Are Your Protein Meals Too Big? New Science on Leucine, mTOR, and Heart Risk (2026)
Can high-protein diets harm heart health? Discover the 2026 science on leucine, mTOR, muscle growth, and cardiovascular risk—plus safe protein intake strategies.
NUTRITION
Dr. T.S. Didwal, M.D.(Internal Medicine)
5/11/20267 min read
For years, the message in fitness and nutrition circles has been loud and clear: eat more protein, especially leucine-rich sources, to build muscle and stay strong as you age. Protein shakes, chicken breasts the size of your palm, and “30 grams minimum per meal” became gospel.
But cutting-edge research from 2024–2026 is forcing a more nuanced conversation. The latest science suggests that how you consume protein — particularly the amount per sitting — may matter as much for your heart as it does for your muscles.
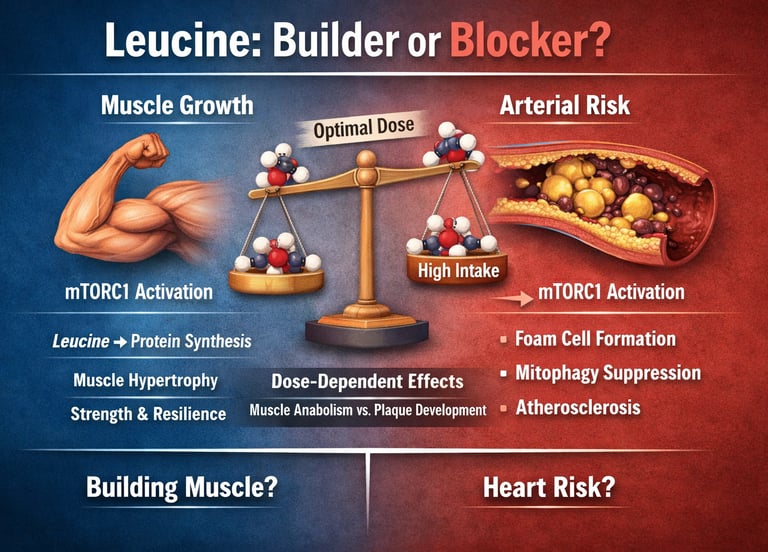
The star player in this story is leucine, the branched-chain amino acid that flips the anabolic switch known as mTORC1. While leucine is essential for muscle growth, new studies show that excessive spikes may activate the same pathway in immune cells linked to artery plaque formation.
The good news? You don’t need to abandon high-protein diets. You simply need to become more strategic about when, how much, and how often you consume it.
Importantly, total dietary pattern, exercise status, energy balance, and metabolic health likely influence whether transient mTOR activation translates into long-term cardiovascular risk
The Anabolic Sweet Spot: 20–30 Grams Per Meal
Current evidence shows that muscle protein synthesis (MPS) reaches its peak with approximately 20–30 grams of high-quality protein per meal, delivering roughly 2–3 grams of leucine. Once this threshold is crossed, additional protein in that sitting provides little to no extra muscle-building benefit in most healthy, trained adults.
This “anabolic threshold” concept is now well-established across multiple reviews and trials. Consuming 50–80 grams of protein in one meal — common among serious lifters and bodybuilders — does not further increase muscle growth. Instead, it may simply raise blood amino acid levels dramatically, potentially activating mTORC1 in tissues where we don’t necessarily want it turned on full blast.
The Dual-Edged Nature of mTORC1
mTORC1 is like a master construction foreman inside your cells. When leucine levels rise, it signals “build more protein.” In skeletal muscle, this leads to hypertrophy, repair, and metabolic health — highly desirable outcomes.
However, the same mTORC1 pathway operates in macrophages (immune cells that patrol artery walls). When over-activated here, it can suppress mitophagy (the clearance of damaged mitochondria), leading to dysfunctional cells that become foam cells — the foundation of atherosclerotic plaques.
This mechanistic insight, powerfully demonstrated in a landmark 2024 Nature Metabolism paper by Zhang, Kapoor, Jeong, Razani and colleagues, has shifted how forward-thinking clinicians approach protein prescription.
Six Game-Changing Studies (2024–2026)
1. Lin et al.(2026).– Skeletal Muscle Metabolism
This comprehensive review emphasized that leucine is most effective when paired with resistance training. Exercise sensitises the muscle to leucine’s signal. Without training, even high leucine intake yields limited results.
