Is Your Muscle Insulin Resistant? 2026 Update
Discover how 2026 research explains muscle insulin resistance, lipotoxicity, inflammation, and how exercise can reverse metabolic dysfunction.
METABOLISMDIABETES
Dr. T.S. Didwal, M.D.(Internal Medicine)
2/27/202613 min read
What if the real driver of type 2 diabetes and metabolic syndrome isn’t just high blood sugar — but a silent metabolic breakdown unfolding inside your skeletal muscle?
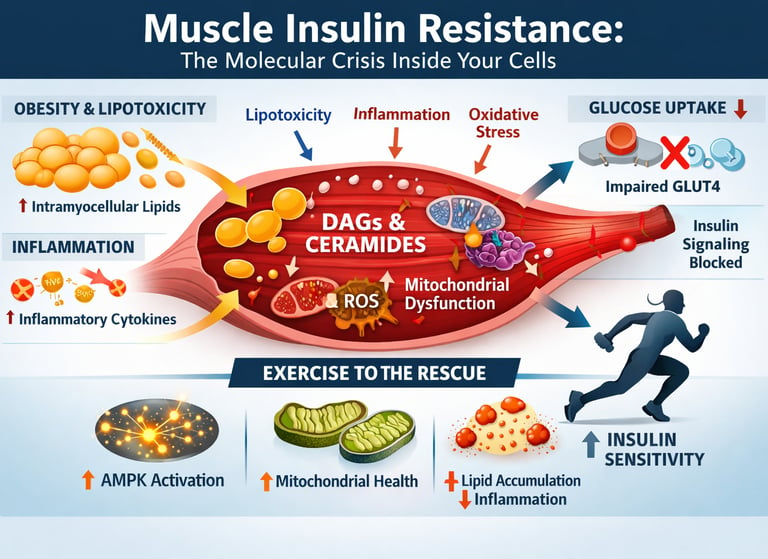
Skeletal muscle is responsible for nearly 70–80% of insulin-stimulated glucose disposal in the human body. When muscle becomes insulin resistant, glucose uptake falters, pancreatic insulin output rises, fat oxidation declines, and a cascade of metabolic dysfunction accelerates. This is not merely a hormonal problem — it is a cellular energy crisis rooted in lipotoxicity, chronic inflammation, oxidative stress, and mitochondrial dysfunction (Razi et al., 2025; Whytock & Goodpaster, 2025).
Emerging research shows that obesity drives excess free fatty acid overflow into muscle tissue, where it accumulates as intramyocellular lipids — particularly diacylglycerols (DAGs) and ceramides. These lipid intermediates activate protein kinase C isoforms that inhibit IRS-1/PI3K/Akt signalling, effectively shutting down insulin sensitivity at the molecular level (Razi et al., 2025; da Silva Rosa et al., 2020). Simultaneously, inflammatory cytokines such as TNF-α and IL-6 amplify serine phosphorylation of IRS-1, while reactive oxygen species impair mitochondrial oxidative capacity, compounding skeletal muscle insulin resistance (Ahn, 2025).
But there is a powerful counterforce.
Exercise activates AMPK and CaMKII pathways that stimulate insulin-independent GLUT4 translocation — bypassing defective insulin signalling entirely. Chronic training reduces harmful lipid species, improves mitochondrial biogenesis, lowers inflammatory signalling, and restores metabolic flexibility (Whytock & Goodpaster, 2025; Yu et al., 2026).
Understanding skeletal muscle insulin resistance is no longer optional — it is central to preventing obesity-related diabetes, cardiometabolic disease, and systemic metabolic dysfunction. The molecular story is complex, but the therapeutic implications are profound.
Cinical pearls
1. The "Bypass" Effect of Muscle Contraction
Muscle contraction stimulates glucose uptake via an insulin-independent pathway. By activating AMPK and CaMKII, exercise triggers GLUT4 translocation to the cell membrane even when the proximal insulin signaling cascade (IRS-1/PI3K/Akt) is severely impaired by lipotoxicity.
Think of exercise as a "back door" into your cells. Even if the front door (the insulin pathway) is jammed or "resistant," moving your muscles creates a temporary side entrance that lets sugar in without needing a key. This is why a simple walk after a meal can lower blood sugar almost instantly.
2. Not All Fat is "Stored" Equally
Systemic insulin resistance is driven by ectopic lipid deposition. Specifically, the accumulation of intramyocellular diacylglycerols (DAGs) and ceramides—rather than neutral triacylglycerols—triggers Protein Kinase C (PKC) isoforms. This leads to inhibitory serine phosphorylation of IRS-1, effectively decoupling the insulin receptor from its downstream metabolic effects.
