Can Aging Cells Cause Diabetes? The Science Behind SASP and Blood Sugar Dysfunction
New research links cellular senescence and SASP to insulin resistance. Understand the mechanisms and future therapies like p38 inhibitors.
AGINGDIABETES
4/23/202616 min read
Can targeting aging cells improve blood sugar control?
Emerging research suggests that targeting pathways such as the p38 MAPK pathway may reduce harmful inflammatory signals from ageing cells and improve insulin sensitivity. However, most evidence is currently based on laboratory studies, and clinical applications are still under investigation.
How do aging cells disrupt blood sugar control?
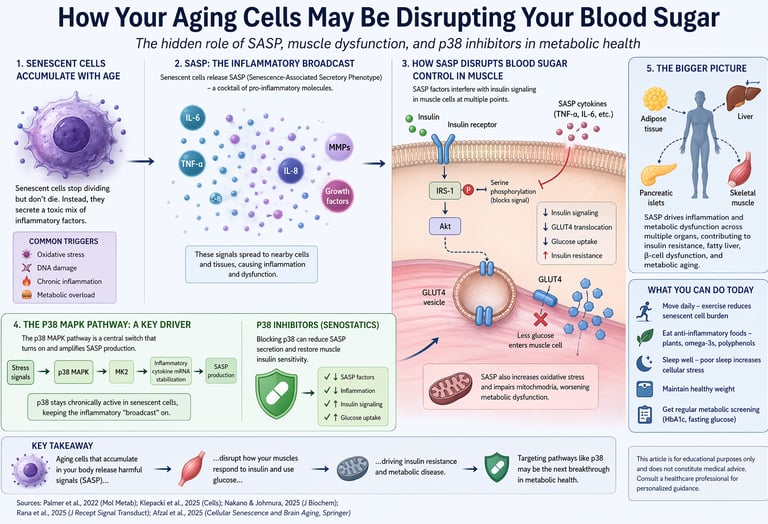
Accumulation of Cellular Senescence in Metabolic Tissues
Release of inflammatory SASP factors (e.g., IL-6, TNF-α)
Impaired insulin signaling in muscle cells
Reduced GLUT4-mediated glucose uptake
Increased oxidative stress and mitochondrial dysfunction
Progressive insulin resistance and metabolic dysfunction
Clinicians Prespectives
Why Aging Cells Matter More Than You Think
Your body is constantly renewing itself — but not all cells follow the rules. Some enter a state called Cellular Senescence, where they stop dividing but don’t get cleared away. Instead, they accumulate silently with age.
These cells release a mix of inflammatory signals known as the Senescence-Associated Secretory Phenotype. Think of it as a persistent “background inflammation” that spreads stress to nearby healthy cells.
How This Affects Your Blood Sugar
Your skeletal muscles are responsible for clearing most of the sugar from your bloodstream after meals.
SASP interferes with this process by disrupting insulin signaling — the very mechanism that allows glucose to enter muscle cells.
Over time, this can lead to insulin resistance, even in individuals who may appear otherwise healthy.
Why Scientists Are Paying Attention
Researchers have identified key pathways, including the p38 MAPK pathway, that regulate these harmful secretions.
Early studies suggest that targeting these pathways may reduce inflammation at its source — not just treat high blood sugar, but address one of its root causes.
What This Means for You Today
This science is promising but still evolving — most findings come from laboratory and early-stage studies.
There are no approved therapies, yet, that specifically target senescent cells in routine clinical care.
What You Can Do Right Now
Regular exercise, balanced nutrition, quality sleep, and weight management remain powerful tools to reduce inflammation and support metabolic health.
These lifestyle strategies may also help limit the accumulation and impact of senescent cells.
Bottom Line
Aging is not just about time — it’s about how your cells behave.
By understanding and eventually targeting these “aging cells,” medicine may shift from managing disease to preventing it at its biological core.
1. How Aging Cells May Be Secretly Disrupting Your Blood Sugar
Imagine a hidden biological process unfolding silently inside your body—one that doesn’t just accompany aging, but may actively increase your risk of insulin resistance and type 2 diabetes. This process is not driven solely by diet or inactivity, but by the gradual accumulation of damaged cells that refuse to die. These cells enter a state known as Cellular Senescence, where they stop dividing but remain metabolically active.
