Can Obesity Weaken Your Bones? The Hidden Fracture Risk Explained
Weight loss can harm bone health without the right plan. Discover expert strategies including nutrition, resistance training, and monitoring
OBESITYSARCOPENIA
Dr. T.S. Didwal, M.D.(Internal Medicine)
4/27/202617 min read
What is the impact of obesity on bone health?
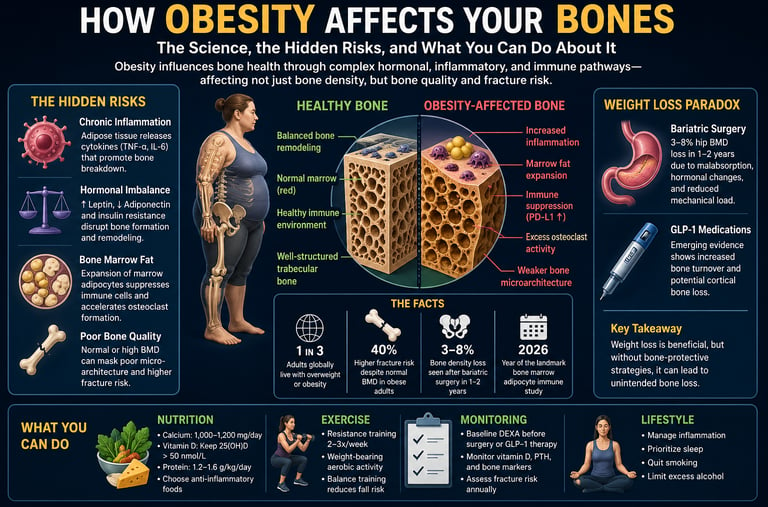
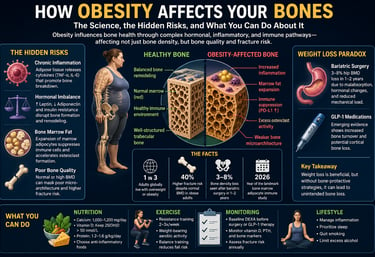
Obesity affects bone health through a combination of chronic inflammation, hormonal imbalance, and altered bone remodeling, rather than simply increasing bone strength. While higher body weight may increase bone mineral density (BMD), it often reduces bone quality, leading to a paradoxical increase in fracture risk.
Key mechanisms include:
Increased body weight → Enhances mechanical loading, which may increase bone mineral density (BMD), but does not guarantee stronger bones
Chronic inflammation → Elevates cytokines (e.g., TNF-α, IL-6), leading to increased bone resorption
Hormonal imbalance → Disrupts leptin, adiponectin, and insulin signaling, reducing bone formation
Bone marrow fat expansion → Causes immune suppression and promotes osteoclast activation, accelerating bone breakdown
Dysregulation of Wnt/β-catenin and RANKL/OPG pathways
Does weight loss affect bones?
Yes. Rapid weight loss—especially after bariatric surgery or GLP-1 therapy—can lead to 3–8% bone density loss, particularly at the hip, unless protective strategies are implemented.
How to protect bone health:
Resistance training (2–3 times/week)
Adequate protein (1.2–1.6 g/kg/day)
Calcium (1000–1200 mg/day) and vitamin D optimization
Bone density monitoring (DEXA) during weight loss
Bottom line:
Obesity does not reliably protect bones. Instead, it alters bone biology at molecular, hormonal, and immune levels, making proactive bone health strategies essential.
