Struggling With Blood Sugar? 4 Hidden Hormones May Be Controlling Your Metabolism
Discover how cortisol, growth hormone, glucagon, and insulin work together to control blood sugar, metabolism, sleep, and stress in diabetes and metabolic health.
DIABETES
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/10/202617 min read
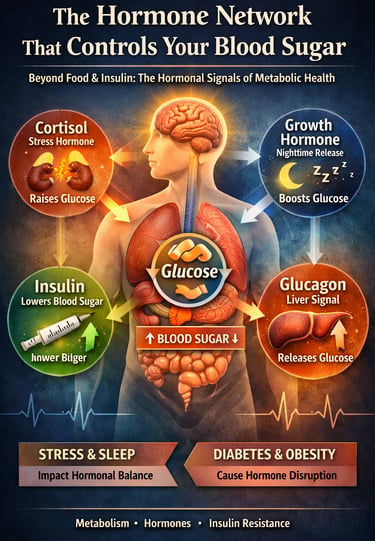
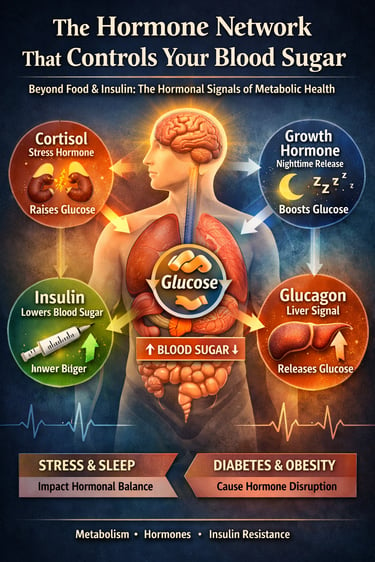
Blood sugar is controlled by more than just insulin. A network of hormones—including cortisol, glucagon, growth hormone, and insulin—works together throughout the day to regulate glucose levels, metabolism, and energy balance. When this hormonal system becomes disrupted, it can contribute to insulin resistance, metabolic syndrome, and type 2 diabetes.
Every moment of the day, your body is performing an extraordinary balancing act. Long before you think about food, exercise, or medication, a complex network of metabolic hormones is already working behind the scenes to keep your blood glucose levels within a narrow and healthy range. This invisible system—often called hormonal glucose regulation—coordinates signals between the pancreas, liver, brain, adrenal glands, and muscle tissue to decide when glucose should be released, stored, or used for energy.
At the center of this metabolic conversation are several powerful chemical messengers: insulin, glucagon, cortisol, and growth hormone. Each of these hormones plays a distinct role in controlling glucose metabolism, insulin sensitivity, and energy balance. Insulin lowers blood sugar by helping cells absorb glucose, while glucagon raises it by signaling the liver to release stored glucose. Meanwhile, cortisol and growth hormone adjust glucose availability during stress, fasting, and sleep, ensuring the brain and muscles have a steady fuel supply (De Fano & Consoli, 2025; Gianotti et al., 2021).
Under normal conditions, this hormonal network operates with remarkable precision. But when these signals become disrupted—because of chronic stress, poor sleep, obesity, insulin resistance, or type 2 diabetes—the system can lose its balance. Instead of maintaining stable glucose levels, the body may begin producing excess glucose, reducing insulin sensitivity, and triggering persistent hyperglycemia.
Recent research has revealed that blood sugar regulation is not controlled by insulin alone. Studies now show that glucagon dysregulation, stress-related cortisol activation, and sleep-dependent growth hormone release all play critical roles in metabolic health and diabetes progression (De Fano & Consoli, 2025; Ding et al., 2025; Jumba, 2025).
Understanding how these hormones interact offers a powerful new perspective on diabetes management, metabolic syndrome, and long-term glucose control. Blood sugar is not simply determined by what you eat—it reflects a sophisticated hormonal system that operates continuously, day and night, to maintain metabolic stability.
In this article, you’ll Learn
Why does cortisol raise blood sugar?