Instead of staying inactive, these senescent cells release a complex mix of inflammatory signals called the Senescence-Associated Secretory Phenotype. This secretome includes cytokines, enzymes, and growth factors that disrupt normal cellular communication. Over time, this creates a state of chronic, low-grade inflammation that interferes with key metabolic processes.
One of the most important targets of this disruption is skeletal muscle—the body’s primary site for glucose disposal. Research shows that SASP-related inflammation can impair insulin signaling pathways, reducing the ability of muscle cells to absorb glucose efficiently (Palmer et al., 2022). The result is a progressive decline in glucose uptake, laying the foundation for systemic insulin resistance and metabolic disease (Klepacki et al., 2025).
What makes this field particularly exciting is its therapeutic potential. Emerging studies point to the p38 MAPK pathway as a key regulator of SASP activity. In experimental models, inhibiting this pathway has been shown to partially restore glucose metabolism in muscle cells exposed to senescent signals, suggesting a novel approach to treating metabolic dysfunction (Rana et al., 2025).
Although most evidence is still preclinical, the implications are significant. Targeting aging cells may allow future therapies to address metabolic disease at its biological root—rather than simply managing its symptoms.
2. Understanding SASP: Your Body’s Hidden Inflammatory Signal Network
The most important feature of senescent cells—and the reason they are so harmful to metabolic health—is what they release. Rather than remaining inactive, these cells actively secrete a complex mixture of inflammatory molecules known as the Senescence-Associated Secretory Phenotype.
This secretome includes pro-inflammatory cytokines such as IL-1α, IL-6, and IL-8, along with tumor necrosis factor-alpha (TNF-α), matrix-degrading enzymes (MMPs), and various growth factors (Afzal et al., 2025). These molecules do not act locally alone—they influence surrounding cells and tissues, amplifying their impact through what is often called the bystander effect.
Importantly, SASP is not a static process. It is a dynamic, tightly regulated signaling network that evolves based on cellular stress and environmental conditions. Research shows that its activity is controlled by key molecular pathways, including NF-κB, mTOR, and the p38 MAPK pathway (Klepacki et al., 2025). These regulatory systems determine not only which inflammatory factors are released, but also how long and how intensely they act.
One of the most concerning aspects of SASP is its ability to spread dysfunction. Certain cytokines, such as IL-1α, can induce nearby healthy cells to enter Cellular Senescence—a phenomenon known as paracrine senescence. Over time, this creates a self-propagating cycle, where a small number of senescent cells can expand into widespread tissue-level disruption.
Why This Matters for Blood Sugar Control
In key metabolic tissues—including fat, liver, and skeletal muscle—SASP-driven inflammation interferes with insulin signaling pathways. This reduces the ability of cells to absorb and utilize glucose effectively, promoting insulin resistance and increasing the risk of type 2 diabetes (Palmer et al., 2022).
Key Takeaway
SASP is a dynamic inflammatory signaling network released by aging cells that spreads dysfunction across
3. How SASP Disrupts Blood Sugar Control in Skeletal Muscle
Skeletal muscle is the single largest site of glucose disposal in the human body. After a meal, roughly 70–80% of the glucose absorbed from your gut is destined for your muscles, where it is either burned for energy or stored as glycogen. This process depends on a tightly coordinated sequence of events — from insulin binding to its receptor on the muscle cell surface, to the intracellular cascades that move glucose transporters (particularly GLUT4) to the cell membrane, to the enzymatic reactions that phosphorylate and ultimately metabolize glucose.
SASP can interfere with this sequence at multiple levels.
Inflammatory cytokines impair insulin receptor signaling
SASP components like TNF-α and IL-6 activate inhibitory kinases (particularly IKKβ and JNK) that phosphorylate insulin receptor substrate-1 (IRS-1) at serine residues — a modification that blocks its normal function and effectively "jams" the insulin signaling cascade before it can fully activate.
