SARMs vs Anabolic Steroids: Are They Really Safer? Benefits, Risks, and Clinical Evidence
SARMs promise muscle growth with fewer side effects—but is it true? Explore clinical evidence on safety, performance, and hormone impact.
EXERCISEAGING
Dr. T.S. Didwal, M.D.(Internal Medicine)
4/25/202616 min read
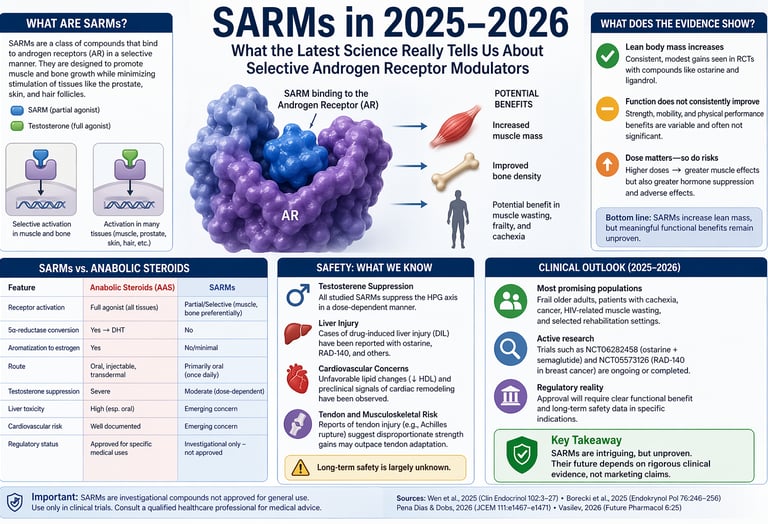
Selective androgen receptor modulators (SARMs) are investigational drugs that can modestly increase lean muscle mass but do not consistently improve strength or physical performance. They suppress natural testosterone and may cause liver and cardiovascular effects. As of 2026, no SARM is approved for clinical use, and long-term safety remains uncertain.
SARMs vs Steroids: Key Differences
Muscle Growth:
SARMs → Modest increase
Steroids → Strong, rapid increaseStrength & Performance:
SARMs → Inconsistent improvement
Steroids → Significant enhancementTestosterone Suppression:
SARMs → Moderate, dose-dependent
Steroids → Severe, prolongedLiver Risk:
SARMs → Possible (emerging DILI cases)
Steroids → High (especially oral forms)Cardiovascular Risk:
SARMs → Emerging (↓ HDL)
Steroids → Well-established riskHormone Conversion:
SARMs → No DHT or estrogen conversion
Steroids → Convert to DHT & estrogenApproval Status:
SARMs → Not approved (investigational)
Steroids → Approved for medical useLong-Term Safety:
SARMs → Unknown
Steroids → Well-documented risks
One-line takeaway:
SARMs are not “safe steroids”—they offer milder anabolic effects with uncertain long-term safety, while steroids are more potent but carry well-established risks.
Clinician’s Perspective on SARMs
SARMs are still experimental—not approved treatments
Despite years of research, no selective androgen receptor modulator has been approved for routine clinical use. Most experts, including recent endocrinology reviews, consider them investigational drugs best limited to clinical trials (Pena Dias & Dobs, 2026).They can increase muscle mass—but function matters more
Clinical studies show modest increases in lean body mass. However, these gains do not consistently improve strength, mobility, or independence, which are the outcomes that truly impact quality of life (Wen et al., 2025).Natural testosterone production is suppressed
A key concern is that SARMs reduce your body’s own testosterone levels, sometimes significantly. Recovery usually occurs after stopping, but the timeline is unpredictable—especially with repeated or prolonged use (Vasilev, 2026).“Safer than steroids” does not mean safe
SARMs may cause fewer androgenic effects (like prostate enlargement or gynecomastia), but they are not risk-free. Reported issues include:Liver injury
Unfavorable cholesterol changes
Possible cardiovascular effects
These risks are still being actively studied (Vasilev, 2026).
Unregulated products are a major hidden danger
Many products sold online as “SARMs” are mislabelled or contaminated, sometimes containing anabolic steroids or other drugs. This makes unsupervised use particularly risky.Where they might have a future
Research suggests potential roles in:Frailty and age-related muscle loss
Cancer-related cachexia
Muscle preservation during weight loss therapies
However, these uses require larger, long-term trials before clinical adoption (Borecki et al., 2025).