Clinician’s Perspective
Reframe the risk narrative
Obesity should no longer be viewed as protective for bone. While bone mineral density (BMD) may be normal or elevated, bone quality is often impaired, leading to disproportionate fracture risk—especially at the ankle, humerus, and hip.Look beyond DEXA alone
Standard BMD assessment may underestimate skeletal fragility in obesity. Consider adjuncts such as trabecular bone score (TBS) or clinical risk tools (e.g., FRAX), particularly in high-risk patients (postmenopausal, sedentary, or on long-term medications).Recognize inflammation-driven bone loss
Chronic low-grade inflammation associated with adiposity promotes osteoclast activation and osteoblast suppression. This shifts remodeling balance toward net bone loss—even in the presence of mechanical loading.Assess bone health before weight-loss interventions
Establish a baseline DEXA scan prior to bariatric surgery or long-term pharmacotherapy (e.g., GLP-1 receptor agonists). This enables meaningful longitudinal monitoring and early intervention.Anticipate post-weight-loss bone decline
Rapid weight loss—particularly after bariatric procedures—is associated with 3–8% hip BMD loss within 1–2 years. Monitor calcium, vitamin D, and parathyroid hormone (PTH), and consider bone turnover markers where available.Prioritize resistance training as first-line therapy
Among lifestyle interventions, progressive resistance exercise remains the most effective strategy to preserve bone mass during caloric restriction. Encourage initiation before or alongside weight loss, not after.Optimize nutritional support
Ensure adequate intake of:Protein: 1.2–1.6 g/kg/day
Calcium: 1000–1200 mg/day
Vitamin D: Maintain serum 25(OH)D > 50 nmol/L
Post-bariatric patients may require calcium citrate supplementation.
Evaluate medication-related bone effects
Review therapies that may influence bone metabolism, including glucocorticoids, proton pump inhibitors, and emerging data on GLP-1 agents. Balance metabolic benefits with skeletal considerations.Adopt a multidisciplinary approach
Coordinate care across endocrinology, nutrition, and physiotherapy. Bone health in obesity is multifactorial, requiring integrated, proactive management rather than reactive treatment.
Clinical Takeaway
In obesity, normal BMD does not equal normal bone health—early assessment, targeted intervention, and continuous monitoring are essential to prevent silent skeletal decline.
Most people think extra body weight protects their bones. On the surface, the logic seems sound: more weight means more mechanical loading, which should stimulate bone formation and increase bone mineral density (BMD). For decades, this assumption shaped both clinical thinking and public perception. But emerging research over the past five years reveals a far more complex—and concerning—reality. Obesity does not simply load the skeleton; it reprograms it at a molecular and cellular level, often in ways that silently weaken bone quality despite normal or even elevated BMD (Piñar-Gutierrez et al., 2022; Bagherifard et al., 2026).
At the center of this shift is a deeper understanding of adipose tissue. Far from being an inert energy store, body fat acts as an active endocrine organ, releasing inflammatory cytokines such as TNF-α and IL-6 that directly suppress bone-forming osteoblasts while accelerating osteoclast-driven bone resorption (Hou et al., 2020). At the same time, hormonal imbalances involving leptin, adiponectin, and insulin resistance disrupt critical signaling pathways—including Wnt/β-catenin and RANKL/OPG—that regulate bone remodeling (Armutcu et al., 2024). The result is a skeleton that may appear dense on imaging but is structurally compromised at the microarchitectural level.
Even more striking are recent discoveries inside the bone marrow itself. Expansion of marrow adipose tissue in obesity has been shown to suppress local immune function and promote excessive osteoclast formation through checkpoint pathways such as PD-L1—mechanisms previously associated with cancer biology (Costa et al., 2026). This reframes obesity-related bone loss not just as a metabolic issue, but as an immunological disorder within the skeletal niche.
Adding to the complexity, rapid weight loss—whether through bariatric surgery or GLP-1–based therapies—can further accelerate bone loss if not carefully managed (Palermo et al., 2025).
In short, the relationship between obesity and bone health is no longer a simple story of protection. It is a biologically intricate paradox—one that demands a more precise, proactive approach to skeletal care.
