Losing Muscle With Age? It May Be Driving Your Insulin Resistance
Hidden sarcopenia may quietly drive insulin resistance and rising blood sugar. Learn how age-related muscle loss disrupts metabolism and increases diabetes risk.
DIABETESSARCOPENIA
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/7/202615 min read
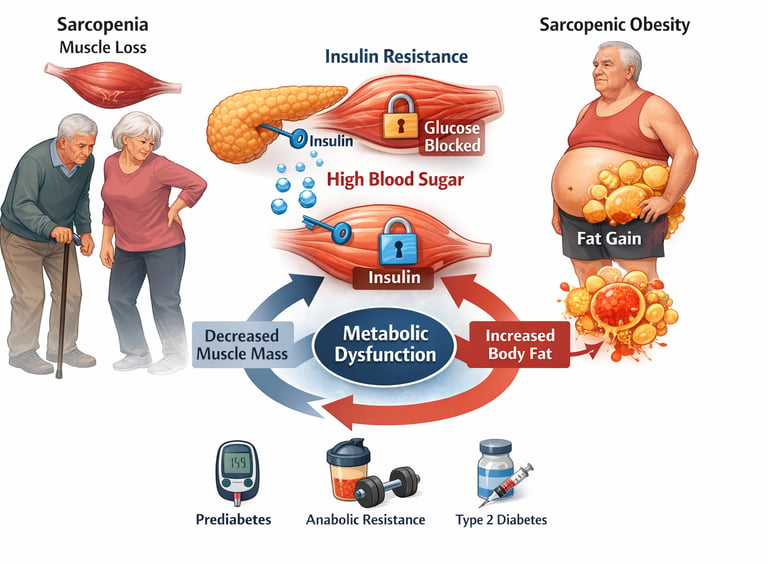
Many people assume that muscle loss and rising blood sugar are simply two separate problems that appear with age. But modern metabolic science tells a very different story. In reality, sarcopenia (age-related muscle loss) and insulin resistance are closely intertwined biological processes that influence each other in powerful ways. When muscle mass begins to decline, the body loses one of its most important metabolic tools for controlling blood sugar — and that change can quietly set the stage for prediabetes, metabolic syndrome, and type 2 diabetes (Du et al., 2026).
Skeletal muscle is far more than a structure that helps us move or lift objects. It is actually the largest glucose-disposal organ in the body, responsible for absorbing the majority of circulating glucose after a meal. In healthy muscle, insulin acts like a key that opens the cellular door, allowing glucose to enter and be stored or used as energy. But when muscle mass shrinks, or muscle cells become less responsive to insulin — a state known as insulin resistance — blood sugar remains elevated for longer periods. Over time, this places increasing stress on the pancreas and accelerates metabolic dysfunction (Ortez Toro, 2025).
Ageing muscles also face another challenge known as anabolic resistance — a reduced ability to respond to the normal signals that stimulate muscle growth, such as dietary protein and resistance exercise. Research suggests that this blunted response involves impaired amino-acid sensing, reduced activation of the mTOR pathway, chronic low-grade inflammation, and alterations in metabolic signalling pathways within muscle tissue (Didier et al., 2025).
As muscle mass declines and body fat gradually increases, many older adults develop a condition called sarcopenic obesity, where reduced muscle and excess fat combine to worsen inflammation, impair glucose metabolism, and increase cardiovascular risk (Benz et al., 2025). Understanding this muscle–metabolism connection is essential because protecting muscle health may be one of the most powerful strategies we have for preserving metabolic health throughout the ageing process.
To synthesize the complex relationship between sarcopenia and metabolism, here are five clinical pearls that bridge the gap between high-level molecular biology and practical patient care.
1. Muscle is a "Metabolic Sponge"
Scientific Perspective: Skeletal muscle accounts for approximately 80% of postprandial glucose disposal. Loss of muscle mass (sarcopenia) directly reduces the volume of the primary "sink" for circulating blood sugar, regardless of diet.