GLUT4 translocation is reduced
With impaired IRS-1 function, downstream Akt activation is blunted. Since Akt phosphorylation is the critical trigger for GLUT4 vesicles to migrate to the cell surface, less GLUT4 reaches the membrane, meaning less glucose can enter the muscle cell.
Oxidative stress compounds insulin resistance
SASP also includes reactive oxygen species (ROS)-inducing factors that damage mitochondria within muscle cells. Mitochondrial dysfunction is strongly associated with impaired fatty acid oxidation and further exacerbates glucose intolerance.
Paracrine senescence spreads the dysfunction
Healthy muscle progenitor cells (satellite cells) exposed to SASP signals may themselves enter senescence, impairing the muscle's regenerative capacity and compounding the metabolic disruption over time.
The landmark study by Rana et al. (2025), published in the Journal of Receptors and Signal Transduction, provides one of the most targeted examinations of this mechanism in the literature. Using C2C12 cells — a well-established model system for studying skeletal muscle physiology — the researchers directly exposed differentiated muscle cells to SASP-conditioned medium (secreted factors from senescent cells) and measured the downstream metabolic consequences.
Their findings confirmed that SASP exposure significantly impaired glucose uptake in C2C12 myotubes. Critically, they also demonstrated that these effects were substantially reversed by the pharmacological inhibition of p38 MAPK — suggesting that this kinase sits at a pivotal intersection between SASP-driven inflammation and glucose metabolic dysfunction.
About C2C12 Cells
C2C12 cells are a mouse-derived myoblast cell line that can be differentiated into mature muscle cells (myotubes) in the laboratory. They are widely used as a model for studying skeletal muscle metabolism, making them ideal for investigating how SASP affects glucose handling in a controlled, reproducible system (Rana et al., 2025).
4. The p38 MAPK Pathway: A Central Switch in Inflammation and Metabolic Dysfunction
To understand why p38 inhibitors are gaining attention, it’s essential to first understand the role of the p38 MAPK pathway. This pathway acts as a critical stress-response system inside cells, activated by triggers such as inflammation, oxidative stress, UV radiation, and cellular damage. Once activated, p38 MAPK orchestrates a cascade of events that regulate gene expression, protein synthesis, and the production of inflammatory cytokines.
A Double-Edged Role in Cellular Senescence
In the context of Cellular Senescence, p38 MAPK has a dual function:
Protective role:
Initially, p38 activation helps damaged cells enter senescence, preventing the uncontrolled division of potentially harmful cells.Pathological role:
When persistently activated, p38 sustains and amplifies the Senescence-Associated Secretory Phenotype, turning a short-term protective mechanism into chronic inflammation.
How p38 Drives SASP at the Molecular Level
Recent research (Klepacki et al., 2025) highlights that p38 MAPK regulates SASP not just by increasing cytokine production, but by enhancing their stability. Through activation of the downstream kinase MK2, p38 stabilizes mRNA transcripts for key inflammatory mediators such as IL-6 and IL-8.
This means the cell continues producing inflammatory signals even after the original trigger is gone — sustaining a long-term inflammatory state.
Why p38 Inhibitors Are a Promising Target
Because p38 sits at a central control point, inhibiting this pathway offers a strategic advantage:
Reduces multiple inflammatory cytokines simultaneously
Modulates SASP without killing senescent cells
Potentially restores tissue function, especially in metabolic organs
This positions p38 inhibitors as senostatics—agents that suppress harmful cellular signaling rather than eliminating cells.
Senostatics vs Senolytics
Senolytics: Remove senescent cells (e.g., dasatinib + quercetin)
Senostatics: Suppress harmful secretions (e.g., p38 inhibitors)
Each approach has distinct clinical implications depending on tissue context and disease stage.
Translational Insight: Muscle Glucose Metabolism
Experimental work by Rana et al. (2025) provides compelling evidence in skeletal muscle models. When muscle cells exposed to SASP were treated with a p38 inhibitor, glucose uptake significantly improved—indicating restored insulin sensitivity.
This suggests that targeting p38 may improve metabolic function not by reducing cell number, but by reprogramming the inflammatory environment.
