Does Muscle Loss Cause Cognitive Decline? What the Latest Science Says
Is Sarcopenia linked to dementia? Evidence from meta-analyses and genetic studies suggests a strong connection. Learn the facts.
SARCOPENIA
Dr. T.S. Didwal, M.D.(Internal Medicine)
5/2/202614 min read
Yes. Multiple 2025–2026 studies, including cohort analyses and Mendelian randomization, show that sarcopenia is associated with a 2–3× higher risk of cognitive impairment, likely mediated through inflammation, insulin resistance, and reduced myokine signaling.
Clinician’s Perspective (Key Points)
Screen early, not just in geriatrics: Begin functional screening for Sarcopenia in midlife (≥40 years), especially in patients with metabolic risk factors or sedentary lifestyles.
Use simple, scalable tools: Grip strength, gait speed, and chair-stand tests are practical bedside measures; escalate to DXA/BIA when diagnosis is uncertain.
Think bidirectional risk: When patients present with Cognitive Impairment, evaluate muscle status; conversely, sarcopenia should prompt cognitive screening.
Prioritize resistance training as therapy: Prescribe progressive resistance exercise 2–3×/week as a first-line, disease-modifying intervention with dual muscle–brain benefits.
Optimise protein intake strategically: Target 1.2–1.6 g/kg/day, distributed across meals; emphasise leucine-rich sources and post-exercise intake.
Address metabolic drivers: Screen for and treat Insulin Resistance, vitamin D deficiency, and chronic inflammation, which accelerate both muscle loss and neurodegeneration.
Stratify high-risk groups: Patients with Parkinson’s disease, diabetes, frailty, or recent hospitalization require proactive muscle and cognitive surveillance
Adopt multidisciplinary care: Integrate physiotherapy, nutrition, and cognitive assessment; align goals with patient function and adherence.
Bottom Line
Sarcopenia is not just a muscle disorder—it is a systemic aging process linked to cognitive decline. Emerging evidence suggests that preserving muscle mass through resistance training, adequate protein intake, and metabolic health may be a key strategy for protecting long-term brain function.
Key Medical Concepts Linked in This Article:
Sarcopenia
Cognitive Impairment
Alzheimer’s Disease
Insulin Resistance
Chronic Inflammation
The Surprising Brain–Muscle Connection You Cannot Afford to Ignore
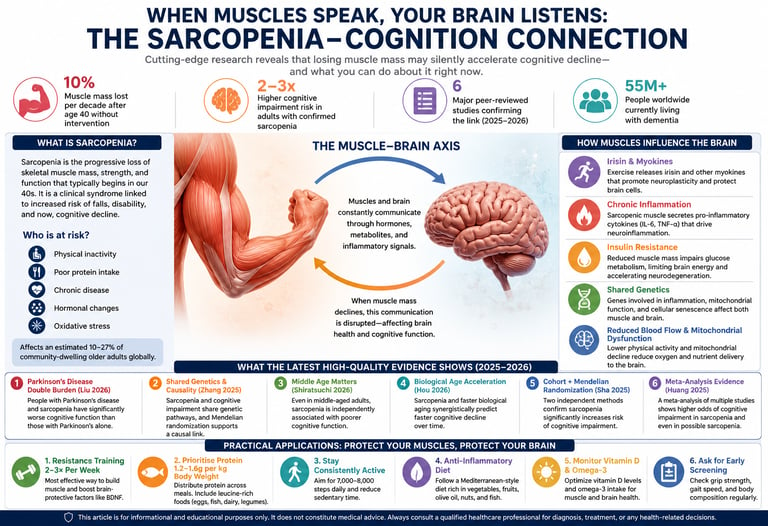
Sarcopenia—the progressive decline in skeletal muscle mass, strength, and function—is increasingly recognized as a contributor to Cognitive Impairment, rather than merely a consequence of aging. Recent high-quality evidence (2025–2026), including a meta-analysis, longitudinal cohort studies, and Mendelian randomization analyses, indicates that individuals with sarcopenia have approximately 2–3-fold higher risk of cognitive impairment (Huang et al., 2025;; Hou et al., 2026) Importantly, causal inference approaches suggest this relationship may extend beyond association (Zhang et al., 2025; Sha et al., 2025)
Biological mechanisms linking muscle and brain include reduced myokine signaling (e.g., irisin), Chronic Inflammation, Insulin Resistance, mitochondrial dysfunction, and impaired cerebral perfusion—shared pathways that accelerate neurodegeneration (Zhang et al., 2025; Hou et al., 2026). Notably, emerging data show associations even in middle-aged populations(Shiratsuchi et al., 2026) and amplified risk in neurological conditions such as Parkinson’s disease (Liu et al., 2026).