Section 01: The Bone–Fat Axis: A Two-Way Street
The skeleton is not a passive structure. Your bones are living tissue, continuously being broken down and rebuilt in a tightly regulated process called bone remodeling. Two key cell types govern this process: osteoblasts, which build new bone, and osteoclasts, which resorb (break down) old bone. When this balance tips — too much breakdown, too little building — bone density falls and fracture risk rises.
What has become clear over the past decade is that adipose tissue is not simply energy storage. Fat cells, called adipocytes, are metabolically active secretory organs. They release hormones, inflammatory proteins (cytokines), and lipid-derived signals that reach virtually every organ system — including the skeleton.
As Hou et al. (2020) demonstrated in their comprehensive review, obesity creates a state of chronic low-grade inflammation mediated primarily by adipose-derived signaling molecules, and this inflammatory environment has direct, measurable effects on bone metabolism. The study identified multiple interconnected pathways linking excess adiposity to impaired bone quality, setting the stage for the molecular investigations that followed.
1 in 3 Adults globally live with overweight or obesity
40% Higher fracture risk despite normal BMD in obese adults
3–5% Bone density loss seen after bariatric surgery in 1–2 years
2026 Year of the landmark bone marrow adipocyte immune study
Piñar-Gutierrez et al. (2022) extended this understanding at the molecular level, showing that the relationship between obesity and bone involves not just inflammation but also endocrine crosstalk — involving leptin, adiponectin, insulin, and sex hormones — as well as direct effects on stem cell differentiation within the bone marrow. Their work made clear that a single mechanism cannot explain the obesity–bone relationship; it is a convergence of many simultaneous dysregulations.
Section 02: The Molecular Pathways: What Goes Wrong Inside the Bone
Understanding how obesity disrupts bone at the molecular level requires a brief tour of the signaling pathways involved. Bagherifard et al. (2026), in their landmark review published in International Journal of Obesity, provide one of the most comprehensive mappings to date of these interactions. Their analysis identified three primary molecular domains of disruption.
Inflammatory Cytokine Cascade
TNF-α, IL-1β, and IL-6 — released by visceral adipose tissue — directly suppress osteoblast function while stimulating osteoclast formation and activity.
Leptin & Adiponectin Imbalance
Elevated leptin and reduced adiponectin in obesity impair bone mineralization and alter the RANKL/OPG ratio, tipping the balance toward bone resorption.
Insulin Resistance & IGF-1 Disruption
Insulin resistance impairs bone anabolic signaling. Disrupted IGF-1 axis blunts osteoblast proliferation and reduces bone matrix protein synthesis.
Wnt & RANKL Pathway Dysregulation
Obesity downregulates Wnt/β-catenin signaling (essential for bone formation) and upregulates RANKL, the primary driver of osteoclast differentiation.
What makes the findings of Bagherifard et al. (2026) particularly striking is the evidence for feedback loops: the bone itself is an endocrine organ, releasing osteocalcin and other factors that regulate adipose tissue. When obesity disrupts bone metabolism, it simultaneously impairs signals that could otherwise help regulate fat accumulation — creating a self-reinforcing cycle of metabolic dysfunction.
Armutcu et al. (2024), writing in the Journal of Cellular Signaling, placed particular emphasis on the RANKL/OPG (osteoprotegerin) system. In a healthy skeleton, OPG acts as a decoy receptor that prevents RANKL from binding to its osteoclast-activating receptor. Obesity shifts this ratio decisively toward RANKL dominance: adipose tissue-derived inflammatory signals suppress OPG production while simultaneously upregulating RANKL. The net result is excessive osteoclast activation — accelerated bone resorption that outpaces formation.