It’s not just about the fat you see in the mirror; it’s about "invisible" fat inside your muscle cells. When your muscles get "marbled" like a steak, certain toxic fat fragments act like gum in a lock, stopping insulin from doing its job. Strength training and a clean diet help "burn off" this internal gum so the lock works again.
3. The "Metabolic Triad" and Visceral Overflow
Skeletal muscle insulin resistance rarely exists in a vacuum. It is part of an inter-organ crosstalk failure. Dysfunctional, inflamed visceral adipose tissue releases a "flood" of Free Fatty Acids (FFAs) and pro-inflammatory cytokines (TNF-α, IL-6). This lipotoxicity simultaneously drives hepatic steatosis (fatty liver) and muscle resistance, creating a self-perpetuating cycle of hyperglycemia.
Your muscles, liver, and belly fat are constantly "talking" to each other. When belly fat becomes inflamed, it starts "shouting" extra fat and stress signals into your blood. This overwhelms your liver and muscles at the same time. To fix the muscle, we often have to calm down the "noise" coming from the belly fat through weight loss and anti-inflammatory foods.
4. Mitochondrial Efficiency as a Metabolic Buffer
Muscle insulin resistance is closely linked to mitochondrial dysfunction and oxidative stress. When mitochondria cannot efficiently oxidize fatty acids, incomplete lipid breakdown products (like the aforementioned acylcarnitines and ROS) accumulate. Increasing mitochondrial biogenesis through Zone 2 aerobic training improves the cell's capacity to "clear" these metabolic bottlenecks.
Your muscle cells have tiny power plants called mitochondria. When these plants are old or sluggish, they leave "trash" (toxic fats) lying around that causes insulin resistance. Consistent, easy cardio (like a light jog or brisk walk) builds more and better power plants, which helps your body burn fuel cleanly and keeps your metabolism running smooth.
5. Epigenetic Flexibility: It’s Not Your Destiny
Scientific Tone: Recent 2026 perspectives highlight that while epigenetic modifications (like DNA methylation) can "lock" a person into an insulin-resistant phenotype, these markers are remarkably plastic. Lifestyle medicine serves as a form of "epigenetic editing," where chronic exercise and caloric restriction can demethylate key metabolic genes, effectively "reprogramming" the muscle toward an insulin-sensitive state.
You might have a family history of diabetes, but your genes aren't a fixed blueprint—they’re more like a series of light switches. Bad habits can turn the "insulin resistance" switches on, but exercise and a healthy diet can actually flip them back off at a molecular level. You have the power to "reprogram" your own biology every time you hit the gym or choose a healthy meal.
Study 1: The Obesity–Muscle Connection — Lipids, Inflammation & Oxidative Stress
This comprehensive review by Razi et a l(2025) draws a molecular roadmap from obesity to skeletal muscle insulin resistance. The authors — a multinational team spanning Iran, Brazil, Cyprus, the UK, and Spain — synthesise evidence on three critical mediating pathways: intramyocellular lipid (IMCL) accumulation, chronic low-grade inflammation, and oxidative stress.
When excess adipose tissue expands during obesity, it releases a flood of free fatty acids (FFAs) into circulation. These FFAs are taken up by skeletal muscle and stored as intramyocellular lipids — specifically as diacylglycerols (DAGs) and ceramides — which directly impair insulin signalling by activating protein kinase C (PKC) and suppressing the critical IRS-1/PI3K/Akt pathway.
Simultaneously, adipose-derived inflammatory cytokines — particularly TNF-α, IL-6, and IL-1β — flood the bloodstream and muscle tissue, further disrupting GLUT4 translocation, the molecular mechanism by which glucose enters muscle cells. Compounding this, reactive oxygen species (ROS) generated by an overburdened mitochondrial network contribute to mitochondrial dysfunction and accelerate insulin resistance development.
Key Takeaways
IMCL accumulation is a central biomarker and mediator of muscle insulin resistance in obesity.
DAGs and ceramides are the molecular culprits that block the insulin signalling cascade.
Chronic inflammation and oxidative stress work synergistically, not in isolation, to drive metabolic dysfunction.
Targeting all three pathways — lipid metabolism, inflammation, and ROS — may be essential for effective therapeutic strategies.