Crucially, interventional evidence strengthens the clinical relevance: systematic reviews and meta-analyses demonstrate that structured exercise—particularly resistance and multimodal training—can significantly improve cognitive function, memory, and executive performance in older adults (Choe et al., 2026; Singh et al., 2025). From a prevention standpoint, resistance training (2–3×/week), adequate protein intake (1.2–1.6 g/kg/day), and early functional screening represent key, evidence-based strategies to preserve both muscle and long-term brain health.
~10% Muscle mass lost per decade after age 40 without intervention
2–3× Higher cognitive impairment risk in adults with confirmed sarcopenia
6 Major peer-reviewed studies confirming the link (2025–2026)
55M+ People worldwide currently living with dementia
What the Science Actually Says: Six Pivotal Studies Decoded
1. Sarcopenia + Parkinson's Disease: A Cognitive Double Burden
Parkinson's disease already challenges motor and cognitive function. Liu and colleagues (2026) conducted a cross-sectional study examining whether sarcopenia compounds cognitive problems in Parkinson's patients — and the findings were striking. Patients with both Parkinson's disease and sarcopenia showed significantly poorer performance on cognitive assessments compared to those with Parkinson's alone. This suggests sarcopenia is not merely a bystander in neurological disease — it may amplify cognitive vulnerability through shared inflammatory and metabolic disruptions (Liu et al., 2026).
2. Shared Genetics: The Chicken-and-Egg Question Answered?
One of the most sophisticated contributions to this field in recent years comes from Zhang and colleagues (2025), published in Frontiers in Aging Neuroscience. Their multidimensional study did three things simultaneously: examined clinical associations, investigated shared genetic architecture, and used Mendelian randomization — a technique that mimics a randomized trial using genetic data — to test causality. The results were compelling: sarcopenia and cognitive impairment share common genetic pathways, and the Mendelian randomization analysis supported a causal relationship rather than mere coincidence. This study moves the conversation from correlation to causation, a critical step in scientific credibility (Zhang et al., 2025).
3. Middle Age Is Not Too Early to Worry
Cognitive decline and muscle loss are usually framed as problems of old age. Shiratsuchi and colleagues (2026) challenged this assumption directly by studying middle-aged adults (not elderly) in a community setting using the updated AWGS 2025 sarcopenia criteria. Their findings revealed that even in this relatively younger group, sarcopenia was independently associated with poorer cognitive function — calling for earlier screening and intervention far before traditional geriatric timelines (Shiratsuchi et al., 2026).
4. Biological Age Acceleration: A Ticking Clock
Hou and colleagues (2026) took a uniquely sophisticated approach in their large longitudinal study published in BMC Public Health. Instead of measuring chronological age, they examined biological age acceleration — how fast a person's body is aging relative to their years — alongside sarcopenia, and tracked cognitive decline over time. Their findings revealed that sarcopenia and biological age acceleration work synergistically to speed up cognitive decline. In other words, muscle loss does not just correlate with a snapshot of poor cognition; it predicts a faster future trajectory of brain deterioration (Hou et al., 2026).
5. Cohort Data + Mendelian Randomization: Two Methods, One Conclusion
Sha and colleagues (2025) published in JMIR Aging a rigorous dual-method study combining a prospective cohort study (tracking real people over time) with Mendelian randomization analysis. Both methods pointed in the same direction: sarcopenia significantly increases the risk of future cognitive impairment. The consistency across two independent analytical approaches strengthens confidence in the causal inference beyond what any single study design could achieve alone (Sha et al., 2025).
6. The Definitive Overview: Systematic Review and Meta-Analysis
When individual studies agree, the most powerful scientific tool is a meta-analysis — a statistical aggregation of all available evidence. Huang and colleagues (2025) published exactly this in PLOS ONE, systematically reviewing and pooling data on sarcopenia, possible sarcopenia, and cognitive impairment across multiple studies and populations. Their meta-analysis confirmed a robust and significant association: individuals with sarcopenia had meaningfully higher odds of cognitive impairment compared to those without. Critically, even possible sarcopenia — a subclinical, early-stage condition — was associated with increased cognitive risk, underscoring the importance of catching this process early (Huang et al., 2025).