Clinical Insight
"Obesity significantly modifies signaling pathways associated with bone remodeling and metabolism — including Wnt/β-catenin, RANKL/OPG, and IGF-1 axis dysregulation — in ways that cumulatively favor bone loss regardless of mechanical loading." — Armutcu, McCloskey, & Ince (2024)
The Wnt/β-catenin pathway deserves special attention. It is arguably the most important bone-anabolic signaling cascade in adult skeletal maintenance. Activation of Wnt signaling promotes osteoblast differentiation, enhances bone matrix production, and suppresses apoptosis of bone-forming cells. Adipose-derived inflammation — particularly through the action of sclerostin and Dickkopf-1, both of which rise with obesity — systematically inhibits this pathway. The practical result is a bone that struggles to repair and renew itself adequately, even when its density appears normal on a DEXA scan.
Section 03: Inside the Bone Marrow: Immune Suppression and Osteoclast Overdrive
Perhaps the most unsettling finding in recent obesity-bone research concerns what is happening not at the surface of bones, but deep within the bone marrow cavity itself.
In a landmark 2026 study published in Bone Research, Costa et al. demonstrated for the first time that the expansion of bone marrow adipocytes (fat cells within the marrow) in obese mice leads to a two-pronged threat: local immune suppression and accelerated osteoclastogenesis — the production of bone-resorbing osteoclast cells.
Bone marrow fat doesn't just take up space. It actively remodels the immunological environment of the marrow cavity — suppressing anti-inflammatory T cells while promoting the differentiation of cells that destroy bone.
The mechanism identified by Costa et al. (2026) centres on PD-L1 — a protein widely known in oncology as a checkpoint molecule that cancer cells exploit to evade immune destruction. In the obese bone marrow, adipocyte-derived signals drive elevated PD-L1 expression on stromal cells. This suppresses cytotoxic T-cell activity, allowing osteoclast precursors to differentiate unchecked. In essence, the immune brake system that might otherwise limit osteoclast overactivation is disabled.
This finding has profound implications. It means that obesity-related bone loss is not simply a mechanical or hormonal phenomenon — it is also an immunological one, driven by a fundamentally altered marrow microenvironment. It also raises intriguing therapeutic possibilities: could checkpoint inhibitors or targeted bone marrow adipocyte interventions protect bone health in obese individuals? These remain active research questions, but the discovery opens an entirely new therapeutic frontier.
Section 04: Debunking the "Obesity Protects Bones" Myth
For years, epidemiological data appeared to show that people with obesity have higher bone mineral density (BMD) and, consequently, lower rates of osteoporosis — compared to leaner individuals. This led to a widespread clinical assumption that, at least from a skeletal standpoint, carrying extra weight was protective.
The picture has changed substantially. As Piñar-Gutierrez et al. (2022) carefully document, BMD is only one determinant of fracture risk. Bone quality — including trabecular microarchitecture, cortical porosity, and material properties — can be significantly compromised in obesity even when DEXA-measured BMD appears normal. People with obesity experience disproportionately high rates of fractures at specific skeletal sites, particularly the ankle, humerus, and non-vertebral axial sites.
Why? Several factors converge. First, adipose tissue infiltrates muscle (sarcopenic obesity), reducing the protective effect of muscle mass on bone. Second, falls are more likely in people with obesity due to impaired balance and altered gait mechanics — and when falls occur, the force of impact is greater. Third, the inflammatory and endocrine dysregulations documented in the molecular studies above compromise bone matrix quality in ways that DEXA simply cannot capture.
The "obesity is protective" narrative was always more nuanced than its popular framing suggested, and modern molecular research has now confirmed its limitations decisively.
Section 05: The Weight-Loss Paradox: When Losing Weight Means Losing Bone
Here lies one of the most clinically important and under-discussed findings in this field: weight loss — even intentional, medically supervised weight loss — can accelerate bone density loss.
Palermo et al. (2025), in their narrative review published in Current Diabetes Reports, synthesize evidence across multiple weight-loss modalities — dietary restriction, physical activity programs, bariatric surgery, and pharmacological intervention including GLP-1 receptor agonists. Their conclusions are sobering for clinicians and patients alike.
