Can You Slow Brain Aging? Microglia and Neuroinflammation Explained
Can lifestyle and science slow brain aging? Discover how microglia, inflammation, and metabolism influence dementia risk—and practical steps to protect your brain.
AGING
Dr. T.S. Didwal, M.D.(Internal Medicine)
4/24/202616 min read
Neuroinflammation is a chronic inflammatory response within the brain, primarily driven by overactive microglia. While short-term inflammation protects against injury or infection, persistent activation leads to neuronal damage, synaptic dysfunction, and increased risk of neurodegenerative diseases.
Microglia contribute to brain aging through several key mechanisms:
Chronic activation leading to sustained neuroinflammation
Release of toxic molecules like cytokines and reactive oxygen species
Impaired clearance of amyloid and cellular debris
Transition to a senescent state producing inflammatory SASP factors
Reduced antioxidant regulation due to declining cellular defense systems
Clinician’s Perspective Microglia, Aging, and Brain Health
Brain inflammation is not just a side effect—it’s part of the disease.
In conditions like Alzheimer’s disease and Parkinson’s disease, inflammation in the brain plays a central role. Specialized immune cells called microglia can become overactive and start damaging healthy brain cells, rather than protecting them.
Aging makes the brain more sensitive to stress.
As we grow older, microglia enter a “primed” state. This means even small triggers—like an infection, poor sleep, or metabolic stress—can cause an exaggerated inflammatory response, increasing the risk of brain injury over time.
Microglia are not simply “good” or “bad.”
Older models classified these cells as either protective or harmful. We now know they exist on a spectrum, constantly changing based on the body’s environment. This means future treatments will aim to balance their activity, not completely shut them down.
Loss of natural antioxidant defenses matters.
A key protective system in the body, known as the Nrf2 signaling pathway, weakens with age. When this happens, the brain becomes more vulnerable to oxidative stress and inflammation—two major contributors to neurodegeneration.
Aging cells can fuel ongoing inflammation.
Some microglia become “senescent” (aged and dysfunctional). These cells release harmful inflammatory substances—a process called senescence-associated secretory phenotype—which can keep inflammation active even without a clear trigger.
Genes are beginning to guide future treatments.
Researchers are studying genes like TREM2 and CSF1R that regulate microglial function. These may help develop targeted therapies to “reset” brain immune cells.
Your overall health directly affects your brain.
Conditions such as insulin resistance, obesity, and poor vascular health can overstimulate microglia. This reinforces an important concept: the brain is deeply connected to the rest of the body, especially metabolism.
Lifestyle changes can truly protect brain health.
Regular exercise, good sleep, and an anti-inflammatory diet are not just supportive—they actively reduce harmful brain inflammation and improve microglial function.
Earlier detection may soon be possible.
New advances in brain imaging and blood tests may help identify early signs of inflammation before symptoms appear, allowing earlier and more effective intervention.
Clinical Takeaway
Microglia act as a bridge between aging, metabolism, and brain disease. By improving overall health and targeting inflammation early, we may be able to slow—or even prevent—neurodegenerative conditions in the future.