The real cause of the dawn phenomenon
How sleep affects insulin resistance
Why is glucagon a hidden driver of diabetes
Clinical pearls
1. The "Dawn Phenomenon" is a Hormonal Wake-up Call
Scientific Perspective: Early morning hyperglycemia is driven by the pulsatile release of Growth Hormone (GH) and Cortisol. These counter-regulatory hormones trigger hepatic gluconeogenesis and temporary insulin resistance to ensure the brain has adequate glucose for the transition from sleep to wakefulness.
If your morning blood sugar is high despite not eating, it isn’t a "failure." Your body is simply over-preparing for the day by dumping stored sugar into your blood. It’s an internal alarm clock that’s set slightly too high.
2. Incretins: The "Smart" Glucose Regulators
Scientific Perspective: The Incretin Effect refers to the significantly higher insulin response elicited by oral glucose compared to intravenous glucose. This is mediated by hormones like GLP-1 and GIP, which stimulate glucose-dependent insulin secretion and, crucially, suppress postprandial Glucagon hypersecretion.
Think of "incretin" medications (like Ozempic or Mounjaro) as volume knobs. They don't just "force" insulin; they help the gut talk to the pancreas so it knows exactly how much insulin is needed, while simultaneously telling the liver to stop "leaking" extra sugar after you eat.
3. Sleep is a Metabolic "Reset" Button
Scientific Perspective: Deep, slow-wave sleep inhibits somatostatin neurons, allowing for a surge in Growth Hormone that aids in muscle repair and metabolic restoration. Fragmentation of this sleep cycle (as seen in Sleep Apnea) keeps the "gate" closed, leading to chronic insulin resistance and elevated HbA1c.
Sleep isn't just "rest"—it's a 7-hour chemical treatment for your metabolism. Getting 7–8 hours of quality sleep can improve your insulin's effectiveness by up to 25% the very next day.
4. Cortisol Turns Stress into Sugar
Scientific Perspective: Cortisol activates the HPA axis, which promotes the breakdown of muscle protein into amino acids for gluconeogenesis (creating new sugar). In patients with visceral obesity, abdominal fat cells actually amplify cortisol activity, creating a feedback loop that worsens "stress-induced" hyperglycemia.
Stress is a physical event, not just a feeling. When you are stressed, your body thinks you are "fighting a tiger" and sends sugar to your muscles for energy. If you’re just sitting at a desk, that sugar stays in your blood, raising your levels.
5. Glucagon: The "Silent" Driver of Highs
Scientific Perspective: In Type 2 Diabetes, the alpha cells of the pancreas lose their "off switch." Even when blood sugar is 200 mg/dL, the pancreas continues to secrete Glucagon, which forces the liver to keep pumping out glucose, contributing heavily to the "unexplained" highs after meals.
Managing diabetes isn't just about lacking insulin; it's also about having too much of insulin's "opposite" hormone, glucagon. Some modern medications work specifically by quietening this "noisy" hormone so your liver stays in "storage mode" instead of "release mode."
6. Muscle is Your Largest Glucose "Sink"
Scientific Perspective: Skeletal muscle is responsible for over 80% of postprandial glucose disposal via GLUT4 translocation. Resistance training increases the density of these transporters, allowing the body to clear blood sugar more efficiently even when insulin levels are low.
Every bit of muscle you build is like adding a larger "drain" to your blood sugar "sink." The more muscle you have, the more places your body has to store sugar safely, making your blood sugar much easier to control after a meal.
What Hormones Control Blood Sugar?
Blood sugar is regulated by four major hormones:
Insulin – lowers blood glucose by helping cells absorb sugar from the bloodstream
Glucagon – raises blood glucose by signaling the liver to release stored sugar
Cortisol – increases glucose during stress and fasting
Growth hormone – influences overnight glucose production and metabolism during sleep
Together, these hormones form a 24-hour metabolic control system that maintains stable blood sugar levels.
Part 1: Cortisol and Blood Sugar — The Stress–Glucose Connection
What Is Cortisol and Why Does It Raise Blood Sugar?
Cortisol is often called the "stress hormone," but that label barely scratches the surface. Produced by the adrenal glands — two small triangular structures sitting atop your kidneys — cortisol is a glucocorticoid steroid hormone that plays a central role in virtually every system of your body. Its primary evolutionary function was to prepare you to respond to threats: to run from a predator, fight an attacker, or survive a famine.
