How to Increase Vagus Nerve Tone to Reduce Chronic Inflammation
How to increase vagus nerve tone to reduce chronic inflammation. Discover research-backed methods to calm immune overactivity naturally
HEART
Dr. T.S. Didwal, M.D.(Internal Medicine)
2/24/202618 min read
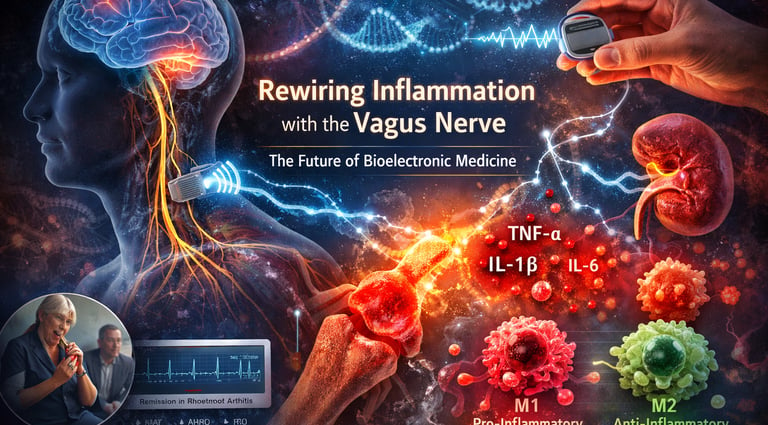
Chronic inflammation is no longer viewed as a background process of disease — it is increasingly recognized as the central driver of modern pathology, linking rheumatoid arthritis, metabolic syndrome, inflammatory bowel disease, neurodegeneration, and even postoperative complications. For decades, medicine has attempted to silence inflammation primarily through pharmacology: corticosteroids, NSAIDs, and biologics targeting TNF-α or IL-6. These agents work — but imperfectly. They suppress immune signaling broadly, carry infection risks, and often fail to induce durable remission. What if, instead of chemically suppressing immunity, we could neurologically recalibrate it?
Emerging research suggests that the answer may lie within the vagus nerve, the longest cranial nerve and the cornerstone of the parasympathetic nervous system. Far from being merely a regulator of heart rate and digestion, the vagus nerve functions as a bidirectional neuroimmune communication network, forming the backbone of what is now termed the inflammatory reflex (Liu et al., 2025). Through the cholinergic anti-inflammatory pathway (CAP), vagal efferent signaling modulates macrophage cytokine release via α7 nicotinic acetylcholine receptor (α7nAChR) activation, suppressing TNF-α, IL-1β, and IL-6 production without global immunosuppression (Han et al., 2026).
Even more compelling, randomized clinical evidence now demonstrates that targeted vagus nerve stimulation (VNS) can significantly reduce disease activity in rheumatoid arthritis patients with inadequate methotrexate response (Tesser et al., 2026). This is the foundation of bioelectronic medicine — a paradigm shift in which electrical neuromodulation, not pharmaceuticals alone, restores immune homeostasis.
The question is no longer whether the nervous system influences inflammation. The question is whether we are ready to treat inflammatory disease through calibrated neural circuitry.
Clinical pearls.
1. The "Brake Pedal" Concept
Scientific Perspective: Vagus nerve stimulation (VNS) activates alpha7nAChR receptors on macrophages, which inhibits the translocation of NF-kappa to the nucleus. This prevents the transcription of pro-inflammatory cytokines, such as TNF-alpha.
Think of your immune system like a car. In diseases like RA, the "gas pedal" is stuck down, causing inflammation. The vagus nerve is your body’s natural brake pedal. VNS therapy helps the body "pump the brakes" to stop the damage without needing to turn the engine off entirely (like some heavy drugs do).
2. Vagal Tone as a Vital Sign
Scientific Perspective: Heart Rate Variability (HRV) serves as a proxy for vagal tone. Low HRV is statistically correlated with high C-Reactive Protein (CRP) and IL-6 levels, marking it as a predictive biomarker for systemic inflammation.
In the future, your "vagal tone" might be as important as your blood pressure. If your vagus nerve is "weak" (low tone), your body stays in a stressed, inflamed state. We can now measure this and use it to see if your treatment is actually working at a nervous-system level.
