Is Type 2 Diabetes an Immune Disease? 5 Breakthrough Studies (2025–2026)
New 2025–2026 research reveals type 2 diabetes as an immune-driven disease. Discover 5 breakthrough studies reshaping diabetes treatmen
DIABETES
Dr. T.S. Didwal, M.D.(Internal Medicine)
2/28/202613 min read
"Diabetes is not just a sugar problem — it's an immune system crisis playing out in slow motion."
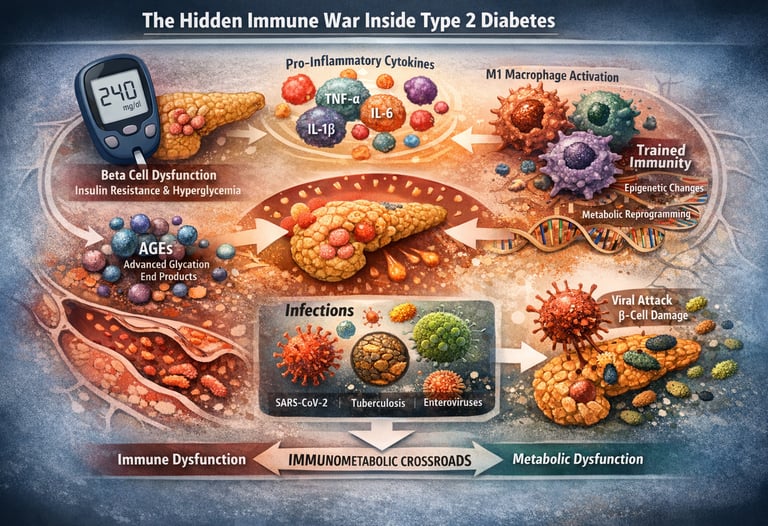
What if type 2 diabetes mellitus (T2DM) is not primarily a disorder of blood sugar, but a chronic, smoldering immune dysfunction unfolding at the cellular level?
For decades, T2DM has been framed as a disease of hyperglycemia, insulin resistance, and pancreatic beta-cell failure. Yet emerging research in immunometabolism, inflammatory signaling, and epigenetic reprogramming suggests a deeper pathology: a persistent immune imbalance driven by metabolic stress. Adipose tissue inflammation, M1 macrophage polarization, elevated pro-inflammatory cytokines (TNF-α, IL-6, IL-1β), and activation of the NF-κB pathway collectively disrupt insulin receptor signaling, impair glucose uptake, and accelerate systemic metabolic dysfunction (Berbudi et al., 2025).
At the same time, chronic hyperglycemia promotes the formation of advanced glycation end-products (AGEs), oxidative stress, and RAGE-mediated inflammatory cascades, further amplifying immune activation and vascular injury (Alexander et al., 2024). Even more striking is the concept of trained immunity — a form of innate immune memory in which monocytes and macrophages undergo epigenetic modifications and metabolic reprogramming toward glycolysis and the mevalonate pathway, sustaining inflammation long after glucose levels improve (Liu et al., 2025). This phenomenon may help explain the persistence of diabetic complications despite optimal glycemic control.
Compounding this immune-metabolic loop, infections such as SARS-CoV-2, Mycobacterium tuberculosis, and enteroviruses can directly impair beta-cell function and worsen insulin resistance, while diabetes itself increases susceptibility to severe infection — a true immunometabolic crossroads (Islam et al., 2025).
Taken together, these findings redefine T2DM as a disorder of chronic low-grade inflammation, immune dysregulation, and disrupted metabolic homeostasis — not merely a disease of elevated glucose, but a systemic immune crisis playing out in slow motion.
Clinical pearls
1. The "Open Door" Policy (Neutrophil Function)
Scientific Perspective: Hyperglycemia induces non-enzymatic glycation of proteins and impairs neutrophil chemotaxis and phagocytosis. Essentially, high sugar "stiffens" the innate immune response, preventing white blood cells from reaching the site of infection.
Think of your white blood cells as a security team. When blood sugar is high, their "GPS" breaks, and they move too slowly to catch intruders. This is why even a small foot cut can become a major issue—the security team simply isn't showing up to work.
2. Metabolic Memory (Trained Immunity)
Scientific Perspective: Through epigenetic reprogramming, myeloid cells (like macrophages) "remember" a hyperglycemic environment via histone modifications. This "trained immunity" leads to a persistent pro-inflammatory state (IL-6 and TNF alpha production) even after reaching euglycemia.