One of cortisol's most immediate effects is to raise blood glucose. It does this through several mechanisms: prompting the liver to release stored glycogen (a process called glycogenolysis), stimulating the production of new glucose from non-sugar molecules like amino acids (gluconeogenesis), and — critically — making cells in your muscles and fat tissue less responsive to insulin. This last effect is known as insulin resistance, and it is at the heart of why chronic stress is so metabolically damaging.
Cortisol, Obesity, and Diabetes — New Research Insights
In a comprehensive review, Gianotti et al. (2021) examined the intricate relationship between the hypothalamic-pituitary-adrenal (HPA) axis — the body's central stress-response system — and the development of obesity and type 2 diabetes. Their findings highlighted a troubling cycle: excess visceral (abdominal) fat itself stimulates increased cortisol activity, which in turn promotes further fat accumulation and worsens insulin resistance.
The researchers noted that the HPA axis is not simply responding to psychological stress. It is also activated by sleep deprivation, poor diet, chronic inflammation, and even social isolation. In other words, many of the conditions that characterize modern life are quietly, persistently elevating cortisol — and with it, blood sugar.
For patients with type 2 diabetes, this has practical implications. Managing stress is not a "soft" lifestyle recommendation — it is a metabolic intervention. Techniques that reduce cortisol, such as mindfulness-based stress reduction, regular moderate exercise, and adequate sleep, can measurably improve glycemic control.
Patient tip: If you notice your fasting blood sugar is higher on particularly stressful days or after poor sleep, cortisol may be the reason. This is not a failure of your medication — it is your stress-response system doing what it was designed to do.
Gianotti et al. (2021) also noted the bidirectional nature of the stress-metabolism relationship in obesity: elevated cortisol suppresses the release of sex hormones and growth hormone while promoting fat storage around the abdomen. This hormonal cascade compounds insulin resistance, making weight management even more difficult for individuals already struggling with metabolic dysfunction.
Part 2: Growth Hormone and Nighttime Glucose — What Happens While You Sleep
The Dawn Phenomenon and Nocturnal Hormonal Surges
Many people with diabetes notice something puzzling: their blood glucose is higher in the morning than when they went to bed — despite not eating anything overnight. This is not a malfunction. It is the "dawn phenomenon," a predictable hormonal event driven largely by the nocturnal surge of growth hormone (GH).
Growth hormone is released in pulses throughout the day, but its largest surge occurs during deep, slow-wave sleep — typically in the early hours of the night. Under normal circumstances, growth hormone promotes tissue repair, muscle growth, and fat metabolism. But it also has an anti-insulin effect: it tells the liver to produce more glucose and reduces the sensitivity of peripheral tissues to insulin. For most healthy people, the body compensates by releasing more insulin. For people with diabetes or insulin resistance, that compensation is inadequate.
A Groundbreaking Discovery: The Neural Circuit Behind GH Release
Until recently, scientists knew that growth hormone was released during sleep but lacked a clear picture of the neural machinery responsible. A landmark 2025 study published in the prestigious journal Cell has now illuminated this process with remarkable precision.
Ding et al. (2025) identified a specific neuroendocrine circuit in the brain responsible for coupling sleep with growth hormone secretion. Using advanced optogenetic and chemogenetic tools in animal models, the researchers traced the pathway from sleep-active neurons in the hypothalamus through a relay of neuroendocrine signals to the pituitary gland — the structure that actually secretes growth hormone into the bloodstream.
The team discovered that a population of neurons expressing the neuropeptide somatostatin plays a gating role: during slow-wave sleep, these neurons are inhibited, allowing the pituitary to release growth hormone. During wakefulness — or when sleep is fragmented — this gate remains closed, and GH secretion is blunted.
Why this matters: This circuit explains why disrupted sleep does not just leave you feeling tired — it actively disrupts the hormonal signals that regulate nighttime glucose, muscle repair, and metabolic recovery.
