Neuromuscular Aging Explained: How Nerve–Muscle Breakdown Causes Weakness After 40
Discover why muscle weakness with age starts in the nervous system—not just muscle loss. Learn how neuromuscular junction decline drives sarcopenia and what science-backed strategies can slow it.
AGING
Dr. T.S. Didwal, M.D.(Internal Medicine)
4/23/202615 min read
What causes muscle weakness with aging?
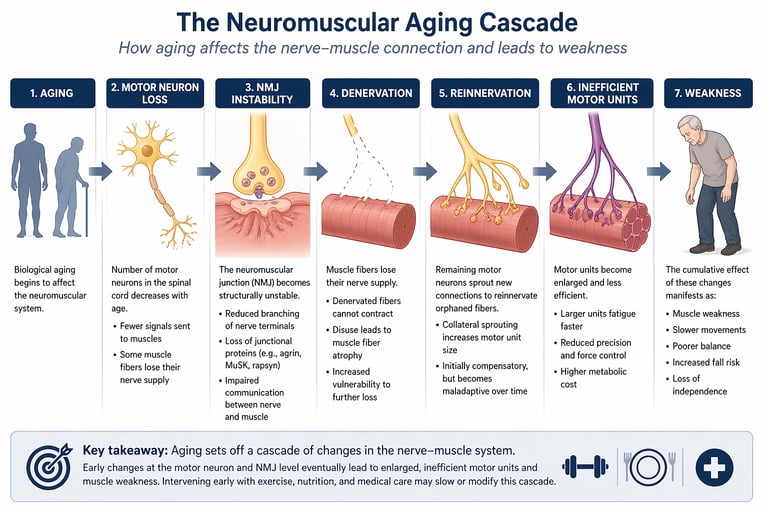
Age-related muscle weakness is not caused by muscle loss alone. Emerging research shows it begins with a decline in the neuromuscular system—particularly at the neuromuscular junction (NMJ), where nerves communicate with muscle fibres. With aging, motor neurons are gradually lost, NMJs become unstable, and some muscle fibres lose their nerve supply (denervation). The body attempts to compensate through reinnervation, but this creates larger, less efficient motor units that fatigue easily. Over time, this leads to reduced strength, poorer balance, and increased fall risk.
Clinician’s Perspective: What his Means for You
Muscle weakness is not just about muscle loss
Clinically, we now understand that declining strength often begins in the nerve–muscle connection, not just the muscle itself. This means early changes may be happening even when muscle size appears normal.Early signs are subtle—but important
Patients often report:Difficulty rising from a chair
Slower walking speed
Reduced balance or increased “unsteadiness”
These are not just “normal aging”—they can reflect early neuromuscular decline.
Strength ≠ muscle size alone
You may have “adequate muscle mass” but still feel weak. This happens when neuromuscular signaling becomes inefficient, reducing coordination and force generation.Simple clinical tests are powerful
In practice, we rely on:Grip strength
Chair stand test
Gait speed
These are quick, evidence-based tools that often predict outcomes better than imaging alone.
Advanced testing may be needed in select cases
If symptoms are disproportionate or unexplained, tests like electromyography (EMG) can detect early nerve–muscle dysfunction before major weakness develops.Intervention works—especially when started early
The most effective strategies we recommend:Resistance training (2–3 times/week) to preserve motor unit function
Adequate protein intake (≈1.2–1.6 g/kg/day) to support muscle repair
Balance and mobility training to maintain coordination
Chronic diseases accelerate decline
Conditions like diabetes, cardiovascular disease, and inactivity can worsen neuromuscular aging. Managing these aggressively is part of preserving strength.The goal is not just strength, but independence
From a clinical standpoint, preventing falls, maintaining mobility, and preserving quality of life are the true endpoints—not just increasing muscle mass.
Bottom line: If you notice even mild changes in strength or balance, early evaluation and intervention can significantly alter your long-term trajectory.
Why Your Muscles and Nervous System Age Together — And What the Latest Science Says You Can Do About It
Muscle weakness is often dismissed as an inevitable consequence of aging—a gradual, almost passive decline in strength and mobility. But emerging science tells a far more complex and clinically important story. What if the earliest drivers of this decline are not within the muscle itself, but at the microscopic junctions where nerves communicate with muscle fibres? These specialised synapses, known as the neuromuscular junction (NMJ), are increasingly recognised as critical control points in the aging process—sites where failure begins long before visible muscle loss occurs.