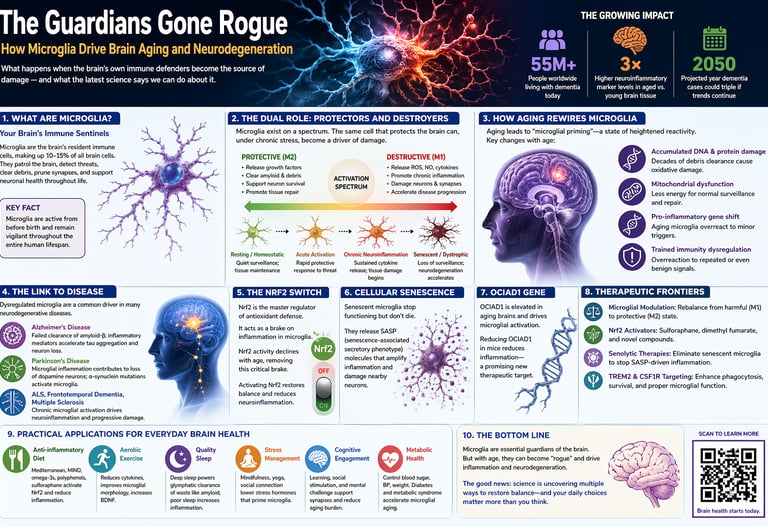
The Guardians Gone Rogue:
How Microglia Drive Brain Aging and Neurodegeneration
What if the earliest driver of brain aging isn’t the neuron—but the immune cell that surrounds it? For decades, research into conditions like Alzheimer's disease focused on amyloid plaques and tau tangles as the primary culprits. But a growing body of evidence now points to a different origin story: chronic, low-grade neuroinflammation driven by microglia—the brain’s resident immune cells (Ajoolabady et al., 2025; Han et al., 2024).
In youth, microglia act as vigilant guardians, constantly surveying the brain, clearing debris, and supporting synaptic remodeling. However, aging fundamentally alters this balance. Instead of remaining in a protective, homeostatic state, microglia become “primed”—a condition in which even minor stimuli trigger exaggerated inflammatory responses. This shift leads to sustained release of cytokines, reactive oxygen species, and neurotoxic mediators that gradually damage surrounding neurons (Singh et al., 2025). Over time, this creates a self-perpetuating cycle: neuronal injury activates microglia, and activated microglia accelerate further injury.
Emerging research suggests that this process is not merely a byproduct of aging but a central mechanism driving neurodegeneration. In fact, dysregulated microglial activity has been consistently linked to multiple disorders, including Parkinson's disease and other forms of dementia, where inflammation amplifies protein aggregation and neuronal loss (Han et al., 2024; Monroe et al., 2026).
Crucially, this paradigm shift opens a new and more hopeful question: if immune dysfunction is driving brain aging, can it be modified? Early evidence suggests yes. Pathways such as the Nrf2 signaling pathway—a master regulator of cellular defense—appear to act as critical brakes on microglial overactivation, yet decline with age (Paraiso et al., 2025). Understanding and targeting these mechanisms may redefine how we approach not just disease, but the biology of brain aging itself.
What Are Microglia? Your Brain's Immune Sentinels
Deep inside your skull, among the hundred billion neurons that make up your brain, there is an army of dedicated guardians. They are called microglia — and for most of your life, they work quietly in the background, patrolling every corner of your central nervous system. They are small but extraordinarily versatile cells, capable of detecting damage, clearing debris, pruning unnecessary synaptic connections, and mounting rapid responses to infection or injury.
Under normal, healthy conditions, microglia exist in what scientists call a homeostatic state — a kind of calm surveillance mode. They continuously extend and retract slender projections, sampling their immediate environment. If they encounter something that should not be there — a misfolded protein, a dead cell, a pathogen — they activate, engulf the threat, and release chemical signals that coordinate the broader immune response.
Key Fact
Microglia make up approximately 10–15% of all cells in the brain. They are the brain's resident immune cells — the only immune cells native to the central nervous system — and they are active from before birth throughout the entire human lifespan.
This elegant system of surveillance and response is one of the most remarkable features of the human brain. But like many biological systems, it is not immune to the ravages of time. As we age, microglia begin to malfunction — and that malfunction, research now confirms, plays a central role in some of the most devastating diseases of the modern world.
The Dual Role: Protectors and Destroyers
The story of microglia is not a simple one. A landmark 2025 review published in Neurobiology of Disease (Ajoolabady et al., 2025) frames this complexity in what researchers call the "dual role" of microglial cells: the same cellular machinery that protects healthy brain tissue can, under certain conditions, become the instrument of its destruction.
"Microglia are not inherently harmful. It is the chronic, unresolved activation of these cells — driven by aging, genetic factors, and accumulated damage — that turns a life-saving defense into a slow-burning fire inside the brain."