Think of your muscles as a sponge for sugar. When you lose muscle, your "sponge" gets smaller, leaving more sugar floating in your blood to cause damage. Building muscle isn't just about strength; it’s about giving your blood sugar a place to go.
2. The "Anabolic Threshold" Shifts Upward
Scientific Perspective: Aging induces anabolic resistance, where the mTORC1 signaling pathway requires a higher concentration of extracellular leucine to initiate muscle protein synthesis (MPS). A "standard" 20g protein dose may be sub-threshold for an older adult.
Your muscles become "hard of hearing" as you age. To get them to listen to the signal to grow, you have to "speak louder" by eating more protein at a single sitting (35–40g) rather than snacking on small amounts throughout the day.
3. BMI is a Mask for Sarcopenic Obesity
Scientific Perspective: Body Mass Index (BMI) cannot distinguish between adipose tissue and lean mass. Myosteatosis (fat infiltration into the muscle) can occur even if weight remains stable, severely impairing insulin sensitivity.
The scale can lie. You can weigh the same at 70 as you did at 30, but if you’ve swapped 10 lbs of muscle for 10 lbs of fat, your risk for diabetes skyrockets. Focus on your strength and how your clothes fit, not just the number on the scale.
4. The "Vicious Cycle" of Inflammation (Inflammaging)
Scientific Perspective: Pro-inflammatory cytokines like TNF-α and IL-6 act as metabolic "noise" that interferes with both insulin signaling and muscle repair. This creates a bidirectional loop where fat-driven inflammation melts muscle, and muscle loss worsens inflammation.
Chronic inflammation is like "metabolic rust." It jams the machinery that builds muscle and manages sugar. Staying active and eating anti-inflammatory foods (like omega-3s) acts like WD-40 for your metabolism, keeping the gears moving smoothly.
5. Resistance Training is "Medicine" for Insulin Receptors
Scientific Perspective: Mechanical loading (weight lifting) increases the expression and translocation of GLUT4 transporters to the cell membrane, improving glucose uptake independently of the insulin pathway.
Lifting weights is like a "backdoor" for blood sugar. It allows your body to process sugar even if your insulin isn't working perfectly. Every time you lift something heavy, you are essentially "cleaning out" your bloodstream.
What Is Sarcopenia — and Why Should You Care?
Sarcopenia is the gradual, age-related loss of skeletal muscle mass, strength, and function. It is not just "getting weaker with age." It is a recognised clinical syndrome with measurable consequences for metabolism, independence, and quality of life. After the age of 30, adults lose approximately 3–8% of their muscle mass per decade, and that rate accelerates significantly after 60.
But here is what makes sarcopenia especially important from a metabolic standpoint: your skeletal muscle is not just responsible for movement. It is the body's largest glucose-disposing organ, responsible for clearing up to 80% of blood sugar after a meal. When muscle mass declines, so does the body's ability to manage glucose effectively — setting the stage for insulin resistance, prediabetes, and eventually type 2 diabetes.
This connection between muscle loss and glucose dysregulation has emerged as one of the most clinically significant relationships in ageing medicine. As Ortez Toro (2025) explains, diabetes and sarcopenia are not separate conditions that happen to coexist in older adults — they are deeply entangled in a bidirectional metabolic relationship that accelerates the progression of both.
The Muscle–Glucose Connection: Why Muscle Mass Is Your Metabolic Engine
Think of skeletal muscle as your body's fuel tank and combustion engine rolled into one. When you eat carbohydrates, your pancreas releases insulin, which signals muscle cells to take up glucose from the bloodstream and store it as glycogen or burn it for energy. In a healthy, muscle-rich body, this process is fast and efficient.
In sarcopenia, that engine is smaller. Less muscle tissue means fewer insulin receptors, reduced glucose transporter (GLUT4) activity, and impaired mitochondrial function. The result? Blood sugar stays elevated for longer after meals, insulin must work harder to do its job, and over time, cells begin to ignore insulin signals altogether — a state known as insulin resistance.