Practical takeaway for patients
If you’re concerned about muscle loss, the current evidence-based approach remains:Resistance training
Adequate protein intake
Management of underlying conditions
Hormone evaluation when appropriate
Monitoring Protocol for Clinicians
Baseline: ALT/AST, Lipid Profile, Total/Free Testosterone, PSA.
During Use (if in trial): Monthly liver enzyme checks and HDL/LDL monitoring.
Bottom line: SARMs are scientifically interesting but clinically unproven. Until stronger safety and functional outcome data emerge, they should be approached with caution and only within medical research settings.
Selective androgen receptor modulators (SARMs) were once heralded as the long-awaited solution to one of medicine’s most persistent dilemmas: how to harness the anabolic power of androgens without their systemic toxicity. In theory, SARMs could stimulate muscle and bone growth while sparing tissues such as the prostate, skin, and cardiovascular system—offering a targeted alternative to traditional anabolic androgenic steroids (AAS). For patients with sarcopenia, cancer cachexia, or chronic illness–related muscle loss, the implications were profound. Yet, despite more than two decades of research, no SARM has received full regulatory approval for routine clinical use as of 2026 (Pena Dias & Dobs, 2026).
Why has such a promising class of compounds failed to translate into clinical practice? The answer lies in a growing disconnect between biological plausibility and clinical reality. Recent high-quality evidence shows that while SARMs consistently increase lean body mass, these gains do not reliably translate into meaningful improvements in strength, mobility, or functional independence—the outcomes that matter most to patients (Wen et al., 2025). At the same time, safety signals once thought to be negligible are becoming increasingly difficult to ignore, including testosterone suppression, adverse lipid changes, and emerging reports of drug-induced liver injury (Vasilev, 2026).
This evolving evidence base forces a critical reassessment. Are SARMs truly “selective,” or simply less potent versions of existing androgens with a different risk profile? And more importantly, where—if anywhere—do they fit in modern clinical medicine? Drawing on the latest systematic reviews, clinical perspectives, and pharmacological analyses, this article examines what the science in 2025–2026 actually tells us—separating enduring promise from persistent limitations (Borecki et al., 2025).
What Are SARMs? A Plain-Language Explanation
Every cell in the body that responds to hormones like testosterone does so through a molecular switch called the androgen receptor (AR). When testosterone binds to the AR, it triggers a cascade of effects — muscle growth, bone strengthening, red blood cell production, and also androgenic effects such as prostate growth, acne, and voice deepening.
Classic anabolic androgenic steroids (AAS) are synthetic versions of testosterone. They are full AR agonists: they activate the receptor powerfully in essentially every tissue it is expressed in. This is why they build muscle so effectively — and also why they cause so many side effects.
SARMs take a fundamentally different approach. As Vasilev (2026) explains in a detailed molecular comparison, SARMs are primarily non-steroidal compounds that bind the AR but induce a slightly different three-dimensional shape in the receptor. This different conformation changes which co-activator proteins are recruited, meaning the gene expression that follows differs by tissue. In theory, a SARM could activate muscle and bone genes strongly while barely touching prostate or skin genes.
Crucially, SARMs also lack the metabolism pathways that amplify steroid effects. Unlike testosterone, they are not converted by the enzyme 5α-reductase into the more potent dihydrotestosterone (DHT), and they do not aromatise into oestrogen. Both of those conversion pathways are responsible for major side effects of classic AAS — DHT drives prostate growth and hair loss, while oestrogen (from aromatisation) can cause gynaecomastia (breast tissue growth in men). By avoiding both, SARMs offer at least a theoretical safety advantage.
0 SARMs with full regulatory approval for clinical use worldwide (as of 2026)
102 Pages of Clinical Endocrinology dedicated to the 2025 SARM physical performance systematic review
24–36 h Elimination half-life of SARMs like ligandrol — enabling convenient once-daily oral dosing
What Do Randomised Controlled Trials Actually Show? Physical Performance Evidence
The most comprehensive evidence synthesis available is the 2025 systematic review by Wen and colleagues published in Clinical Endocrinology. Examining randomised controlled trials of SARMs and their effects on physical performance, this review provides the most rigorous picture of what these compounds actually do — and what they do not do — in controlled human studies.