Study 2: Molecular Mechanisms and Therapeutic Targets of Insulin Resistance
This review by Ahn (2025).provides an in-depth, mechanistically rigorous examination of the entire insulin resistance landscape — from cellular signalling to pharmacological intervention. It is a tour de force for anyone seeking to understand the latest developments in molecular insulin resistance research.
Ahn's review centres on the dysfunction of the insulin receptor substrate (IRS) proteins and downstream cascades. Key among these is the impairment of PI3K/Akt signalling, which normally drives GLUT4 vesicle translocation to the cell membrane to facilitate glucose uptake. The review also examines the role of endoplasmic reticulum (ER) stress, which triggers the unfolded protein response (UPR) and contributes to the phosphorylation of IRS-1 on serine residues — effectively switching off insulin sensitivity.
From a therapeutic standpoint, Ahn evaluates promising molecular targets including AMPK activators, PPAR-γ agonists, sodium-glucose cotransporter-2 (SGLT2) inhibitors, and novel GLP-1 receptor agonists — discussing both their clinical utility and the mechanistic rationale behind their efficacy in improving insulin sensitivity. Emerging targets like mitochondrial biogenesis pathways and gut microbiome modulation are also discussed as future frontiers.
Study 3: Skeletal Muscle Insulin Resistance and the Restorative Role of Exercise
A review by Whytock and Goodpaster(2025) represents one of the most comprehensive contemporary analyses of how exercise counteracts skeletal muscle insulin resistance. It bridges molecular science with clinical exercise physiology in remarkable detail.
The authors detail the precise molecular pathways through which physical exercise restores insulin sensitivity. During and after exercise, muscle contractions stimulate AMPK (AMP-activated protein kinase) and CaMKII (calcium/calmodulin-dependent protein kinase II) pathways — independent of insulin — to drive GLUT4 translocation and glucose uptake. This insulin-independent glucose transport is critical: it means exercise can bypass the very defects that cause insulin resistance.
Whytock and Goodpaster also show that chronic exercise training improves mitochondrial density and function, reduces intramyocellular lipid content (particularly harmful lipid species like DAGs and ceramides), decreases skeletal muscle inflammation, and upregulates oxidative metabolism. Both aerobic exercise and resistance training are highlighted as potent modulators of insulin sensitivity, with distinct but complementary molecular mechanisms.
Key Takeaways
Exercise is arguably the most powerful tool for reversing skeletal muscle insulin resistance — and it works through distinct, insulin-independent molecular pathways.
AMPK activation during exercise directly drives glucose uptake without requiring a functional insulin signalling cascade.
Both aerobic and resistance training improve insulin sensitivity, but via different molecular mechanisms — ideally, both should be combined.
Exercise-induced reductions in IMCL particularly target the harmful ceramide and DAG fractions, not the metabolically benign IMCL in trained athletes.
Study 4: Insulin Resistance Mechanisms and Therapeutic Interventions — A 2026 Perspective
The review by Yu et al.( 2026) represents the most current synthesis available on insulin resistance mechanisms and emerging therapies. Yu and colleagues offer a new perspective, connecting intracellular signalling defects to whole-body metabolic consequences and cutting-edge interventions.
The authors provide a multilayer analysis of insulin resistance causation, covering epigenetic modifications, microRNA dysregulation, post-translational protein modifications, and the role of lipotoxicity and glucotoxicity in perpetuating the insulin-resistant phenotype. A notable focus is placed on the crosstalk between skeletal muscle, liver, and adipose tissue in propagating systemic insulin resistance — particularly through exosome-mediated signalling and myokine release during muscle activity.
Therapeutically, Yu et al. evaluate lifestyle interventions (exercise, caloric restriction, and dietary patterns like Mediterranean and ketogenic diets), as well as pharmacological agents including metformin, thiazolidinediones, GLP-1 agonists, and exploratory therapies like intermittent fasting mimetics and natural polyphenols (e.g., resveratrol, berberine). The review highlights that combining lifestyle and pharmacological strategies yields synergistic benefits for insulin sensitivity restoration.
Key Takeaways
Epigenetic reprogramming — including DNA methylation changes and histone modifications — can perpetuate insulin resistance across generations.
Myokines secreted by exercising muscle act as natural insulin sensitisers for liver and adipose tissue.
Polyphenols and nutraceuticals (e.g., berberine, resveratrol) show emerging promise as adjunct therapies in insulin resistance management.
The future of treatment lies in personalised, multi-targeted intervention strategies rather than single-drug approaches.
