moves smoothly from the bloodstream into muscle tissue, where it is stored or used for energy. Blood sugar rises modestly and returns to baseline within a few hours. However, when muscle becomes insulin resistant — due to inactivity, aging, excess lipid accumulation, or inflammation — this process becomes inefficient (Drobiova et al., 2025). Glucose remains elevated for longer periods, increasing metabolic stress and contributing over time to type 2 diabetes and cardiovascular disease.
The encouraging news is that muscle is highly adaptable. Even short bouts of movement after meals can significantly reduce glucose and insulin spikes (Petrie et al., 2025). Resistance training and aerobic conditioning can improve muscle quality, mitochondrial function, and glucose uptake capacity across the lifespan (Horlem et al., 2025; Opazo-Díaz et al., 2025).
Your muscles are not just for movement. They are your body’s largest glucose-regulating organ — and one of the most powerful tools you have for protecting your metabolic health
Clinical Pearls: Skeletal Muscle & Metabolic Health
1. The "Hybrid" Muscle Crisis (Type IIa Fiber Loss)
Sarcopenia is characterised not just by atrophy but also by a specific fibre-type shift. Type IIa "fast-oxidative" fibers are the metabolic workhorses; they possess high mitochondrial density (like Type I) and high glycolytic capacity (like Type IIb). The age-related decline in these hybrid fibers reduces metabolic flexibility, making the muscle less adept at switching between burning fats and sugars.
Think of Type IIa fibers as the 'hybrid engine' of your body. Just like a hybrid car uses both gas and electricity to be efficient, these muscles can burn both sugar and fat. As we age, we lose these versatile engines first, which is why your blood sugar might spike more easily even if your weight hasn't changed."
2. The "Back Door" Glucose Entry (AMPK Pathway)
While insulin resistance "jams" the PI3K/Akt signaling pathway, the AMP-activated protein kinase (AMPK) pathway remains largely functional. Muscle contraction triggers AMPK, which stimulates GLUT4 translocation to the cell membrane independently of insulin. This creates a physiological "bypass" for glucose disposal.
When you have insulin resistance, the 'front door' to your cells is stuck. But exercise acts like a 'back door.' When you move your muscles, you don't need insulin's 'key' to let the sugar in; the movement itself pulls the sugar out of your blood and into the muscle to be used as fuel."
3. Postprandial Water Shifting (Cellular Hydration)
As noted in Petrie et al. (2025), the ratio of extracellular to intracellular water (ECW/ICW) predicts the glucose-lowering success of post-meal exercise. Intracellular dehydration can impair the molecular signaling required for GLUT4 docking, suggesting that "cellular turgor" is a prerequisite for optimal metabolic response.
Muscle is mostly water. If you are even slightly dehydrated, your muscle cells 'shrink' and become less responsive to the signals that tell them to absorb sugar. Drinking a glass of water with your meal isn't just for digestion; it literally primes your muscles to be better sugar sponges."
4. Cardiorespiratory Fitness as a "Multiplier"
Lean mass quantity is a poor predictor of fat oxidation if Maximal Oxygen Consumption (VO 2 max) is low. Cardiorespiratory fitness (CRF) mediates muscle quality by ensuring adequate capillary density and mitochondrial efficiency. Without CRF, added muscle mass may remain "metabolically dormant."
Having big muscles is like having a large factory, but cardiorespiratory fitness is the delivery system that brings in the oxygen and carries out the waste. If the delivery system is broken, the size of the factory doesn't matter. You need both strength training and 'huff-and-puff' cardio to make your muscles truly healthy."
5. The "Muscle-First" Diagnostic (Postprandial Continuity)
Fasting blood glucose is a "lagging indicator" that often stays normal until significant beta-cell exhaustion occurs. Joshi et al. (2025) argue that postprandial glucose variability is the "leading indicator" of muscle-specific insulin resistance, reflecting a failure of the body's largest glucose-disposal organ long before the pancreas fails.
