GLP-1 Muscle Loss: You’re Losing Weight—But Are You Losing Strength Too?
GLP-1 drugs like semaglutide can cause muscle loss along with fat loss. Learn the science, risks, and proven strategies to preserve muscle and metabolism.
SARCOPENIAOBESITY
Dr. T.S. Didwal, M.D.(Internal Medicine)
4/27/202616 min read
Do GLP-1 weight loss injections cause muscle loss?
Yes—GLP-1 receptor agonists (such as semaglutide and tirzepatide) can lead to some loss of lean body mass, including skeletal muscle, during weight reduction. Current evidence suggests that ~25–40% of total weight lost may come from lean mass, although this varies based on age, baseline muscle, diet, and physical activity (Sanchis-Gomar et al., 2025; Rossi et al., 2026).
Why this matters
Skeletal muscle is not just for strength—it is a metabolic organ that:
Regulates blood sugar (glucose disposal)
Maintains resting metabolic rate
Protects against frailty and long-term weight regain
Loss of muscle during rapid weight loss may slow metabolism and increase the risk of regaining weight after stopping therapy (Kwan et al., 2026).
Is this unique to GLP-1 drugs?
No. All weight loss methods (dieting, bariatric surgery, medications) result in some muscle loss. However, because GLP-1 drugs produce larger total weight loss, the absolute muscle loss can be clinically significant, especially in:
Older adults
Sedentary individuals
Patients with sarcopenic obesity or MASLD (Iorra et al., 2026)
How to prevent muscle loss on GLP-1 therapy
Evidence-based strategies:
Resistance training (essential): 2–3 times per week
High protein intake: ~1.2–1.6 g/kg/day (up to 2.0 g/kg in high-risk groups)
Track body composition: DEXA or BIA, not just weight
Stay physically active daily
Consider creatine supplementation (if appropriate)
Clinician’s Perspective: Practical Guidance for GLP-1 Therapy and Muscle Preservation
Reframe the treatment goal:
Shift the focus from total weight loss to body composition optimization. Emphasize fat loss while actively preserving lean mass, as muscle is a key determinant of long-term metabolic health (Barber et al., 2025).Risk-stratify patients early:
Identify individuals at higher risk of clinically significant muscle loss:Older adults
Sedentary patients
Sarcopenic obesity
Chronic disease (e.g., MASLD)
These groups require more aggressive muscle-preservation strategies (Iorra et al., 2026).
Set expectations before initiation:
Counsel patients that 25–40% of weight loss may include lean mass, especially without intervention. This improves adherence to exercise and nutrition plans (Sanchis-Gomar et al., 2025).Prescribe resistance training as standard of care:
Treat strength training like a “co-therapy,” not optional advice.
Minimum: 2–3 sessions/week, progressive overload, major muscle groups.Prioritize protein intake clinically:
Recommend 1.2–1.6 g/kg/day, higher in older or high-risk patients.
Address appetite suppression proactively—many patients unintentionally under-eat protein.Monitor beyond the scale:
Incorporate body composition tracking (DEXA preferred, BIA acceptable).
Functional metrics (grip strength, sit-to-stand tests) add real-world relevance.Anticipate metabolic adaptation:
Lean mass loss lowers resting metabolic rate, increasing the risk of post-treatment weight regain. Plan mitigation strategies early (Kwan et al., 2026).Adopt a phased treatment approach:
Loss phase: minimise muscle loss
Protection phase: stabilize lean mass
Maintenance phase: prevent regain
Consider adjuncts where appropriate:
Creatine supplementation and leucine-rich protein sources may support muscle preservation, particularly in older adults.Deliver multidisciplinary care:
Integrate input from physicians, dietitians, and exercise professionals. GLP-1 therapy is most effective when embedded in a comprehensive metabolic program.
Clinical Bottom Line:
GLP-1 receptor agonists are powerful tools—but without deliberate muscle-preserving strategies, they risk trading short-term weight loss for long-term metabolic vulnerability.