Study 5: Muscle Insulin Resistance and Cross-Talk With Liver and Adipose Tissue
da Silva Rosa (2020).established foundational understanding of how skeletal muscle insulin resistance does not occur in isolation but is fundamentally intertwined with hepatic and adipose tissue metabolism through complex inter-organ crosstalk. Da Silva Rosa and colleagues provide an elegant framework that continues to guide research today.
The authors illuminate how lipid overflow from dysfunctional adipose tissue — especially from visceral fat — feeds both skeletal muscle and liver with excess FFAs, triggering parallel insulin resistance in both organs. In muscle, this manifests as increased IMCL accumulation and impaired insulin receptor signalling. In the liver, it drives hepatic steatosis and increased hepatic glucose production, worsening systemic hyperglycaemia.
The study places particular emphasis on the role of diacylglycerol-PKCε signalling in liver insulin resistance and DAG-PKCθ signalling in muscle, illustrating that while the downstream consequences are similar, the specific PKC isoforms and lipid species involved differ between tissues. The authors also highlight adipokine dysregulation — particularly reduced adiponectin and elevated leptin resistance — as major inter-organ drivers of systemic insulin resistance.
Key Takeaways
Skeletal muscle, liver, and adipose tissue form an interconnected metabolic triad — treating insulin resistance requires addressing all three, not just one.
Visceral adiposity is the primary driver of systemic lipid overflow and multi-organ insulin resistance.
Adiponectin deficiency is a critical hormonal signal linking adipose dysfunction to muscle and liver insulin resistance.
PKC isoform specificity (PKCθ in muscle vs PKCε in liver) offers tissue-specific therapeutic targeting opportunities.
Connecting the Dots: A Unified Picture of Skeletal Muscle Insulin Resistance
Taken together, these five studies paint a remarkably cohesive molecular picture. Insulin resistance in skeletal muscle is not a single-pathway problem — it is a multidimensional metabolic crisis driven by:
Ectopic lipid accumulation (specifically DAGs and ceramides) that poisons the insulin signalling cascade (Razi et al., 2025; da Silva Rosa et al., 2020)
Chronic inflammation driven by adipose-derived cytokines disrupting GLUT4 trafficking (Razi et al., 2025; Yu et al., 2026)
Oxidative stress and mitochondrial dysfunction that impair cellular energy handling (Razi et al., 2025; Whytock & Goodpaster, 2025)
Molecular signalling defects — particularly IRS-1 serine phosphorylation and ER stress — that cut the insulin signalling wire (Ahn, 2025)
Inter-organ dysregulation via lipid overflow, adipokine imbalance, and myokine signalling failures (da Silva Rosa et al., 2020; Yu et al., 2026)
Importantly, exercise emerges as a universal corrector across nearly every pathway identified — reducing IMCL, activating AMPK, improving mitochondrial health, secreting beneficial myokines, and reducing inflammation simultaneously (Whytock & Goodpaster, 2025; Yu et al., 2026).
Clinical Implications: What This Means for Prevention and Treatment
Skeletal Muscle Is the Metabolic Gatekeeper
Skeletal muscle is responsible for 70–80% of insulin-stimulated glucose disposal. When it becomes insulin resistant, systemic metabolic dysfunction follows. Hyperinsulinaemia, impaired glucose uptake, reduced metabolic flexibility, and accelerated fat storage are not isolated abnormalities — they are downstream consequences of muscle signalling failure.
Lipotoxicity Is the Primary Molecular Insult
Obesity drives excess free fatty acid overflow into skeletal muscle, where lipids accumulate as diacylglycerols (DAGs) and ceramides. These toxic lipid intermediates activate protein kinase C (PKC) isoforms, inhibit IRS-1/PI3K/Akt signalling, and effectively “switch off” insulin sensitivity at the cellular level. This is not passive fat storage — it is active biochemical sabotage.
Inflammation and Oxidative Stress Amplify the Damage
Adipose-derived cytokines such as TNF-α and IL-6 trigger serine phosphorylation of IRS-1, while mitochondrial overload generates reactive oxygen species that impair oxidative phosphorylation. Together, chronic inflammation and mitochondrial dysfunction create a feed-forward loop that entrenches skeletal muscle insulin resistance.
Insulin Resistance Is a Multi-Organ System Disorder
Muscle dysfunction does not occur in isolation. Visceral adipose tissue, liver steatosis, adipokine imbalance, and lipid spillover form a tightly integrated metabolic triad. Addressing glucose alone without targeting adiposity and ectopic lipid deposition is biologically incomplete medicine.