"Checking your blood sugar only when you wake up is like checking your bank balance only once a year. It doesn't tell you how you're spending. Seeing how your body handles a meal tells us exactly how well your 'muscle engine' is running right now, allowing us to fix it years before a diabetes diagnosis."
6. The Lipid-Induced "Signaling Jam" (KLF9/GRB14)
:Saturated fat overload (palmitic acid) triggers the KLF9–GRB14 axis, which directly inhibits the insulin receptor. This "lipotoxicity" means that even in the presence of high insulin, the signal never reaches the GLUT4 vesicles. Silencing this axis can restore sensitivity, highlighting the importance of dietary fat quality.
High levels of certain fats can actually 'gum up' the sensors on your muscle cells. It’s like putting the wrong oil in an engine; eventually, the parts stop communicating. By choosing healthier fats and moving more, we can 'clean' those sensors and get the signal moving again."
Section 1: What Happens to Your Blood Sugar After You Eat?
Postprandial glucose muscle uptake
The period after a meal — roughly 2–4 hours — is one of the most metabolically active windows in your day. When carbohydrates are digested and absorbed, glucose floods into the bloodstream. The pancreas senses this rise and releases insulin, the key hormone that signals cells throughout your body to open their doors and let glucose in.
In healthy individuals, blood glucose peaks gently and returns to baseline within about two hours. But what makes this possible isn't just insulin — it's the enormous capacity of skeletal muscle to absorb that glucose wave.
A landmark 2025 review by Joshi and colleagues titled "Postprandial Glucose: A Variable in Continuum" reframes how clinicians and patients should think about after-meal blood sugar. Rather than treating postprandial glucose as a single snapshot number, the authors argue it is a continuous variable — one that reflects the dynamic interplay between carbohydrate absorption, insulin secretion, hepatic (liver) glucose output, and peripheral tissue uptake, with muscle at the center of the story.
Key Takeaway — Joshi et al. (2025): Postprandial glucose should not be viewed as a single data point but as a dynamic continuum. Muscle tissue is the primary peripheral destination for glucose after meals, and even modest disruptions in muscle insulin sensitivity can tip this continuum toward metabolic disease. Monitoring and managing postprandial glucose variability — not just fasting glucose — is essential for truly understanding metabolic health.
What makes this clinically important for you as a patient is simple: if your muscles are not responding properly to insulin's signal, glucose that should enter muscle cells stays circulating in your blood instead. Over time, that pattern — high postprandial glucose spikes — is associated with inflammation, oxidative stress, cardiovascular disease, and type 2 diabetes.
Exercise as a Postprandial Reset Button
How does muscle control blood sugar?
One of the most exciting recent findings in this space comes from a 2025 study by Petrie and colleagues, published in Physiological Reports. Their research examined how postprandial exercise — physical activity taken after a meal — affects blood glucose and insulin levels, and whether these effects differ by age and body composition.
The results were striking. Exercise performed after a meal significantly attenuated (reduced) the glucose and insulin spike that typically follows eating. But the story didn't stop there. The researchers found that the beneficial response was associated with age, cognitive function, and the ratio of extracellular to intracellular body water — a marker of cellular health and hydration status.
What this tells us is that post-meal movement doesn't just burn a few extra calories; it fundamentally alters how your muscles interact with insulin and glucose. When you contract your muscles — even with a brisk 10–15 minute walk — a second pathway for glucose uptake is activated that works independently of insulin. This is mediated by a protein called AMPK (AMP-activated protein kinase), which we'll revisit in the next section.
Key Takeaway — Petrie et al. (2025): Postprandial exercise is a powerful, drug-free tool for blunting blood glucose spikes. Its effectiveness varies by age and cellular hydration, suggesting that maintaining muscle quality and staying well-hydrated may enhance the metabolic benefits of after-meal activity. Even a short walk after meals can meaningfully improve glucose regulation.
Section 2: Inside the Muscle Cell — The GLUT4 Story
What Is GLUT4 and Why Should You Care?