GLP-1 receptor agonists such as semaglutide and tirzepatide have transformed obesity treatment, delivering unprecedented weight loss and cardiometabolic benefits. For many patients, these medications represent the first truly effective, sustainable option after years of unsuccessful dieting. However, emerging evidence highlights a critical nuance that both clinicians and patients must understand: weight loss is not synonymous with fat loss. A substantial proportion of the weight loss during GLP-1 therapy may be attributable to lean body mass, including skeletal muscle (Sanchis-Gomar et al., 2025; Rossi et al., 2026).
This distinction matters because skeletal muscle is not merely structural—it is a central regulator of metabolic health. Muscle tissue drives glucose disposal, supports resting energy expenditure, and protects against functional decline, particularly with aging. Loss of muscle during rapid weight reduction can therefore blunt metabolic improvements and increase the risk of long-term weight regain (Barber et al., 2025; Kwan et al., 2026). Importantly, this phenomenon is not unique to GLP-1 therapies; any significant caloric deficit leads to some degree of lean mass loss. However, the magnitude becomes clinically relevant given the substantial total weight loss these agents produce.
Recent literature suggests that approximately 25–40% of weight lost may be lean mass, though this varies widely depending on baseline muscle status, age, physical activity, and nutritional intake (Sanchis-Gomar et al., 2025). In vulnerable populations—such as older adults or those with sarcopenic obesity or metabolic dysfunction-associated steatotic liver disease (MASLD)—even modest muscle loss may have disproportionate consequences (Iorra et al., 2026).
This evolving evidence shifts the clinical focus from simply achieving weight loss to optimizing body composition. The most effective approach combines pharmacotherapy with targeted strategies—particularly resistance training and adequate protein intake—to preserve muscle while reducing fat. Ultimately, the success of GLP-1 therapy should not be judged solely by kilograms lost, but by the extent to which it delivers durable metabolic health and functional resilience.
Why Skeletal Muscle Matters More Than You Think
Most people think about muscle in terms of gym performance — can you lift heavy, do you look toned? But skeletal muscle is far more than an aesthetic asset. It is a metabolically active endocrine organ that plays a central role in glucose regulation, energy expenditure, immune function, and even hormonal signalling.
Barber et al. (2025), writing in Metabolites, articulate this comprehensively. Their review argues that optimising skeletal muscle mass — not simply reducing body weight — should be positioned as a key pillar of obesity management. They point out that excess fat mass and insufficient muscle mass often co-exist in people with obesity, a condition sometimes called "sarcopenic obesity," and that this combination is associated with significantly worse cardiometabolic outcomes than obesity alone.
In practical terms, here is what adequate skeletal muscle does for you:
Insulin sensitivity: Muscle is the primary tissue responsible for clearing glucose from the blood after meals. More muscle = better blood sugar control.
Resting metabolic rate: Muscle tissue burns calories even at rest. Losing muscle slows your metabolism, making future weight maintenance harder.
Functional independence: Muscle strength protects against falls, fractures, and disability — especially as you age.
Cardiovascular protection: Higher muscle mass is independently associated with lower risk of heart disease and all-cause mortality.
Given all of this, any weight loss strategy that significantly erodes muscle mass carries real long-term risks — even if it produces excellent short-term results on the scale.
GLP-1 Receptor Agonists: The Weight Loss Breakthrough
Before examining the concern, it is worth briefly appreciating the achievement. GLP-1 receptor agonists work by mimicking glucagon-like peptide-1, a gut hormone that regulates appetite, slows gastric emptying, and improves insulin secretion. The result is a powerful reduction in caloric intake, leading to sustained weight loss that was previously only achievable through bariatric surgery.
Clinical trials have demonstrated weight reductions of 15–22% of total body weight with newer agents like semaglutide and tirzepatide. For a person weighing 100 kg, that can mean losing 15–22 kg — a transformation with profound health benefits.
However, Rossi et al. (2026), publishing in Acta Diabetologica, are among the researchers now drawing attention to the composition of that weight loss. Their study examined muscle loss in patients treated with GLP-1 receptor agonists, finding that a significant proportion of the weight lost was lean body mass — including skeletal muscle — rather than exclusively adipose (fat) tissue.