Key Takeaway (Snippet-Optimized)
The p38 MAPK pathway acts as a central regulator of inflammation and SASP. Its chronic activation drives insulin resistance, while targeted inhibition may restore glucose metabolism without eliminating senescent cells.
5. SASP in Metabolic Disease and Aging: The Missing Link in Whole-Body Glucose Control
Modern metabolic disease cannot be explained by a single pathway. The real clinical impact of Senescence-Associated Secretory Phenotype becomes clear only when viewed through the lens of multi-organ aging and systemic glucose regulation.
A growing body of research shows that Cellular Senescence is not confined to one tissue—it affects fat, liver, pancreas, and skeletal muscle simultaneously. According to Palmer et al. (2022), senescent cell burden increases in parallel with obesity, insulin resistance, and type 2 diabetes severity. Importantly, experimental removal of these cells in preclinical models leads to measurable improvements in metabolic function.
How SASP Disrupts Key Metabolic Organs
Adipose tissue (body fat):
SASP drives chronic low-grade inflammation, impairs adipogenesis, and reduces adiponectin levels—directly worsening insulin sensitivity.Liver:
Senescence-associated inflammation contributes to hepatic insulin resistance and the development of fatty liver disease (NAFLD).Pancreatic islets:
Accumulation of senescent beta cells reduces insulin secretion, compounding blood sugar dysregulation.Skeletal muscle:
SASP interferes with insulin signaling, limiting glucose uptake where it matters most.
The Aging–Metabolism Connection
The rise in type 2 diabetes with age mirrors the accumulation of senescent cells. This is not coincidence—it reflects a shared biology driven by oxidative stress, chronic inflammation, and impaired cellular repair mechanisms (Palmer et al., 2022; Afzal et al., 2025).
Crucially, SASP is not just a local phenomenon. Circulating inflammatory factors can act systemically, meaning dysfunction in one tissue—such as visceral fat—can trigger metabolic disturbances across the entire body.
A Vicious Cycle of Senescence and Metabolic Dysfunction
Emerging evidence supports a self-reinforcing loop:
Senescent cells accumulate in metabolic tissues
SASP spreads inflammation to nearby and distant cells
Insulin resistance worsens across organs
Ectopic fat and oxidative stress increase
More cells enter senescence
Therapeutic Insight: Not All Senescence Is Harmful
Not all senescent cells are detrimental. As highlighted by Nakano and Johmura (2025), some play roles in tissue repair and regeneration. This shifts the therapeutic goal:
Not elimination, but modulation
Reducing harmful SASP while preserving beneficial functions
Why This Matters Clinically
Targeting SASP—through emerging therapies like senolytics, senostatics, or lifestyle interventions—offers a system-wide strategy to improve metabolic health. Instead of treating isolated symptoms, this approach addresses one of the root biological drivers of insulin resistance and aging-related disease.
Key Takeaway
SASP links aging to metabolic disease by driving inflammation across fat, liver, muscle, and pancreas—creating a self-reinforcing cycle of insulin resistance that may be reversible with targeted therapies and lifestyle interventions.
6. Practical Applications: What This Research Means for Your Health Today
Understanding the science of SASP is not merely an academic exercise. The growing body of evidence linking cellular senescence to metabolic dysfunction has direct and actionable implications — both for individuals managing their health now, and for the direction of medical therapies in the coming years.
Exercise Reduces Senescent Cell Burden
Regular aerobic and resistance exercise has been shown to decrease markers of cellular senescence in skeletal muscle and adipose tissue, likely through enhanced autophagy (cellular "self-cleaning") and reduced oxidative stress. Targeting 150–300 minutes of moderate-intensity exercise weekly is one of the most evidence-based strategies for limiting SASP-related metabolic damage.
Anti-Inflammatory Nutrition Matters
Diets rich in polyphenols (berries, green tea, extra-virgin olive oil), omega-3 fatty acids (fatty fish, flaxseed), and diverse plant fiber reduce circulating inflammatory cytokines that mirror the SASP profile. Caloric restriction and intermittent fasting have also shown effects on reducing senescent cell accumulation in animal models.