Recent evidence suggests that aging initiates a cascade of neuromuscular disruption: motor neurons are progressively lost, NMJs become structurally unstable, and muscle fibres are left without proper neural input—a process termed denervation (Moss et al., 2025). In response, surviving neurons attempt to compensate through collateral reinnervation, creating larger but less efficient motor units that fatigue more easily and generate less precise force (Cui et al., 2025). This adaptive mechanism, while initially protective, ultimately contributes to the functional decline observed in older adults.
This paradigm shift is now influencing how clinicians and researchers define and diagnose sarcopenia. Rather than viewing it solely as a problem of reduced muscle mass, leading experts advocate for a broader concept of “muscle failure,” encompassing the entire neuromuscular system, including neural input and synaptic integrity (Cruz-Jentoft et al., 2025). Longitudinal studies such as the TRAJECTOR-AGE trial are further reinforcing this integrative model by tracking how neuromuscular changes evolve and interact with factors such as physical activity, nutrition, and comorbidity (Lauretani et al., 2025).
Understanding this neuromuscular cascade is not merely academic—it has profound implications for early detection, prevention, and targeted intervention in aging populations.
30% Muscle mass lost between age 40 and 80 on average
50M+ People estimated to live with sarcopenia globally
40% Of adults over 80 affected by clinically significant sarcopenia
4× Increased fall risk associated with sarcopenic muscle weakness
What Eactly Is the Neuromuscular Junction?
The neuromuscular junction is one of the most elegantly engineered structures in the human body. When a motor neuron — a nerve cell originating in your spinal cord — fires, it releases a chemical called acetylcholine into the tiny gap separating it from the surface of a muscle fibre. The muscle fibre's surface is lined with specialized receptors that catch this chemical signal and translate it into an electrical impulse, which causes the muscle to contract. This entire sequence — from nerve signal to muscle movement — takes only milliseconds.
What makes the NMJ particularly significant in aging is its structural complexity. Both the nerve terminal and the muscle surface must be precisely aligned and maintained for reliable signalling to occur. When either side deteriorates — as happens progressively with age — the communication breaks down. Fewer nerve signals get through. Muscle fibres that fail to receive signals begin to weaken, shrink, and eventually die.
"The neuromuscular junction is not merely a relay station — it is an active, dynamic interface that must be constantly renewed. Its failure is not incidental to aging; it may be foundational to it."
The NMJ as a "Shared Vulnerability" in Aging and Disease
In what may become one of the most cited neuroscience papers of 2025, Moss, Darvishi, Badawi, Fish, and colleagues at multiple leading institutions published a comprehensive review arguing that the neuromuscular junction represents a shared point of vulnerability across both normal aging and a range of neuromuscular diseases (Moss et al., 2025).
The authors documented the structural and molecular changes that occur at the NMJ as humans age: the nerve terminal becomes smaller and less branched, the muscle surface becomes simplified and less receptive, and the precise molecular scaffolding that holds both sides in alignment — proteins such as agrin, MuSK, and rapsyn — begins to degrade. The result is what researchers call NMJ instability: a state where communication between nerve and muscle is intermittent rather than reliable.
Crucially, this same pattern of NMJ deterioration was found to occur — often far more rapidly and severely — in diseases such as amyotrophic lateral sclerosis (ALS), spinal muscular atrophy (SMA), myasthenia gravis, and Lambert-Eaton myasthenic syndrome. This overlap between aging and disease is what the authors characterize as a "shared vulnerability": the NMJ appears to be a weak link in the neuro-muscular chain, one that fails under the dual pressures of time and pathology (Moss et al., 2025).
For patients and the public, this finding carries an important implication: therapies being developed to protect or restore NMJ function in disease — some of which are already in clinical trials — may one day have powerful applications in healthy aging populations as well. The boundary between "treating disease" and "treating aging" is becoming thinner.