Exercise Is Molecular Therapy — Not Lifestyle Advice
Exercise activates AMPK and CaMKII pathways that stimulate insulin-independent GLUT4 translocation. It reduces harmful lipid species, enhances mitochondrial biogenesis, suppresses inflammatory signalling, and restores metabolic flexibility. No pharmacologic agent currently matches its multi-pathway impact.
The Therapeutic Future Is Multi-Targeted
GLP-1 receptor agonists, SGLT2 inhibitors, AMPK activators, and emerging nutraceuticals show promise. But the next frontier lies in integrated strategies — combining exercise prescription, dietary lipotoxicity reduction, visceral fat targeting, and precision pharmacotherapy.
Dietary Strategies Targeting Lipotoxicity and Inflammation
Reducing saturated fat and refined carbohydrate intake limits the FFA overflow that drives IMCL accumulation. Anti-inflammatory dietary patterns (Mediterranean diet, whole-food plant-rich diets) may reduce TNF-α and IL-6 levels. Emerging evidence supports intermittent fasting as a tool to reduce DAG and ceramide content in muscle.
Targeting the Adipose-Muscle-Liver Axis
Visceral fat reduction — achieved through caloric restriction, exercise, or pharmacological intervention — is foundational. Addressing adiponectin deficiency (through exercise and weight loss) and restoring leptin sensitivity represent meaningful therapeutic targets.
Emerging Pharmacological and Nutraceutical Approaches
GLP-1 receptor agonists, SGLT2 inhibitors, and AMPK activators (including metformin) are increasingly understood not just as glucose-lowering agents but as insulin sensitisers at the molecular level. Adjuncts like berberine and resveratrol show promise but await larger clinical trials.
To Summarize
Skeletal muscle insulin resistance accounts for the majority of whole-body insulin dysfunction, making it the key therapeutic target in metabolic disease.
Intramyocellular lipids (DAGs and ceramides specifically) are central molecular mediators linking obesity to insulin resistance.
Chronic inflammation and oxidative stress act synergistically to impair insulin signalling — they must be targeted together, not in isolation.
Skeletal muscle does not act alone — inter-organ crosstalk with liver and adipose tissue via lipid overflow, adipokines, and myokines shapes the overall metabolic phenotype.
Exercise is the single most powerful intervention — it corrects insulin resistance through at least 5 distinct molecular pathways simultaneously.
Future therapies will likely combine pharmacological, dietary, and exercise-based approaches targeting the epigenetic, inflammatory, and mitochondrial dimensions of insulin resistance.
Frequently Asked Questions (FAQs)
Q1: What exactly is skeletal muscle insulin resistance?
Skeletal muscle insulin resistance occurs when muscle cells fail to respond normally to insulin's signal to take up glucose. This leads to elevated blood glucose levels, increased pancreatic insulin secretion, and over time, the development of type 2 diabetes. Since skeletal muscle is responsible for ~70–80% of insulin-stimulated glucose disposal, its dysfunction has outsized metabolic consequences.
Q2: What are intramyocellular lipids, and why are they dangerous?
Intramyocellular lipids (IMCLs) are fat droplets stored within skeletal muscle fibres. In trained athletes, certain IMCLs serve as fuel — but in obesity and inactivity, excess IMCLs accumulate as diacylglycerols (DAGs) and ceramides, which activate PKC enzymes that phosphorylate IRS-1 on serine residues, effectively switching off insulin signalling (Razi et al., 2025; da Silva Rosa et al., 2020).
Q3: How does inflammation cause insulin resistance in muscle?
Inflammatory cytokines such as TNF-α, IL-6, and IL-1β — released primarily from dysfunctional adipose tissue in obesity — activate NF-κB and JNK signalling pathways inside muscle cells. These pathways phosphorylate IRS-1 at inhibitory serine sites, impair GLUT4 vesicle trafficking, and reduce glucose uptake capacity (Razi et al., 2025; Ahn, 2025).
Q4: Can exercise really reverse insulin resistance at the molecular level?
Yes — and the evidence from multiple studies is compelling. Exercise activates AMPK and CaMKII pathways that drive insulin-independent GLUT4 translocation, directly bypassing the broken insulin signalling cascade. Chronic training also reduces harmful IMCL fractions, improves mitochondrial biogenesis, decreases inflammatory cytokines, and stimulates beneficial myokine secretion — making it a comprehensive metabolic reset (Whytock & Goodpaster, 2025; Yu et al., 2026).
Q5: Is insulin resistance in muscle linked to liver and fat tissue problems?