The Muscle Loss Problem: What the Evidence Actually Shows
Lean Mass Loss Is Real — But Contextual
Sanchis-Gomar, Neeland, and Lavie (2025), in a commentary published in Nature Reviews Endocrinology, frame the issue with clinical precision. They note that while GLP-1 receptor agonists do result in loss of lean body mass, this is not unique to these medications — any significant caloric restriction produces some lean mass loss, regardless of how it is achieved.
The critical question is whether the proportion of muscle lost during GLP-1 therapy is disproportionately high relative to the fat lost. Early data suggest that somewhere between 25–40% of total weight lost on these agents may be lean mass, which is broadly consistent with other weight loss strategies. However, the absolute amounts may be larger simply because total weight loss is larger.
What makes this clinically meaningful is the interaction with pre-existing risk factors. People with obesity who are already older, sedentary, or have low baseline muscle mass may be especially vulnerable to the functional consequences of even modest muscle loss. For them, losing an additional 3–5 kg of muscle mass alongside 20 kg of fat could meaningfully increase their risk of sarcopenia and its consequences.
GLP-1 and Muscle in Liver Disease: A Special Case
The picture becomes even more complex in patients with metabolic dysfunction-associated steatotic liver disease (MASLD) — formerly known as non-alcoholic fatty liver disease (NAFLD). Iorra et al. (2026), in a systematic review published in Liver International, examined the effects of GLP-1 receptor agonists specifically on muscle mass, strength, and quality in people with MASLD.
Their findings are nuanced. While GLP-1 receptor agonists produced beneficial reductions in liver fat and inflammation, the effects on skeletal muscle were variable across studies. Some studies showed preservation of muscle function despite reductions in muscle mass; others raised concerns about strength decrements. The authors called for standardised outcome measures and longer-term follow-up studies to clarify the picture.
This is an important finding because MASLD itself is associated with muscle dysfunction — the two conditions share metabolic pathways — and clinicians managing patients with both conditions need to be especially vigilant.
The Broader Metabolic Implications
Kwan, Chan, and colleagues (2026), in a paper published in Clinical Nutrition, provide perhaps the most comprehensive synthesis of the metabolic implications of GLP-1-associated muscle atrophy. Their analysis examines not just what happens to muscle during treatment, but what happens after — and here the concern deepens.
They note that if patients discontinue GLP-1 therapy without having built protective muscle mass (through resistance training and adequate protein intake), the combination of a slower resting metabolism and reduced lean mass creates a setup for rapid weight regain. The muscle loss that occurred during treatment effectively undermines the long-term sustainability of results.
This insight reframes how we should think about GLP-1 therapy: not as a standalone intervention, but as a tool that must be paired with deliberate lifestyle strategies to preserve and build muscle.
Enter Amycretin: The Next Generation Looks Promising
One of the most exciting developments in obesity pharmacology in 2026 is the emergence of amycretin — a novel dual-acting molecule that combines GLP-1 receptor agonism with amylin receptor agonism in a single compound. Fu et al. (2026), writing in Metabolism, provide a comprehensive review of amycretin's mechanisms, clinical efficacy data, and prospects.
What makes amycretin particularly interesting from a muscle health perspective is its distinct mechanism of action. Amylin — the other component of amycretin's dual action — has been shown to influence body composition differently from GLP-1 alone, with some preliminary evidence suggesting more favourable lean mass preservation during weight loss. Additionally, early clinical trial data indicate that amycretin may produce superior overall weight loss compared to GLP-1 agonists alone, potentially allowing for similar fat reduction with less lean mass sacrifice.
It is important to emphasise that amycretin is still in clinical development, and long-term data on muscle outcomes are not yet available. However, the emergence of next-generation agents that are designed with body composition in mind — not just total weight — represents a significant and welcome evolution in the field.
As Fu et al. (2026) conclude, the future of obesity pharmacotherapy likely lies in precision approaches that simultaneously target fat reduction, muscle preservation, and metabolic health — rather than optimising for weight loss in isolation.
Practical Applications: How to Protect Your Muscles on GLP-1 Therapy
1. Prioritise Resistance Training — Non-Negotiable
The single most powerful intervention for preserving muscle mass during caloric restriction is resistance (strength) training. This means lifting weights, using resistance bands, or performing bodyweight exercises like squats, push-ups, and rows — with enough challenge to stimulate muscle protein synthesis.