Bariatric surgery, particularly Roux-en-Y gastric bypass, is consistently associated with significant bone density loss — 3–8% at the hip within the first two years post-operation. The mechanisms include calcium and vitamin D malabsorption, dramatic reductions in mechanical loading, and secondary hyperparathyroidism. Even sleeve gastrectomy, once considered more bone-friendly, produces measurable BMD reductions.
The growing use of GLP-1 receptor agonists (such as semaglutide and tirzepatide) for weight management introduces new concerns. While these agents produce significant weight loss, the bone effects remain incompletely characterized. Palermo et al. (2025) note emerging evidence of accelerated bone turnover markers and potential cortical bone deficits, though longer-term data are still accumulating. Clinicians prescribing these therapies should consider baseline bone density assessment and proactive monitoring.
Important Note for Patients
This does not mean weight loss is bad for your bones. What it means is that how you lose weight — and what protective strategies accompany it — matters enormously. The risks are not inevitable; they are manageable with the right approach.
Lifestyle-based weight loss that incorporates adequate dietary protein, calcium, vitamin D, and particularly resistance exercise appears to attenuate bone loss during caloric restriction. The central message from Palermo et al. (2025) is not that weight loss should be avoided, but that it must be paired with bone health strategies from the outset — not as an afterthought.
Section 06: Practical Applications: Protecting Your Bones While Managing Weight
Translating this science into daily action is where research meets real life. Based on the six studies reviewed here, the following evidence-based strategies represent the current best practice for protecting skeletal health in the context of obesity and weight management.
Nutrition Essentials
Aim for 1,000–1,200 mg calcium daily from food sources (dairy, leafy greens, fortified foods)
Supplement vitamin D3 to maintain serum 25(OH)D above 50 nmol/L; levels often fall post-surgery
Prioritize protein: 1.2–1.6 g/kg body weight supports both muscle and bone preservation
Limit ultra-processed foods — their inflammatory load mimics adipose-derived cytokine effects on bone
After bariatric procedures, work with a dietitian on calcium citrate supplementation specifically
Exercise Prescription
Resistance training (2–3 sessions/week) is the single most effective intervention for preserving BMD during weight loss
Weight-bearing aerobic activity (walking, jogging, dancing) stimulates osteoblast activity via mechanical signals
Balance training reduces fall risk, which is disproportionately elevated in obesity
Begin structured exercise before or concurrent with caloric restriction — not after
Even seated or aquatic resistance work confers bone benefit in those with limited mobility
Medical Monitoring
Request a DEXA scan baseline before bariatric surgery or initiating GLP-1 therapy
Monitor vitamin D, PTH, and bone turnover markers (P1NP, CTX) every 6–12 months post-surgery
Discuss bisphosphonate or denosumab options with your doctor if post-surgical BMD loss is significant
Review all medications for bone-adverse effects (steroids, PPIs, certain diabetes drugs)
Annual fracture risk assessment using FRAX tool is appropriate for adults with obesity
Lifestyle & Inflammation Control
Prioritize 7–9 hours of quality sleep — poor sleep elevates cortisol, which suppresses osteoblast activity
Smoking cessation is critical: nicotine directly impairs osteoblast function and increases oxidative stress in bone
Moderate alcohol intake: chronic excess disrupts the Wnt signaling pathway central to bone formation
Manage metabolic syndrome components — insulin resistance has direct anti-anabolic effects on bone
Anti-inflammatory dietary patterns (Mediterranean diet) may attenuate adipose-derived cytokine burden
Section 07: Frequently Asked Questions
1 Does obesity cause osteoporosis?
Not in a straightforward, direct sense — but it significantly increases the risk of poor bone quality and atypical fractures. Obesity elevates inflammatory signals (TNF-α, IL-6) and disrupts hormones like adiponectin and IGF-1 that are essential for bone maintenance. While BMD may appear normal on a DEXA scan, bone microarchitecture and material strength can be compromised. Piñar-Gutierrez et al. (2022) document that people with obesity fracture at higher-than-expected rates at the ankle, humerus, and hip, even without a formal osteoporosis diagnosis.