Key findings from Moss et al. (2025)
NMJ structural changes in aging mirror — and sometimes overlap with — changes seen in ALS, SMA, and myasthenia gravis
Both sides of the junction deteriorate: pre-synaptic (nerve) and post-synaptic (muscle)
Molecular scaffolding proteins that maintain NMJ architecture decline with age
Therapeutic targets in neuromuscular disease may translate to aging interventions
The NMJ may be a rate-limiting factor in age-related physical decline
Reading the Electrical Signature of Aging Muscles
If Moss and colleagues told us why the NMJ fails with age, Cui, Hu, Wong, Zhang, Guan, and Cheung's 2025 systematic review in GeroScience showed us how to measure that failure in living people — using a tool called electromyography (EMG) (Cui et al., 2025).
Electromyography works by recording the electrical activity of muscle fibres through tiny electrodes placed on or inserted into the skin. In a healthy young muscle, each motor neuron controls a relatively modest bundle of muscle fibres — a group called a motor unit. The electrical signal recorded from this unit has a characteristic shape, size, and timing. As aging and NMJ dysfunction progress, however, the picture changes dramatically.
Motor neurons begin to die with age, orphaning the muscle fibres they once controlled. Neighbouring healthy motor neurons may attempt to "adopt" these fibres by sprouting new connections — a process called collateral reinnervation. While this rescue mechanism is initially beneficial, it creates larger, more ungainly motor units that fire less efficiently and fatigue more easily. EMG can detect this remodelling: the electrical signals from aging motor units are larger in amplitude and longer in duration, a signature the researchers call motor unit enlargement (Cui et al., 2025).
The systematic review found consistent evidence across multiple studies that people with sarcopenia show significantly greater motor unit enlargement, more signs of NMJ instability — measurable as "jitter" in the timing of muscle fibre responses — and fewer functional motor units overall. The exciting clinical implication is that EMG-based assessments could serve as early biomarkers of sarcopenia, potentially allowing intervention before significant muscle loss has occurred.
"Motor unit remodelling may be one of the earliest detectable signs that the neuromuscular system is beginning to fail — years before muscle loss becomes visible on a scan or apparent in daily movement."
Redefining What We Mean by "Muscle Failure"
Before we can fully appreciate the impact of NMJ research on aging, we need to grapple with a fundamental challenge: defining sarcopenia itself. A landmark 2025 paper by Cruz-Jentoft, Bahat, Bauer, and a distinguished international consortium, published in The Lancet Healthy Longevity, undertook a thorough critical appraisal of existing sarcopenia definitions and found them wanting (Cruz-Jentoft et al., 2025).
The paper argues that current definitions — which typically focus on three measurable parameters: muscle mass (measured by DEXA scan or bioelectrical impedance), muscle strength (measured by handgrip dynamometry), and physical performance (measured by gait speed or chair stand tests) — are necessary but insufficient. They capture the downstream consequences of muscle failure but do not fully account for its upstream causes, including NMJ dysfunction and motor unit remodelling (Cruz-Jentoft et al., 2025).
The authors propose the framing of "muscle failure" as a broader and more clinically meaningful concept — one that encompasses not just the muscles themselves but the entire neuromuscular system that supports them. This shift in language is not merely semantic: it fundamentally changes the scope of what should be assessed, monitored, and treated in aging patients with muscle decline.
For clinicians, the paper calls for a new generation of diagnostic tools — including neurophysiological measures like EMG — to be incorporated into sarcopenia assessment. For patients, it underscores that muscle weakness is rarely about muscle alone, and that any effective approach to treatment must address the nervous system as well as the muscle.
The evolving language of sarcopenia
Old frame: muscle mass loss + weakness + poor performance = sarcopenia
New frame: "muscle failure" — a systemic breakdown involving nerves, junctions, and fibres
Neurophysiological markers (EMG, NMJ assessment) increasingly seen as essential
Functional outcomes — falls, mobility loss, disability — must anchor any definition
Biological sex, ethnicity, and comorbidities require tailored diagnostic thresholds
From the Lab to the Clinic: The TRAJECTOR-AGE Trial
Research into aging muscles and nerves has historically been dominated by cross-sectional studies — snapshots of different people at different ages. What has been missing is a rigorous longitudinal view: tracking the same individuals over time to understand how and when the neuromuscular system begins to falter, and what factors accelerate or slow that process. The TRAJECTOR-AGE clinical trial, designed by Lauretani, Maggio, Pilotto, and colleagues, aims to fill precisely this gap (Lauretani et al., 2025).