Aim for at least two to three resistance training sessions per week, targeting all major muscle groups. You do not need to become a competitive powerlifter; even moderate resistance training consistently outperforms cardio alone for muscle preservation during weight loss.
If you are new to resistance training, consider working with a qualified fitness professional, at least initially, to learn proper form and build a sustainable routine.
2. Eat Enough Protein — More Than You Probably Think
GLP-1 receptor agonists powerfully suppress appetite, which is part of how they work. But when appetite is dramatically reduced, it becomes very easy to under-eat protein — the macronutrient most critical for maintaining muscle tissue.
Current evidence supports protein intakes of 1.2–1.6 grams per kilogram of body weight per day during active weight loss — and potentially higher (up to 2.0 g/kg) for older adults or those with sarcopenic obesity (Barber et al., 2025). For a 90 kg person, that means consuming 108–144 grams of protein daily — an amount that requires deliberate planning when appetite is suppressed.
Practical strategies include:
Prioritising protein at every meal (eggs, chicken, fish, Greek yoghurt, legumes, cottage cheese)
Using protein shakes or powders if meeting targets through whole foods alone is difficult
Spreading protein intake across 3–4 meals rather than loading it into one or two
3. Monitor Body Composition, Not Just Body Weight
The bathroom scale tells you total weight — it cannot tell you whether you are losing fat or muscle. Ask your doctor or healthcare provider about DEXA scanning (dual-energy X-ray absorptiometry), the gold standard for measuring lean mass and fat mass separately. Even if DEXA is not accessible, bioelectrical impedance analysis (BIA) scales — increasingly affordable for home use — can provide useful tracking data.
Monitoring body composition allows you and your healthcare team to identify early muscle loss and adjust your approach accordingly, before it becomes clinically significant.
4. Consider Leucine-Rich and Creatine Supplementation
Among nutritional supplements, creatine monohydrate has the most robust evidence base for supporting muscle mass and strength, particularly in conjunction with resistance training. It is safe, inexpensive, and well-studied across age groups.
Leucine, an essential amino acid, is particularly important for stimulating muscle protein synthesis. Ensuring your protein sources are leucine-rich (whey protein, dairy, meat, eggs) or supplementing with leucine directly may support muscle maintenance during the caloric deficit created by GLP-1 therapy.
Always discuss supplementation with your healthcare provider, particularly if you have kidney disease or other conditions that affect protein metabolism.
5. Stay Active Between Formal Exercise Sessions
Beyond formal resistance training, general daily movement matters. Sitting for long periods, even with regular workouts, is associated with muscle atrophy and metabolic dysfunction. Aim to break up periods of sitting, walk regularly throughout the day, and maintain an active lifestyle beyond the gym. The goal is to send your muscles a consistent signal that they are needed — a signal that protects them during caloric restriction.
6. Work With a Multidisciplinary Team
GLP-1 therapy is most effective and safest when delivered as part of a comprehensive obesity management programme. Ideally, your team should include:
A prescribing physician experienced in obesity medicine
A registered dietitian to help you optimise protein intake and overall nutrition
A physiotherapist or certified exercise professional to guide resistance training
Regular monitoring of weight, body composition, and metabolic markers
Frequently Asked Questions
Q1: How much muscle will I lose on GLP-1 medications like Ozempic or Wegovy? The amount varies between individuals, but research suggests that approximately 25–40% of total weight lost may be lean mass, including muscle (Sanchis-Gomar et al., 2025). In absolute terms, this depends on how much total weight you lose. The good news is that with resistance training and adequate protein intake, this proportion can be meaningfully reduced.
Q2: Is muscle loss on GLP-1 therapy dangerous? For most healthy adults, the muscle loss associated with GLP-1 therapy is manageable and does not cause immediate harm. However, for older adults, those with pre-existing low muscle mass, or individuals with conditions like MASLD, muscle loss can have more significant functional and metabolic consequences (Iorra et al., 2026; Kwan et al., 2026). This is why proactive prevention strategies are essential.