2 Will losing weight make my bones weaker?
Weight loss without bone-protective strategies can reduce bone density, particularly rapid weight loss through surgery or aggressive caloric restriction. Palermo et al. (2025) found that bariatric surgery is associated with 3–8% hip BMD loss within two years. However, gradual, nutrition-supported weight loss combined with resistance exercise can preserve or even improve bone quality. The key is to treat bone health as an active priority during weight loss, not an afterthought.
3 Are GLP-1 drugs like semaglutide safe for my bones?
The long-term bone effects of GLP-1 receptor agonists are still being studied. Palermo et al. (2025) note emerging signals of elevated bone turnover markers and potential cortical bone changes with sustained use, though this is not yet fully characterized. If you are using these medications, speak with your doctor about baseline bone density assessment and calcium and vitamin D optimization. Pairing GLP-1 therapy with resistance exercise is strongly advisable to mitigate potential bone loss.
4 What is bone marrow fat, and why does it matter?
Bone marrow fat (marrow adipose tissue) fills the interior of bones and expands substantially in obesity. Unlike subcutaneous fat, bone marrow adipocytes directly interact with the bone's immune and structural cells. Costa et al. (2026) demonstrated that expanded marrow fat in obese mice suppresses immune surveillance via PD-L1 signaling and dramatically accelerates osteoclast production — the cells that break down bone. This means obesity doesn't just affect bone from the outside through inflammation; it fundamentally alters the bone's internal environment.
5 Which exercises are best for protecting bone health in obesity?
Resistance (strength) training is the gold standard for preserving bone density, particularly at the hip and spine. Weight-bearing aerobic activity — walking, low-impact aerobics, stair climbing — provides additional osteogenic stimulus through mechanical loading. For individuals with joint limitations common in obesity, aquatic resistance training and seated strength exercises also confer measurable bone benefit. The evidence strongly supports starting resistance training before or concurrent with caloric restriction, not after weight has been lost, as this is when the bone-protective effect is most needed.
6 Should I get a bone density scan if I am obese?
Increasingly, the answer is yes — particularly if you are planning bariatric surgery, long-term GLP-1 therapy, or if you have other risk factors (low physical activity, vitamin D deficiency, corticosteroid use, postmenopausal status). Standard DEXA scanning may underestimate fracture risk in people with high BMI due to technical limitations, so discuss with your doctor whether additional imaging (such as trabecular bone score or high-resolution peripheral quantitative CT) may be appropriate. Baseline measurement before any weight-loss intervention allows meaningful comparison over time.
7 Can reducing inflammation through diet help protect my bones?
Yes, and the mechanism is well-supported by the molecular evidence. Bagherifard et al. (2026) and Hou et al. (2020) both document how adipose-derived inflammatory cytokines — TNF-α, IL-1β, IL-6 — directly suppress bone formation and accelerate resorption. Anti-inflammatory dietary patterns, particularly the Mediterranean diet (rich in omega-3 fatty acids, polyphenols, fiber, and antioxidants), have been shown to reduce circulating inflammatory markers. While this alone will not reverse obesity-related bone changes, it meaningfully attenuates one of the key drivers of bone loss and supports overall metabolic health.
8 Does obesity weaken bones?
Obesity does not directly cause osteoporosis but impairs bone quality through inflammation, hormonal disruption, and increased bone resorption. This leads to a higher fracture risk despite normal bone density.
9 . Does losing weight cause bone loss?
Yes, especially with rapid weight loss. Studies show 3–8% bone loss at the hip within 1–2 years after bariatric surgery. However, resistance training and proper nutrition can significantly reduce this risk.