Published in the Journal of the American Geriatrics Society, the TRAJECTOR-AGE protocol describes an ambitious multi-centre study designed to map the trajectories of neuromuscular aging across a large cohort of older adults. The trial will follow participants through repeated assessments of muscle mass, strength, motor unit function, NMJ integrity, gait analysis, cognitive performance, and nutritional status, among other markers (Lauretani et al., 2025).
What makes TRAJECTOR-AGE particularly significant is its integrative design. Rather than examining any single biomarker in isolation, the study will attempt to model the relationships between different aspects of neuromuscular decline — including the interplay between NMJ dysfunction and cognitive aging, the influence of comorbidities such as diabetes and cardiovascular disease, and the potential protective effects of physical activity, nutrition, and pharmacological interventions.
For patients and caregivers, the practical value of this trial is immense. Its results — expected over the coming years — are likely to inform which combinations of interventions most effectively preserve neuromuscular function in older age, and at what stage of life those interventions should ideally begin. It represents the kind of rigorous, long-term clinical evidence that can translate NMJ science from the laboratory bench into the doctor's surgery.
Practical Applications: What Can You Do Right Now?
Science that stays in academic journals helps no one. The four studies reviewed here, while complex in their methodology, point toward actionable strategies that older adults, caregivers, and healthcare providers can begin implementing today. Here are the most evidence-informed practical takeaways:
Prioritise resistance training
Progressive resistance exercise is the most robustly proven intervention for preserving both motor unit function and muscle mass. Aim for 2–3 sessions per week targeting all major muscle groups. Even light resistance training with bands or bodyweight is beneficial
Optimise protein intake
Older adults require more dietary protein than current general guidelines suggest — current evidence supports 1.2–1.6g per kilogram of body weight daily, distributed across meals. Leucine-rich sources such as eggs, dairy, legumes, and lean meats stimulate muscle protein synthesis most effectively.
Walk — and balance train
Daily walking activates motor units in the lower limbs, maintaining the nerve-muscle communication pathways identified in the Cui et al. EMG research. Balance and coordination exercises (yoga, tai chi, standing on one leg) specifically challenge and preserve the precision of NMJ signalling.
Neuromuscular Aging: Frequently Asked Questions
1. What is the neuromuscular junction, and why is it the "new frontier" of aging?
The Science: The neuromuscular junction (NMJ) is the chemical synapse where a motor neuron meets a muscle fiber. 2025 research by Moss et al. demonstrates that aging isn't just about the muscle "melting away"; it's about the communication link snapping. When the NMJ degrades, the muscle fiber becomes "de-innervated" (disconnected), leading to rapid atrophy.
The Bottom Line: Think of the NMJ as the "aux cable" between your brain and your body. If the cable is frayed, it doesn't matter how good the speakers (muscles) are—the music won't play.
2. Is sarcopenia different from just "getting weaker" as I age?
The Science: Yes. "Weakness" is a symptom; sarcopenia is a clinical condition. According to the 2025 Cruz-Jentoft framework, sarcopenia is now viewed as "muscle failure." It involves a measurable loss of muscle quantity and quality, often driven by the nervous system’s inability to recruit muscle fibers effectively.
The Bottom Line: Normal aging involves slight slowing, but sarcopenia is a functional breakdown that significantly increases your risk of falls, hospitalizations, and loss of independence.
3. Can I actually reverse NMJ decline or muscle loss once it starts?
The Science: While you cannot stop biological time, the NMJ is remarkably plastic. Studies (Cui et al., 2025) show that resistance training and specific nutrients can stabilize the NMJ by "re-anchoring" the nerve to the muscle. This process, called reinnervation, can actually restore function to fibers that were on the verge of being lost.
The Bottom Line: It is rarely "too late." Even in your 80s, targeted exercise can strengthen the neural connections you still have and potentially "rescue" fading muscle fibers.
4. How does a doctor test for this "nerve-muscle" connection?
The Science: The gold standard is Electromyography (EMG). As highlighted in GeroScience (2025), EMG can detect "jitter" (timing errors in the nerve signal) and "motor unit enlargement" (where one nerve tries to control too many fibers). This provides a signature of how much "remodeling" your nervous system has done to compensate for age.