Du et al. (2026) provide a comprehensive mechanistic breakdown of this relationship in elderly patients, identifying impaired insulin signalling pathways, increased intramuscular fat infiltration (myosteatosis), and chronic low-grade inflammation as the three major drivers of insulin resistance in sarcopenic older adults. Crucially, their work confirms that this is a bidirectional relationship: not only does muscle loss worsen insulin resistance, but insulin resistance accelerates muscle breakdown, creating a self-perpetuating cycle that is very difficult to interrupt once established.
This is why treating blood sugar and preserving muscle cannot be separated. They are two sides of the same coin.
Anabolic Resistance: Why Your Muscles Stop Responding to Protein
In a young, healthy body, eating a protein-rich meal or completing a resistance training session sends a clear signal: build more muscle. Amino acids flood into the bloodstream, mTOR (the master growth regulator) switches on, muscle protein synthesis ramps up, and new fibres are laid down. This is anabolism — the constructive side of metabolism.
In older adults, this response is blunted. The muscle becomes progressively less sensitive to the two most powerful anabolic stimuli — dietary protein and physical exercise. This phenomenon is called anabolic resistance, and it is now recognised as the central mechanism driving sarcopenia.
Didier, Messaoudi, and Magne (2025) offer one of the most detailed explorations of anabolic resistance to date, identifying multiple overlapping factors:
Impaired amino acid sensing: Older muscle cells are less efficient at detecting rising amino acid levels in the blood, meaning the "build signal" is delayed or weakened.
Blunted mTORC1 activation: The intracellular signalling cascade that triggers muscle protein synthesis becomes sluggish with age.
Increased splanchnic extraction: A greater proportion of ingested amino acids is captured by the gut and liver before reaching muscle tissue, reducing the amino acid "dose" available for muscle building.
Chronic low-grade inflammation: Elevated inflammatory markers — especially IL-6 and TNF-α — directly interfere with anabolic signalling, essentially jamming the "build" signal at the cellular level.
Gut microbiome dysbiosis: Emerging evidence suggests that age-related shifts in gut bacteria further impair amino acid absorption and inflammatory regulation, adding another layer of complexity.
The practical implication of anabolic resistance is significant: older adults need more dietary protein, not less, to achieve the same muscle-building stimulus as younger people. Current evidence suggests a threshold of 35–40 grams of high-quality protein per meal (compared to 20–25 grams for younger adults) to meaningfully activate muscle protein synthesis — a recommendation that challenges the traditionally modest protein targets in many clinical nutrition guidelines.
Hormonal Modulation: The Endocrine Forces Governing Muscle and Metabolism
Hormones are the chemical messengers that coordinate muscle maintenance, fat distribution, and glucose regulation. In ageing, a cascade of hormonal changes tips the balance away from muscle building and toward muscle loss and metabolic dysfunction.
Insulin and IGF-1
Beyond its role in glucose uptake, insulin is an anabolic hormone that promotes muscle protein synthesis and inhibits protein breakdown. When muscles become insulin resistant, they lose this anabolic benefit too — a double blow that accelerates sarcopenia. Similarly, insulin-like growth factor 1 (IGF-1), which works in tandem with insulin to promote muscle growth, declines significantly with age.
Sex Hormones
Testosterone and oestrogen both support muscle mass maintenance. Their age-related decline — gradual in men (andropause), more abrupt in women (menopause) — contributes meaningfully to accelerated muscle loss in midlife and beyond.
Adiponectin and the Fat–Muscle Axis
One of the most illuminating recent findings concerns adiponectin, a hormone secreted by fat tissue that normally has insulin-sensitising and anti-inflammatory effects in muscle. In healthy adults, adiponectin acts as a protective signal — improving glucose uptake, reducing inflammation, and supporting muscle protein metabolism.