From a patient perspective, the implications of this research are significant. It is not enough to simply "get some sleep." The quality of that sleep — particularly the amount of deep, uninterrupted slow-wave sleep — determines how much growth hormone is released, which in turn shapes blood sugar, body composition, and tissue repair overnight.
People with obstructive sleep apnea, insomnia, or those who routinely get fewer than seven hours of sleep may be chronically underproducing growth hormone. This helps explain why these populations tend to have higher rates of insulin resistance, increased abdominal fat, and impaired recovery from exercise (Ding et al., 2025).
Part 3: The Role of Glucagon in Diabetes — From Supporting Actor to Centre Stage
Glucagon Reconsidered: More Than Just Insulin's Opposite
For decades, glucagon was understood primarily as insulin's counterpart: while insulin lowers blood sugar by helping cells absorb glucose, glucagon raises it by telling the liver to release stored glucose. In the classic model of type 2 diabetes, insulin deficiency and resistance were the primary drivers of hyperglycemia, and glucagon was considered a secondary concern.
That understanding is now being revised. A pivotal 2025 review by De Fano and Consoli, published in Nutrition, Metabolism and Cardiovascular Diseases, argues compellingly that glucagon deserves recognition as a key pathophysiological player in diabetes — one that has been underappreciated for too long.
Glucagon's Expanded Biological Role
In healthy individuals, glucagon and insulin exist in careful balance. After eating, insulin rises and glucagon falls, ensuring that glucose from the meal is absorbed into cells and stored, not added to an already-adequate blood supply. Between meals and during exercise, glucagon rises to prevent blood sugar from dropping dangerously low.
But in type 2 diabetes, this regulation breaks down in a specific way: glucagon levels remain inappropriately elevated even when blood sugar is already high. Instead of falling after a meal, glucagon stays high, continuously driving the liver to produce and release more glucose. This phenomenon — called "glucagon hypersecretion" — can account for a substantial portion of the postprandial (after-meal) hyperglycemia that characterises type 2 diabetes.
De Fano and Consoli (2025) further detail that the problem is not confined to the pancreatic alpha cells that produce glucagon. The receptors for glucagon are expressed broadly — in the liver, heart, kidneys, brain, and adipose tissue — meaning that dysregulated glucagon has systemic metabolic consequences far beyond blood glucose elevation.
Glucagon as a Therapeutic Target
This expanding understanding of glucagon's role has opened new avenues in diabetes therapy. Several classes of medications now target the glucagon system, most notably the glucagon-like peptide-1 receptor agonists (GLP-1 RAs) — drugs like semaglutide and liraglutide that have reshaped diabetes and obesity treatment in recent years.
GLP-1 receptor agonists work partly by suppressing inappropriate glucagon secretion after meals, in addition to stimulating insulin release and slowing gastric emptying. This dual effect on both arms of the insulin-glucagon axis helps explain their impressive glycemic control and, for some patients, their ability to support substantial weight loss.
Key takeaway: If you are on a GLP-1 medication, it is working on multiple hormonal fronts — including reducing the glucagon excess that has been silently driving up your blood sugar.
De Fano and Consoli (2025) conclude their review by calling for greater clinical attention to glucagon measurement and management, noting that current standard diabetes monitoring — which focuses primarily on insulin and blood glucose — may be missing an important piece of the metabolic puzzle.
Part 4: How Sleep Affects Blood Sugar — The Evidence Is Compelling
Sleep Duration, Sleep Quality, and Glycemic Control
The relationship between sleep and blood sugar is bidirectional and well-established. Poor blood sugar control can disrupt sleep through nocturia (nighttime urination), hypoglycemic episodes, and discomfort. But the reverse is equally true — and often more actionable: poor sleep directly impairs blood sugar control.
A 2025 longitudinal study by Jumba examined the effect of sleep duration and quality on glycemic control in adults with type 2 diabetes. Following participants over time, the study found that both short sleep duration (less than six hours per night) and poor sleep quality were independently associated with higher HbA1c levels — the key long-term marker of blood sugar control.