Imagine insulin as a key, and your muscle cells as houses with locked doors. The "lock" that insulin opens is a receptor on the surface of muscle cells. When insulin binds this receptor, it sends a signal cascade inward — telling the cell to move special glucose transporters called GLUT4 to the cell surface. Only once GLUT4 proteins arrive at the membrane can glucose actually enter the cell.
This process — called GLUT4 trafficking — is one of the most studied and most critical molecular events in metabolic biology. When it works properly, glucose moves efficiently from the bloodstream into muscle cells after meals. When it breaks down, insulin resistance begins.
Under resting conditions, GLUT4 proteins are stored inside the cell in specialized compartments called GLUT4 storage vesicles (GSVs). Think of these as sealed cargo containers waiting in a warehouse. When insulin arrives, an elaborate signaling cascade — involving proteins like IRS-1, PI3K, Akt, and AS160 — essentially orders the warehouse doors to open, loading the cargo containers onto "delivery trucks" (cytoskeletal motors) that transport GLUT4 to the cell surface.
Drobiova et al. (2025) identified several key regulatory nodes in this process that are disrupted in insulin resistance. Critically, in states like obesity, chronic inflammation, or lipid accumulation in muscle cells, the signaling pathway becomes "jammed." Akt — a pivotal protein in the insulin-signaling cascade — becomes less active. AS160, the gatekeeper that releases GLUT4 vesicles toward the membrane, stays in its "closed" position. The result: GLUT4 never makes it to the surface, and glucose cannot enter the muscle cell despite insulin being present.
Key Takeaway — Drobiova et al. (2025): GLUT4 trafficking is a multi-step, tightly regulated molecular process. Disruptions at multiple points — from insulin receptor signaling to vesicle docking — can cause insulin resistance in skeletal muscle. Understanding these nodes offers potential targets for therapeutic intervention and highlights why both exercise (which activates GLUT4 independently) and lifestyle changes that reduce cellular lipid accumulation are metabolically powerful.
The Exercise Bypass — An Insulin-Independent Route
Here is where the muscle-exercise connection becomes particularly empowering. Exercise activates AMPK, which can independently stimulate GLUT4 movement to the muscle cell surface — bypassing the jammed insulin pathway entirely. This is why people with type 2 diabetes, whose insulin signaling is significantly impaired, still benefit enormously from physical activity. They are essentially using a back door to achieve what the front door can no longer accomplish.
This dual-pathway system — insulin-dependent and exercise-dependent GLUT4 translocation — is one of the most elegant designs in human physiology. It also explains why combining dietary management with physical activity produces effects that neither approach achieves alone.
Section 3: Does Muscle Mass or Muscle Quality Matter More?
The Quantity vs. Quality Debate
For years, the medical and fitness community focused primarily on muscle mass — the sheer amount of muscle tissue you have. More muscle = better metabolism, the thinking went. But an important 2025 study challenges this oversimplification in an illuminating way.
Opazo-Díaz and colleagues, publishing in the Journal of the International Society of Sports Nutrition, investigated whether lean mass quantity or lean mass quality is a stronger determinant of maximal fat oxidation capacity — the body's ability to burn fat as a fuel source, a key marker of metabolic flexibility.
Their findings pointed to a nuanced answer: while the amount of lean (muscle) mass matters, cardiorespiratory fitness (CRF) — a measure of how well your heart, lungs, and muscles work together — serves as a critical mediating variable. In other words, having more muscle only translates to better fat-burning capacity if that muscle is metabolically fit and well-supplied with cardiovascular support.
Muscles that are large but poorly conditioned — low in mitochondrial density, weak in oxidative capacity — do not perform nearly as well metabolically as leaner, well-conditioned muscles. The quality of the metabolic machinery inside each muscle fiber matters enormously.
Key Takeaway — Opazo-Díaz et al. (2025): Muscle quantity alone is insufficient for optimal metabolic health. Cardiorespiratory fitness — which reflects muscle metabolic quality — mediates the relationship between lean mass and fat oxidation capacity. This means that building muscle through resistance training and improving aerobic fitness through cardio exercise are both necessary for full metabolic benefit.