Your immune cells have a memory. If they spent years in a high-sugar environment, they may stay "angry" and inflamed even after their A1c is back to normal. This is why long-term consistency matters more than a single "good" week of blood sugar readings.
3. The "Clogging" of the Signal (IRS-1 Inhibition)
Scientific Perspective: Pro-inflammatory cytokines (specifically IL-1beta) activate kinases like JNK and KK beta, which lead to the inhibitory serine phosphorylation of Insulin Receptor Substrate-1 (IRS-1). This is the primary molecular link between inflammation and insulin resistance.
Imagine your cells have a lock (the insulin receptor) and a key (insulin). Inflammation acts like gum jammed into that lock. No matter how many keys (insulin) you have, you can't open the door to let the energy in because the "gum" of inflammation is blocking the mechanism.
4. Beyond the A1c (The Biomarker Shift)
Scientific Perspective: HbA1c is a 3-month retrospective of glucose, but it doesn't measure active tissue damage. Incorporating hs-CRP (high-sensitivity C-reactive protein) and Galectin-3 provides a "real-time" look at systemic inflammation and the risk of organ scarring (fibrosis).
Patient Perspective: The A1c test is like a "summary" of your car's fuel efficiency, but an inflammation test (like hs-CRP) is like a "check engine" light. You can have a full tank of gas, but still have an engine that is overheating and causing damage. Ask your doctor about checking both.
5. The Gut-Immune Highway
Scientific Perspective: Diabetic dysbiosis leads to increased intestinal permeability (leaky gut), allowing Lipopolysaccharides (LPS) from bacteria to enter the bloodstream. This triggers Toll-like Receptor 4 (TLR4), igniting systemic metabolic inflammation.
Your gut is the "border control" for your immune system. When the gut lining is weak, bacterial toxins leak into your blood and put your immune system on high alert. Eating fibre and probiotics isn't just for digestion; it’s about keeping those toxins out of your blood so your insulin can work better.
6. The Bidirectional Viral Threat
Scientific Perspective: SARS-CoV-2 and other viruses utilize the ACE2 receptor, which is highly expressed in pancreatic beta-cells. Viral entry can trigger insulitis (inflammation of the islets), leading to rapid metabolic decompensation or "post-COVID" new-onset diabetes.
Getting sick with a virus isn't just a temporary setback; it can be a direct attack on the cells that make your insulin. Protecting yourself from infections (via vaccines and hygiene) is actually a vital part of "diabetes management" because it protects your pancreas from direct viral damage
.
Current 2025–2026 research suggests type 2 diabetes is not only a metabolic disease but also a chronic immune-driven inflammatory disorder characterised by cytokine activation, trained immunity, and metabolic–immune dysregulation.
Study 1: The Insulin Resistance–Immune Dysregulation Loop
This comprehensive review by Berbudi et al.(2025) maps out the intricate two-way relationship between insulin resistance and immune system dysfunction. Rather than viewing these as separate problems, the authors argue compellingly that they form a self-reinforcing pathological loop — one that is central to the progression of T2DM.
The paper examines how chronic low-grade inflammation, driven largely by pro-inflammatory macrophages (the so-called M1 macrophage polarization), contributes directly to impaired insulin signaling. Key inflammatory mediators — including tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), and interleukin-1 beta (IL-1β) — interfere with the insulin receptor substrate (IRS-1) pathway, effectively blocking glucose uptake in peripheral tissues.
Simultaneously, hyperglycemia and dyslipidemia in diabetic patients promote a pro-inflammatory immune environment, creating a feedback cycle that perpetuates both insulin resistance and immune dysregulation.
The review also highlights the roles of T regulatory cells (Tregs), natural killer (NK) cells, and dendritic cells in modulating metabolic outcomes, and evaluates the potential of immunomodulatory therapies — including anti-IL-1β agents and PPAR-γ agonists — as adjunctive treatments for T2DM.
Key Takeaways
Insulin resistance is not just a metabolic defect — it is fundamentally immunological in nature.
M1 macrophage polarization and pro-inflammatory cytokines directly impair insulin receptor signaling.
Therapeutic strategies targeting immune pathways (e.g., IL-1β blockade) may hold promise as treatments for T2DM.
The relationship between metabolism and immunity is bidirectional and cyclical — not linear.