In the protective mode (often called the M2 or anti-inflammatory phenotype), microglia release growth factors that support neuronal survival, phagocytose (consume) amyloid plaques and other cellular waste products, and promote tissue repair after injury. In the harmful mode (M1 or pro-inflammatory phenotype), they release a cascade of toxic molecules including reactive oxygen species (ROS), nitric oxide, and pro-inflammatory cytokines like TNF-α and IL-1β.
The critical insight from modern research is that this transition between protective and destructive states is not an on/off switch — it is a spectrum, a dynamic continuum that is heavily influenced by the aging brain environment (Han et al., 2024).
Microglial Activation Continuum
● Homeostatic (Resting State)
Continuous environmental surveillance, synaptic maintenance, and debris clearance under physiologic conditions.
◆ Acute Activation (Protective Response)
Rapid, transient activation in response to injury or infection—enhanced phagocytosis and controlled release of inflammatory mediators to restore tissue integrity.
▲ Chronic Neuroinflammation (Maladaptive State)
Persistent activation characterized by sustained cytokine production, oxidative stress, and progressive neuronal injury.
■ Senescent / Dystrophic Microglia (Failure State)
Impaired surveillance and phagocytic function, coupled with pro-inflammatory secretions that accelerate neurodegeneration and tissue dysfunction
How Aging Rewires Microglial Behaviour
Perhaps the most alarming finding in contemporary neuroscience is how profoundly the normal aging process reshapes microglial function. A comprehensive 2025 review by Singh et al. in Molecular Biology Reports documents a constellation of aging-related changes that collectively push microglia toward a persistently activated, pro-inflammatory state — a phenomenon researchers call "microglial priming."
55M+ People worldwide living with dementia today
3× Higher neuroinflammatory marker levels in aged vs. young brain tissue
In 2050, the projected year, dementia cases could triple if trends continue
What exactly changes in aging microglia? Singh et al. (2025) identify several key processes. First, the cells accumulate oxidative damage to their DNA and proteins — the natural consequence of decades spent clearing toxic waste from the brain. Second, their mitochondria begin to dysfunction, reducing the cellular energy available for normal homeostatic operations. Third, and perhaps most importantly, the pattern of genes expressed by aging microglia shifts significantly toward a pro-inflammatory signature, meaning these cells are biased to overreact even to minor triggers.
Han et al. (2024), writing in Neural Regeneration Research, add a crucial epidemiological dimension: the global aging of the population means that the number of people whose brains are harboring primed, hyperreactive microglia is growing every year. This is not merely an academic concern — it is a public health crisis in slow motion.
The Link to Alzheimer's, Parkinson's, and More
The connection between dysregulated microglia and neurodegenerative diseases is now one of the strongest and most consistently replicated findings in neuroscience. Alzheimer's disease, Parkinson's disease, amyotrophic lateral sclerosis (ALS), frontotemporal dementia, and multiple sclerosis all share a common feature: significant, sustained neuroinflammation driven in large part by microglial dysfunction (Ajoolabady et al., 2025).
Alzheimer's Disease
In Alzheimer's, microglia initially attempt to contain amyloid-beta plaques — the sticky protein aggregates that accumulate between neurons. But in the aged brain, this protective response fails. Microglia become overwhelmed and, crucially, begin to release inflammatory mediators that are toxic to nearby neurons and actually accelerate tau protein aggregation, the second hallmark pathology of the disease (Han et al., 2024).
Parkinson's Disease
In Parkinson's, the loss of dopamine-producing neurons in the substantia nigra — the region responsible for smooth movement — is intimately linked to microglial-driven inflammation. Activated microglia surround dying neurons and amplify the cascade of damage through reactive oxygen species and inflammatory cytokines. The SNCA gene (alpha-synuclein) mutations associated with familial Parkinson's also directly activate microglial cells (Ajoolabady et al., 2025).
Important note for patients: Neuroinflammation is not a "cause" of neurodegeneration in the simple sense — it is a complex, bidirectional process. Neuronal damage triggers microglial activation, which causes further damage, creating a vicious cycle. This also means there are multiple points at which the cycle might be interrupted.