Senolytic Therapies in Clinical Trials
The combination of dasatinib (a chemotherapy drug) and quercetin (a plant flavonoid) is among the most studied senolytic regimens. Early clinical trials — including the landmark Mayo Clinic work — have shown improvements in physical function and some metabolic markers in older adults. These remain experimental and should not be self-administered.
p38 Inhibitors on the Horizon
The findings of Rana et al. (2025) add to growing preclinical evidence that p38 MAPK inhibitors may represent a new class of senostatic therapies for metabolic disease. While no p38 inhibitor is yet approved for this indication, several compounds have passed Phase I and II safety trials for inflammatory conditions like rheumatoid arthritis and COPD, meaning the translational pathway is shorter than for entirely novel drug classes.
Sleep Quality and Cellular Aging
Chronic sleep deprivation is a potent inducer of cellular stress and senescence markers. Prioritizing 7–9 hours of quality sleep per night is not merely about feeling rested — it is one of the most accessible interventions for reducing the oxidative and inflammatory burden that drives SASP in the first place.
Regular Metabolic Screening
Given the bidirectional relationship between SASP and insulin resistance, individuals with risk factors for type 2 diabetes — particularly those with central obesity, family history, or age over 45 — should undergo regular HbA1c and fasting glucose screening. Early detection allows early intervention, before SASP-driven damage becomes self-reinforcing.
It is worth emphasizing that the majority of this evidence comes from preclinical (cell and animal) models, with human clinical data still emerging. The science is compelling, but patients should not attempt to self-administer drugs like dasatinib or experimental p38 inhibitors outside of supervised clinical settings. The safest, most evidence-based actions currently available are the lifestyle interventions described above.
Frequently Asked Questions About SASP and Glucose Homeostasis
What is the simplest way to explain what SASP is to a non-scientist?
Think of SASP as your body's "distress broadcast." When a cell is old or damaged and stops working properly, instead of quietly shutting down, it starts releasing hundreds of chemical signals — like a factory sounding every alarm at once. These signals are designed to call for help, but when too many cells do this for too long, the constant "noise" of inflammation damages the surrounding healthy tissue. In metabolic terms, this chronic noise interferes with the signals that normally tell muscle cells to take up and burn sugar, contributing to insulin resistance and blood sugar dysregulation.
Can I have my "SASP levels" tested at a clinic?
Not yet — at least not routinely. While researchers can measure specific SASP components like IL-6, IL-8, and TNF-α in blood or tissue samples, there is currently no validated, standardized clinical test for "SASP burden." However, high-sensitivity C-reactive protein (hs-CRP) is a widely available blood marker for chronic low-grade inflammation that can partially reflect SASP activity. Research-grade senescence biomarkers (such as p16INK4a expression in circulating T cells) exist, but are not yet part of standard clinical care. This is an active area of development in precision medicine.
Are senescent cells always harmful?
No — and this is one of the most important nuances in this field. As Nakano and Johmura (2025) emphasize, senescent cells serve important biological roles in embryonic development, wound healing, and cancer suppression. A young, healthy person clears senescent cells efficiently through immune surveillance, so they appear briefly, do their job, and are removed. The problem in aging and metabolic disease is that this clearance mechanism becomes impaired, causing senescent cells to accumulate, persist, and cause harm through chronic SASP. The therapeutic goal is therefore not to eliminate all senescence, but to restore healthy senescence dynamics — minimizing pathological accumulation while preserving transient, beneficial senescent signaling.
What makes skeletal muscle particularly important in blood sugar control?
Skeletal muscle is responsible for the disposal of 70–80% of glucose following a meal, making it the dominant site of postprandial glucose clearance. This happens primarily through insulin-stimulated glucose uptake, where the hormone insulin binds to receptors on muscle cells and triggers a signaling cascade that brings glucose transporters (GLUT4) to the cell surface. When SASP-derived inflammatory signals disrupt this cascade — as demonstrated experimentally by Rana et al. (2025) using C2C12 muscle cells — the consequence is impaired glucose uptake even in the presence of normal or elevated insulin levels. This is the cellular definition of insulin resistance in muscle tissue, which is a key early event in the progression to type 2 diabetes.
How do p38 inhibitors differ from existing diabetes medications?
