Why Skeletal Muscle Is the Body’s Most Powerful Metabolic Organ
Your muscles are more than movement. Discover how skeletal muscle acts as a metabolic and endocrine organ controlling glucose, fat burning, and brain health.
METABOLISMDIABETES
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/12/202611 min read
Skeletal Muscle: The Metabolic Engine You Cannot Afford to Ignore
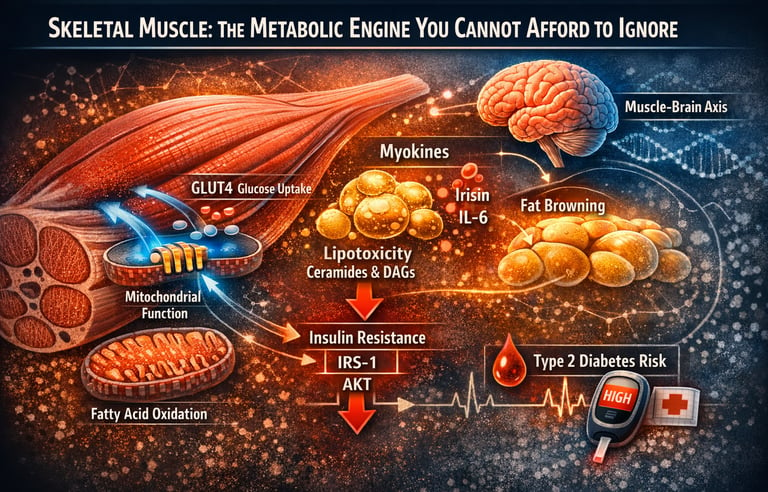
Skeletal muscle is not merely a contractile tissue responsible for movement — it is the body’s dominant metabolic organ and the primary regulator of postprandial glucose disposal, accounting for nearly 70–80% of insulin-stimulated glucose uptake in healthy individuals (Richter et al., 2025). In the context of modern metabolic disease, this fact changes everything. Long before fasting glucose rises or HbA1c drifts into the prediabetic range, subtle impairments in skeletal muscle insulin signaling, GLUT4 translocation, mitochondrial oxidative capacity, and lipid handling begin to reshape systemic metabolism (Whytock & Goodpaster, 2025).
Emerging research reframes skeletal muscle as a dynamic endocrine and metabolic command center. Beyond glucose uptake, muscle regulates fatty acid oxidation, secretes metabolically active myokines such as irisin and IL-6, influences adipose tissue browning, and communicates directly with the brain through the muscle–brain axis (Arosio & Picca, 2026). Disturbances in intramyocellular lipid metabolism — particularly the accumulation of ceramides and diacylglycerols — initiate lipotoxic signaling cascades that impair IRS-1 and AKT phosphorylation, forming the molecular foundation of insulin resistance (Cheng et al., 2025).
Even more striking, metabolic remodeling can occur independently of classical muscle fiber type switching, revealing that contractile identity and metabolic phenotype are governed by dissociable regulatory programs (Pereyra et al., 2022). This challenges long-standing assumptions in exercise physiology and deepens our understanding of how skeletal muscle dysfunction precedes overt type 2 diabetes.
In an era of escalating obesity, metabolic syndrome, and cardiometabolic disease, skeletal muscle health is not a secondary concern — it is the central battlefield upon which metabolic resilience is either preserved or lost.
Clinical pearls
1. The "Second Door" for Glucose
Contraction-mediated glucose uptake is mechanistically distinct from insulin-mediated signaling. While insulin requires the PI3K/AKT pathway, exercise activates AMPK and CaMKII, which independently trigger GLUT4 translocation to the sarcolemma.
Your muscle cells have two different "doors" to let sugar in. One requires a key (insulin), but the other opens automatically when you move. If your "insulin key" stops working well (insulin resistance), you can still lower your blood sugar simply by using the "movement door" through a post-meal walk or a workout.
2. The "Athlete’s Paradox" and Lipotoxicity
Intramyocellular lipid (IMCL) accumulation is not inherently pathological. Insulin resistance is driven by bioactive lipid intermediates—specifically diacylglycerols (DAGs) and ceramides—which interfere with IRS-1 signaling, rather than the total volume of stored triacylglycerol.