Surina et al. (2026) present compelling in vivo and in vitro evidence that ageing is associated with adiponectin resistance in skeletal muscle — meaning that even when adiponectin levels are adequate, the muscle stops responding to it properly. Their work identifies downregulation of adiponectin receptors (AdipoR1 and AdipoR2) in aged skeletal muscle as the likely mechanism, implicating the AMPK and PPARα signalling pathways as key disrupted nodes. This finding is particularly important because it suggests that sarcopenia is not simply a problem of too little anabolic stimulation — it also involves the loss of protective metabolic signalling that normally keeps muscle healthy throughout life. Restoring adiponectin sensitivity may emerge as a novel therapeutic target in the years ahead.
Cortisol and the Catabolic Burden
Cortisol, the primary stress hormone, promotes muscle protein breakdown. Chronic stress, poor sleep (both common in ageing), and systemic inflammation all elevate baseline cortisol, imposing a persistent catabolic load on muscle tissue that even robust anabolic signals struggle to overcome.
Sarcopenic Obesity: The Most Dangerous Body Composition Phenotype
Perhaps no concept better illustrates the convergence of muscle loss and metabolic dysfunction than sarcopenic obesity — a condition in which reduced muscle mass coexists with excess body fat, particularly visceral (abdominal) fat.
At first glance, sarcopenic obesity seems paradoxical: how can someone be both too thin (in terms of muscle) and too heavy (in terms of fat) at the same time? The answer lies in a concept called body composition shift. As we age, muscle tissue is gradually replaced by adipose (fat) tissue — often without a dramatic change in total body weight. Someone who weighs the same at 70 as they did at 40 may have lost 10 kilograms of muscle and gained 10 kilograms of fat. Standard BMI measurements would miss this entirely.
Sarcopenic obesity is metabolically devastating for several reasons:
Less muscle means reduced glucose disposal capacity, driving insulin resistance.
Excess visceral fat secretes inflammatory cytokines that further impair anabolic signalling and insulin sensitivity.
The combination creates a pro-inflammatory, pro-diabetic state that dramatically elevates cardiovascular risk.
Physical function declines faster, reducing activity levels and accelerating the cycle.
Benz et al. (2025) tackle this challenge head-on with the development of the Sarcopenic Obesity Phenotype Index (SOPi) — a novel population-based composite measure designed to identify individuals at the highest risk. Using data from a large European cohort, the SOPi integrates measures of appendicular lean mass, waist circumference, and functional performance to create a single clinically usable score. The index significantly outperforms either measure alone in predicting metabolic complications and disability outcomes, offering a practical screening tool for clinicians working with ageing populations. This work represents a meaningful step toward standardising diagnosis and treatment thresholds for sarcopenic obesity in routine clinical care.
Mitochondrial Dysfunction and Insulin Resistance
Age-related decline in mitochondrial function reduces the muscle cell’s ability to efficiently oxidize fatty acids. When fatty acid oxidation becomes impaired, excess lipid intermediates such as diacylglycerols and ceramides accumulate within skeletal muscle fibers, a phenomenon known as intramyocellular lipid accumulation. These lipid metabolites interfere with key steps in the insulin signaling pathway, particularly the insulin receptor substrate (IRS) and downstream signaling cascades, ultimately impairing glucose uptake. As a result, mitochondrial dysfunction contributes directly to insulin resistance in aging skeletal muscle, reinforcing the metabolic cycle that links sarcopenia with impaired glucose regulation.
The Vicious Cycle: How It All Connects
Understanding sarcopenia and insulin resistance together requires appreciating how each condition feeds the other in a compounding loop:
Muscle loss → reduced glucose disposal → elevated blood sugar → compensatory hyperinsulinemia → anabolic resistance worsens → more muscle loss
Inflammation → disrupts anabolic signalling → accelerates muscle breakdown → increases fat infiltration → worsens insulin resistance → more inflammation
Hormonal decline → reduced anabolic stimulation → muscle loss accelerates → adiposity increases → adiponectin resistance develops → worsens metabolic signalling
Each arrow in this cycle can become a therapeutic entry point. Interrupting the cycle at multiple points simultaneously — through nutrition, exercise, hormonal optimisation, and anti-inflammatory strategies — is more effective than targeting any single pathway in isolation.