The Mechanisms: How Poor Sleep Raises Blood Sugar
Sleep deprivation affects blood glucose through multiple converging pathways. First, as established in the cortisol literature, sleep loss activates the HPA axis, elevating cortisol and driving up blood sugar. Second, it blunts the release of growth hormone (as demonstrated in the Ding et al., 2025 circuit research), disrupting overnight metabolic regulation. Third, sleep deprivation reduces insulin sensitivity in peripheral tissues — studies have shown that just two to three nights of restricted sleep can reduce insulin sensitivity by as much as 25%.
Jumba (2025) noted that sleep quality may be as important as sleep quantity. Fragmented sleep — characterised by frequent awakenings, light sleep, or untreated sleep apnea — is associated with worse glycemic outcomes even when total sleep time appears adequate. This makes clinical sense in light of the Ding et al. (2025) findings: it is specifically deep, slow-wave sleep that drives growth hormone release and metabolic restoration.
How Hormones Control Blood Sugar Throughout the Day
Morning (Early Wake-Up Phase)
Cortisol increases
Signals the liver to release stored glucose
Helps prepare the brain and muscles for the day’s energy demands
Contributes to the “dawn phenomenon” seen in many people with diabetes
After Meals (Post-Meal Phase)
Insulin increases
Moves glucose from the bloodstream into muscle and fat cells
Promotes glucose storage in the liver as glycogen
Glucagon decreases
Prevents the liver from releasing additional glucose
Helps stabilise post-meal blood sugar levels
Night (During Deep Sleep)
Growth hormone increases
Supports tissue repair, muscle recovery, and fat metabolism
Slightly raises glucose production overnight
Plays an important role in metabolic recovery during slow-wave sleep
Key Takeaway
Blood sugar regulation is a 24-hour hormonal process.
Cortisol prepares the body for daytime energy needs, insulin manages glucose after meals, glucagon prevents low blood sugar between meals, and growth hormone coordinates metabolic recovery during sleep.
Practical Strategies for Better Sleep and Better Blood Sugar
For patients with type 2 diabetes or prediabetes, optimising sleep hygiene is a legitimate and evidence-based part of metabolic management. Some practical steps include:
Maintaining a consistent sleep and wake time, even on weekends
Keeping the bedroom cool, dark, and quiet to promote slow-wave sleep
Limiting screen exposure for at least an hour before bed, as blue light suppresses melatonin
Avoiding large meals, alcohol, and caffeine in the hours before sleep
Discussing symptoms of sleep apnea with your doctor if you snore, feel unrested, or frequently wake in the night
Engaging in regular moderate-intensity exercise, which has been shown to improve both sleep quality and insulin sensitivity
Jumba (2025) also highlighted the importance of screening for sleep disorders as part of routine diabetes care, noting that sleep apnea — which is significantly more prevalent in people with obesity and type 2 diabetes — is a frequently undiagnosed contributor to poor glycemic control.
Connecting the Dots: A Systems View of Hormones and Blood Sugar
1. Blood Sugar Regulation Is a Hormonal System, Not Just a Dietary Issue
Blood glucose control is often portrayed as a simple matter of diet and insulin. In reality, it is governed by a sophisticated endocrine network involving multiple hormones that continuously communicate with the liver, pancreas, brain, and muscle tissue. Insulin lowers blood glucose by facilitating cellular uptake, while glucagon, cortisol, and growth hormone act as counter-regulatory hormones that increase glucose availability during fasting, stress, or sleep. Disruption of this balance lies at the heart of metabolic disease and type 2 diabetes.
2. Cortisol Links Modern Stress to Metabolic Dysfunction
Chronic activation of the hypothalamic–pituitary–adrenal (HPA) axis has become a defining feature of modern metabolic disorders. Elevated cortisol promotes hepatic gluconeogenesis, glycogen breakdown, and peripheral insulin resistance, creating sustained hyperglycemia. Research suggests that visceral adiposity can further amplify cortisol activity, establishing a vicious cycle between stress hormones, obesity, and impaired glucose metabolism (Gianotti et al., 2021).
3. Sleep Physiology Plays a Critical Role in Glucose Regulation
Sleep is not a passive metabolic state. During deep slow-wave sleep, the brain activates neuroendocrine circuits that stimulate growth hormone secretion, a process that influences overnight glucose production and tissue repair. Disrupted sleep architecture can impair this process, leading to reduced metabolic recovery and worsening insulin sensitivity (Ding et al., 2025).