Storing fat in your muscles isn't always bad; elite athletes actually store a lot of it. The problem arises when that fat isn't "burned" and turns into metabolic "sludge." This sludge blocks the cell's internal signaling, making it harder for your body to manage energy and insulin.
3. Muscle as a "Chemical Pharmacy" (Myokines)
Skeletal muscle is an endocrine organ that secretes myokines such as IL-6, Irisin, and BDNF. These molecules exert systemic anti-inflammatory effects and facilitate inter-organ crosstalk, modulating hepatic glucose output and adipose tissue browning.
Every time you lift weights or run, your muscles act like a natural pharmacy. They release specialized "messenger chemicals" into your blood that tell your brain to stay sharp, your liver to stay healthy, and your body fat to burn more efficiently.
4. Metabolic vs. Contractile Fiber Switching
Metabolic reprogramming in muscle can occur independently of Myosin Heavy Chain (MHC) isoform switching. Pathological states (like defective fatty acid oxidation) can induce a "glycolytic shift" in Type I fibers without changing their physical contraction speed.
A muscle fiber can change its "metabolism" before it changes its "strength." Even if your muscles still look the same or feel as strong, they can lose their ability to burn fat and switch to burning only sugar if they aren't challenged correctly, which is an early warning sign of metabolic trouble.
5. The Muscle-Brain Connection (Irisin)
The myokine Irisin, cleaved from the FNDC5 protein during exercise, crosses the blood-brain barrier to upregulate Brain-Derived Neurotrophic Factor (BDNF) in the hippocampus, linking physical exertion to cognitive resilience and neuroprotection.
There is a direct "hotline" between your muscles and your brain. When you work out, your muscles send a signal (Irisin) that acts like "fertilizer" for your brain cells, helping you learn faster, remember more, and protect against aging-related memory loss.
I. Muscle Fiber Types: Beyond Slow and Fast-Twitch
While we traditionally classify muscle as Type I (Oxidative) or Type II (Glycolytic), recent data suggests a more complex reality. The metabolic identity of these fibers is more flexible than their physical contraction speed.
The Metabolic Switch: A landmark study (Pereyra et al., 2022) proved that muscle can undergo a "metabolic fiber type switch"—losing its ability to burn fat—without changing its physical "twitch" speed.
Clinical Insight: You cannot judge a muscle's health solely by its strength. A fiber may look normal under a microscope but function poorly metabolically, leading to early-stage "hidden" insulin resistance.
II. Glucose Uptake: The Dual-Gateway Mechanism
How does sugar move from your blood into your muscles? There are two distinct "gateways" that converge on the GLUT4 transporter.
1. The Insulin Gateway (The Chemical Key)
When you eat, insulin activates the PI3K/AKT pathway. This signals GLUT4 vesicles to move to the cell surface. In metabolic disease, this "key" often gets stuck (Insulin Resistance).
2. The Contraction Gateway (The Movement Bypass)
Exercise activates AMPK and CaMKII independently of insulin (Richter et al., 2025).
Why it matters: This "bypass" allows exercise to lower blood sugar even when insulin signaling is broken. This makes physical activity a non-negotiable "metabolic medicine."
III. Myokine Signaling: How Muscles Talk to the Brain
Skeletal muscle acts as a "chemical factory," secreting myokines that coordinate whole-body health.
IL-6: When released during exercise, it acts as an anti-inflammatory and boosts fat burning.
Irisin (The Muscle-Brain Link): Research by Arosio & Picca (2026) shows that Irisin crosses the blood-brain barrier. It stimulates BDNF (Brain-Derived Neurotrophic Factor), acting as "fertilizer" for the hippocampus to prevent cognitive decline.
Metabolic Browning: Irisin also turns "white fat" into calorie-burning "brown fat," increasing your resting metabolic rate.
IV. The Lipotoxicity Paradox: When Fat Blocks Fuel
Muscle stores fat as Intramyocellular Lipid (IMCL). While athletes store a lot of fat (The Athlete's Paradox), they burn it cleanly. The danger arises when fat isn't burned and turns into toxic intermediates:
DAGs (Diacylglycerols): These physically block the insulin receptor.