Key Takeaways: Sarcopenia, Aging, and Insulin Resistance
Skeletal muscle is the body’s primary glucose-disposal organ. After a meal, nearly 70–80% of circulating glucose is cleared by skeletal muscle. When muscle mass declines with age, the body loses a major metabolic sink for glucose, increasing the risk of insulin resistance, prediabetes, and type 2 diabetes (Du et al., 2026).
Sarcopenia is not simply weakness—it is a metabolic disorder. Age-related loss of muscle mass and strength affects not only mobility and physical function but also whole-body metabolic regulation, including glucose control, lipid metabolism, and inflammatory balance.
Muscle loss and insulin resistance reinforce each other in a vicious cycle. Reduced muscle mass limits glucose uptake, raising blood sugar levels. In response, the pancreas produces more insulin, leading to chronic hyperinsulinemia, which further impairs anabolic signalling and accelerates muscle breakdown (Ortez Toro, 2025).
Anabolic resistance is a central driver of sarcopenia. With ageing, skeletal muscle becomes less responsive to the two strongest muscle-building signals: dietary protein and resistance exercise. Impaired amino-acid sensing, reduced activation of the mTOR pathway, and chronic inflammation all contribute to this blunted anabolic response (Didier et al., 2025).
Intramuscular fat accumulation worsens insulin signalling. As muscle tissue declines, fat increasingly infiltrates muscle fibres—a condition known as myosteatosis. This lipid accumulation disrupts insulin signalling pathways and contributes to metabolic dysfunction.
Chronic low-grade inflammation accelerates both conditions. Elevated inflammatory cytokines such as IL-6 and TNF-α interfere with insulin receptor signalling and suppress muscle protein synthesis, promoting both insulin resistance and muscle atrophy.
Hormonal changes amplify metabolic decline. Age-related reductions in testosterone, oestrogen, growth hormone, and IGF-1 shift the body’s metabolic balance away from muscle preservation and toward fat accumulation and catabolism.
Sarcopenic obesity represents the highest-risk phenotype. When low muscle mass coexists with excess visceral fat, the combined metabolic effects dramatically increase the risk of cardiovascular disease, metabolic syndrome, disability, and premature mortality (Benz et al., 2025).
Lifestyle interventions remain the most effective therapy. Resistance training, higher protein intake (approximately 1.2–1.6 g/kg/day), improved sleep, and anti-inflammatory dietary patterns can significantly improve muscle health and insulin sensitivity.
Protecting muscle is protecting metabolism. Preserving skeletal muscle mass throughout adulthood is one of the most powerful strategies for maintaining metabolic health, preventing diabetes, and promoting healthy ageing.
Frequently Asked Questions (FAQs)
FAQ 1: What is the difference between normal age-related muscle loss and sarcopenia?
Some degree of muscle loss is a natural part of ageing — most adults lose a small amount of muscle mass each decade after 30. Sarcopenia, however, is a clinical syndrome defined by low muscle mass combined with reduced muscle strength or impaired physical performance. It goes beyond what is expected for age and is associated with measurable functional decline, increased fall risk, and metabolic complications. If you find that everyday tasks like climbing stairs, carrying groceries, or rising from a chair are becoming significantly harder, it is worth discussing a formal assessment with your doctor.
FAQ 2: Can I have sarcopenia if I am not underweight or "thin"?
Absolutely — and this is one of the most important misconceptions to address. Many people with sarcopenia have a normal or even elevated BMI because fat tissue has gradually replaced lost muscle tissue. This is called sarcopenic obesity, and it is particularly common and dangerous. Someone can appear a healthy weight while having significantly depleted muscle reserves. Standard BMI does not detect this shift in body composition, which is why more specialised assessments (like DEXA scans or grip strength tests) are increasingly recommended for older adults.