Q3: Will I regain weight if I stop GLP-1 medications? Many patients do regain weight after stopping GLP-1 therapy, partly due to the return of appetite but also because of metabolic changes — including reduced muscle mass and a slower resting metabolic rate — that occurred during treatment (Kwan et al., 2026). Building and maintaining muscle during treatment is one of the most important strategies for long-term weight maintenance.
Q4: Can I build muscle while losing weight on GLP-1 therapy? Yes, though it is challenging. Achieving significant net muscle gain during a caloric deficit is difficult even for highly trained individuals. However, consistent resistance training and high protein intake can absolutely preserve existing muscle mass and in some cases produce modest gains, particularly in individuals who are untrained at baseline (Barber et al., 2025).
Q5: What is amycretin and is it better than current GLP-1 medications for muscle health? Amycretin is a next-generation obesity medication combining GLP-1 receptor agonism with amylin receptor agonism. Early data suggest it may offer superior weight loss with potentially more favourable body composition outcomes compared to GLP-1 agonists alone (Fu et al., 2026). However, it is still in clinical development and not yet widely available. Your doctor can advise you on emerging treatment options.
Q6: Does GLP-1 therapy affect people with liver disease (MASLD) differently? Yes. People with MASLD face a more complex picture, as both the disease and the treatment affect the muscle and liver simultaneously. GLP-1 receptor agonists do provide meaningful liver benefits in MASLD, but their effects on muscle mass and strength in this population are variable and require more research (Iorra et al., 2026). If you have MASLD, closer monitoring and a strong focus on exercise and protein are especially important.
Q7: How much protein should I eat while on GLP-1 therapy? Current research supports 1.2–1.6 grams of protein per kilogram of body weight per day during active weight loss, and up to 2.0 g/kg for older adults (Barber et al., 2025). Since GLP-1 medications suppress appetite strongly, many patients struggle to meet these targets. Working with a registered dietitian can help you develop practical strategies to hit your protein goals without forcing yourself to eat uncomfortably.
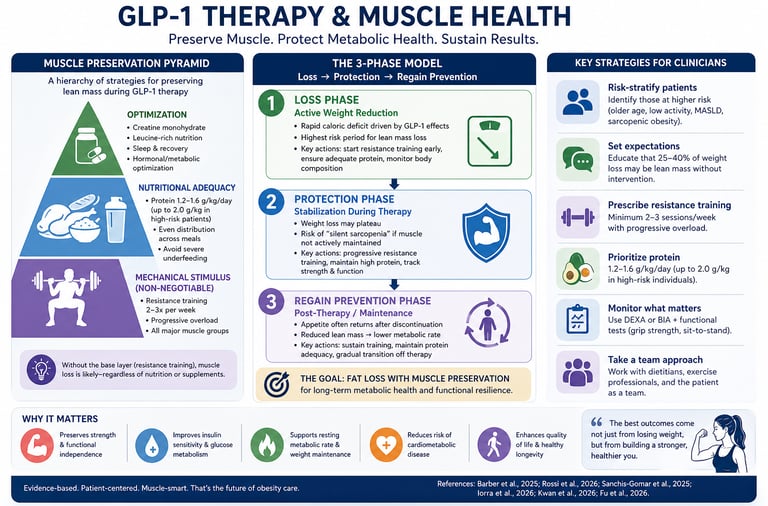
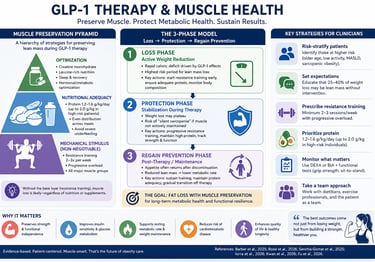
Muscle Preservation Pyramid in GLP-1 Therapy
The Muscle Preservation Pyramid is a practical, hierarchy-based framework that prioritizes interventions according to their physiological impact on maintaining lean mass during GLP-1–induced weight loss.
Top Tier: Optimization (Advanced Support)
Creatine monohydrate supplementation
Leucine-rich amino acid intake
Sleep quality and recovery optimization
Hormonal/metabolic fine-tuning (e.g., vitamin D, testosterone, where appropriate)
These strategies enhance muscle retention, but only work effectively when foundational layers are in place.