3. The Spleen: The Immune "Command Center"
Scientific Perspective: The anti-inflammatory effect of the vagus nerve is largely indirect; it signals the celiac ganglion, which then tells the splenic nerve to release norepinephrine, triggering T-cells to produce acetylcholine within the spleen.
Even though we stimulate the nerve in your neck or ear, the "magic" happens in your spleen. The nerve acts like a telephone line, sending a "stand down" order to the command center where your immune cells are being "armed" to fight.
4. Ear vs. Implant: Accessibility of the "Cymba Conchae"
Scientific Perspective: The Cymba Conchae of the external ear is the only place on the human body where the sensory (afferent) branch of the vagus nerve is located directly under the skin, making taVNS a viable alternative to surgery.
You don’t always need surgery to reach this "anti-inflammatory highway." We can reach the nerve through a specific spot in your outer ear. It’s like using a remote control for your immune system instead of having to rewire the house.
5. Breaking the "Stress-Gut-Inflammation" Loop
Scientific Perspective: Chronic stress lowers vagal efferent activity, which increases intestinal permeability ("leaky gut") and allows microbial metabolites to trigger systemic immune responses. Restore the vagus, and you restore the gut barrier.
Stress isn't just "in your head"; it physically weakens your gut's defenses. By stimulating the vagus nerve, we can help "seal" the gut and stop the cycle where stress makes your inflammation worse, and your inflammation makes you feel more stressed.
6. Polarization: From "War" to "Repair"
Scientific Perspective: VNS promotes macrophage polarization, shifting cells from the M1 phenotype (pro-inflammatory/tissue damaging) to the M2 phenotype (anti-inflammatory/tissue-healing).
Your immune cells have two modes: "Soldier" and "Janitor." In chronic illness, you have too many soldiers causing a mess. Vagus stimulation tells those cells to stop fighting and start cleaning up, shifting your body from a state of attack to a state of repair.
What Is the Vagus Nerve, and Why Does It Matter for Inflammation?
The vagus nerve (cranial nerve X) is the longest cranial nerve in the body. Traveling from the brainstem down through the neck, chest, and abdomen, it innervates the heart, lungs, stomach, intestines, liver, and spleen. Approximately 80% of its fibers are afferent — meaning they carry signals to the brain, not away from it — making the vagus nerve a sophisticated sensory highway as much as a motor command line.
Its relevance to immune regulation lies in what researchers call the inflammatory reflex, a neural circuit first described by Kevin Tracey's foundational work in the early 2000s. The reflex works like this: when the brain detects inflammatory signals from peripheral tissues (via vagal afferents), it can send efferent signals back through the vagus nerve to the spleen and other immune organs, instructing immune cells to reduce cytokine production. This circuit — brain detects inflammation, brain sends "stand down" signal through vagus — is the biological engine behind an entire new class of therapies.
Liu et al. (2025), in their comprehensive review published in International Immunopharmacology, describe this as a bidirectional neuro-immune dialogue. The vagus nerve does not merely react; it participates actively in immune surveillance, detecting cytokine levels and modulating macrophage behavior in real time. Understanding this dialogue is the first step toward exploiting it therapeutically.
The Cholinergic Anti-Inflammatory Pathway: Molecules Behind the Magic
At the molecular level, the vagus nerve's anti-inflammatory effects are mediated primarily through the cholinergic anti-inflammatory pathway (CAP). When vagal efferent fibers signal the spleen via the celiac ganglion and splenic nerve, they trigger the release of acetylcholine from a specialized population of T cells that express choline acetyltransferase (ChAT). This acetylcholine binds to alpha-7 nicotinic acetylcholine receptors (α7nAChR) on macrophages, suppressing the production of pro-inflammatory cytokines including TNF-α, IL-1β, IL-6, and IL-18.
Han et al. (2026), writing in Frontiers in Immunology, provide an elegant mechanistic breakdown of this reflex specifically in the context of transcutaneous auricular vagus nerve stimulation (taVNS). Their review confirms that activation of α7nAChR not only reduces cytokine storm activity but also promotes macrophage polarization from the pro-inflammatory M1 phenotype to the anti-inflammatory M2 phenotype. This shift has downstream consequences: reduced tissue damage, decreased oxidative stress, and improved resolution of inflammation.