Lean Body Mass: A Consistent Gain, But Is It Enough?
Across multiple trials reviewed by Wen et al. (2025), SARMs consistently produced modest but measurable increases in lean body mass. This finding holds across the most commonly studied compounds — ostarine (enobosarm/MK-2866) and ligandrol (LGD-4033). In cancer patients with muscle wasting, phase II studies of ostarine showed dose-dependent increases in lean mass. In healthy young men given ligandrol, lean mass increased in a dose-dependent manner over short trial periods.
However, the critical finding — echoed by both Wen et al. (2025) and Vasilev (2026) — is that these lean mass gains do not reliably translate into proportional improvements in functional outcomes. Strength gains were variable, and improvements in physical performance metrics such as stair-climb power or walking speed were inconsistent across trials.
Key Research Finding
Pena Dias and Dobs (2026) of Johns Hopkins University note that while multiple SARMs have shown efficacy in increasing lean body mass in clinical trials, few have demonstrated consistent improvements in functional outcomes such as strength or mobility. This distinction between surrogate endpoints (lean mass) and clinical endpoints (function) is central to the ongoing regulatory challenge.
The Dose–Response Relationship
One important pattern across the RCT evidence is dose-dependence: higher doses of SARMs tend to produce greater anabolic effects but also greater endocrine suppression. This is not the clean tissue-selectivity that was originally hoped for — it suggests that the HPG (hypothalamic–pituitary–gonadal) axis is being activated centrally regardless of the peripheral tissue profile.
SARMs for Cachexia and Frailty: The Most Promising Clinical Territory
Borecki, Byczkiewicz, and Słowikowska-Hilczer (2025) published a focused review in Endokrynologia Polska examining SARMs specifically for cachexia (severe muscle loss associated with cancer and chronic disease) and frailty syndrome (the age-related decline in physical reserve). This represents arguably the most compelling potential clinical niche for these drugs.
Why Cachexia and Frailty Are So Hard to Treat
Cachexia affects millions of cancer patients, causing devastating loss of muscle mass that worsens prognosis independent of the underlying disease. Frailty — characterised by weakness, exhaustion, slow gait, low activity, and unintentional weight loss — affects a substantial proportion of adults over 65 and significantly increases the risk of falls, hospitalisation, and death.
Existing treatments are limited. Traditional anabolic steroids carry cardiovascular and hepatic risks that make them unsuitable for frail or seriously ill patients. This is precisely why SARMs have attracted such interest in these populations: if tissue-selectivity holds, patients might gain muscle and strength without the serious side effects.
Current Evidence in Cachexia and Frailty
The 2025 review by Borecki et al. confirms that SARMs show measurable anabolic activity in preclinical and early clinical models of muscle wasting. The oral route of administration is particularly valuable for ill or elderly patients who would struggle with injections. The relatively predictable pharmacokinetics of SARMs — once-daily dosing, stable plasma concentrations — are also clinically attractive in these populations.
That said, Borecki et al. (2025) are careful to note that the evidence base remains limited by short-duration trials, small sample sizes, and inconsistent functional endpoints. The clinical relevance of lean mass gains in frail elderly patients depends on whether those gains actually translate into meaningful improvements in daily function — and this has yet to be convincingly demonstrated at scale.
"SARMs are likely to enter clinical practice in select niches of men and women, especially among the frail, the elderly, or individuals with excessive loss of lean body mass."— Pena Dias & Dobs, The Journal of Clinical Endocrinology & Metabolism, 2026
SARMs vs. Classic Anabolic Steroids: A Head-to-Head Clinical Comparison
A detailed pharmacological review by Vasilev (2026) highlights an important reality: while selective androgen receptor modulators (SARMs) were designed to improve safety through tissue selectivity, current evidence suggests a more nuanced risk–benefit profile rather than a clear advantage over traditional anabolic androgenic steroids (AAS).
Mechanism of Action
Anabolic Steroids (AAS):
Act as full agonists at the androgen receptor, producing widespread activation across multiple tissues—including muscle, bone, prostate, skin, and cardiovascular system.SARMs:
Function as partial or tissue-selective agonists, aiming to preferentially stimulate muscle and bone while minimizing activation in androgen-sensitive tissues. However, this selectivity is incomplete in practice.