Study 2: Diabetes-Induced Immune Dysfunction at the Molecular Level
Published in the International Journal of Molecular Sciences, this study by Alexander et al.(2024) studied how diabetes reshapes the immune landscape at the molecular and cellular level. It is one of the most detailed explorations of diabetes-induced immune pathology available in the current literature.
The authors systematically dissect how hyperglycemia impairs the function of multiple immune cell populations. Neutrophils, for instance, exhibit defective chemotaxis and reduced phagocytic activity in diabetic individuals — a phenomenon that directly explains why people with diabetes face significantly elevated risks of bacterial and fungal infections. T-cell dysfunction in T2DM includes impaired proliferation, skewed Th1/Th2 ratios, and reduced cytotoxic CD8+ T-cell activity.
Beyond cellular dysfunction, the study spotlights the role of advanced glycation end-products (AGEs) — molecules formed when excess glucose reacts with proteins and lipids. AGEs activate receptors (particularly RAGE) that trigger NF-κB signaling, generating a persistent inflammatory environment that impairs immune surveillance and accelerates vascular complications.
The study also underscores the significance of complement system dysregulation in diabetes, which contributes to tissue damage and impaired pathogen clearance — a less-discussed but critically important aspect of diabetic immune dysfunction.
Key Takeaways
Hyperglycemia systematically cripples innate and adaptive immune cells, increasing vulnerability to infections.
Advanced glycation end-products (AGEs) drive persistent inflammation via RAGE/NF-κB signaling.
Complement system dysregulation in diabetes contributes to both immune impairment and organ damage.
Understanding molecular immune pathology is essential for developing targeted therapies for diabetic complications.
Study 3: Trained Immunity — The Immune System's Metabolic Memory
Liu et al.(2025) introduced the concept of trained immunity as a key player in the pathophysiology of diabetes and its complications. Published in Frontiers in Immunology, it offers a genuinely novel lens through which to understand why diabetic inflammation can become self-sustaining even after glucose levels are controlled.
Trained immunity refers to the ability of innate immune cells — primarily monocytes, macrophages, and natural killer cells — to undergo epigenetic reprogramming following an initial stimulus (such as persistent hyperglycemia), causing them to mount exaggerated inflammatory responses to subsequent stimuli. This is distinct from traditional immunological memory, which was thought to be exclusive to adaptive immune cells (T and B lymphocytes).
In the context of T2DM, the authors demonstrate how metabolic reprogramming — particularly shifts in cellular energy metabolism toward glycolysis and the mevalonate pathway — drives trained immunity phenotypes in myeloid cells. These epigenetically altered cells produce excessive amounts of IL-6, TNF-α, and IL-1β even under conditions of normalized glycemia, which may help explain the concept of "metabolic memory" and why tight glucose control alone does not always prevent diabetic complications.
The paper also explores exciting therapeutic angles, including the potential of mTOR inhibitors, AMPK activators, and epigenetic modifiers to reverse trained immunity phenotypes in diabetic patients.
Study 4: Inflammation and Immune Biomarkers in Diabetic Complications
While the previous studies illuminate mechanisms, the review by Rao et al.(2025) in Inflammopharmacology brings us closer to clinical translation — specifically, how inflammatory and immune biomarkers can transform the way we predict, monitor, and treat diabetic complications.
The study comprehensively catalogues biomarkers associated with major diabetic complications, including diabetic nephropathy, diabetic retinopathy, diabetic neuropathy, and cardiovascular disease. Among the most clinically actionable findings are the roles of high-sensitivity C-reactive protein (hs-CRP), interleukin-18 (IL-18), monocyte chemoattractant protein-1 (MCP-1), adiponectin, and galectin-3 as indicators of disease severity and progression.
The authors argue forcefully for integrating multi-biomarker panels into routine diabetic care, moving beyond HbA1c as the singular measure of disease management. They also review evidence supporting immunomodulatory pharmacotherapy — including SGLT-2 inhibitors, GLP-1 receptor agonists, and colchicine — as agents that reduce systemic inflammation and confer organ-protective effects that exceed their glucose-lowering properties.
Particularly notable is the paper's emphasis on the gut microbiome-immune axis in modulating inflammation in T2DM, with microbial dysbiosis emerging as both a biomarker and a therapeutic target.
Multiple immune biomarkers (hs-CRP, IL-18, MCP-1, galectin-3) can predict and track diabetic complications more precisely than HbA1c alone.