4. Glucagon Is Emerging as a Central Player in Diabetes
For decades, diabetes research focused primarily on insulin deficiency and resistance. However, recent evidence indicates that inappropriate glucagon secretion is a major contributor to persistent hyperglycemia. Elevated glucagon levels stimulate hepatic glucose output even when blood sugar is already high, highlighting the importance of therapies that target the insulin–glucagon axis (De Fano & Consoli, 2025).
5. Sleep Quality Influences Long-Term Glycemic Control
Epidemiological and longitudinal data increasingly show that short sleep duration and poor sleep quality correlate with higher HbA1c levels in people with type 2 diabetes. Sleep deprivation simultaneously increases cortisol, reduces insulin sensitivity, and disrupts growth hormone rhythms—creating a multi-hormonal pathway to metabolic deterioration (Jumba, 2025).
6. The Future of Diabetes Care Requires a Systems Perspective
Blood sugar regulation is a 24-hour hormonal process shaped by stress, sleep, and metabolic signaling. Understanding this interconnected system is essential for designing more effective strategies to prevent and treat diabetes, obesity, and metabolic syndrome.
Frequently Asked Questions (FAQs)
Q1. Why is my blood sugar higher in the morning even though I didn't eat anything overnight?
This is known as the "dawn phenomenon." During the early morning hours, your body releases hormones — including cortisol and growth hormone — as part of its natural wake-up process. These hormones signal the liver to release stored glucose, preparing your body for the day ahead. In people with diabetes, the usual insulin response to counteract this glucose release is inadequate, resulting in elevated fasting blood sugar. This is especially linked to the neuroendocrine circuit described by Ding et al. (2025), which governs growth hormone release during sleep.
Q2. Can stress really raise my blood sugar? How much does it matter?
Yes, stress can raise blood sugar significantly, and it matters more than many people realise. Cortisol — released during both psychological and physical stress — directly promotes glucose production in the liver and reduces insulin sensitivity in muscles and fat tissue. Research by Gianotti et al. (2021) highlights that people with obesity and type 2 diabetes often have a dysregulated cortisol response, meaning their baseline cortisol can be elevated even in the absence of obvious stressors. Over time, chronically high cortisol contributes meaningfully to poor glycemic control.
Q3. What is glucagon, and how does it affect diabetes differently from insulin?
Glucagon is a hormone produced by alpha cells in the pancreas. Its job is to raise blood sugar by stimulating the liver to release stored glucose — essentially the opposite of insulin's role. In type 2 diabetes, glucagon secretion becomes dysregulated: it remains inappropriately high even when blood sugar is already elevated after a meal, which continually pushes blood glucose higher. De Fano and Consoli (2025) argue that this glucagon excess is a key driver of diabetes that has historically been underappreciated in clinical practice.
Q4. How does poor sleep affect my blood sugar the next day?
Poor sleep raises blood sugar through several mechanisms: it elevates cortisol, blunts growth hormone release (which impairs overnight glucose regulation), and directly reduces insulin sensitivity. Research by Jumba (2025) found that both short sleep duration and poor sleep quality are independently associated with worse long-term blood sugar control (as measured by HbA1c) in adults with type 2 diabetes. Even a few nights of disrupted sleep can measurably reduce insulin sensitivity.
Q5. How many hours of sleep should a person with type 2 diabetes aim for?
Most evidence — including the findings of Jumba (2025) — supports seven to nine hours of quality sleep per night for optimal metabolic health. However, quality is as important as quantity. Deep, uninterrupted slow-wave sleep is particularly important because it is during this phase that growth hormone is released and overnight metabolic restoration occurs. People experiencing fragmented sleep due to conditions like sleep apnea may not benefit sufficiently even if their total sleep time appears adequate.
Q6. Are GLP-1 medications (like Ozempic or Wegovy) related to the glucagon research discussed in this article?