Ceramides: These deactivate AKT, stopping glucose from entering the cell.
Pan-Lipotoxicity: Cheng et al. (2025) introduced this concept, showing that toxic fats in muscle don't stay there—they leak out and damage the heart, liver, and kidneys in a destructive loop.
V. Clinical Correlation: Muscle as an Early Sentinel
Whytock & Goodpaster (2025) argue that muscle insulin resistance is the earliest detectable sign of metabolic failure.
The 10-Year Window: Muscle dysfunction often appears a decade before blood sugar rises on a standard test.
Emerging Biomarkers: We are moving beyond the A1c. Future screenings will look at circulating acylcarnitines and Irisin levels to catch disease in the "muscle phase" before it becomes diabetes.
Synthesis: The Muscle–Metabolism Interface
1. Muscle Is the Body’s Largest Glucose Regulator
Skeletal muscle accounts for nearly 70–80% of insulin-stimulated glucose uptake. At the molecular level, insulin activates the IRS-1 → PI3K → AKT pathway, driving GLUT4 transporters to the muscle cell surface. When this signaling cascade is impaired, glucose cannot efficiently enter the cell — a defect that often appears years before blood sugar levels rise.
Your blood tests can look “normal” while your muscles are already becoming resistant to insulin. This silent phase is where prevention is still powerful.
2. Lipotoxicity Drives Early Insulin Resistance
The issue is not fat stored in muscle — it is toxic lipid intermediates such as ceramides and diacylglycerols. These molecules disrupt insulin signaling and reduce mitochondrial efficiency, creating a state of metabolic inflexibility.
It’s not just about body weight. Even people who are not visibly obese can accumulate harmful fat inside muscle cells if they are sedentary or metabolically unhealthy.
3 Exercise Activates an Independent Rescue Pathway
Muscle contraction activates AMPK, bypassing defective insulin signaling and restoring GLUT4 movement to the cell membrane. Over time, regular training increases mitochondrial density, improves fatty acid oxidation, and reduces toxic lipid buildup.
Even if you have insulin resistance or prediabetes, exercise still works. Your muscles have a backup system — and movement turns it on.
4. Muscle Is an Endocrine Organ
Contracting muscle releases myokines such as irisin and IL-6, influencing adipose tissue metabolism, liver glucose production, and even brain health through the muscle–brain axis.
Exercise is not just about burning calories. Your muscles release protective hormones that improve energy, cognition, and metabolic balance.
5. Clinical Implication
Skeletal muscle insulin resistance often precedes type 2 diabetes by years. Protecting muscle metabolic health through resistance training, aerobic exercise, and adequate protein intake is not optional — it is foundational preventive medicine.
Bottom Line:
Metabolic disease does not begin with high blood sugar. It begins when skeletal muscle stops responding efficiently. The good news? Muscle is highly adaptable. And adaptation begins with movement.
To provide the most utility for a clinical or high-level health platform, this checklist translates the molecular research of your article into a diagnostic framework. This helps bridge the gap between "knowing the science" and "identifying the patient."
Skeletal Muscle Metabolic Assessment Checklist
Use these five criteria to evaluate the functional metabolic state of skeletal muscle before overt hyperglycemia (High A1c/Glucose) manifests.
1. Metabolic Flexibility (Substrate Switching)
Assess the patient’s ability to transition between carbohydrate and lipid oxidation.
Clinical Sign: Postprandial lethargy or "sugar crashes" (reactive hypoglycemia) despite normal fasting levels.
The Science: This indicates a "stuck" Randle Cycle, where intramyocellular lipids (IMCL) are blocking the efficient switch to glucose oxidation.
Red Flag: Inability to perform low-intensity fasted exercise without immediate fatigue.
2. Myokine Proxy: The Muscle-Brain Axis
Evaluate cognitive "sharpness" as a reflection of exercise-induced secretome health.
Clinical Sign: Reports of "brain fog" that improve significantly immediately following vigorous physical activity.
The Science: This suggests a reliance on acute Irisin and BDNF pulses to overcome baseline neuro-inflammation.
Red Flag: Cognitive decline or "fog" specifically associated with periods of physical inactivity.