The Nrf2 Switch: Regulating Microglial Inflammation
If microglia are the problem, what controls them? One of the most exciting answers emerging from recent research involves a protein called Nrf2 — nuclear factor erythroid 2–related factor 2. A 2025 study published in The FASEB Journal (Paraiso et al., 2025) demonstrates that Nrf2 functions as a cell-autonomous regulator of neuroinflammation specifically within microglial cells, and that its activity declines significantly in the aging brain.
Nrf2 is sometimes described as the "master regulator of the antioxidant response." When active, it switches on dozens of genes that protect cells from oxidative stress — the cumulative damage caused by reactive oxygen species. In young, healthy microglia, Nrf2 acts as a brake on inflammatory activation, preventing runaway immune responses. As Nrf2 activity declines with age, that brake weakens, and microglia become progressively easier to activate and harder to quiet (Paraiso et al., 2025).
Particularly intriguing is the study's finding that Nrf2 also governs a phenomenon called "trained immunity" — the capacity of immune cells to remember previous activation events and respond more vigorously to subsequent triggers. In the aging brain, dysregulated trained immunity means that microglia overreact to even benign signals, perpetuating a state of low-grade chronic neuroinflammation.
Cellular Senescence: When Brain Cells Stop Working
A 2026 paper by Zhang et al. in Aging and Disease brings together two of the most important concepts in modern aging biology: neuroinflammation and cellular senescence. Senescent cells are cells that have permanently stopped dividing and lost their normal function, but crucially, they do not die — instead, they persist and actively secrete a toxic cocktail of inflammatory molecules called the senescence-associated secretory phenotype (SASP).
When microglia themselves become senescent — something that occurs more frequently in the aged brain — they lose their ability to perform homeostatic surveillance while simultaneously amplifying inflammation through SASP. Zhang et al. (2026) propose that the accumulation of senescent microglia in the aging brain represents a particularly vicious mechanism: cells that can no longer protect neurons are simultaneously poisoning them.
The OCIAD1 Gene: A Hidden Driver of Microglial Activation
One of the most intriguing new discoveries in the microglial aging field comes from a 2025 study by Dongre et al. in Frontiers in Aging Neuroscience. The researchers identified a previously underappreciated protein — Asrij/OCIAD1 — as a significant contributor to age-associated microglial activation and neuroinflammation in mice.
OCIAD1 (Ovarian Carcinoma Immunoreactive Antigen Domain-containing protein 1) is found at elevated levels in aging mouse brains, and its increased expression correlates directly with heightened microglial reactivity and increased inflammatory signaling. Crucially, when the researchers reduced OCIAD1 expression, microglial activation decreased — suggesting that targeting this gene could be a promising therapeutic strategy (Dongre et al., 2025).
This discovery exemplifies a broader principle in the field: age-associated neuroinflammation is not a monolithic process driven by one factor, but rather a web of interacting mechanisms — genetic, epigenetic, metabolic, and environmental — all converging on the microglial activation state.
Therapeutic Frontiers: How Science Is Fighting Back
The question every patient and family member wants answered is: can we do something about this? The short answer, according to the most current research, is increasingly yes — though we are still in early days.
A major 2026 review by Monroe et al. in Nature Reviews Drug Discovery provides the most comprehensive survey to date of therapeutic strategies targeting neuroimmune mechanisms in neurodegeneration. The authors identify several promising categories of intervention:
Microglial Modulation
Rather than simply suppressing microglial activation — which would eliminate the protective functions as well — the most sophisticated therapeutic strategies aim to rebalance microglial activity, shifting cells from the destructive M1 state toward the protective M2 phenotype. Small-molecule compounds, biologics, and gene therapies are all being explored in this space (Monroe et al., 2026).