SGLT-2 inhibitors and GLP-1 receptor agonists have significant anti-inflammatory effects beyond glucose control.
The gut microbiome is an emerging source of immune biomarkers and a therapeutic target in T2DM.
Multi-biomarker approaches represent the future of personalized diabetes complication management.
Study 5: Infections as Two-Way Traffic Between Diabetes and Immunity
The final study in our review — and possibly the most globally relevant given the aftermath of COVID-19 — is this paper by Islam et al. (2025) in Frontiers in Endocrinology. It tackles a profoundly important and often underappreciated question: Do infections cause diabetes, or does diabetes cause infections — or both?
The answer, as the authors demonstrate with exhaustive evidence, is unequivocally both. This is the concept of the "immunometabolic crossroads" — the intersection of infectious disease, immune function, and metabolic dysregulation.
The paper reviews how specific pathogens including SARS-CoV-2, Mycobacterium tuberculosis (the TB bacterium), Helicobacter pylori, and various enteroviruses can directly induce beta-cell destruction, insulin resistance, and systemic metabolic dysfunction through mechanisms including molecular mimicry, cytokine storms, and direct pancreatic viral invasion.
Conversely, the diabetic immune environment — characterised by impaired neutrophil function, reduced T-cell immunity, elevated pro-inflammatory cytokine levels, and disrupted mucosal immunity — dramatically increases susceptibility to bacterial, viral, and fungal infections, creating a vicious and often lethal cycle.
The study gives special attention to COVID-19-induced new-onset diabetes (sometimes called "pancreatogenic diabetes" or "post-COVID diabetes"), presenting compelling evidence that SARS-CoV-2 can trigger T2DM and worsen existing metabolic disease through direct ACE-2 receptor-mediated pancreatic injury and immune-mediated insulitis.
Infections and T2DM have a bidirectional relationship — each worsens the other.
SARS-CoV-2, M. tuberculosis, and enteroviruses can directly induce or accelerate beta-cell dysfunction and insulin resistance.
Diabetic immune dysfunction creates a perfect storm of increased infection susceptibility and impaired pathogen clearance.
Post-COVID diabetes is a real and concerning phenomenon driven by direct viral and immune-mediated pancreatic injury.
Connecting the Dots: The Unified Picture
1️⃣ Type 2 Diabetes Is Not Just Hyperglycemia
For decades, type 2 diabetes mellitus (T2DM) has been reduced to a disorder of elevated blood glucose and insulin resistance. This framing is incomplete. Emerging evidence positions T2DM as a state of chronic immune dysregulation, characterized by persistent low-grade inflammation, altered innate immune activation, and disrupted metabolic–immune crosstalk.
2️⃣ The Inflammation–Insulin Resistance Loop
Pro-inflammatory cytokines — including TNF-α, IL-6, and IL-1β — impair insulin receptor signaling through activation of stress kinases and the NF-κB pathway, directly worsening insulin resistance. Meanwhile, hyperglycemia and lipotoxicity amplify M1 macrophage polarization, sustaining a self-reinforcing inflammatory loop. Metabolic stress drives immune activation; immune activation deepens metabolic dysfunction.
3️⃣ Hyperglycemia Rewrites Immune Memory
The concept of trained immunity represents a paradigm shift. Chronic glucose exposure induces epigenetic reprogramming in monocytes and macrophages, shifting cellular metabolism toward glycolysis and the mevalonate pathway. Even after glycemic normalization, these immune cells remain primed for exaggerated inflammatory responses — a potential explanation for “metabolic memory” and persistent diabetic complications.
4️⃣ Immune Biomarkers Redefine Risk Assessment
HbA1c alone is an insufficient lens. Biomarkers such as hs-CRP, IL-18, MCP-1, galectin-3, and adiponectin provide insight into inflammatory burden and organ-specific risk. The future of diabetes care lies in multi-biomarker panels that integrate metabolic and immune parameters.
5️⃣ Infection and Diabetes: A Bidirectional Axis
Diabetes impairs neutrophil function, T-cell responsiveness, and mucosal immunity, increasing susceptibility to infection. Conversely, pathogens — including SARS-CoV-2 and Mycobacterium tuberculosis — can accelerate beta-cell dysfunction and insulin resistance. This is the true immunometabolic crossroads.