FAQ 3: How does sarcopenia actually cause insulin resistance?
Skeletal muscle is the body's primary site for disposing of blood sugar after meals. When muscle mass declines, there is simply less tissue available to absorb and process glucose efficiently. Additionally, ageing muscle accumulates more intramuscular fat, which interferes with insulin signalling at the cellular level. Chronic inflammation associated with sarcopenia also disrupts the insulin signalling cascade. The result is that blood sugar stays elevated for longer after meals, insulin must be secreted in larger amounts to compensate, and eventually cells become desensitised to insulin's signals — a state we call insulin resistance.
FAQ 4: What is anabolic resistance, and can it be reversed?
Anabolic resistance means that older muscles respond less strongly to the two main stimuli that normally trigger muscle growth: protein intake and physical exercise. It does not mean the muscle cannot grow at all — it means the threshold for triggering growth is higher. In practical terms, this means older adults need larger amounts of high-quality protein per meal (particularly leucine-rich sources) and more consistent resistance training to achieve the same muscle-building effect as younger people. While anabolic resistance cannot be completely "cured," its effects can be significantly countered with appropriate nutrition, exercise, and management of the underlying inflammatory and hormonal factors that drive it.
FAQ 5: What role do hormones play in muscle loss with age?
Several key hormones decline with age in ways that directly impair muscle maintenance. Testosterone and oestrogen both promote muscle protein synthesis and are lost gradually (in men) and more rapidly (in women around menopause). Growth hormone and IGF-1 — which stimulate muscle repair and growth — also decline. A less widely known but important finding is that skeletal muscle can develop adiponectin resistance with age, meaning the muscle stops responding to this normally protective metabolic hormone even when blood levels are adequate. The combined effect of these hormonal changes is a shift in the body's metabolic balance away from muscle building and toward fat accumulation and catabolism.
FAQ 6: Is sarcopenic obesity more dangerous than either sarcopenia or obesity alone?
Yes — research consistently shows that the combination of low muscle mass and excess body fat carries greater health risks than either condition in isolation. Sarcopenic obesity is associated with significantly higher rates of cardiovascular disease, type 2 diabetes, functional disability, and mortality compared to sarcopenia or obesity alone. This is because the two conditions compound each other metabolically: less muscle worsens glucose metabolism, while excess fat drives the inflammation that further impairs muscle function. The Sarcopenic Obesity Phenotype Index (SOPi) developed by Benz et al. (2025) was specifically designed to help clinicians identify and risk-stratify individuals with this combined phenotype, which had previously been underdiagnosed due to limitations of standard BMI-based assessments.
FAQ 7: What are the most effective lifestyle strategies to protect muscle mass and insulin sensitivity as I age?
The most evidence-backed strategies are:
Resistance training at least two to three times per week — this is the single most effective tool for both building muscle and improving insulin sensitivity at any age.
Higher protein intake distributed across meals, with particular attention to leucine-rich sources (eggs, dairy, lean meat, legumes) to overcome anabolic resistance.
Reducing prolonged sedentary behaviour — even light physical activity throughout the day (walking, standing) meaningfully improves glucose metabolism.
Anti-inflammatory nutrition — Mediterranean-style eating patterns rich in omega-3s, colourful vegetables, and polyphenols help reduce the chronic inflammation that drives both sarcopenia and insulin resistance.
Prioritising sleep and stress management — both poor sleep and chronic stress elevate cortisol, which promotes muscle breakdown and worsens blood sugar control.
Regular metabolic monitoring — knowing your fasting glucose, HbA1c, and if possible, your muscle-to-fat ratio- helps you and your healthcare team catch problems early and intervene before the cycle becomes entrenched.
Starting any of these changes — even one — is worthwhile. The biology is clear: it is never too early, and never too late, to invest in your muscle health.