Metabolic Pathways
AAS:
Undergo conversion via:5α-reductase → DHT (linked to prostate growth, hair loss)
Aromatase → Estrogen (gynecomastia, fluid retention)
SARMs:
Do not convert to DHT or estrogen, reducing these specific side effects
Still exert systemic endocrine effects through androgen receptor signaling
Administration & Pharmacokinetics
AAS:
Available as oral (often hepatotoxic), injectable, or transdermal formulations with variable half-lives depending on esterification.SARMs:
Primarily oral agents with relatively predictable pharmacokinetics and half-lives of ~12–36 hours, allowing convenient once-daily dosing.
Endocrine Effects (HPG Axis)
AAS:
Cause profound suppression of endogenous testosterone, often requiring prolonged recovery and posing risks to fertility.SARMs:
Also suppress the hypothalamic–pituitary–gonadal axis, though typically in a dose-dependent and somewhat less severe manner. Recovery patterns remain insufficiently characterized.
⚠️ Safety Profile
Liver Effects:
AAS: High risk, particularly with 17α-alkylated oral forms
SARMs: Initially thought safer, but cases of drug-induced liver injury (DILI) have been reported
Cardiovascular Risk:
AAS: Well-established risks (dyslipidemia, cardiac remodeling)
SARMs: Emerging concerns, including reduced HDL cholesterol and preclinical cardiac changes
Regulatory Status & Evidence Base
AAS:
Approved for specific medical indications (e.g., hypogonadism), with decades of safety data—though misuse remains problematic.SARMs:
Remain investigational compounds with no regulatory approvals as of 2026. Long-term safety data are limited due to short-duration trials.
Clinical Takeaway
SARMs were developed to separate anabolic benefits from androgenic harm. However, current evidence suggests they are not simply “safe steroids”. Instead, they represent a pharmacological middle ground—offering modest anabolic effects with a still-evolving and incompletely understood safety profile.
The Safety Picture: What We Know and What We Don't
Safety is the axis on which the entire SARM story turns. The safety profile of classic AAS is well understood after decades of clinical observation. SARMs, by contrast, remain poorly characterised in terms of long-term outcomes. Here is what the 2025–2026 evidence tells us.
Testosterone Suppression: A Universal Finding
Despite not converting to DHT or oestrogen, SARMs consistently suppress the HPG axis in a dose-dependent manner. Vasilev (2026) highlights that this effect has been clearly demonstrated in clinical studies of ligandrol — central AR activation alone is sufficient to inhibit the body's own testosterone production. Although suppression is typically reversible after stopping a SARM, recovery kinetics after prolonged use are poorly defined. For younger men who may desire future fertility, this is a clinically significant concern.
Liver Safety: Not as Clean as Expected
⚠️ Safety Caution
Despite the absence of the 17α-alkylation that makes classic oral steroids hepatotoxic, cases of drug-induced liver injury (DILI) have been documented with SARMs including ostarine and RAD-140, in both clinical trial settings and real-world use. Presentations range from asymptomatic liver enzyme elevations to cholestatic injury. The mechanism is not yet fully understood (Vasilev, 2026).
Cardiovascular Signals: An Emerging Concern
Vasilev (2026) highlights emerging preclinical evidence of cardiac remodelling with ostarine in rodent models, including increased myocardial collagen deposition and signs of cardiac hypertrophy. Multiple SARMs also reduce HDL ("good") cholesterol, paralleling — though to a lesser degree — the cardiometabolic harms seen with AAS. The long-term clinical significance of these signals is unknown, but they underscore that SARMs are not cardiovascularly neutral.
Musculoskeletal Risk: A Paradox
One counterintuitive finding noted in Vasilev's (2026) review is that SARMs may predispose users to tendon injury — the same class of complication associated with AAS. A case of bilateral Achilles tendon rupture in a competitive powerlifter using SARMs is described, possibly related to disproportionate rapid muscle strength gains outpacing tendon adaptation. This matters practically for patients undertaking rehabilitation or exercise programmes.