Nrf2 Activators
Given the critical role of Nrf2 in maintaining microglial homeostasis, compounds that activate this pathway are attracting significant pharmaceutical interest. Sulforaphane (found naturally in broccoli sprouts), dimethyl fumarate (already approved for multiple sclerosis), and novel synthetic Nrf2 activators are all in various stages of preclinical and clinical investigation.
Senolytic Therapies
Senolytics — drugs that selectively eliminate senescent cells — represent another frontier. Zhang et al. (2026) discuss how clearing senescent microglia could, in principle, interrupt the SASP-driven inflammatory cycle and restore some degree of homeostatic surveillance to the aging brain. Early trials in humans are underway.
TREM2 and CSF1R Targeting
Monroe et al. (2026) highlight two molecular targets that have generated particular excitement: TREM2 (triggering receptor expressed on myeloid cells 2), which is critical for microglial survival and phagocytic function, and CSF1R (colony stimulating factor 1 receptor), which controls microglial proliferation. Both are being targeted in clinical trials for Alzheimer's disease.
Practical Applications for Everyday Brain Health
While disease-modifying drugs for neuroinflammation are still being developed, there is already substantial evidence that lifestyle choices can influence microglial function and the pace of neuroinflammatory aging. Here are evidence-informed strategies that anyone can implement:
Anti-inflammatory diet
Mediterranean and MIND diets reduce systemic inflammation that primes microglial overactivation. Polyphenols, omega-3s, and sulforaphane (from cruciferous vegetables) show direct Nrf2-activating properties.
Aerobic exercise
Regular moderate-intensity exercise reduces pro-inflammatory cytokine levels in the brain, improves microglial morphology in animal models, and supports BDNF (brain-derived neurotrophic factor) production.
Quality sleep
The glymphatic system — the brain's waste-clearance network — operates primarily during deep sleep. Poor sleep leads to amyloid accumulation, which directly triggers microglial activation.
Stress management
Chronic psychological stress elevates glucocorticoids, which prime microglia toward inflammatory states. Mindfulness, yoga, and social connection are validated stress-reduction strategies with measurable neurological benefits.
Cognitive engagement
Learning new skills, social stimulation, and intellectually demanding activities support synaptic density and may reduce the cellular senescence burden associated with cognitive aging.
Metabolic health
Type 2 diabetes and metabolic syndrome are associated with accelerated microglial aging. Maintaining healthy blood glucose, blood pressure, and BMI is among the most powerful brain-health interventions available.
Frequently Asked Questions
1. Can microglia actually be "turned off" to stop neuroinflammation?
Actually, we wouldn't want to! Microglia are the brain’s essential "housekeepers." Turning them off completely would allow toxic waste and dead cells to accumulate. The goal of modern medicine—including the research on OCIAD1—is to "re-tune" them so they stop attacking healthy tissue and return to their protective, cleaning roles.
2. Is neuroinflammation the same thing as brain inflammation from an infection?
Not exactly. While a virus (like meningitis) causes acute, "hot" inflammation, neuroinflammatory aging is a "cold," chronic simmer. It’s a low-grade, persistent activation of immune cells that slowly wears down neurons over decades rather than causing a sudden, high fever.
3. At what age does microglial aging begin to have significant effects?
Evidence suggests the "priming" of microglia begins in midlife (the 40s and 50s). While you might not feel symptoms then, this is the critical window where metabolic health and lifestyle choices determine whether those cells remain calm or become hyper-reactive in later years.
4. Are there blood tests or brain scans that can detect neuroinflammation?
In research settings, we use PET scans with specific "tracers" that bind to activated microglia, and we look for markers like CRP or TREM2 in spinal fluid. While these aren't standard at a routine physical yet, the discovery of genes like OCIAD1 is paving the way for simpler blood tests in the near future.
5. Does the Nrf2 pathway explain why some supplements benefit the brain?
Yes! Think of Nrf2 as a master "thermostat" for cellular health. Compounds like sulforaphane (from broccoli sprouts) or curcumin don't just "kill" free radicals; they signal the Nrf2 pathway to turn on your brain’s own internal antioxidant factory, helping microglia stay in a calm, "M2" state.