The KLF9-GRB14 Axis — A New Villain in Diabetic Muscle
While Opazo-Díaz and colleagues looked at muscle quality from a functional perspective, a remarkable 2026 molecular study by Zhu and colleagues zeroed in on a specific molecular mechanism that degrades muscle insulin sensitivity in type 2 diabetes.
Published in Current Proteomics, the study examined what happens in undifferentiated myoblasts (early-stage muscle cells) when they are exposed to palmitic acid — a saturated fat that mimics the lipid-overload environment common in obesity and type 2 diabetes. The researchers found that a transcription factor called KLF9 (Krüppel-Like Factor 9) was significantly upregulated under these conditions, and that KLF9 activated a downstream target called GRB14 — a known insulin signaling inhibitor.
When KLF9 was silenced (its gene turned off), both insulin resistance and inflammatory markers in the muscle cells were significantly reduced. This identifies the KLF9–GRB14 axis as a novel pathogenic pathway in type 2 diabetic myopathy — muscle dysfunction driven by diabetes.
Key Takeaway — Zhu et al. (2026): A newly identified KLF9–GRB14 molecular pathway appears to drive insulin resistance and inflammation in skeletal muscle under high-fat conditions. Silencing KLF9 reversed these effects in cell models, revealing a potential therapeutic target for diabetic myopathy. This research highlights how dietary fat quality can directly alter gene expression inside muscle cells, reinforcing the importance of nutrition alongside exercise.
Section 4: Aging, Sarcopenia, and the Silent Metabolic Crisis
How Muscle Mass Shapes Your Metabolic Future
Here is an uncomfortable truth: starting around age 30, most people lose 3–8% of their muscle mass per decade. After age 60, the rate accelerates. This progressive loss of muscle mass and function is called sarcopenia, and its metabolic consequences are profound.
Because skeletal muscle is responsible for the majority of postprandial glucose uptake, losing muscle mass means you progressively lose metabolic capacity. Each kilogram of muscle lost reduces your body's ability to buffer blood sugar after meals. Over decades, this contributes to worsening insulin sensitivity and, in many cases, type 2 diabetes — even in people who never significantly gain weight.
The Molecular Biology of Muscle Aging
A detailed 2025 molecular review by Horlem and colleagues in the International Journal of Molecular Sciences, titled "Molecular Framework of the Onset and Progression of Skeletal Muscle Aging," maps the key biological processes that drive sarcopenia at the cellular level.
The authors identify several interconnected mechanisms: mitochondrial dysfunction (aging muscle cells produce energy less efficiently), impaired protein synthesis (muscle is slower to repair and rebuild), chronic low-grade inflammation — sometimes called "inflammaging" — and disrupted satellite cell activity (the stem cells responsible for muscle regeneration become less responsive with age).
Particularly relevant to glucose metabolism is the finding that aged muscle shows measurable reductions in GLUT4 expression and trafficking efficiency. The very molecular machinery we discussed in Section 2 gradually degrades with aging. Insulin signaling proteins become less responsive, mitochondria are less able to oxidize glucose efficiently, and the muscle fiber composition shifts away from the metabolically active Type IIa (fast-oxidative) fibers.
Key Takeaway — Horlem et al. (2025): Skeletal muscle aging is driven by multiple interacting molecular mechanisms, including mitochondrial decline, impaired protein turnover, chronic inflammation, and reduced stem cell activity. These changes collectively degrade the muscle's ability to respond to insulin and take up glucose — directly contributing to age-associated metabolic disease. Interventions that target these pathways (resistance training, adequate protein intake, anti-inflammatory diets) may slow this decline.
The Petrie Study Revisited — Age and the Postprandial Response
Recall the finding from Petrie et al. (2025) that the glucose-lowering effect of postprandial exercise was associated with age. This takes on new meaning in the context of sarcopenia. Older adults not only have less muscle mass but also have muscle cells that are less responsive to both insulin and exercise stimuli. The combination — reduced mass and reduced sensitivity — creates a compounding metabolic disadvantage that becomes increasingly difficult to overcome without deliberate, targeted intervention.