Will SARMs Ever Be Approved? The Regulatory Reality
The Johns Hopkins perspective published in 2026 by Pena Dias and Dobs directly confronts this question, and their answer is carefully calibrated: possibly, but narrowly. They conclude that widespread approval is unlikely unless future trials can demonstrate both meaningful functional efficacy and robust long-term safety in targeted populations.
The path most likely to succeed, they suggest, involves specific, narrow indications — frail older adults with proven sarcopenia, patients with cancer-related cachexia, or individuals with conditions causing severe lean mass loss. These are populations where the potential benefit is greatest and the risk–benefit calculation tips more favourably.
Active clinical research continues. A trial (NCT06282458) evaluating ostarine for preventing muscle loss in patients receiving semaglutide (Ozempic/Wegovy) was completed in 2025, with results pending. Another trial (NCT05573126) is still recruiting, evaluating RAD-140 (vosilasarm) in advanced androgen-receptor positive breast cancer — a genuinely novel therapeutic direction.
✅ Areas of Genuine Promise
Pena Dias and Dobs (2026) identify several areas where SARMs show realistic clinical potential: older adults with frailty and sarcopenia, patients with cancer or HIV-related muscle wasting, post-surgical rehabilitation support in selected patients, and potentially as adjuncts in androgen receptor-positive breast cancer. The oral route and favourable pharmacokinetics make them particularly suited to populations who cannot tolerate injections.
Practical Applications: What This Means in Real Life
Frail Older Adults
If confirmed by larger trials, SARMs may help preserve independence in elderly patients facing sarcopenia. Current evidence suggests modest lean mass gains — discuss monitoring protocols with your physician if you are at high fall risk.
Cancer and Cachexia Patients
For oncology patients losing significant muscle mass, SARMs remain investigational. Enrol in clinical trials where possible; avoid unregulated market products, which are frequently mislabelled or contaminated.
Fitness and Athletic Context
SARMs are prohibited in competitive sport by WADA. Non-medical recreational use carries real risks — including liver injury, testosterone suppression, and cardiovascular effects — without the protection of medical supervision.
Patients on GLP-1 Drugs (Semaglutide)
Emerging research (including the 2025 NCT06282458 trial) explores whether SARMs can protect lean mass during weight loss on GLP-1 agonists. Results are eagerly awaited — this could represent a clinically important application.
Frequently Asked Questions About SARMs
1. Are SARMs legal to buy and use?
The legal status of SARMs varies by country. In the United States and most of Europe, SARMs are not approved for human use and are classified as investigational drugs. They cannot legally be sold as dietary supplements. The FDA has issued specific warnings against body-building products containing SARMs. Outside of formally approved clinical trials, there is no legal route to access them as a patient (Vasilev, 2026; Pena Dias & Dobs, 2026).
2. Do SARMs work for building muscle and improving physical performance?
Yes — but with important caveats. Clinical trials confirm that SARMs including ostarine and ligandrol produce modest, dose-dependent increases in lean body mass. However, the systematic review by Wen et al. (2025) found that these gains do not consistently translate into meaningful improvements in strength or physical function. They are not as potent as classic anabolic steroids and fall well short of what some marketing claims suggest.
3. Are SARMs safer than anabolic steroids?
SARMs appear to cause fewer androgenic side effects than classic AAS — they do not convert to DHT or oestrogen, making them less likely to cause prostate effects, hair loss, or gynaecomastia. However, they are not risk-free. Evidence documents testosterone suppression, adverse lipid changes, emerging liver injury signals, and preclinical cardiovascular concerns. Their long-term safety profile is largely unknown (Vasilev, 2026).
4. Can SARMs be used to treat cancer-related muscle wasting?
SARMs represent a promising — though still investigational — approach to cachexia and frailty. The 2025 review by Borecki et al. confirms biologically plausible mechanisms and early clinical signals supporting their potential in this context. However, no SARM is currently approved for this indication, and patients should ideally access them only through formal clinical trials where safety monitoring is in place.
5. Will SARMs suppress my body's natural testosterone production?
Yes — this is one of the most consistent findings in the clinical literature. All studied SARMs suppress the HPG axis in a dose-dependent manner. Your body's own testosterone production will decline during use. For most people, this recovers after stopping, but recovery can be slow, and the kinetics after prolonged or repeated use are not well characterised. Men concerned about fertility should consider this carefully (Vasilev, 2026; Wen et al., 2025).