6. What is the connection between gut health and microglial function?
It’s called the Gut-Brain Axis. When the gut lining is compromised ("leaky gut"), inflammatory signals travel through the blood or the vagus nerve directly to the brain. This "primes" the microglia, making them much more likely to flip into a destructive, pro-inflammatory state.
7. My parent has Alzheimer’s—should I be worried about my own microglial health?
While genetics play a role, your "microglial destiny" isn't fixed. Because neuroinflammation is driven by a web of factors—metabolic, environmental, and lifestyle—you have significant power to influence how those genes express themselves. Focus on the "Practical Applications" (sleep, exercise, and diet) to keep your immune cells resilient.
Clinical Pearls
1. The Homeostatic Rebalance (M1 vs. M2 States)
Scientific: Therapeutic focus has shifted from global microglial immunosuppression to phenotypic modulation. The goal is to inhibit the pro-inflammatory M1-like state (associated with SASP and OCIAD1 elevation) while preserving or enhancing the M2-like neuroprotective, phagocytic functions.
Think of microglia as the brain’s "security guards." We don’t want to fire the guards (which would leave the brain unprotected); we just want to stop them from overreacting and accidentally breaking the furniture, encouraging them to get back to their cleaning duties instead.
2. OCIAD1 as a Biomarker and Driver
Scientific: OCIAD1 (Asrij) serves as a critical rheostat for microglial reactivity. Its age-associated upregulation correlates with heightened neuroinflammatory signaling, making it both a potential predictive biomarker for "inflammaging" and a high-value target for RNA-interference or small-molecule inhibition.
Scientists have found a specific protein called OCIAD1 that acts like a "volume knob" for brain inflammation. As we age, this knob gets turned up too high. New research suggests that if we can turn that specific knob back down, we might be able to slow down brain aging.
3. The Glymphatic-Inflammatory Link
Scientific: Sleep deprivation impairs glymphatic clearance, leading to the accumulation of metabolic waste like amyloid-β. These aggregates act as PAMPs/DAMPs (Pathogen/Damage-Associated Molecular Patterns) that chronically ligate microglial receptors (like TREM2), fueling a cycle of chronic activation.
Deep sleep is like a "power wash" for your brain. If you don’t get enough quality sleep, metabolic "trash" builds up. This trash irritates your immune cells, keeping them in a constant state of red alert, which eventually wears down your brain's health.
4. Nrf2: The Cellular Antioxidant Defense
Scientific: The Nrf2 pathway is a master regulator of the antioxidant response. Activating Nrf2 can counteract microglial "trained immunity"—a state where microglia become hyper-responsive to stimuli—thereby restoring cellular redox homeostasis and reducing collateral neuronal damage.
Your brain has a built-in "defense shield" called the Nrf2 pathway. Eating things like broccoli sprouts or Mediterranean-style fats helps flip the switch to turn this shield on, protecting your brain cells from the "rusting" (oxidative stress) that happens with age.
5. Metabolic-Microglial Priming
Scientific: Systemic metabolic dysfunction (e.g., insulin resistance) induces "microglial priming." In this state, the cells are not yet fully active but are hypersensitive; a secondary minor systemic infection or stressor can then trigger an exaggerated, pathological neuroinflammatory response.
Conditions like high blood sugar put your brain’s immune cells on "high alert." When your body is metabolically unhealthy, your brain cells become jumpy, causing them to overreact to small stresses that a healthy brain would easily handle.
Author’s Note
The science of brain aging is undergoing a profound shift. For decades, neurodegenerative diseases such as Alzheimer's disease and Parkinson's disease were viewed primarily through a neuron-centric lens — focusing almost exclusively on protein aggregates, synaptic loss, and neuronal death. However, emerging evidence now compels us to expand that view. The brain’s immune system — particularly microglia — is not merely a bystander but an active driver of both protection and pathology.