Middle Tier: Nutritional Adequacy
Protein intake: 1.2–1.6 g/kg/day (up to 2.0 g/kg in high-risk individuals)
Even protein distribution across meals
Prevention of severe caloric underfeeding (common with appetite suppression)
This layer ensures substrate availability for muscle protein synthesis, a critical requirement during caloric deficit.
Base Tier: Mechanical Stimulus (Non-Negotiable)
Resistance training: 2–3 sessions/week minimum
Progressive overload targeting all major muscle groups
This is the cornerstone. Without mechanical loading, muscle loss is biologically inevitable—regardless of nutrition or pharmacotherapy.
Clinical Insight:
Most patients focus on diet alone. However, without resistance training (base of the pyramid), muscle preservation strategies are fundamentally compromised.
3-Phase Model: Loss → Protection → Regain Prevention
This model reframes GLP-1 therapy as a dynamic metabolic journey, not a static treatment.
Phase 1: Loss (Active Weight Reduction)
Goal: Maximize fat loss while minimizing muscle loss
Rapid caloric deficit driven by GLP-1 effects
The highest risk period for lean mass reduction
Key interventions:
Initiate resistance training early
Ensure adequate protein intake
Monitor body composition
Clinical priority: Prevent disproportionate muscle loss during rapid weight reduction.
Phase 2: Protection (Stabilization During Therapy)
Goal: Preserve muscle and maintain metabolic rate
Weight loss may plateau
Risk of “silent sarcopenia” if muscle is not actively maintained
Key strategies:
Progressive resistance training (increase intensity over time)
Maintain high protein intake
Functional strength monitoring
Clinical priority: Shift from passive weight loss to active body composition management.
Phase 3: Regain Prevention (Post-Therapy or Maintenance)
Goal: Prevent weight regain and metabolic rebound
Appetite often returns after discontinuation
Reduced lean mass → lower resting metabolic rate
Key strategies:
Sustain resistance training as a long-term habit
Maintain protein adequacy
Gradual transition off pharmacotherapy where appropriate
Clinical priority: Muscle becomes the primary defense against weight regain.
Clinical Takeaway:
Patients who exit GLP-1 therapy without adequate muscle mass are biologically predisposed to regain weight. Those who preserve muscle create a metabolic buffer that supports long-term success
Clinical Pearls: GLP-1s & Muscle Preservation
1. The "25% Rule" of Weight Quality
Scientific Perspective: In standard weight loss, roughly 25% of weight lost is lean mass. With GLP-1s, if protein and resistance training are ignored, that number can climb to 40%.
For every 10 pounds you lose on the scale, you want at least 7 or 8 of those to be fat. If you aren't eating protein, you’re essentially "digesting" your own muscles to make up the difference.
2. The Appetite Paradox
Scientific Perspective: GLP-1s induce profound satiety, often leading to a caloric intake below the basal metabolic requirement for protein, triggering muscle catabolism despite high fat stores.
The medicine makes you not want to eat—which is great for fat loss—but you have to treat protein like medicine. Even if you aren't hungry, hitting your protein goal is what keeps your metabolism from crashing.
3. Strength vs. Size (The MASLD Insight)
Scientific Perspective: Research in liver disease (MASLD) shows that muscle quality (strength) can sometimes be preserved even when muscle quantity (mass) drops.
Don't just watch the size of your arms; watch how much you can lift. If you are losing weight but getting "weaker" in your daily life, your dosage or nutrition needs immediate adjustment.
4. The "Sarcopenic Obesity" Trap
Scientific Perspective: Patients with high BMI but low baseline muscle are at the highest risk for "medical frailty" during rapid GLP-1 weight loss.
If you haven't exercised in years, you are at the highest risk for side effects. You shouldn't just "lose weight"; you should "rebuild your body." Strength training isn't an extra—it’s a safety requirement for the drug.
5. Creatine as a "Muscle Insurance Policy"
Scientific Perspective: Creatine monohydrate increases cellular hydration and ATP availability, which can mitigate the catabolic signals sent during a 20%+ caloric deficit.