6. What is the difference between SARMs and testosterone replacement therapy (TRT)?
TRT uses testosterone itself (or closely related compounds) to restore physiological hormone levels in genuinely deficient men. SARMs are experimental compounds intended to deliver selective anabolic effects without full androgenic activation. TRT has decades of safety data and regulatory approval for hypogonadism. SARMs have neither. If you are experiencing genuine testosterone deficiency, TRT under medical supervision is the evidence-based choice — SARMs are not a substitute.
7. Can SARMs help with the muscle loss that occurs with GLP-1 weight loss drugs like semaglutide?
This is an active and exciting area of research. A clinical trial (NCT06282458) specifically evaluated whether ostarine could prevent lean mass loss in patients taking semaglutide for weight loss; results had not been published as of this article. Given that GLP-1 drugs can cause significant lean mass reduction alongside fat loss, SARMs are being seriously considered as a potential protective strategy — but this remains investigational and requires further evidence before it can be recommended clinically (Vasilev, 2026).
Clinical Pearls: Selective Androgen Receptor Modulators (SARMs)
1. The Function-Mass Paradox
Scientific Tone: Clinical trials demonstrate that while SARMs increase lean body mass (LBM) in a dose-dependent manner, these gains frequently fail to reach statistical significance in functional outcomes, such as grip strength, gait speed, or stair-climb power.
Just because a drug helps you build muscle on the scale doesn't mean it automatically makes that muscle stronger or more useful. Growing muscle is only half the battle; training that muscle to perform better is a separate process that SARMs don't always guarantee.
2. The "Tissue-Selectivity" Myth at High Doses
Scientific Tone: The touted "tissue-selectivity" of SARMs—preferring muscle/bone over the prostate or HPG axis—is relative rather than absolute. At supratherapeutic or even "bodybuilding" doses, SARMs often exhibit loss of selectivity, leading to significant hypothalamic-pituitary-gonadal (HPG) axis suppression.
SARMs are marketed as "side-effect-free" because they target muscle specifically. However, if you take higher doses to see faster results, the drug loses its "aim" and starts affecting your hormones and other organs just like traditional steroids would.
3. Hepatotoxicity Without 17α-Alkylation
Scientific Tone: Despite lacking the 17α-alkylated structure of traditional oral steroids, SARMs are increasingly linked to Drug-Induced Liver Injury (DILI). Presentations often include cholestatic patterns or idiosyncratic hepatocellular damage, even at modest dosages.
Many believe SARMs are "liver-safe" because they aren't structured like old-school pills. This is false. There are many documented cases of people developing serious liver stress and yellowing of the eyes (jaundice) while using these compounds.
4. The "Dry Muscle" Lipid Trade-off
Scientific Tone: SARMs consistently produce a pro-atherogenic lipid profile, characterized by significant reductions in high-density lipoprotein (HDL) and, occasionally, elevations in low-density lipoprotein (LDL). This increases the cardiovascular risk-to-benefit ratio in long-term users.
While SARMs might give you a "lean" or "dry" look, they often do this at the expense of your internal health. They can significantly lower your "good" cholesterol, which is vital for protecting your heart and clearing out your arteries.
5. Pharmacokinetic Stability vs. Natural Pulsatility
Scientific Tone: SARMs possess extended elimination half-lives (e.g., 24–36 hours for Ligandrol), leading to continuous androgen receptor saturation. This contrasts with the natural circadian pulsatility of endogenous testosterone, potentially leading to receptor downregulation over time.
Your body is used to hormones that go up and down throughout the day. SARMs stay in your system at a high level 24/7. This constant "on" switch can eventually make your body less sensitive to its own muscle-building signals.
6. The Contamination and Mislabeling Reality
Scientific Tone: Chemical analysis of products sold as SARMs reveals high rates of ingredient substitution. Many "SARM" products contain unlisted substances, including classic anabolic steroids, growth hormone secretagogues, or vastly different dosages than what is stated on the certificate of analysis.
Buying SARMs online is a gamble. Research shows that nearly half of the products tested don't actually contain what the label says. You might think you're taking a "modern" SARM when you are actually taking a cheap, harsh, old-fashioned steroid that was hidden in the bottle.