The Bottom Line: Most GPs start with functional tests like grip strength or the Timed Up and Go (TUG) test. If you have unexplained weakness, an EMG can provide a deeper "under-the-hood" look at your electrical wiring.
5. Why is 2025 research saying I need more protein than I used to?
The Science: Older adults suffer from anabolic resistance, meaning the "signal" to build muscle is muted. To overcome this, the TRAJECTOR-AGE trial supports a higher threshold of 1.2–1.6g of protein per kg of body weight. Furthermore, you need a large "bolus" (30-40g) in a single meal to trigger the mTOR pathway—the body's master switch for muscle growth.
The Bottom Line: A light sprinkle of protein throughout the day isn't enough for aging muscles. You need a "loud" protein signal—think a significant portion of protein at breakfast and dinner—to tell your body to keep its muscle mass.
6. What diseases share the same "vulnerabilities" as aging muscles?
The Science: The Moss et al. (2025) study found that the structural breakdown of the NMJ in "normal" aging looks surprisingly similar to the early stages of ALS (Motor Neuron Disease), Myasthenia Gravis, and SMA. This suggests that aging might be a "low-grade" version of these neuromuscular pathologies.
The Bottom Line: This is actually good news for medicine. It means that drugs and therapies currently being developed to treat rare nerve diseases might eventually be used to help everyone maintain strength and mobility as they age.
Quick Reference: The 3-Step Protection Plan
Resistance Training: (The "Glue") 2–3x a week to keep nerves attached to muscles.
Protein Bolus: (The "Signal") 30g+ of protein in a single sitting to trigger growth.
Balance Work: (The "Fine-Tuning") Yoga or Tai Chi to keep the electrical signals precise.Evidence-based answers to the questions we hear most often about neuromuscular aging and sarcopenia
Clinical pearls
1. The "Orphaned Fiber" Phenomenon
Scientific Tone: Age-related sarcopenia is characterized by motor neuron dropout, leading to "orphan" muscle fibers. While collateral reinnervation (neighboring nerves "adopting" fibers) maintains mass temporarily, it results in motor unit enlargement, which reduces fine motor control and increases metabolic fatigue.
Think of your nerves like power lines. As we age, some lines go down. Neighboring lines try to pick up the slack, but they get overloaded. You might still have the "muscle," but it becomes clunkier and tires out faster because the wiring is stretched too thin.
2. NMJ Instability as a "Pre-Sarcopenia" Marker
Scientific Tone: Electromyographic (EMG) "jitter"—a variation in the interval between firing and contraction—serves as a proxy for NMJ instability. This instability often precedes measurable atrophy, suggesting that neuromuscular transmission failure is a primary driver, not a secondary symptom, of muscle loss.
Before a muscle actually shrinks, the "handshake" between the nerve and the muscle gets shaky. If you feel "fumble-fingered" or slightly off-balance even though your muscles look the same size, your nervous system might be sending early warning signals that it needs more stimulation.
3. The "Muscle Failure" Diagnostic Shift
Scientific Tone: The 2025 Cruz-Jentoft framework shifts the clinical focus from static muscle mass (quantity) to muscle failure (quality and connectivity). Diagnosis should prioritize functional assessments like the chair-stand test and gait speed over DEXA scans alone, as mass does not always correlate with NMJ integrity.
Having big muscles doesn't always mean they work well. Doctors are moving away from just "measuring your biceps" and moving toward "measuring your movement." How fast you can get out of a chair is a much better map of your health than how much you weigh on a scale.
4. Resistance Training as "Synaptic Glue"
Scientific Tone: Progressive resistance exercise (PRE) upregulates the expression of scaffolding proteins like agrin and MuSK. These proteins are essential for maintaining the structural alignment of the NMJ, effectively "anchoring" the nerve terminal to the muscle fiber and preventing denervation.
Lifting weights does more than build muscle; it acts like "superglue" for your nerves. It forces the nerve and muscle to stay tightly connected. Every time you challenge your muscles with resistance, you are telling your nervous system that the connection is still vital and needs to be maintained.
5. The Protein-Distribution Threshold
Scientific Tone: To overcome "anabolic resistance" in the aging NMJ-muscle complex, a bolus of 0.4g/kg per meal (or ~30-40g of high-leucine protein) is required to trigger muscle protein synthesis. Sub-threshold protein intake fails to activate the mTOR pathway effectively in older adults compared to younger cohorts.