This molecular mechanism is important for one critical reason — it is targetable. Unlike systemic immunosuppression, which blunts the entire immune response and leaves the patient vulnerable to infection, cholinergic neuromodulation offers a localized and calibrated suppression of excessive inflammatory signaling. The immune system remains functional; only its runaway activity is reined in.
Key Takeaway — Han et al. (2026): Transcutaneous auricular vagus nerve stimulation activates the cholinergic anti-inflammatory pathway via α7nAChR signaling, promoting macrophage polarization from M1 to M2 phenotypes and suppressing TNF-α, IL-1β, and IL-6 across rheumatic, metabolic, and postoperative conditions. The review underscores the mechanistic breadth and translational momentum of taVNS as a non-invasive neuromodulatory approach.
Neuro-Immune Interactions: A Bidirectional Conversation
Liu et al. (2025) frame vagal anti-inflammation within a broader narrative of neuro-immune crosstalk — a recognition that the nervous system and immune system are not parallel but deeply intertwined. Their review in International Immunopharmacology catalogues the various anatomical and molecular pathways through which the vagus nerve shapes immune cell behavior, including direct innervation of lymphoid organs, regulation of the hypothalamic-pituitary-adrenal (HPA) axis, and modulation of gut-associated lymphoid tissue (GALT).
One particularly fascinating observation from this review is the role of vagal tone — the baseline level of vagal activity — as a biomarker for inflammatory status. Low vagal tone has been associated with elevated levels of circulating inflammatory markers such as C-reactive protein (CRP) and IL-6 in multiple population studies. In other words, a quieter vagus nerve correlates with a louder inflammatory voice. Conversely, interventions that increase vagal tone — whether electrical stimulation, mindfulness-based stress reduction, or even aerobic exercise — appear to dampen inflammatory activity across the board.
This bidirectional framing matters clinically. It suggests that vagal health is not just a consequence of disease but potentially a cause. Patients with chronically low vagal tone may be physiologically predisposed to inflammatory dysregulation — a finding with enormous implications for prevention, early intervention, and personalized medicine.
Key Takeaway — Liu et al. (2025): The vagus nerve engages in active, bidirectional neuro-immune communication. Low vagal tone functions as both a consequence and potential driver of systemic inflammation, implicating vagal health as a modifiable risk factor across multiple chronic disease states and positioning vagal monitoring as a meaningful clinical biomarker.
Non-Invasive Vagus Nerve Stimulation: Technology Meets Biology
The idea of using the vagus nerve therapeutically is not new — surgically implanted VNS devices have been approved for epilepsy and treatment-resistant depression since the 1990s. But the last decade has witnessed an explosion of interest in non-invasive alternatives that achieve similar neural effects without the cost, risk, or permanence of surgery.
Liu et al. (2024), in a dedicated review published in Frontiers in Neuroscience, provide a thorough examination of non-invasive vagus nerve stimulation (nVNS) modalities, including transcutaneous cervical VNS (tcVNS) and transcutaneous auricular VNS (taVNS). Both approaches deliver electrical stimulation to vagal nerve branches accessible through the skin — the cervical branch near the neck or the auricular branch through the outer ear — thereby activating the broader vagal network without any surgical implantation.
Their mechanistic analysis reveals that nVNS reduces NF-κB activation, a master regulator of inflammatory gene transcription. By blunting NF-κB signaling in macrophages and monocytes, nVNS effectively turns down the volume on inflammatory gene expression across multiple pathways simultaneously. The review also discusses emerging evidence that nVNS modulates the gut-brain axis, potentially influencing intestinal permeability, microbial composition, and intestinal immune homeostasis — three factors increasingly recognized as systemic inflammation amplifiers.
From a clinical translation perspective, Liu et al. (2024) highlight key challenges remaining: optimal stimulation parameters (frequency, intensity, pulse width, session duration) remain incompletely characterized across different conditions, and the long-term durability of anti-inflammatory effects requires further study in controlled populations.