Absolutely. Da Silva Rosa et al. (2020) demonstrated that these three tissues form an interconnected metabolic triad. Visceral adipose tissue overflows FFAs into the circulation, which triggers ectopic lipid storage in both muscle and liver — causing insulin resistance in both simultaneously. Dysfunctional adipose tissue also reduces adiponectin secretion and increases leptin resistance, further disrupting whole-body insulin sensitivity.
Q6: What are the most promising new treatments for insulin resistance?
Based on Ahn (2025) and Yu et al. (2026), the most promising developments include GLP-1 receptor agonists (which improve insulin sensitivity beyond just glucose lowering), SGLT2 inhibitors, AMPK activators like metformin and its mimetics, and emerging nutraceuticals such as berberine and resveratrol. Future personalised medicine approaches will likely combine pharmacological agents with precision exercise and dietary interventions tailored to individual molecular profiles.
Author’s Note
As a physician trained in internal medicine, I have long been struck by how often insulin resistance is discussed as a laboratory abnormality rather than a cellular disease process. We focus on fasting glucose, HbA1c, and medication titration — yet the real story unfolds inside skeletal muscle, at the level of lipid intermediates, mitochondrial energetics, inflammatory signalling, and insulin receptor substrate biology. This article was written to bridge that gap.
The five studies synthesised here represent a convergence of molecular physiology, metabolic medicine, and exercise science. Together, they make one principle unmistakably clear: skeletal muscle insulin resistance is not a passive consequence of aging or genetics — it is a dynamic, modifiable, biologically reversible state. Lipotoxicity, oxidative stress, ER stress, and adipose–liver–muscle crosstalk are not abstract pathways; they are therapeutic entry points.
Equally important, this body of research reframes exercise from a generic lifestyle recommendation to a form of molecular medicine. AMPK activation, mitochondrial biogenesis, GLUT4 translocation, myokine signalling — these are measurable, reproducible biological adaptations. When prescribed appropriately, physical activity is not adjunctive therapy; it is foundational treatment.
My goal in writing this piece was twofold:
To provide clinicians and researchers with a systems-level synthesis of emerging insulin resistance mechanisms.
To translate complex molecular science into actionable clinical and lifestyle strategies grounded in evidence.
Metabolic disease is advancing globally at an unprecedented pace. The response must be equally sophisticated — integrating pharmacology, exercise physiology, nutritional science, and personalised medicine.
Skeletal muscle remains one of the most adaptable tissues in the human body. The science suggests that, with targeted intervention, metabolic resilience can be restored.
The responsibility now lies with us — as clinicians, scientists, and informed individuals — to act on that knowledge.metabolic health, insulin sensitivity, and the remarkable resilience of skeletal muscle.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Fuel Partitioning Explained: How Your Body Decides to Burn Fat or Sugar | DR T S DIDWAL
The Fat Inside Your Muscles: More Dangerous Than We Thought? | DR T S DIDWAL
Your Body Fat Is an Endocrine Organ—And Its Hormones Shape Your Heart Health | DR T S DIDWAL
hsCRP Explained: What Inflammation Means for Your Heart | DR T S DIDWAL
References
Ahn, B. (2025). Advances in insulin resistance — Molecular mechanisms, therapeutic targets, and future directions. International Journal of Molecular Sciences, 26(6), 2574. https://doi.org/10.3390/ijms26062574
da Silva Rosa, S. C., Nayak, N., Caymo, A. M., & Gordon, J. W. (2020). Mechanisms of muscle insulin resistance and the cross-talk with liver and adipose tissue. Physiological Reports, 8(19), e14607. https://doi.org/10.14814/phy2.14607
Razi, O., de Moraes, C., Zamani, N., Saeidi, A., Hadjicharalambous, M., Hackney, A. C., Del Coso, J., Laher, I., & Zouhal, H. (2025). From obesity to muscle insulin resistance: The mediating roles of intramyocellular lipids, inflammation, and oxidative stress. Diabetes/Metabolism Research and Reviews, 41(7), e70094. https://doi.org/10.1002/dmrr.70094
Whytock, K. L., & Goodpaster, B. H. (2025). Unraveling skeletal muscle insulin resistance: Molecular mechanisms and the restorative role of exercise. Circulation Research, 137(2), 184–204. https://doi.org/10.1161/CIRCRESAHA.125.325532
Yu, L., Qian, J., Li, X., et al. (2026). Insulin resistance: Mechanisms and therapeutic interventions. Molecular Biomedicine, 7, 12. https://doi.org/10.1186/s43556-026-00408-5