6️⃣ Therapeutic Implications: Beyond Glucose Control
Modern therapies such as SGLT-2 inhibitors and GLP-1 receptor agonists exert anti-inflammatory effects independent of glucose-lowering. Future strategies may include targeted cytokine inhibition, epigenetic modulation, microbiome-directed therapy, and precision immune profiling.
7️⃣ The Clinical Imperative
The next decade of diabetes management will not focus solely on glycemic targets. It will focus on restoring immune-metabolic homeostasis. Recognizing T2DM as a systemic inflammatory condition reframes prevention, treatment, and long-term risk reduction.
The conclusion is clear: Type 2 diabetes is not merely a metabolic disorder — it is a chronic immune crisis unfolding at cellular scale.
Therapeutic Frontiers: Where Science Is Heading
Anti-inflammatory agents targeting specific cytokines (IL-1β, IL-6, TNF-α) to disrupt the insulin resistance–inflammation cycle. Epigenetic therapies that reverse trained immunity in macrophages and monocytes. Multi-biomarker monitoring panels that go beyond HbA1c to track immune inflammation across target organs. Gut microbiome modulation through probiotics, prebiotics, or fecal microbiota transplantation to reduce systemic immunometabolic dysregulation. Infection prevention and early treatment as a first-line strategy for preventing metabolic deterioration in at-risk populations.
Recent findings by Anderson et al. (2026), published in Nature Cardiovascular Research, have sent shockwaves through the metabolic community. For years, the drug AZD1656 was considered a "failed" medication because it was inconsistent at lowering blood glucose. However, researchers at Queen Mary University have discovered that it does something arguably better: it heals the diabetic heart by retraining the immune system. This landmark 2026 research identifies AZD1656 as a pioneer in "immunometabolic" therapy, shifting the focus of Type 2 Diabetes (T2DM) treatment from mere glucose lowering to active immune restoration. While originally designed to activate glucokinase for glycemic control, studies demonstrate that AZD1656’s true value lies in its ability to selectively enhance the migration of Regulatory T cells (Tregs) into the diabetic heart.
The study found that diabetic cardiomyopathy is driven by a "perfect storm" of mitochondrial dysfunction and chronic inflammation. This metabolic stress triggers the NLRP3 inflammasome, turning the heart into an immunogenic site that attracts proinflammatory T and B cells. AZD1656 effectively "retrains" this environment; by fueling Treg motility through glucokinase-regulated glycolysis, it floods the myocardium with anti-inflammatory signals like IL-10.
Remarkably, these benefits occurred independently of blood sugar levels. AZD1656 reduced cardiac fibrosis, lowered myocardial oxygen consumption by 68%, and restored metabolic flexibility. This represents a paradigm shift: the first "sugar-neutral" diabetes drug that treats the immune crisis at the heart of the disease, proving that repairing the body’s "internal repair crew" (Tregs) can reverse organ damage even when hyperglycemia persists.
Frequently Asked Questions (FAQs)
Q1: Can improving immune function help control type 2 diabetes? Yes — emerging evidence strongly suggests that immunomodulatory strategies (reducing inflammation, targeting specific cytokines) can improve insulin sensitivity and slow the progression of diabetic complications. This is an active and rapidly advancing area of clinical research.
Q2: What is trained immunity, and why does it matter in diabetes? Trained immunity is the ability of innate immune cells to "remember" prior inflammatory exposures through epigenetic changes, leading them to respond more aggressively in the future. In T2DM, hyperglycemia can train macrophages to stay chronically inflamed — even when blood sugar is later controlled. This may explain why complications persist despite good glycemic management.
Q3: How does COVID-19 relate to diabetes? SARS-CoV-2 can infect pancreatic beta cells via ACE-2 receptors and trigger immune-mediated insulitis, leading to new-onset T2DM or worsening of existing disease. Post-COVID diabetes is now recognized as a distinct clinical phenomenon requiring careful monitoring and management.
Q4: What immune biomarkers should people with diabetes ask their doctors about? Beyond HbA1c, emerging biomarkers like high-sensitivity CRP (hs-CRP), IL-18, MCP-1, adiponectin, and galectin-3 can provide important insights into inflammatory status and risk of diabetic complications. Discuss with your healthcare provider whether expanded biomarker testing is appropriate for your situation.