Key Takeaway — Liu et al. (2024): Non-invasive VNS modalities (tcVNS and taVNS) suppress inflammation by inhibiting NF-κB signaling, reducing cytokine production, and modulating gut-brain axis dynamics. While mechanistic understanding is advancing rapidly, standardization of stimulation protocols remains a primary unresolved challenge for clinical translation.
Clinical Breakthrough: Vagus Nerve Stimulation in Rheumatoid Arthritis
The most headline-worthy development in this field came with a pivotal randomized controlled trial published in Nature Medicine by Tesser et al. (2026). The study enrolled patients with active rheumatoid arthritis (RA) who had an inadequate response to methotrexate, one of the cornerstones of RA pharmacotherapy. Participants were randomized to receive implanted bioelectronic vagus nerve stimulation or a sham procedure over a 12-week period, with DAS28-CRP (a standard RA disease activity score) as the primary outcome.
The results were striking. Active VNS produced statistically significant reductions in disease activity scores compared to sham, with a meaningful proportion of patients achieving clinical remission — a threshold rarely reached with first-line pharmacotherapy alone. Crucially, the biomarker data corroborated the mechanistic pathway: responders showed significant reductions in TNF-α and IL-6 levels, consistent with cholinergic anti-inflammatory pathway activation.
This trial is pivotal not simply because it demonstrated efficacy, but because it did so in a rigorously controlled, double-blind setting using objective biomarker endpoints. It marks the first high-level clinical evidence that intentional, device-mediated vagal stimulation can produce disease-modifying effects in an established autoimmune condition — setting a precedent that will almost certainly drive trials in related conditions, including lupus, Sjögren's syndrome, and inflammatory bowel disease.
Key Takeaway — Tesser et al. (2026): This landmark randomized controlled trial in Nature Medicine demonstrated that implanted vagus nerve stimulation significantly reduced RA disease activity and inflammatory biomarkers (TNF-α, IL-6) in patients with inadequate methotrexate response, providing the strongest clinical evidence to date that bioelectronic neuromodulation can serve as a disease-modifying intervention in autoimmune disease.
The Gut-Brain-Vagus Axis: Microbiota as a Neuroimmune Modulator
No discussion of vagal anti-inflammation would be complete without addressing the gut microbiome — an ecosystem of trillions of microorganisms that profoundly influences both nervous system function and immune tone. Kurhaluk et al. (2025), in a narrative review published in the International Journal of Molecular Sciences, explore the three-way relationship between the parasympathetic nervous system, the vagus nerve, and gut microbial communities under conditions of psychological stress.
Their analysis reveals a compelling feedback architecture. The vagus nerve continuously samples gut microbial metabolites — including short-chain fatty acids (SCFAs) such as butyrate, acetate, and propionate — via mucosal afferents. These metabolites act as molecular messengers, communicating the immunological state of the gut lumen to the central nervous system. In return, vagal efferents regulate gut motility, intestinal barrier integrity, and mucosal immune responses, which in turn shape the microbial environment.
Critically, chronic psychological stress disrupts this equilibrium at multiple levels. Stress reduces vagal tone, elevates glucocorticoid levels, alters gut motility, and shifts the microbiome toward pro-inflammatory dysbiosis — creating a vicious cycle in which stress begets inflammation, and inflammation begets further stress. Kurhaluk et al. (2025) argue that interventions targeting vagal tone — whether pharmacological, behavioral, or neuromodulatory — may interrupt this cycle by simultaneously restoring neural and microbial balance.
This systems-level perspective is increasingly relevant in the context of conditions like irritable bowel syndrome, inflammatory bowel disease, depression with inflammatory comorbidities, and metabolic syndrome — all of which feature disrupted gut-brain-vagal crosstalk as a common thread.
Key Takeaway — Kurhaluk et al. (2025): Chronic psychological stress disrupts the vagus-gut microbiota axis by reducing vagal tone, promoting dysbiosis, and amplifying inflammatory signaling in a self-reinforcing cycle. Interventions targeting vagal activity may restore neuro-microbial balance, offering a systems-level therapeutic approach to stress-related inflammatory conditions.
taVNS Across Disease States: Rheumatic, Metabolic, and Postoperative Applications
Han et al. (2026) extend the clinical landscape considerably by reviewing evidence for taVNS across three broad disease categories: rheumatic disorders, metabolic conditions, and postoperative inflammatory complications. This breadth of application reflects the universal nature of the cholinergic anti-inflammatory pathway, because inflammation is a common feature across all three domains, a mechanism that suppresses inflammatory signaling has the potential to be broadly therapeutic.