10 Why does fat affect bone health?
Fat tissue releases inflammatory and hormonal signals that:
Suppress bone-forming cells (osteoblasts)
Activate bone-resorbing cells (osteoclasts)
Disrupt key pathways like Wnt and RANKL/OPG
Clinical pearls
1. The Density vs. Quality Delusion
Scientific Perspective: High Body Mass Index (BMI) often correlates with elevated Bone Mineral Density (BMD) due to mechanical loading, but this frequently masks microarchitectural deterioration. The inflammatory milieu (TNF-α, IL-6) and marrow adiposity impair bone "quality" and material strength.
"Heavy" bones aren't always "strong" bones. Just because a bone looks dense on a standard scan doesn't mean it’s healthy. Think of it like a piece of wood: it might be thick, but if termites (inflammation) have hollowed out the inside, it will still snap easily under pressure.
2. The Bone Marrow "Takeover"
Scientific Perspective: Obesity triggers the expansion of Marrow Adipose Tissue (MAT). These marrow fat cells aren't just fillers; they promote local immunosuppression via the PD-L1 pathway, which disables the "brakes" on osteoclast activity, leading to accelerated bone resorption.
Fat doesn't just sit under the skin; it can move inside your bones. When too much fat crowds the inside of your bone marrow, it interferes with your body’s ability to repair the bone. It essentially "bullies" your bone-building cells, making it harder for your skeleton to stay renewed.
3. The "Weight-Loss Tax" on Skeletal Health
Scientific Perspective: Rapid weight loss—especially via bariatric surgery or GLP-1 receptor agonists—induces a "calcium-stealing" effect. Mechanical unloading combined with hormonal shifts can lead to a $3\text{--}8\%$ drop in hip BMD within 12–24 months if not mitigated.
Losing weight is great for your heart, but your bones pay a "tax" for it. When you lose weight quickly, your body thinks it doesn't need as much "scaffolding" to hold you up, so it starts thinning out the bone. You have to actively tell your body to keep that bone by doing the right exercises.
4. Resistance Training as "Skeletal Insurance"
Scientific Perspective: Progressive resistance training is the most effective non-pharmacological stimulus for the Wnt/β-catenin signaling pathway, which is essential for osteoblast differentiation. It provides the mechanical "strain" necessary to counteract the catabolic effects of caloric restriction.
Lifting weights is a "Keep" signal for your skeleton. While walking is good, lifting something heavy (resistance training) sends a loud, clear message to your brain: "I still need these bones, don't break them down!" It’s the best way to protect your frame while the fat melts away.
5. The Inflammation-Fracture Link
Scientific Perspective: Obesity is a state of chronic low-grade systemic inflammation. Adipokines like leptin (in excess) and reduced adiponectin disrupt the RANKL/OPG ratio, favoring the formation of osteoclasts (bone-destroyers) over osteoblasts (bone-builders).
Body fat acts like a chemical factory. This "factory" pumps out inflammatory chemicals that travel through your blood and irritate your bones. This irritation speeds up how fast your body dissolves old bone, often faster than it can build new bone to replace it.
6. Protein: The Overlooked Bone Builder
Scientific Perspective: Bone is roughly $50\%$ protein by volume. In the context of weight loss, an intake of 1.2--1.6 g/kg/day is required to maintain the IGF-1 axis, which stimulates bone-forming cells and maintains the structural collagen matrix of the bone.
Your bones are made of more than just calcium. People usually only think about milk, but your bones have a huge "netting" of protein that holds everything together. If you don't eat enough protein while losing weight, that netting gets weak, and the calcium has nothing to stick to.
Author’s Note
The relationship between obesity and bone health is often misunderstood—sometimes even within clinical practice. For years, the prevailing assumption was that higher body weight offered a protective advantage to the skeleton. While partially true from a mechanical loading perspective, this view is now clearly incomplete. As emerging evidence shows, obesity introduces a complex interplay of inflammatory, hormonal, metabolic, and immunological factors that can quietly undermine bone integrity.