The silver lining: resistance training and regular aerobic exercise have been shown to partially reverse sarcopenia's molecular damage. Even in adults over 70, exercise interventions can increase GLUT4 expression, improve mitochondrial function, and restore some degree of insulin sensitivity. It is never too late to start.
Practical Implications: What You Can Do
Move after meals. Even a 10–15 minute walk after eating engages GLUT4 through the insulin-independent (AMPK) pathway and measurably reduces postprandial glucose spikes
Prioritise resistance training. Building and preserving muscle mass protects your body's glucose disposal capacity. Aim for 2–3 strength training sessions per week.
Add aerobic exercise. Since cardiorespiratory fitness mediates the metabolic quality of your muscle, aerobic conditioning is not optional — it amplifies the benefits of the muscle you build.
Eat adequate protein. Protein intake (especially essential amino acids and leucine) stimulates muscle protein synthesis and supports satellite cell function, helping to counter the molecular aging of muscle
Minimise saturated fat excess. Palmitic acid-driven activation of the KLF9–GRB14 axis shows that what you eat changes gene expression in your muscle cells. A dietary pattern rich in unsaturated fats, fiber, and whole foods supports muscle insulin sensitivity at the molecular level.
Monitor postprandial glucose, not just fasting. Postprandial glucose variability may be a more sensitive early marker of metabolic dysfunction than fasting glucose alone. Talk to your doctor about whether continuous or postprandial monitoring is right for you.
Patient Worksheet: Your Muscle-First Exercise Prescription
Goal: To optimize skeletal muscle for maximum blood sugar disposal and metabolic resilience.
Phase 1: The "Post-Meal Reset" (Daily)
The goal is to activate the AMPK "Back Door" to pull sugar out of your blood without needing extra insulin.
The Action: 10–15 minutes of light-to-moderate activity.
The Timing: Start within 20–30 minutes of finishing your largest meal of the day.
Options: * Brisk walk around the block.
Bodyweight squats or wall push-ups (2 sets of 15).
Stair climbing (3–5 flights).|
Phase 2: Building the "Glucose Sponge" (2–3x Weekly)
The goal is to maintain and grow Type IIa "Hybrid" fibers to increase your total glucose-buffering capacity.
The Protocol: Full-body resistance training focusing on large muscle groups (legs, back, chest).
Intensity: Choose a weight where the last 2 reps of a set are challenging but maintain good form.
The Prescription:
Lower Body: Goblet squats or lunges (3 sets of 10–12).
Push: Overhead press or floor press (3 sets of 10–12).
Pull: Seated rows or lat pulldowns (3 sets of 10–12).
Hinge: Glute bridges or kettlebell deadlifts (3 sets of 12).
Phase 3: Improving "Engine Quality" (2x Weekly)
The goal is to increase Mitochondrial Density and Cardiorespiratory Fitness (CRF) to ensure your muscles are metabolically flexible.
The Action: "Zone 2" Steady-State Cardio.
The Intensity: You should be able to maintain a conversation but feel slightly breathless (the "Talk Test").
Duration: Aim for 30–45 minutes.
Options: Cycling, swimming, incline walking, or rowing.
The "Hydration Anchor"
Remember: Muscle is 75% water. Dehydration "jams" the signaling process.
Prescription: Drink 300–500ml of water with every workout and every post-meal walk.
Physician’s Note: "Think of these movements as doses of medicine. Each rep 'greases the gears' of your GLUT4 warehouse, making your body more resilient to the next meal you eat."
Frequently asked questions
1️⃣ “If I take diabetes medicine, do I still need to exercise?”
Yes — absolutely.
Medication helps control blood sugar, but exercise helps your body use sugar more efficiently. When your muscles contract, they pull glucose out of the bloodstream even if insulin isn’t working perfectly. Think of movement as improving the “machinery,” not just lowering the number.
Medicine manages the condition.
Muscle improves the system.
2️⃣ “How soon will I see results if I start walking after meals?”
Often within days.