Author’s Note
The intersection between sarcopenia, aging, and insulin resistance represents one of the most important yet often overlooked areas in modern metabolic medicine. For decades, muscle loss in older adults was largely accepted as an unavoidable consequence of aging. At the same time, rising blood sugar and insulin resistance were typically viewed as separate metabolic disorders. However, emerging research now shows that these conditions are deeply interconnected and frequently reinforce each other through shared biological pathways.
Skeletal muscle is not merely a structural tissue responsible for strength and mobility. It is also a central metabolic organ that plays a critical role in regulating glucose metabolism, insulin sensitivity, and overall energy balance. When muscle mass declines with age, the body loses a major site for glucose disposal, which can contribute to rising blood sugar levels and metabolic dysfunction. Conversely, insulin resistance itself can impair anabolic signaling within muscle cells, accelerating muscle breakdown and reducing the body’s ability to maintain healthy muscle tissue.
This chapter was written with two goals in mind. First, to help readers understand the biological mechanisms that connect muscle health with metabolic health, and second, to translate complex scientific research into clear, practical insights that patients and clinicians can use. While the molecular pathways involved—such as anabolic resistance, inflammation, mitochondrial dysfunction, and hormonal changes—may seem complex, the practical message is straightforward: protecting muscle mass is one of the most powerful strategies for preserving metabolic health across the lifespan.
Fortunately, research consistently shows that lifestyle interventions—particularly resistance exercise, adequate protein intake, and metabolic health management—can meaningfully slow or even partially reverse the trajectory of muscle loss and insulin resistance. The science continues to evolve rapidly, and future therapies may further enhance our ability to maintain muscle function and metabolic resilience well into older age.
Understanding this muscle–metabolism connection is essential not only for preventing chronic disease but also for promoting healthy, functional aging and long-term independence.your metabolism — at every age.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
The 5 Pillars of Muscle Health: How to Prevent Muscle Loss as You Age | DR T S DIDWAL
Can Your Brain Actually Regrow New Cells? What the Latest Neuroscience Reveals | DR T S DIDWAL
Can Healthy Mitochondria Protect Against Sarcopenia and Age-Related Decline? | DR T S DIDWAL
Stopping GLP-1: Will the Weight Come Back? | DR T S DIDWAL
Sarcopenic Obesity: How to Lose Fat Safely Without Losing Muscle | DR T S DIDWAL
References
Benz, E., Pinel, A., Guillet, C., Capel, F., Pereira, B., Rizopoulos, D., Cruz-Jentoft, A. J., Eglseer, D., Topinkova, E., Barazzoni, R., Donini, L. M., Rivadeneira, F., Steur, M., Voortman, T., Weijs, P. J. M., Schoufour, J. D., & Boirie, Y. (2025). Sarcopenic obesity phenotype index (SOPi): A population-based study. Journal of Cachexia, Sarcopenia and Muscle, 16(5), e70099. https://doi.org/10.1002/jcsm.70099
Didier, C., Messaoudi, I., & Magne, H. (2025). Exploring the multifactorial causes and therapeutic strategies for anabolic resistance in sarcopenia (Version 2). bioRxiv. https://doi.org/10.1101/2025.09.12.675977
Du, H., Qu, Y., Huai, Z., & Wang, S. (2026). Mechanisms of insulin resistance in elderly sarcopenia patients and their bidirectional relationship. Geriatric Nursing, 69, 103907. https://doi.org/10.1016/j.gerinurse.2025.103907
Ortez Toro, J. J. (2025). Diabetes and sarcopenia: Unraveling the metabolic crossroads of muscle loss and glycemic dysregulation. Endocrines, 6(3), 47. https://doi.org/10.3390/endocrines6030047
Surina, S., Scisciola, L., Basilicata, M. G., et al. (2026). Age-related adiponectin resistance in human skeletal muscle dysfunction: In vivo and in vitro evidence. Journal of Translational Medicine. https://doi.org/10.1186/s12967-026-07959-9