This article was written to bridge that gap between traditional assumptions and modern scientific understanding. The goal is not to oversimplify, but to translate evolving research—from molecular pathways like RANKL/OPG and Wnt signaling to newer insights into bone marrow adiposity—into clinically meaningful, actionable knowledge.
A particularly important motivation behind this piece is the growing use of weight-loss interventions, including bariatric surgery and GLP-1 receptor agonists. While these therapies offer substantial metabolic benefits, their effects on bone health are still being fully understood. Clinicians and patients alike need to approach weight loss with a dual focus: metabolic improvement and skeletal preservation.
It is also important to emphasize that bone health is dynamic and modifiable. The risks outlined here are not inevitable. With appropriate strategies—resistance training, nutritional optimization, and timely monitoring—bone loss can be mitigated, and in some cases, prevented.
Finally, this work reflects a broader shift in medicine: moving from organ-specific thinking to systems biology. Bone is not an isolated structure—it is deeply connected to metabolism, immunity, and endocrine function. Understanding these connections allows us to practice more precise, preventive, and personalized care.
Obesity doesn’t just affect metabolism—it reprograms bone biology at a cellular and molecular level
You can have normal bone density but still be at high fracture risk due to poor bone quality
Fat tissue acts as an endocrine organ, releasing signals that weaken bone over time
Bone loss in obesity is driven not only by hormones but also by chronic inflammation and immune dysfunction
Rapid weight loss can improve metabolic health—but without a proper strategy, it may silently reduce bone density
The real risk is hidden: bone microarchitecture deteriorates even when standard scans appear normal
Protecting bone during weight loss requires a targeted approach combining nutrition, resistance training, and monitoring
This article is for informational purposes only and does not constitute medical advice. Always consult a qualified healthcare professional before making changes to your diet, exercise routine, or medical treatment plan.
Related Articles
Can Healthy Mitochondria Protect Against Sarcopenia and Age-Related Decline? | DR T S DIDWAL
Stopping GLP-1: Will the Weight Come Back? | DR T S DIDWAL
Sarcopenic Obesity: How to Lose Fat Safely Without Losing Muscle | DR T S DIDWA
Obesity Isn’t Just Fat: How Muscle Quality Controls Fat Loss and Metabolism
References
Armutcu, F., McCloskey, E., & Ince, M. (2024). Obesity significantly modifies signaling pathways associated with bone remodeling and metabolism. Journal of Cellular Signaling, 5(4), 183–194. https://doi.org/10.33696/Signaling.5.124
Bagherifard, A., Hemmatyar, A., Khosravi, K., et al. (2026). The impact of obesity on bone health: molecular pathways, metabolic interactions, and associated pathologies. International Journal of Obesity, 50, 87–115. https://doi.org/10.1038/s41366-025-01907-1
Costa, S. N., Chlebek, C., Gray, L., et al. (2026). Expansion of bone marrow adipocytes in obese mice leads to PD-L1-driven bone marrow immunosuppression and osteoclastogenesis. Bone Research, 14, 32. https://doi.org/10.1038/s41413-026-00509-5
Hou, J., He, C., He, W., Yang, M., Luo, X., & Li, C. (2020). Obesity and bone health: a complex link. Frontiers in Cell and Developmental Biology, 8, 600181. https://doi.org/10.3389/fcell.2020.600181
Palermo, A., Tsourdi, E., Yavropoulou, M. P., et al. (2025). The effects of obesity and weight loss interventions on bone health: a narrative review. Current Diabetes Reports, 25, 52. https://doi.org/10.1007/s11892-025-01609-y
Piñar-Gutierrez, A., García-Fontana, C., García-Fontana, B., & Muñoz-Torres, M. (2022). Obesity and bone health: a complex relationship. International Journal of Molecular Sciences, 23(15), 8303. https://doi.org/10.3390/ijms23158303