Q5: Are anti-inflammatory drugs used to treat diabetes? Some yes — SGLT-2 inhibitors (like empagliflozin) and GLP-1 receptor agonists (like semaglutide) have demonstrated significant anti-inflammatory effects beyond glucose control. Clinical trials of dedicated anti-inflammatory agents (such as canakinumab, an anti-IL-1β antibody) in T2DM have shown promising results and are shaping future treatment guidelines.
Q6: Does the gut microbiome affect diabetes through the immune system? Yes. Gut microbial dysbiosis — an imbalance in intestinal bacterial populations — activates systemic immune responses and promotes insulin resistance and chronic inflammation. The gut-immune-metabolic axis is now considered a critical pathway in T2DM pathogenesis and a genuine therapeutic target.
Author’s Note
As a physician trained in internal medicine and deeply engaged in metabolic research, I have long believed that type 2 diabetes mellitus (T2DM) cannot be fully understood through the lens of glucose alone. Over the past decade, the scientific literature has increasingly revealed that diabetes is not merely a disorder of insulin and carbohydrates — it is a condition rooted in immune–metabolic dysregulation, chronic inflammation, and cellular stress signaling.
This article was written to bridge an important gap: the space between highly technical immunology research and practical clinical understanding. The studies referenced here reflect a growing consensus that insulin resistance, trained immunity, inflammatory cytokine activation, and infection susceptibility are not parallel phenomena — they are biologically intertwined. The immune system does not sit on the sidelines of metabolic disease; it participates actively in shaping its onset, progression, and complications.
Importantly, this discussion does not diminish the central role of lifestyle interventions, pharmacotherapy, or glycemic monitoring. Rather, it expands the framework. When we understand diabetes as a disorder of immune–metabolic homeostasis, new therapeutic possibilities emerge — from anti-inflammatory strategies and biomarker-guided risk assessment to microbiome modulation and precision immunotherapy.
My goal in writing this piece is not to provoke alarm, but to encourage a more integrated view of chronic disease — one grounded in systems biology, translational medicine, and evidence-based care. For clinicians, I hope it stimulates broader diagnostic thinking. For patients, I hope it offers clarity: managing diabetes is not only about lowering sugar — it is about restoring balance across the body’s interconnected metabolic and immune networks.
Medicine evolves. Our frameworks must evolve with it.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
The Fat Inside Your Muscles: More Dangerous Than We Thought? | DR T S DIDWAL
Your Body Fat Is an Endocrine Organ—And Its Hormones Shape Your Heart Health | DR T S DIDWAL
hsCRP Explained: What Inflammation Means for Your Heart | DR T S DIDWAL
References
Alexander, M., Cho, E., Gliozheni, E., Salem, Y., Cheung, J., & Ichii, H. (2024). Pathology of diabetes-induced immune dysfunction. International Journal of Molecular Sciences, 25(13), 7105. https://doi.org/10.3390/ijms25137105
Berbudi, A., Khairani, S., & Tjahjadi, A. I. (2025). Interplay between insulin resistance and immune dysregulation in type 2 diabetes mellitus: Implications for therapeutic interventions. ImmunoTargets and Therapy, 14, 359–382. https://doi.org/10.2147/ITT.S499605
Islam, M. S., Monir, S. B., Haque, N., Vabna, M. A., Fan, J., Li, Y., Nime, I., Feroz, F., Acharjee, M., & Pan, F. (2025). Immunometabolic crossroads: Infections as bidirectional modulators in diabetes and metabolic syndromes. Frontiers in Endocrinology, 16, 1710157. https://doi.org/10.3389/fendo.2025.1710157
Liu, Y., Lei, Y., Dai, Z., Luo, C., Gong, Q., Li, Y., Xu, Y., & Huang, W. (2025). Trained immunity: Novel perspectives in diabetes and associated complications. Frontiers in Immunology, 16, 1613602. https://doi.org/10.3389/fimmu.2025.1613602
Rao, P. P., Mishra, S., Gupta, J., Vyas, M., & Babu, M. R. (2025). Inflammation and immune biomarkers: New frontiers in understanding and managing diabetes complications. Inflammopharmacology, 33(11), 6507–6534. https://doi.org/10.1007/s10787-025-01996-4
Anderson, S., Karlstaedt, A., Young, M. et al. Targeting immunometabolic pathways with AZD1656 alleviates inflammation and metabolic dysfunction in type 2 diabetic cardiomyopathy. Nat Cardiovasc Res 5, 138–154 (2026). https://doi.org/10.1038/s44161-025-00769-0