Even a 10–15 minute walk after eating can lower your blood sugar spike that same day. If you check your glucose 1–2 hours after a meal, you may notice it doesn’t rise as high when you move compared to when you sit.
The effect is immediate — and cumulative over time.
3️⃣ “I’m over 60. Is it too late to improve my muscle health?”
Not at all.
Muscle remains adaptable at any age. Studies show people in their 70s and beyond can increase strength, improve insulin sensitivity, and build healthier muscle with resistance training and regular walking.
It’s never too late to strengthen your metabolic “engine.”
4️⃣ “Do I need to lift heavy weights to improve blood sugar?”
No.
You need consistent muscle engagement, not extreme workouts. Bodyweight exercises, resistance bands, light dumbbells, or even chair squats can improve muscle function. The key is regular practice, not intensity alone.
5️⃣ “Is cardio enough, or do I need strength training too?”
Both help — but they do different things.
Cardio improves how efficiently your muscles use oxygen (quality).
Strength training increases the amount of muscle you have (capacity).
Together, they give the best blood sugar control.
6️⃣ “What if I can’t exercise because of joint pain?”
Start small and choose low-impact options:
Walking at a comfortable pace
Stationary cycling
Swimming
Chair-based strength exercises
Even gentle movement improves glucose handling. The goal is progress, not perfection.
7️⃣ “If my fasting sugar is normal, should I still worry?”
Possibly.
Fasting glucose can stay normal for years while post-meal spikes are already elevated. Watching how your body handles meals may reveal early insulin resistance before fasting numbers change.
8️⃣ “Can diet alone fix insulin resistance?”
Diet is powerful — but muscle is the main site where sugar is stored and used. Without movement, you’re only solving half the problem.
Food controls input.
Muscle controls disposal.
9️⃣ “How much exercise is enough to make a difference?”
A practical starting point:
Walk 10–15 minutes after meals
Strength train 2–3 times per week
Add 30 minutes of moderate cardio twice weekly
Small, consistent steps matter more than intense bursts.
🔟 “What’s the most important thing I should remember?”
Your muscles are not just for movement.
They are your body’s most powerful blood sugar regulator.
Every step, every squat, every walk after dinner is a small investment in long-term metabolic health.
And those investments add up.
Key Takeaways: What This Means for You
1. Your muscles control most of your blood sugar after meals.
After you eat, up to 70–80% of the glucose cleared from your bloodstream goes into skeletal muscle. If your muscles are healthy and responsive, blood sugar rises gently and returns to normal efficiently.
2. Insulin doesn’t work alone — your muscles must respond.
Insulin is the signal, but muscle is the engine. If muscle cells become resistant to insulin, glucose stays in your bloodstream longer, increasing metabolic stress.
3. Movement after meals is powerful medicine.
Even a 10–15 minute walk after eating activates a separate pathway inside muscle cells that helps pull glucose out of the blood — even if insulin isn’t working perfectly.
4. Muscle quality matters as much as muscle size.
It’s not just about having bigger muscles. Muscles that are well-conditioned, rich in mitochondria, and supported by good cardiovascular fitness handle glucose far more efficiently.
5. Aging naturally weakens this system — but it’s reversible.
After age 30, muscle mass gradually declines. Over time, this reduces your body’s glucose-buffering capacity. However, resistance training and aerobic exercise can rebuild muscle function at almost any age.
6. What you eat affects your muscle cells at the gene level.
Excess saturated fat can impair insulin signaling inside muscle. A diet rich in whole foods, fiber, lean protein, and healthy fats supports muscle insulin sensitivity.
7. Fasting glucose is only part of the story.
Post-meal glucose spikes may reveal early metabolic dysfunction before fasting numbers change. Monitoring after-meal responses provides a clearer picture of metabolic health.
Bottom line:
Strong, metabolically fit muscles are one of the most powerful tools you have to prevent insulin resistance, protect against type 2 diabetes, and maintain long-term metabolic resilience.