Current diabetes medications — including metformin, GLP-1 receptor agonists, SGLT2 inhibitors, and insulin — work by improving insulin sensitivity, stimulating insulin secretion, or directly lowering blood glucose levels. They treat the metabolic consequence, not its upstream cellular cause. p38 inhibitors, by contrast, would intervene at the level of the SASP itself — reducing the inflammatory secretome of senescent cells and thereby removing one of the fundamental drivers of insulin resistance. This positions them as potential disease-modifying agents rather than symptomatic treatments. Of course, they remain investigational for this indication, and much more clinical evidence is needed before they could be recommended as a metabolic
therapy (Rana et al., 2025; Palmer et al., 2022).
Is SASP relevant to type 1 diabetes as well as type 2?
The evidence is most developed for type 2 diabetes, obesity, and age-related insulin resistance — conditions where senescent cell accumulation in metabolic tissues is well-documented. In type 1 diabetes, the primary driver is autoimmune destruction of pancreatic beta cells. However, SASP may still play a role in the broader inflammatory milieu of the pancreas during disease onset, and in the long-term complications of type 1 diabetes (cardiovascular disease, kidney disease, neuropathy) — all of which involve inflammatory pathways that overlap with SASP biology. Research into SASP's role in type 1 diabetes is ongoing and represents an important frontier.
When might SASP-targeting therapies become available as standard treatments?
This is difficult to predict with certainty, but the trajectory is encouraging. Several senolytic compounds are already in Phase II clinical trials for conditions including diabetes, pulmonary fibrosis, kidney disease, and Alzheimer's disease. p38 inhibitors have existing safety data from trials in inflammatory diseases like rheumatoid arthritis, which accelerates their translational pathway for new indications. Optimistically, if ongoing clinical trials produce strong efficacy data, some form of SASP-targeting therapy could reach approved clinical use within the next 5–10 years for specific metabolic or geriatric indications. That said, the science is still maturing, and patient safety — particularly ensuring that suppressing SASP does not impair beneficial senescent cell functions — must be rigorously established first.
Clinical Pearls
1. The "Bystander Effect": Paracrine Senescence
Scientific Tone: Cellular senescence is not a sequestered event; SASP factors like IL-1α and TGF-β induce "paracrine senescence" in neighboring healthy cells. This "bystander effect" amplifies tissue dysfunction beyond the initial site of damage, creating a self-propagating inflammatory circuit.
Think of a senescent cell as a "bad apple" in a crate. It doesn't just sit there; it releases signals that cause the healthy apples next to it to start spoiling too. This is why a small number of aging cells can eventually cause big problems for your whole muscle or organ.
2. Muscle as a Metabolic Sink
Scientific Tone: Skeletal muscle accounts for approximately 80% of postprandial glucose disposal. SASP-mediated inhibition of the PI3K/Akt pathway blunts GLUT4 translocation, effectively closing the primary metabolic sink for systemic glucose and driving hyperinsulinemia.
Your muscles act like a giant sponge for blood sugar. When aging cells release inflammatory "noise," they essentially "clog" the pores of that sponge. Even if your body produces enough insulin, the sugar can't get inside the muscle to be burned for energy, leaving it stuck in your bloodstream.
3. p38 MAPK: The SASP Volume Knob
Scientific Tone: The p38 MAPK pathway acts as a critical transcriptional and post-transcriptional regulator of the SASP. Unlike senolytics which eliminate cells, p38 inhibitors act as senostatics, modulating the "secretome" to restore insulin signaling without requiring cell death.
Researchers have found a "volume knob" inside these aging cells called p38. When the volume is turned all the way up, the cell screams inflammatory signals. New drugs under study aim to turn that volume down, keeping the cell quiet so it stops interfering with your metabolism.
4. Selective Senescence: The "Double-Edged Sword"
Scientific Tone: Senescence is a pleiotropic state; while chronic SASP is pathological, transient senescence is vital for myogenic differentiation and wound repair. Therapeutic interventions must target "accumulated" senescence rather than physiological, programmed senescence to avoid impairing tissue regeneration.