In rheumatic disorders, taVNS has shown efficacy in pilot trials for RA, with reductions in joint tenderness, morning stiffness, and circulating inflammatory cytokines. In metabolic disease — particularly type 2 diabetes and non-alcoholic fatty liver disease — taVNS appears to reduce hepatic inflammatory gene expression and improve insulin sensitivity through a combination of vagal and metabolic mechanisms. In the postoperative setting, where systemic inflammatory responses can complicate recovery and contribute to delirium, organ dysfunction, and prolonged ICU stays, taVNS has demonstrated promising early results in reducing postoperative inflammatory burden and supporting faster recovery.
The review by Han et al. also addresses the translational infrastructure needed to move taVNS from bench to bedside: standardized electrode placement protocols, optimal auricular stimulation targets (the cymba conchae, for its particularly rich vagal innervation), waveform characteristics, and patient stratification strategies. This operationalization of taVNS is the bridge between mechanistic understanding and routine clinical use.
Key Takeaway — Han et al. (2026, extended): taVNS demonstrates cross-disease anti-inflammatory efficacy spanning rheumatic, metabolic, and postoperative domains, underscoring the universality of cholinergic pathway modulation. Translational success will depend on developing standardized protocols that account for disease-specific stimulation parameters and patient-level response predictors.
Brief Summary
1️⃣ Inflammation Is the Common Denominator of Modern Disease
Chronic low-grade inflammation now underpins rheumatoid arthritis, metabolic syndrome, type 2 diabetes, inflammatory bowel disease, atherosclerosis, neurodegeneration, and even depression.
For decades, medicine has treated inflammation chemically — with corticosteroids, NSAIDs, biologics, and monoclonal antibodies.
These therapies are powerful but blunt: they suppress immune signaling broadly, increase infection risk, and are not universally effective.
The unmet need is clear: precision immune regulation without global immunosuppression.
2️⃣ The Vagus Nerve: From Parasympathetic Bystander to Immune Regulator
The vagus nerve (cranial nerve X) is the longest cranial nerve, innervating the heart, lungs, gut, liver, and spleen.
Roughly 80% of its fibers are afferent, making it a sensory highway monitoring inflammatory signals in real time.
Through the inflammatory reflex, the brain detects peripheral cytokines and dispatches efferent signals to suppress excessive immune activation.
This discovery reframes inflammation as a neuroimmune circuit, not merely a molecular cascade.
3️⃣ The Cholinergic Anti-Inflammatory Pathway (CAP): Precision Control
Vagal efferents influence splenic immune cells via acetylcholine signaling.
Activation of α7 nicotinic acetylcholine receptors (α7nAChR) on macrophages suppresses TNF-α, IL-1β, and IL-6 production.
Unlike systemic immunosuppression, CAP modulation is calibrated — reducing excessive cytokine release while preserving host defense.
This is not immune shutdown; it is immune recalibration.
4️⃣ Bioelectronic Medicine: The Rise of Device-Based Immunomodulation
Vagus nerve stimulation (VNS) has been used for decades in epilepsy and depression.
Now, invasive and non-invasive modalities (tcVNS, taVNS) are being explored for inflammatory disease.
Randomized clinical evidence in rheumatoid arthritis demonstrates meaningful reductions in disease activity and inflammatory biomarkers in methotrexate-inadequate responders.
Electrical neuromodulation is emerging as a disease-modifying strategy, not merely symptomatic relief.
5️⃣ Systems Biology: The Gut–Brain–Immune Axis
The vagus nerve continuously samples gut microbial metabolites such as short-chain fatty acids.
Vagal tone influences intestinal barrier integrity, motility, and mucosal immune balance.
Chronic stress lowers vagal tone, promotes dysbiosis, and amplifies systemic inflammation.
Thus, inflammation is not only immunologic — it is neural, microbial, and behavioral.
6️⃣ Why This Paradigm Matters
Pharmacology modifies molecules; neuromodulation modifies regulatory circuits.