Taking 3–5g of creatine daily is like an insurance policy for your muscles. It’s one of the cheapest and safest ways to make sure your workouts actually "stick" while you're eating less.
6. The "Exit Strategy" Principle
Scientific Perspective: GLP-1 discontinuation leads to a rapid return of "hedonic hunger." Without a high BMR (driven by muscle), the body will prioritize fat storage over lean mass during the regain phase.
Think of muscle as your "metabolic retirement fund." If you stop the injections one day, your muscle is the only thing that will keep the weight from coming right back. Build that fund now while the drug is helping you lose the fat.
Author’s Note
The rapid rise of GLP-1 receptor agonists marks a pivotal moment in the management of obesity and metabolic disease. As a clinician, I have seen firsthand how these therapies can transform lives—improving glycemic control, reducing cardiovascular risk, and achieving weight loss that was once difficult to sustain. These are not incremental advances; they represent a genuine shift in therapeutic capability.
However, medicine evolves not just by embracing innovation, but by refining how we use it. The emerging evidence around lean mass loss during GLP-1–induced weight reduction is not a reason for alarm—but it is a reason for precision. It challenges us to move beyond a simplistic “weight-centric” model and adopt a more nuanced, physiology-driven approach focused on body composition, functional capacity, and long-term metabolic resilience.
This article was written to bridge that gap—between exciting pharmacological progress and the practical realities of patient care. The goal is not to discourage the use of GLP-1 therapies, but to ensure they are used intelligently and comprehensively, alongside resistance training, adequate protein intake, and appropriate monitoring.
For clinicians, this is an opportunity to lead—to integrate pharmacotherapy with lifestyle science in a way that maximizes benefit and minimizes unintended consequences. For patients, it is a reminder that sustainable health is not defined by the number on the scale alone, but by the strength, function, and metabolic health of the body.
This blog post is for educational purposes only and does not constitute medical advice. Always consult a qualified healthcare professional before starting, stopping, or adjusting any medication or exercise programme.
Related Articles:
Can Obesity Weaken Your Bones? The Hidden Fracture Risk Explained
Incretin Therapy Explained: GLP-1, Tirzepatide & Next-Gen Drugs
Can Healthy Mitochondria Protect Against Sarcopenia and Age-Related Decline? | DR T S DIDWAL
Stopping GLP-1: Will the Weight Come Back? | DR T S DIDWAL
Sarcopenic Obesity: How to Lose Fat Safely Without Losing Muscle | DR T S DIDWAL
References
Barber, T. M., Kabisch, S., Pfeiffer, A. F. H., & Weickert, M. O. (2025). Optimised skeletal muscle mass as a key strategy for obesity management. Metabolites, 15(2), 85. https://doi.org/10.3390/metabo15020085
Fu, L., Ding, R., Xu, G., Hu, J., & Yang, P. (2026). Amycretin in obesity: Mechanisms, clinical efficacy, and future perspectives. Metabolism, 179, 156594. https://doi.org/10.1016/j.metabol.2026.156594
Iorra, F., Jayakar, T., Yee, M., Thursz, M. R., Schaan, B. D., & Manousou, P. (2026). Effects of GLP-1 receptor agonists on muscle mass, strength, and quality in MASLD: A systematic review. Liver International, 46(5), e70643. https://doi.org/10.1111/liv.70643
Kwan, A. T. H., Chan, K. K. C., & colleagues. (2026). Muscle atrophy associated with glucagon-like peptide-1 receptor agonists: Implications for skeletal muscle and metabolism. Clinical Nutrition. Advance online publication. https://doi.org/10.1016/j.clnu.2026.106620
Rossi, G., Bucciarelli, L., Mananguite, C. L., et al. (2026). Muscle loss and GLP-1R agonists use. Acta Diabetologica, 63, 333–342. https://doi.org/10.1007/s00592-025-02611-2
Sanchis-Gomar, F., Neeland, I. J., & Lavie, C. J. (2025). Balancing weight and muscle loss in GLP1 receptor agonist therapy. Nature Reviews Endocrinology, 21, 584–585. https://doi.org/10.1038/s41574-025-01160-6