Not all "retired" cells are villains. Some show up briefly to help heal a cut or repair a strained muscle before disappearing. The goal of modern medicine isn't to get rid of every aging cell, but to clear out the ones that have "overstayed their welcome" and started causing chronic inflammation.
5. Exercise as a Biological "Senolytic"
Scientific Tone: Mechanical loading and aerobic conditioning stimulate autophagy and reduce mitochondrial ROS production, naturally lowering the threshold for senescence induction. Evidence suggests regular exercise reduces p16INK4a expression (a marker of aging) in human skeletal muscle.
Exercise is essentially a "deep clean" for your cells. It triggers a process in which your body recycles damaged parts and flushes out the inflammatory signals produced by ageing cells. It is currently the most effective way we have to keep our cellular "neighborhood" quiet and healthy.
Author’s Note
This article was written to bridge an important gap between rapidly evolving laboratory science and real-world clinical understanding. Concepts such as Cellular Senescence and the Senescence-Associated Secretory Phenotype are transforming how we think about aging and metabolic disease, yet they often remain confined to highly technical research settings. My goal has been to translate these complex mechanisms into clear, clinically meaningful insights that both patients and healthcare professionals can engage with.
The discussion around pathways such as the p38 MAPK pathway reflects an exciting frontier in medicine — one that aims to address disease at its biological roots rather than only managing downstream effects. However, it is equally important to emphasize that much of this evidence is still emerging, with a significant portion derived from preclinical models. Careful interpretation and rigorous clinical validation remain essential before these findings can be applied broadly in patient care.
As a clinician, I believe the future of metabolic medicine lies in integrating molecular biology with lifestyle and preventive strategies. While we await targeted therapies, foundational interventions — physical activity, nutrition, sleep, and early screening — remain the most powerful tools we have.
This piece is not intended to offer medical advice, but to inform, stimulate curiosity, and encourage evidence-based conversations between patients and their healthcare providers.
Medical Disclaimer
This article is intended for educational purposes only and does not constitute medical advice. The research discussed reflects findings from peer-reviewed studies published up to 2025 and should not be used to guide personal health decisions without consulting a qualified healthcare professional. Do not self-administer any pharmaceutical compound mentioned in this article.
Related Articles
Mitochondria, Motor Units, and Muscle Aging: A Complete Guide | DR T S DIDWAL
Exercise and Longevity: The Science of Protecting Brain and Heart Health as You Age | DR T S DIDWAL
The Science of Healthy Brain Aging: Microglia, Metabolism & Cognitive Fitness | DR T S DIDWAL
The Aging Muscle Paradox: How Senescent Cells Cause Insulin Resistance and The Strategies to Reverse It | DR T S DIDWAL
References
Afzal, M., Zeyadi, M., Hosawi, S., Kazmi, I., & Gupta, G. (2025). Introduction to Senescence-Associated Secretory Phenotype (SASP). In G. Gupta (Ed.), Cellular Senescence and Brain Aging. Springer. https://doi.org/10.1007/978-981-96-8873-9_3
Klepacki, H., Kowalczuk, K., Łepkowska, N., & Hermanowicz, J. M. (2025). Molecular regulation of SASP in cellular senescence: Therapeutic implications and translational challenges. Cells, 14(13), 942. https://doi.org/10.3390/cells14130942
Nakano, Y., & Johmura, Y. (2025). Functional diversity of senescent cells in driving ageing phenotypes and facilitating tissue regeneration. The Journal of Biochemistry, 177(3), 189–195. https://doi.org/10.1093/jb/mvae098
Palmer, A. K., Tchkonia, T., & Kirkland, J. L. (2022). Targeting cellular senescence in metabolic disease. Molecular Metabolism, 66, 101601. https://doi.org/10.1016/j.molmet.2022.101601
Rana, K. S., Marwah, M. K., Raja, F. N. S., Dias, I., Hindalekar, Y. S., Al Tahan, M. A., … & Bellary, S. (2025). The influence of senescent associated secretory phenotype on glucose homeostasis in C2C12 muscle cells: Insights into potential p38 inhibitor interventions. Journal of Receptors and Signal Transduction, 45(2), 118–127. https://doi.org/10.1080/10799893.2025.2475441