Electrical stimulation parameters can be adjusted in real time: frequency, pulse width, and intensity.
Future closed-loop systems may monitor inflammatory biomarkers and auto-adjust stimulation to maintain homeostasis.
This represents a shift from reactive treatment to dynamic physiologic control
.
7️⃣ Caution and Clinical Responsibility
VNS is not a universal replacement for biologics or DMARDs.
Long-term durability, optimal patient selection, and stimulation protocols require rigorous trials.
Overenthusiasm without evidence risks undermining credibility in a rapidly evolving field.
8️⃣ The Broader Implication
Inflammation is not merely a biochemical storm — it is partly a misfiring neural reflex.
If neural circuits can amplify inflammation, they can also restrain it.
The nervous system may be the most underutilized therapeutic organ in modern medicine.
We stand at a transition point in medicine. The future of inflammatory disease management may not depend solely on discovering the next biologic molecule, but on understanding — and ethically harnessing — the electrical language of the body itself.
The vagus nerve is no longer just anatomy. It is a therapeutic frontier.
Key Takeaways from Each Study
Liu, L. et al. (2025) — International Immunopharmacology: The vagus nerve participates in real-time, bidirectional neuro-immune communication. Low vagal tone correlates with elevated inflammatory markers, positioning vagal tone as both a disease risk factor and a clinically measurable intervention target.
Han, R. et al. (2026) — Frontiers in Immunology: taVNS activates the cholinergic anti-inflammatory pathway, driving macrophage polarization from M1 to M2 and suppressing multiple pro-inflammatory cytokines. Its efficacy spans rheumatic, metabolic, and postoperative inflammatory disorders, with translational protocols under active refinement.
Liu, F-J. et al. (2024) — Frontiers in Neuroscience: Non-invasive VNS modalities suppress inflammation via NF-κB inhibition and gut-brain axis modulation. Mechanistic clarity is advancing, but optimal stimulation parameters across disease states remain an open research question requiring standardized clinical trials.
Tesser, J.R.P. et al. (2026) — Nature Medicine: A pivotal randomized controlled trial confirmed that implanted vagus nerve stimulation significantly reduces RA disease activity and associated biomarkers in methotrexate-inadequate responders, establishing the first high-level clinical proof-of-concept for bioelectronic anti-inflammatory therapy in autoimmune disease.
Kurhaluk, N. et al. (2025) — International Journal of Molecular Sciences: Chronic psychological stress disrupts the vagus-gut microbiota axis through a vicious cycle of reduced vagal tone, dysbiosis, and amplified inflammation. Vagal-targeted interventions offer a systems-level approach to breaking this cycle in stress-mediated inflammatory conditions.
Frequently Asked Questions (FAQs)
1. What is the vagus nerve's primary role in inflammation? The vagus nerve serves as the neural backbone of the inflammatory reflex, a circuit through which the brain detects peripheral inflammatory signals and dispatches anti-inflammatory commands via cholinergic pathways. When activated, vagal efferents suppress macrophage production of TNF-α, IL-1β, and IL-6 through alpha-7 nicotinic acetylcholine receptor signaling in the spleen and other immune organs. This mechanism makes the vagus nerve a natural, built-in brake on runaway inflammatory activity.
2. What is vagus nerve stimulation (VNS), and how does it work? Vagus nerve stimulation involves delivering controlled electrical impulses to the vagus nerve — either via a surgically implanted device or, increasingly, through non-invasive skin-surface electrodes applied at the neck (tcVNS) or the outer ear (taVNS). These impulses activate vagal afferent and efferent fibers, triggering the cholinergic anti-inflammatory pathway and suppressing pro-inflammatory cytokine production. The technique essentially "turns up the volume" on the body's existing anti-inflammatory neural circuitry.
3. Is vagus nerve stimulation safe? Generally, VNS has a well-established safety profile. Non-invasive forms (tcVNS and taVNS) carry minimal risk and are well tolerated in trials, with side effects typically limited to mild local skin discomfort at the electrode site. Implanted VNS devices carry the risks associated with any surgical procedure, including infection and device-related complications, but have been used safely for decades in epilepsy patients. The clinical trials reviewed here reported no serious adverse events attributable to the stimulation itself.