Author’s Note
This article was written to bridge the gap between emerging molecular science and real-world clinical decision-making. Selective androgen receptor modulators (SARMs) are often discussed in polarized terms—either as breakthrough therapies or as performance-enhancing shortcuts. The reality, as current evidence suggests, lies somewhere in between.
As a clinician, one of the most important responsibilities is to distinguish between biological promise and clinically meaningful outcomes. While SARMs demonstrate measurable anabolic effects at the molecular and body composition level, medicine ultimately prioritizes outcomes that improve function, independence, and long-term health. This distinction is central to understanding why enthusiasm for SARMs has not yet translated into widespread clinical adoption.
Another key motivation behind this article is patient safety. In recent years, there has been a noticeable rise in unsupervised use of investigational compounds, often sourced from unregulated markets. These products frequently lack quality control, accurate labelling, and safety monitoring—introducing risks that extend beyond the pharmacology of SARMs.
At the same time, it is equally important not to dismiss their potential. In carefully selected populations—such as individuals with severe muscle wasting or frailty—SARMs may eventually find a role, provided that ongoing and future trials demonstrate clear functional benefits and acceptable long-term safety.
Medicine evolves through evidence, not expectation. As newer data from ongoing trials become available, our understanding of SARMs will continue to refine. Until then, this topic demands a balanced approach—scientifically grounded, clinically cautious, and patient-centered.
Medical Disclaimer: This article is for educational and informational purposes only. It does not constitute medical advice and should not be used as a substitute for professional clinical consultation. SARMs are investigational compounds not approved for general clinical use. Always consult a qualified healthcare provider before making any decisions about treatments or supplements. The authors of this article have no financial conflicts of interest related to any pharmaceutical compounds discussed.
Related Articles
Can Healthy Mitochondria Protect Against Sarcopenia and Age-Related Decline? | DR T S DIDWAL
Stopping GLP-1: Will the Weight Come Back? | DR T S DIDWAL
Sarcopenic Obesity: How to Lose Fat Safely Without Losing Muscle | DR T S DIDWAL
How to Prevent and Reverse Muscle Wasting in Chronic Disease (2025 Guide) | DR T S DIDWAL
Vitamin D Deficiency and Sarcopenia: The Critical Connection | DR T S DIDWAL
References
Wen, J., Syed, B., Leapart, J., Shehabat, M., Ansari, U., Akhtar, M., Razick, D., & Pai, D. (2025). Selective androgen receptor modulators (SARMs) effects on physical performance: A systematic review of randomized control trials. Clinical Endocrinology, 102, 3–27. https://doi.org/10.1111/cen.15135
Borecki, R., Byczkiewicz, P., & Słowikowska-Hilczer, J. (2025). Selective androgen receptor modulators (SARMs) – potential anabolic drugs for the treatment of cachexia and frailty syndrome. Endokrynologia Polska, 76(3), 246–256. https://doi.org/10.5603/ep.104795
Pena Dias, J., & Dobs, A. S. (2026). Will selective androgen receptor modulators ever reach the clinic? The Journal of Clinical Endocrinology & Metabolism, 111(5), e1467–e1471. https://doi.org/10.1210/clinem/dgaf700
Vasilev, V. (2026). SARMs vs. classic anabolic androgenic steroids: Molecular, pharmacokinetic and safety differences: A narrative review. Future Pharmacology, 6(2), 25. https://doi.org/10.3390/futurepharmacol6020025
Bond, P., Smit, D. L., Verdegaal, T., & de Ronde, W. (2025). Selective androgen receptor modulators: a critical appraisal. Frontiers in endocrinology, 16, 1634799. https://doi.org/10.3389/fendo.2025.1634799
Hall, E., & Vrolijk, M. F. (2023). Androgen Receptor and Cardiovascular Disease: A Potential Risk for the Abuse of Supplements Containing Selective Androgen Receptor Modulators. Nutrients, 15(15), 3330. https://doi.org/10.3390/nu15153330
Malave, B. Metabolic and hormonal dysfunction in asymptomatic patient using selective androgen receptor modulators: a case report. Bull Natl Res Cent 47, 11 (2023). https://doi.org/10.1186/s42269-023-00989-z