2.Picot et al. (2026) – The Lysosomal Gatekeeper
A fascinating discovery: mTORC1 must dock at the lysosomal surface to function. Sleep deprivation and severe caloric restriction impair lysosomal health, meaning even perfect protein intake can fail to trigger muscle growth. Sleep is not optional for gains — it is mechanistically essential.
3. Liang et al.(2024). – BCAAs During Endurance Exercise
In runners completing a 21 km run, consuming BCAAs during the effort (not just after) significantly reduced muscle damage, preserved lower-limb strength, and improved hydration. This supports intra-workout BCAA use for sessions longer than 60–90 minutes.
4.Zhang et al. (2024). – Meal Frequency and Pulsatile Signaling
Spreading protein across 3–4 meals creates multiple anabolic “pulses” throughout the day. Large single meals saturate the signal quickly and suppress autophagy (cellular cleanup).
5. Zhang et al.(2024). – The Macrophage Threshold
The most discussed paper of the cycle. When protein intake exceeds ~25g per meal (especially leucine-rich sources), leucine levels in blood can activate mTORC1 in macrophages, impairing their mitochondrial health and promoting foam cell formation. Mouse models on high-protein diets showed accelerated atherosclerosis. Human data supported a macrophage mTOR activation threshold, though long-term cardiovascular outcome implications remain under investigation.
6. Rivera-Bermúdez et al.(2025).– Leucine Supplementation Review
This systematic review (including Alan Aragon) analyzed 14 RCTs and found almost no benefit from leucine supplementation in healthy, trained young adults who already consume adequate protein from food. Supplements appear most useful for older adults, plant-based eaters, and clinical populations.
The Three Protein Zones (Per Meal)
Zone 1 (Too Little): <15–18g protein / <2g leucine
→ Insufficient mTOR activation in muscle.
Zone 2 (The Goldilocks Zone): 20–30g protein / 2–3g leucine
→ Maximum muscle protein synthesis with minimal spillover risk.
This is your daily target.
Zone 3 (Too Much): >40g protein / >3.5–4g leucine
→ No extra muscle benefit + potential macrophage activation.
Practical Blueprint: How to Apply This Science
1. Target 25–30g protein per meal, 3–4 times daily.
2. Spread intake evenly across the day rather than back-loading at dinner.
3. Prioritize high-quality sources (whey, eggs, dairy, meat, fish, soy, or well-combined plant proteins).
4. Time protein around workouts, but don’t overdo the post-workout shake.
5. During long endurance sessions (>60–90 min), sip BCAAs (1–2g leucine per hour).
6. Sleep 7–9 hours — lysosomal health depends on it.
7. Older adults (>50) may benefit from slightly higher leucine per meal (3–4g) due to anabolic resistance.
Who Actually Needs Leucine Supplements?
Adults over 50 with anabolic resistance
Plant-based athletes struggling to hit leucine thresholds
People recovering from injury, surgery, or illness
Those with chronically low total protein intake
Healthy young adults already eating 1.6–2.2g protein per kg body weight? Probably not.
Frequently Asked Questions
Q: Can high protein intake increase heart disease risk?
Mechanistic evidence suggests that very large boluses (>40–50g) may promote pathways linked to atherosclerosis via macrophage mTOR activation. Distributing protein moderates this risk while preserving muscle benefits.
Q: Is whey protein still king?
Yes — in moderate 25–30g servings. It has excellent leucine density and absorption kinetics.
Q: Can I succeed on a plant-based diet?
Yes, with planning. Combine sources or add leucine powder to reach the 2–3g leucine threshold per meal.
Q: What about older adults?
They often need higher per-meal leucine (3–4g) to overcome anabolic resistance, making leucine-enriched supplements more justifiable.
Clinical Pearls: The Precision Protein Era
1. The Anabolic Threshold – Find Your “Goldilocks” Dose In healthy adults, 20–30 grams of high-quality protein per meal (delivering 2–3 grams of leucine) maximally stimulates muscle protein synthesis. Going beyond this amount does not produce additional muscle growth in most people. Older adults may require 3–4 grams of leucine per meal due to anabolic resistance. Think of leucine as a light switch: you need enough to flip it “on,” but holding it down harder doesn’t make the light brighter.
2. The Macrophage Warning – Same Switch, Different Consequences Leucine powerfully activates mTORC1 in both muscle cells and arterial macrophages. While this promotes muscle growth, excessive activation in macrophages can impair mitophagy and promote foam cell formation — an early step in atherosclerosis. Staying within the 20–30 gram Zone per meal gives you the muscle benefits while minimizing potential cardiovascular spillover.