4. Can vagus nerve stimulation replace medications for conditions like rheumatoid arthritis? Based on current evidence, VNS is best understood as a complementary or adjunct therapy rather than a replacement for established treatments. The Tesser et al. (2026) trial enrolled patients who had inadequate responses to methotrexate, suggesting VNS's most immediate clinical utility may be in augmenting partial pharmaceutical responses. Whether VNS can serve as first-line monotherapy in appropriate patients remains an important open question requiring further trials.
5. How does the gut microbiome connect to the vagus nerve and inflammation? The gut microbiome influences vagal activity through metabolites — particularly short-chain fatty acids like butyrate — that are sampled by vagal afferents in the gut wall. Healthy microbial communities tend to support vagal tone and anti-inflammatory signaling, while dysbiosis (microbial imbalance) can reduce vagal activity and amplify inflammatory responses. Chronic stress worsens this dynamic by simultaneously lowering vagal tone and shifting microbial composition toward pro-inflammatory species, creating a cycle that vagal-targeted interventions may be able to interrupt.
6. What conditions might benefit from vagus nerve stimulation in the future? Given the universal role of the cholinergic anti-inflammatory pathway, the range of conditions that might benefit is extensive. Active or emerging areas of investigation include: rheumatoid arthritis, inflammatory bowel disease (Crohn's disease and ulcerative colitis), lupus, type 2 diabetes, non-alcoholic fatty liver disease, post-surgical inflammatory complications, sepsis, long COVID, and neuroinflammatory conditions such as multiple sclerosis. The common denominator is dysregulated systemic inflammation, which VNS may help recalibrate regardless of the specific organ system involved.
7. What does "bioelectronic medicine" mean, and is it accessible today? Bioelectronic medicine refers to the use of electrical devices that modulate neural circuits to treat disease — a field in which VNS is currently the most clinically advanced application. Implanted VNS devices are already approved and commercially available for epilepsy, depression, and, in some regulatory jurisdictions, inflammatory conditions. Non-invasive taVNS devices are in late-stage clinical development and available in research settings. While the full promise of personalized, closed-loop neuromodulation is still on the horizon, the foundational technology is real, regulated, and rapidly evolving.
Roadmap for increasing vagal tone to combat chronic inflammation.
1. Electrical Neuromodulation (The "Direct" Route)
The most potent way to increase vagal tone is through Vagus Nerve Stimulation (VNS). As of 2026, we categorize these into two main methods:
Transcutaneous Auricular VNS (taVNS): This non-invasive method targets the Cymba Conchae of the ear. Research by Han et al. (2026) confirms that stimulating this area activates the cholinergic anti-inflammatory pathway, shifting macrophages from a "war" (M1) to a "repair" (M2) state.
Transcutaneous Cervical VNS (tcVNS): A handheld device applied to the side of the neck. It inhibits NF-kappa, the "master switch" for inflammatory genes.
2. Respiratory Biofeedback: The "Breathing" Brake
You can manually "hack" your vagus nerve through your lungs. Because the vagus nerve innervates the diaphragm, specific breathing patterns increase Heart Rate Variability (HRV), a direct proxy for vagal tone.
Resonant Frequency Breathing: Breathing at a rate of approximately 5.5 to 6 breaths per minute.
The Technique: Inhale for 4 seconds, exhale for 6 seconds. The elongated exhale signals the brainstem to increase parasympathetic (vagal) outflow, immediately lowering systemic inflammatory markers like IL-6.
3. The Gut-Microbiome Axis
As noted by Kurhaluk et al. (2025), the vagus nerve "tastes" the chemicals produced by your gut bacteria. To increase vagal tone through the gut:
Short-Chain Fatty Acids (SCFAs): Increase intake of prebiotic fibers (inulin, chicory, acacia). Bacteria ferment these into butyrate, which binds to vagal afferent receptors, sending anti-inflammatory signals to the brain.
Targeted Probiotics: Certain "psychobiotics" (e.g., L. rhamnosus) have been shown to modulate GABA receptors in the brain specifically via the vagus nerve.
4. Physical & Thermal Hormesis
Extreme temperatures and physical movement act as "exercises" for the vagus nerve.