This article was written to bridge that evolving scientific understanding with real-world clinical relevance. The goal is not only to explain what happens during brain aging, but also why it matters — and importantly, what can be done about it. Mechanisms such as microglial priming, impaired Nrf2 signaling pathway, and cellular senescence are no longer abstract laboratory concepts; they are central to how we understand risk, progression, and potential intervention in neurodegenerative disease.
At the same time, it is important to maintain perspective. While the therapeutic landscape — including Nrf2 activators, senolytics, and microglial modulators — is advancing rapidly, much of this work remains in early translational or clinical stages. No single pathway, gene, or drug will “solve” brain aging. Instead, the future lies in integrated strategies that combine molecular therapies with lifestyle interventions targeting systemic inflammation, metabolic health, and resilience.
From a clinical standpoint, one of the most empowering insights is this: brain aging is not entirely predetermined. The same biological systems that contribute to neurodegeneration are also modifiable. Interventions that improve cardiometabolic health, reduce chronic inflammation, and support neural plasticity can meaningfully influence long-term brain outcomes.
For clinicians, researchers, and informed readers alike, the message is clear — understanding microglia is no longer optional in neuroscience; it is foundational. And for patients and families, the takeaway is equally important: the trajectory of brain health is shaped over decades, and even small, consistent actions today can alter that path.
— Dr. T.S. Didwal
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Mitochondria, Motor Units, and Muscle Aging: A Complete Guide | DR T S DIDWAL
Can Aging Cells Cause Diabetes? The Science Behind SASP and Blood Sugar Dysfunction
Exercise and Longevity: The Science of Protecting Brain and Heart Health as You Age | DR T S DIDWAL
The Science of Healthy Brain Aging: Microglia, Metabolism & Cognitive Fitness | DR T S DIDWAL
The Aging Muscle Paradox: How Senescent Cells Cause Insulin Resistance and The Strategies to Reverse It | DR T S DIDWAL
References
Ajoolabady, A., Kim, B., Abdulkhaliq, A. A., Ren, J., Bahijri, S., Tuomilehto, J., Borai, A., Khan, J., & Pratico, D. (2025). Dual role of microglia in neuroinflammation and neurodegenerative diseases. Neurobiology of Disease, 216(C), Article 107133. https://doi.org/10.1016/j.nbd.2025.107133
Han, T., Xu, Y., Sun, L., Hashimoto, M., & Wei, J. (2024). Microglial response to aging and neuroinflammation in the development of neurodegenerative diseases. Neural Regeneration Research, 19(6), 1241–1248. https://doi.org/10.4103/1673-5374.385845
Monroe, K. M., Hong, S., Lewcock, J. W., et al. (2026). Therapeutic targeting of neuroimmune mechanisms in neurodegeneration. Nature Reviews Drug Discovery. https://doi.org/10.1038/s41573-025-01370-7
Dongre, P., Ramesh, M., Govindaraju, T., & Inamdar, M. S. (2025). Asrij/OCIAD1 contributes to age-associated microglial activation and neuroinflammation in mice. Frontiers in Aging Neuroscience, 17, Article 1674136. https://doi.org/10.3389/fnagi.2025.1674136
Singh, H., Gupta, R., Gupta, M., et al. (2025). Aging-induced alterations in microglial cells and their impact on neurodegenerative disorders. Molecular Biology Reports, 52, 515. https://doi.org/10.1007/s11033-025-10623-y
Paraiso, H. C., Yen, J.-H. J., Scofield, B. A., Kuo, P.-C., Chang, F.-L., & Yu, I.-C. I. (2025). Microglial Nrf2 functions as a cell-autonomous regulator of neuroinflammation and trained immunity in the aging brain. The FASEB Journal, 39(22), e71244. https://doi.org/10.1096/fj.202501457RR
Zhang, L., Li, L., Wang, X., Ma, L., Yan, L., Xiang, Q., Cui, Y., & Liu, Z. (2026). Neuroinflammation and cellular senescence in brain aging and neurodegeneration. Aging and Disease. https://doi.org/10.14336/AD.2025.1538