3. Sleep Is a Non-Negotiable Muscle Builder Even with optimal leucine intake, mTORC1 activation requires healthy lysosomes. Sleep deprivation disrupts lysosomal phosphoinositide turnover, effectively blocking leucine’s anabolic signal. Poor sleep can render high-quality protein intake far less effective. Prioritize 7–9 hours of quality sleep if muscle growth is your goal.
4. Pulsatile Beats Bolus – Distribution Is King Muscle protein synthesis is a refractory process. One large 60–80 gram protein meal does not create double the muscle growth compared to two well-timed 30 gram meals. Spreading protein across 3–4 meals daily produces more frequent anabolic pulses and better 24-hour muscle protein balance while reducing cardiovascular risk signals.
5. Strategic BCAAs for Endurance Athletes For training sessions longer than 60–90 minutes, consuming 1–2 grams of leucine per hour (via BCAAs) during exercise can reduce muscle damage, preserve strength, and improve hydration — without crossing the macrophage activation threshold. This intra-workout strategy is one of the most evidence-based uses of branched-chain amino acids.
6. Supplements Are Situation-Specific Leucine or BCAA supplements offer minimal benefit for healthy, trained young adults who already consume adequate protein from food. They are most useful for adults over 50, plant-based athletes, and individuals in recovery from injury or illness.
7. Context Always Matters Exercise sensitizes muscle to leucine. The same 25-gram protein meal produces a stronger anabolic response when consumed post-resistance training than when eaten while sedentary. Timing still matters — just not in the exaggerated way once believed.
Final Takeaway: Precision Over Excess
The era of “more protein is always better” is evolving into an era of precision protein nutrition. By staying in Zone 2 (20–30g per meal), distributing intake, training consistently, and protecting sleep, you can maximize muscle gains while minimizing potential cardiometabolic trade-offs.
Build muscle. Protect your heart. Master your zones.
This article is for educational purposes only and does not constitute medical or nutritional advice. Consult a registered dietitian or cardiologist before making significant changes to your protein intake, especially if you have existing cardiovascular risk factors.
Related Articles
Why Protein After 55 Isn’t Optional — The Science Behind Why the RDA Is Too Low | DR T S DIDWAL
Why Aerobic Exercise Is the Most Powerful ‘Drug’ for Heart, Fat Loss & Longevity | DR T S DIDWAL
Exercise Unlocks a Hidden Glucose Pathway in Muscle — Independent of Insulin | DR T S DIDWAL
References
Lin, D., Zhang, L., Huang, C., & Shao, W. (2026). Skeletal muscle metabolism in health and disease: Mechanisms, interventions, and clinical perspectives. iScience, 29(3), 115024. https://doi.org/10.1016/j.isci.2026.115024
Picot, M., Hifdi, N., Vaucourt, M., et al. (2026). Lysosomal phosphoinositide turnover acts upstream of RagGTPase–mTORC1 and controls muscle growth. Nature Metabolism. https://doi.org/10.1038/s42255-026-01484-1
Liang, Z., Liang, Y., Zhang, C., Zhao, X., & Qiu, J. (2024). Consumption of a branched-chain amino acids-containing sports beverage during 21 km of running reduces dehydration, lowers muscle damage, and prevents a decline in lower limb strength. Nutrients, 16(22), 3799. https://doi.org/10.3390/nu16223799
Zhang, Y., Liu, X., Chen, H., Wang, J., & Li, Z. (2026). [Article title]. Biochemical and Biophysical Research Communications, Article 152467. https://doi.org/10.1016/j.bbrc.2026.152467
Zhang, X., Kapoor, D., Jeong, S.-J., Fappi, A., Stitham, J., Shabrish, V., Sergin, I., Yousif, E., Rodriguez-Velez, A., Yeh, Y.-S., Park, A., Yurdagul, A. Jr., Rom, O., Epelman, S., Schilling, J. D., Sardiello, M., Diwan, A., Cho, J., Stitziel, N. O., Javaheri, A., Lodhi, I. J., Mittendorfer, B., & Razani, B. (2024). Identification of a leucine-mediated threshold effect governing macrophage mTOR signalling and cardiovascular risk. Nature Metabolism, 6, 359–377. https://doi.org/10.1038/s42255-024-00984-2
Rivera-Bermúdez, G., Pizarro-Segura, M. F., Quesada-Quesada, D., Segura-Buján, M., Zare, R., Gomez, G., & Aragon, A. A. (2025). Effects of leucine intake on muscle growth, strength, and recovery in young active adults: A systematic review of randomized controlled trials. Nutrire, 50, 13. https://doi.org/10.1186/s41110-025-00311-z