Yes, directly. GLP-1 receptor agonists — which include semaglutide (brand names Ozempic and Wegovy) and liraglutide (Victoza) — work in part by suppressing the inappropriate glucagon hypersecretion that occurs after meals in people with type 2 diabetes. This is precisely the mechanism highlighted in De Fano and Consoli's (2025) review. By reducing excess glucagon, these medications help prevent the liver from releasing unnecessary glucose after eating — one reason they are so effective at lowering postprandial blood sugar and supporting weight management.
Q7. What practical steps can I take today to better manage the hormonal factors affecting my blood sugar?
There are several evidence-informed steps you can take: (1) Prioritise 7–9 hours of quality sleep each night — consistent sleep timing and a cool, dark bedroom help promote deep slow-wave sleep. (2) Manage stress actively, whether through exercise, mindfulness, social connection, or working with a mental health professional. (3) Ask your doctor whether your diabetes management plan accounts for glucagon dysregulation — newer medication classes may be appropriate. (4) Get screened for sleep apnea if you snore or feel unrefreshed in the morning. (5) Recognise that blood sugar management is a 24-hour process — what you do overnight matters as much as your daytime habits.
Author’s Note
This article was written to translate complex discoveries in endocrinology and metabolic physiology into language that patients, caregivers, and health-conscious readers can understand and use. Modern research increasingly shows that blood sugar regulation is not controlled by insulin alone. Instead, it reflects the coordinated actions of several hormones—including insulin, glucagon, cortisol, and growth hormone—that interact continuously with sleep patterns, stress responses, and metabolic health.
The goal of this article is to bridge the gap between emerging scientific literature and practical patient awareness. The explanations presented here are based on peer-reviewed research in endocrinology, metabolism, and diabetes science. Wherever possible, findings from recent studies have been summarized to illustrate how hormonal networks influence glucose metabolism, insulin sensitivity, and long-term metabolic outcomes.
It is important to note that the field of metabolic health is evolving rapidly. New discoveries—particularly in areas such as glucagon physiology, neuroendocrine sleep circuits, and hormonal regulation of glucose production—are reshaping how clinicians understand diabetes and related metabolic disorders. As such, the discussion presented here reflects current scientific knowledge but should not be interpreted as individualized medical guidance.
Readers living with diabetes, prediabetes, or other metabolic conditions should always consult their healthcare provider before making changes to medications, diet, sleep routines, or lifestyle interventions.
Ultimately, improving metabolic health requires both scientific understanding and practical action. By understanding how hormones influence blood sugar throughout the day and night, patients and clinicians alike can take a more integrated and informed approach to diabetes management and long-term metabolic wellness.Have you noticed a pattern between your stress levels and your morning glucose readings? Share your observations in the comments below
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Managing Diabesity: A Complete Guide to Weight Loss and Blood Sugar Control | DR T S DIDWAL
The BMI Paradox: Why "Normal Weight" People Still Get High Blood Pressure | DR T S DIDWAL
Breakthrough Research: Leptin Reduction is Required for Sustained Weight Loss | DR T S DIDWAL
References
De Fano, M., & Consoli, A. (2025). A new role for glucagon: From secondary hormone to key player in the pathogenesis and treatment of diabetes mellitus. Nutrition, Metabolism and Cardiovascular Diseases. Advance online publication. https://doi.org/10.1016/j.numecd.2025.06.014
Ding, X., Hwang, F.-J., Silverman, D., Zhong, P., Li, B., Ma, C., Lu, L., Jiang, G., Zhang, Z., Huang, X., Tu, X., Tian, Z. M., Ding, J., & Dan, Y. (2025). Neuroendocrine circuit for sleep-dependent growth hormone release. Cell, 188(18), 4968–4982. https://doi.org/10.1016/j.cell.2025.05.039
Gianotti, L., Belcastro, S., D'Agnano, S., & Tassone, F. (2021). The stress axis in obesity and diabetes mellitus: An update. Endocrines, 2(3), 334–347. https://doi.org/10.3390/endocrines2030031
Jumba, K. K. (2025). The effect of sleep duration and quality on glycemic control in adults with type 2 diabetes: A longitudinal study. Newport International Journal of Public Health and Pharmacy, 6(1), 95–99. https://doi.org/10.59298/NIJPP/2025/619599