3. Skeletal Muscle Density & Quality (Lipotoxicity)
Look beyond the BMI to evaluate the presence of "metabolic sludge" (DAGs/Ceramides).
Clinical Sign: High "Body Roundness Index" (BRI) or waist-to-height ratio in a "Normal BMI" individual (The TOFI Phenotype).
The Science: High visceral fat almost always correlates with Pan-Lipotoxicity, where ceramides spill over into skeletal muscle, deactivating the AKT pathway.
Red Flag: Decreased muscle "tone" or firmness despite adequate protein intake, suggesting intramyocellular lipid infiltration.
4. GLUT4 Gateway Efficiency (Post-Meal Response)
Evaluate the efficiency of the "Second Door" (Contraction-mediated uptake).
Clinical Sign: Blood glucose that remains elevated 2 hours post-meal but drops rapidly with a 10-minute walk.
The Science: A dramatic drop with light movement suggests the Insulin Gateway is failing (AS160 impairment), but the Contraction Gateway (AMPK/TBC1D1) is still functional.
Red Flag: High postprandial glucose that is only responsive to intense movement, suggesting advanced insulin resistance.
5. Fiber Type Resilience (Mitochondrial Capacity)
Assess the oxidative capacity of Type I fibers independent of MHC strength.
Clinical Sign: Poor "Zone 2" endurance (getting winded at low heart rates) despite having high absolute strength in the gym.
The Science: This indicates a Metabolic Fiber Type Switch (Pereyra et al., 2022). The muscle has lost its mitochondrial oxidative "engine" even if the contractile "frame" is still strong.
Red Flag: Rapid accumulation of lactate during submaximal, everyday activities (e.g., climbing a single flight of stairs).
Frequently Asked Questions (FAQs)
1. What makes skeletal muscle an endocrine organ? Skeletal muscle releases bioactive peptides called myokines — including IL-6, irisin, and BDNF — during and after contraction. These molecules travel through the bloodstream to influence metabolism, inflammation, neuroplasticity, and energy balance in distant tissues, qualifying muscle as an endocrine organ with wide-ranging effects beyond locomotion.
2. Can you have muscle insulin resistance with normal blood sugar? Yes. Research demonstrates that skeletal muscle insulin resistance — characterized by impaired GLUT4 translocation, serine phosphorylation of IRS-1, and reduced mitochondrial oxidative capacity — can be present and measurable years before fasting glucose or HbA1c become abnormal. This makes it a potentially powerful early biomarker for predicting future type 2 diabetes risk (Whytock & Goodpaster, 2025).
3. How does exercise improve insulin sensitivity in muscle? Exercise activates AMPK independently of insulin, driving GLUT4 to the plasma membrane through the TBC1D1 pathway. Over time, regular training increases GLUT4 protein expression, enhances mitochondrial density and oxidative capacity, reduces toxic lipid intermediates (DAGs, ceramides), and normalizes IRS-1 signaling — collectively restoring insulin sensitivity through mechanisms that complement or exceed those of pharmacological agents (Richter et al., 2025).
4. What is the "athlete's paradox" in muscle lipid metabolism? Endurance-trained athletes store more intramyocellular lipid (IMCL) than sedentary individuals, yet maintain excellent insulin sensitivity — the opposite of what would be expected if IMCL alone caused insulin resistance. This paradox is explained by the high lipid oxidative capacity of trained muscle: athletes efficiently oxidize IMCL, preventing accumulation of the toxic DAG and ceramide intermediates that drive insulin resistance in untrained, metabolically compromised muscle.
5. What is pan-lipotoxicity and why does it matter? Pan-lipotoxicity, as described by Cheng et al. (2025), refers to the coordinated lipotoxic injury of multiple organ systems — liver, heart, pancreas, kidney, and skeletal muscle — driven by shared circulating bioactive lipid species including ceramides and DAGs. It reframes lipotoxicity as a systemic, self-amplifying process rather than a tissue-isolated phenomenon, with important implications for understanding multi-organ metabolic failure in obesity.