Cold Water Immersion: Splashing freezing water on the face or taking a cold shower triggers the "Dive Reflex." This causes an immediate spike in vagal activity to slow the heart rate and stabilize the system.
Aerobic Zone 2 Training: Consistent, low-intensity steady-state exercise improves baseline vagal tone, making the "inflammatory reflex" more sensitive to small spikes in cytokines.
5. Mindfulness & The HPA Axis
Chronic stress creates a "leaky" vagus nerve. When the Hypothalamic-Pituitary-Adrenal (HPA) axis is overactive (high cortisol), it suppresses the vagus.
Vagus-Specific Meditation: Practices like Loving-Kindness Meditation (LKM) have been clinically shown to increase HRV more effectively than standard mindfulness, likely by activating the "social engagement" portion of the vagus nerve complex.
Author’s Note
As a physician trained in internal medicine and deeply engaged in metabolic and inflammatory research, I have long been struck by a recurring pattern: we often treat inflammation as an isolated biochemical event rather than as a systems-level phenomenon. The more one studies immunology, neurophysiology, and metabolism together, the clearer it becomes that inflammation is not merely a molecular cascade — it is a networked response shaped by neural input, endocrine signaling, microbial ecology, and behavioral stress.
The science of the vagus nerve represents a pivotal shift in how we conceptualize immune regulation. What was once considered primarily a parasympathetic conduit for heart rate and digestion is now understood as a central regulator of inflammatory tone through the inflammatory reflex and the cholinergic anti-inflammatory pathway. This reframing challenges the traditional drug-centric model of immunomodulation and opens the door to bioelectronic medicine — an approach that leverages neural circuitry rather than systemic pharmacologic suppression.
This article is written not to sensationalize neuromodulation, but to contextualize it within rigorous mechanistic science and emerging randomized clinical evidence. Vagus nerve stimulation is not a miracle cure. It is, however, a scientifically grounded therapeutic frontier that demands thoughtful investigation, cautious optimism, and disciplined clinical translation.
If there is one central message, it is this: the nervous system is not merely a bystander in inflammatory disease. It is an active participant — and potentially, a powerful therapeutic ally.
Medical Disclaimer
The information in this article, including the research findings, is for educational purposes only and does not constitute medical advice, diagnosis, or treatment.
Related Articles
Metabolic Plasticity: Epigenetic Adaptations to Calorie Restriction | DR T S DIDWAL
References
Han, R., Peng, Z., Zhuo, M., Song, Y., Liu, Y., Zhang, X., Deng, Z., Xia, L., & Zhong, M. L. (2026). Cholinergic reflex control of inflammation: mechanistic and translational advances in transcutaneous auricular vagus nerve stimulation across rheumatic, metabolic, and postoperative disorders. Frontiers in Immunology, 16, 1702185. https://doi.org/10.3389/fimmu.2025.1702185
Kurhaluk, N., Kołodziejska, R., Kamiński, P., & Tkaczenko, H. (2025). Integrative neuroimmune role of the parasympathetic nervous system, vagus nerve and gut microbiota in stress modulation: A narrative review. International Journal of Molecular Sciences, 26(23), 11706. https://doi.org/10.3390/ijms262311706
Liu, F-J., Wu, J., Gong, L-J., Yang, H-S., & Chen, H. (2024). Non-invasive vagus nerve stimulation in anti-inflammatory therapy: Mechanistic insights and future perspectives. Frontiers in Neuroscience, 18, 1490300. https://doi.org/10.3389/fnins.2024.1490300
Liu, L., Lou,Liu, L., Lou, S., Fu, D., Ji, P., Xia, P., Shuang, S., Dong, W., Yuan, X., Wang, J., Xie, K., Wang, D., & Shen, R. (2025). Neuro-immune interactions: Exploring the anti-inflammatory role of the vagus nerve. International Immunopharmacology, 159, 114941. https://doi.org/10.1016/j.intimp.2025.114941
Tesser, J. R. P., Crowley, A. R., Box, E. J., et al. (2026). Vagus nerve-mediated neuroimmune modulation for rheumatoid arthritis: A pivotal randomized controlled trial. Nature Medicine, 32, 369–378. https://doi.org/10.1038/s41591-025-04114-7