Author’s Note
As a physician and student of metabolic science, I have spent decades treating patients with type 2 diabetes, cardiovascular disease, obesity, and age-related metabolic decline. What continues to strike me is how often skeletal muscle — one of the most powerful regulators of glucose metabolism — is overlooked in routine discussions about blood sugar control.
In clinical practice, we frequently focus on medications, lab numbers, and dietary restrictions. While these are important, they represent only part of the equation. The science is clear: skeletal muscle is not merely a structure for movement; it is a dynamic metabolic organ. It responds to lifestyle, adapts to training, and directly determines how efficiently your body handles glucose after every meal.
This chapter was written to bridge the gap between molecular research and practical application. The studies referenced here are drawn from current peer-reviewed literature in physiology, endocrinology, and metabolic science. My goal is not only to explain what happens inside muscle cells, but to help you understand why it matters and how you can act on that knowledge.
Whether you are a patient seeking better glucose control, a healthcare professional refining your understanding, or someone committed to long-term healthy aging, I hope this chapter reinforces one essential truth:
Muscle is not optional in metabolic health — it is foundational.
Small, consistent actions — walking after meals, strength training regularly, eating thoughtfully — can reshape the biology inside your cells. The human body remains remarkably adaptable, even later in life.
Your muscles are not just part of your body. They are part of your medicine.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Losing Muscle With Age? It May Be Driving Your Insulin Resistance | DR T S DIDWAL
The Fat Inside Your Muscles: More Dangerous Than We Thought? | DR T S DIDWAL
Your Body Fat Is an Endocrine Organ—And Its Hormones Shape Your Heart Health | DR T S DIDWAL
hsCRP Explained: What Inflammation Means for Your Heart | DR T S DIDWAL
References
Drobiova, H., Alhamar, G., Ahmad, R., Al-Mulla, F., & Al Madhoun, A. (2025). GLUT4 trafficking and storage vesicles: Molecular architecture, regulatory networks, and their disruption in insulin resistance. International Journal of Molecular Sciences, 26(15), 7568. https://doi.org/10.3390/ijms26157568
Horlem, T., Carvalhal, S. R. S., Bonatto, S. J. R., & Fernandes, L. C. (2025). Molecular framework of the onset and progression of skeletal muscle aging. International Journal of Molecular Sciences, 26(20), 10145. https://doi.org/10.3390/ijms262010145
Joshi, S., Kesavadev, J., K M, P. K., Saboo, B., Mehta, A., Bhattacharyya, A., Sosale, A., Jabbar, P. K., Santosh, R., Deshmukh, V., Deka, N., & Samajdar, S. S. (2025). Postprandial glucose: A variable in continuum. Clinical Medicine Insights: Endocrinology and Diabetes, 18, 11795514251370507. https://doi.org/10.1177/11795514251370507
Opazo-Díaz, E., Corral-Pérez, J., Pérez-Bey, A., Marín-Galindo, A., Montes-de-Oca-García, A., Rebollo-Ramos, M., Velázquez-Díaz, D., Casals, C., & Ponce-González, J. G. (2025). Is lean mass quantity or quality the determinant of maximal fat oxidation capacity? The potential mediating role of cardiorespiratory fitness. Journal of the International Society of Sports Nutrition, 22(1), 2455011. https://doi.org/10.1080/15502783.2025.2455011
Petrie, M. A., Johnson, K. A., McCue, P. M., Aggarwal, A., Brown, T., Grevengoed, K., Hoyman, J., Kallner, A., Klein, J., Lloyd, M., Patterson, M., & Shields, R. K. (2025). Postprandial exercise attenuates glucose and insulin and is associated with age, cognitive function, and extracellular/intracellular body water. Physiological Reports, 13(22), e70657. https://doi.org/10.14814/phy2.70657
Zhu, M., Zhang, F., Mao, Y., & Kang, Z. (2026). Silencing of KLF9 alleviates palmitic acid-induced insulin resistance and inflammation in undifferentiated myoblasts by targeting GRB14, unveiling a pathogenic axis in Type 2 diabetic myopathy. Current Proteomics, 23(1), 100077. https://doi.org/10.1016/j.curpro.2026.100077