6. What is the muscle–brain axis and how does irisin mediate it? The muscle–brain axis describes the bidirectional hormonal communication between contracting skeletal muscle and the central nervous system. Irisin, a myokine cleaved from FNDC5 during exercise, crosses the blood–brain barrier and stimulates hippocampal BDNF expression, promoting neuroplasticity, synaptic connectivity, and neuroprotection against age-related neurodegeneration. Declining irisin levels with aging and inactivity may thus contribute to cognitive decline as well as metabolic dysfunction (Arosio & Picca, 2026).
7. Can fiber type composition be changed through training or disease? Yes, though the relationship is nuanced. Endurance training promotes a shift toward more oxidative fiber characteristics, while chronic disease states and fatty acid oxidation defects can impose a metabolic switch toward glycolytic profiles without necessarily altering myosin heavy chain composition (Pereyra et al., 2022). This means metabolic fiber type and contractile fiber type can be independently regulated — and independently targeted — making metabolic characterization of muscle more clinically meaningful than MHC-based histology alone.
Author’s Note
As a physician trained in internal medicine, I was taught to diagnose metabolic disease through laboratory thresholds — fasting glucose, HbA1c, triglycerides, LDL cholesterol. Over time, however, clinical experience and a deep engagement with emerging metabolic research reshaped my understanding. Hyperglycemia is often the final chapter of a much longer story. The earlier chapters unfold silently inside skeletal muscle.
This article was written to bridge that gap — between molecular physiology and everyday clinical reality. The science of skeletal muscle metabolism is no longer confined to exercise laboratories. It has become central to understanding insulin resistance, type 2 diabetes, obesity, cardiovascular risk, and even neurodegeneration. The muscle cell is not simply a contractile unit; it is a metabolic sensor, a substrate regulator, and an endocrine communicator.
My goal in writing this piece was twofold.
First, to present the mechanistic science with precision — honoring the complexity of insulin signaling, lipotoxic intermediates, mitochondrial adaptation, and myokine biology without oversimplification.
Second, to translate that science into a clinically meaningful message: prevention begins before glucose rises. Muscle insulin resistance precedes overt disease, and skeletal muscle remains one of the most modifiable tissues in the human body.
If there is one idea I hope readers carry forward, it is this: metabolic resilience is not built in the pancreas alone. It is cultivated in skeletal muscle — through movement, structured training, adequate nutrition, and metabolic awareness.
The encouraging truth is that muscle adapts. At nearly any age, and across a wide spectrum of metabolic states, skeletal muscle retains a remarkable capacity for recovery and remodeling.
Understanding that biology changes how we practice medicine — and how we live it.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Is Your Muscle Insulin Resistant? 2026 Update | DR T S DIDWAL
Activate Your Brown Fat: A New Pathway to Longevity and Metabolic Health | DR T S DIDWAL
Leptin vs. Adiponectin: How Your Fat Hormones Control Weight and Metabolic Health | DR T S DIDWAL
References
Arosio, B., & Picca, A. (2026). Irisin and the muscle–brain axis: Mechanisms and translational potential. Experimental Gerontology, 214, 113028. https://doi.org/10.1016/j.exger.2026.113028
Cheng, Y., Shao, S., Wang, Z., et al. (2025). From lipotoxicity to pan-lipotoxicity. Cell Discovery, 11, 27. https://doi.org/10.1038/s41421-025-00787-z
Pereyra, A. S., Lin, C.-T., Mesa Sanchez, D., Laskin, J., Spangenburg, E. E., Neufer, P. D., Fisher–Wellman, K., & Ellis, J. M. (2022). Skeletal muscle undergoes fiber type metabolic switch without myosin heavy chain switch in response to defective fatty acid oxidation. Molecular Metabolism, 59, 101456. https://doi.org/10.1016/j.molmet.2022.101456
Richter, E. A., Bilan, P. J., & Klip, A. (2025). A comprehensive view of muscle glucose uptake: regulation by insulin, contractile activity, and exercise. Physiological Reviews, 105(3), 1867–1945. https://doi.org/10.1152/physrev.00033.2024
Whytock, K. L., & Goodpaster, B. H. (2025). Unraveling skeletal muscle insulin resistance: Molecular mechanisms and the restorative role of exercise. Circulation Research, 137(2), 184–204. https://doi.org/10.1161/CIRCRESAHA.125.325532