Older muscles are "hard of hearing" when it comes to protein. If you only eat a little protein at a time, your muscles don't even realize it's there. You need to eat a solid "dose" of protein (like a chicken breast or a large Greek yogurt) in one sitting to actually "turn on" the growth machinery.
Author’s Note
This article was written with a clear purpose: to bridge the gap between rapidly evolving neuromuscular science and its real-world relevance for patients, clinicians, and caregivers. Much of what we traditionally understood about age-related muscle loss focused on the muscle itself. However, the emerging evidence—particularly from recent high-quality studies published in journals such as Journal of Neuroscience and The Lancet Healthy Longevity—compels us to think more broadly, and more precisely, about the entire neuromuscular system.
In developing this piece, care was taken to synthesise findings from multiple peer-reviewed sources while maintaining clarity and clinical applicability. The aim was not only to explain complex mechanisms—such as neuromuscular junction instability and motor unit remodelling—but also to translate them into meaningful, actionable insights. For clinicians, this means re-evaluating how we assess and monitor sarcopenia. For patients, it means understanding that early changes in strength and balance are biologically significant—and, importantly, modifiable.
It is equally important to acknowledge that this field is still evolving. Longitudinal trials and interventional studies are ongoing, and while current evidence strongly supports the role of neuromuscular dysfunction in aging, many questions remain regarding optimal diagnostic tools and targeted therapies. Readers are encouraged to view this article not as a final word, but as part of an ongoing scientific conversation.
Ultimately, the central message is one of cautious optimism: while aging is inevitable, the trajectory of neuromuscular decline is not fixed. With informed strategies grounded in science, there is substantial potential to preserve strength, function, and independence across the lifespan
Medical Disclaimer
This article is intended for educational purposes only and does not constitute medical advice. The research discussed reflects findings from peer-reviewed studies published up to 2025 and should not be used to guide personal health decisions without consulting a qualified healthcare professional. Do not self-administer any pharmaceutical compound mentioned in this article.
Related Articles
Can You Slow Aging at the Cellular Level? The Autophagy Breakthrough Explained
Mitochondria, Motor Units, and Muscle Aging: A Complete Guide | DR T S DIDWAL
Exercise and Longevity: The Science of Protecting Brain and Heart Health as You Age | DR T S DIDWAL
The Science of Healthy Brain Aging: Microglia, Metabolism & Cognitive Fitness | DR T S DIDWAL
The Aging Muscle Paradox: How Senescent Cells Cause Insulin Resistance and The Strategies to Reverse It | DR T S DIDWAL
References
Cruz-Jentoft, A. J., Bahat, G., Bauer, J., Boirie, Y., Bruyère, O., Cederholm, T., Landi, F., Rolland, Y., Sayer, A. A., Schneider, S. M., Sieber, C. C., Topinková, E., Vandewoude, M., & Visser, M. (2025). Capturing what counts in muscle failure: A critical appraisal of the current definitions of sarcopenia. The Lancet Healthy Longevity, 6(8), e1000753. https://doi.org/10.1016/S2666-7568(25)00075-3
Cui, C., Hu, Y., Wong, R. M. Y., Zhang, N., Guan, Y.-Z., & Cheung, W. H. (2025). Exploring motor unit and neuromuscular junction dysfunction in aging and sarcopenia: Insights from electromyography in systematic review. GeroScience. https://doi.org/10.1007/s11357-025-01760-0
Lauretani, F., Maggio, M., & Pilotto, A. M., et al. (2025). The Trajectories of Neuromuscular Aging (TRAJECTOR-AGE Clinical Trial): Study rationale and methodological protocol. Journal of the American Geriatrics Society, 73(9), 2819–2827. https://doi.org/10.1111/jgs.70005
Moss, K. R., Darvishi, F. B., Badawi, Y., Fish, L. A., Funke, J. R., Pedersen, T. H., Robitaille, R., Arnold, W. D., Burgess, R. W., Meriney, S. D., Nishimune, H., & Saxena, S. (2025). The neuromuscular junction: A shared vulnerability in aging and disease. Journal of Neuroscience, 45(46), e1353252025. https://doi.org/10.1523/JNEUROSCI.1353-25.2025