Why Do Muscles Affect the Brain? The Biological Pathways
Understanding the mechanisms transforms this from an abstract statistical association into a biologically coherent story — and helps motivate real behavioural change.
Irisin — The Exercise Hormone
Muscles release a protein called irisin during exercise that crosses the blood-brain barrier and promotes neuroplasticity. Sarcopenia reduces irisin production, potentially depriving the brain of this neuroprotective signal.
Chronic Systemic Inflammation
Sarcopenic muscle is a source of pro-inflammatory cytokines (IL-6, TNF-α). These inflammatory molecules cross into the brain and promote neuroinflammation, a key driver of Alzheimer's and vascular dementia.
Insulin Resistance
Skeletal muscle is the body's primary site of glucose disposal. As muscle mass falls, insulin resistance rises — reducing the brain's ability to use glucose efficiently and accelerating neurodegeneration.
Shared Genetic Architecture
Zhang et al. (2025) found that genes involved in inflammation, mitochondrial function, and cellular senescence are jointly implicated in both muscle loss and cognitive decline — the same genetic "fault lines" affect both organs.
Reduced Cerebral Blood Flow
Lower physical activity in sarcopenic individuals reduces cerebrovascular fitness, diminishing oxygen and nutrient delivery to the brain, particularly affecting regions associated with memory and executive function.
Mitochondrial Dysfunction
Both neurons and muscle cells rely heavily on mitochondria. Sarcopenia and cognitive impairment share impaired mitochondrial energetics as a common pathological thread, amplifying cellular aging in both tissues simultaneously.
Who Is Most Vulnerable? Recognising the Warning Signs
Sarcopenia does not announce itself with a dramatic symptom. It creeps in gradually. The following changes may warrant a conversation with your healthcare provider:
Difficulty rising from a chair without using your arms
Grip strength that feels noticeably weaker than it used to
Walking slower than peers of your age
Unexplained weight loss with visible muscle thinning in arms or legs
Increasing fatigue with daily activities that were once effortless
Subtle memory lapses, brain fog, or difficulty concentrating
Important NoteExperiencing these symptoms does not confirm sarcopenia or cognitive impairment. Many conditions cause similar presentations. A proper clinical evaluation — including grip strength testing, gait speed measurement, and body composition assessment — is needed for diagnosis. Always consult a qualified healthcare professional.
Practical Applications: Protect Your Muscles, Protect Your Brain
These evidence-aligned strategies are supported by the studies reviewed — always discuss with your doctor before starting a new health programme.
Resistance Training 2–3× Per Week
Progressive resistance exercise is the single most effective intervention against sarcopenia. Even light resistance bands or bodyweight exercises improve muscle mass and simultaneously elevate BDNF — a brain growth factor.
Prioritise Protein: 1.2–1.6g per kg Body Weight
Distribute protein evenly across meals. Leucine-rich sources (eggs, fish, legumes, dairy) maximally stimulate muscle protein synthesis. Protein timing after exercise enhances absorption.
Stay Consistently Active
The longitudinal data from Hou et al. (2026) shows that physical activity buffers biological age acceleration. Even 7,000–8,000 steps daily reduces sarcopenia risk meaningfully.
Anti-Inflammatory Diet
The Mediterranean dietary pattern — rich in olive oil, fish, vegetables, nuts, and whole grains — reduces systemic inflammation, addressing a key shared pathway between sarcopenia and cognitive decline.
Monitor Vitamin D & Omega-3
Vitamin D deficiency accelerates both muscle loss and neuroinflammation. Omega-3 fatty acids (from oily fish or supplements) support both muscle protein synthesis and neuronal membrane integrity.
Ask for Early Screening
Especially if you have Parkinson's disease or other neurological conditions (Liu et al., 2026), proactively request grip strength and gait speed assessment. Catching sarcopenia early preserves cognitive reserve.
Special Considerations: Parkinson's Disease and Neurological Conditions
For individuals already living with Parkinson's disease, the research from Liu et al. (2026) carries particular urgency. Parkinson's itself disrupts motor pathways and, in many patients, accelerates cognitive decline into Parkinson's disease dementia. The addition of sarcopenia to this clinical picture appears to create a compounding burden — and clinicians managing Parkinson's patients may need to integrate muscle health assessment as a routine part of cognitive monitoring.
This same principle likely extends to other neurological conditions. Multiple sclerosis, post-stroke recovery, and traumatic brain injury all alter muscle function through reduced mobility and systemic inflammation. Proactively managing muscle health in these populations may offer a meaningful lever for slowing cognitive deterioration — an area ripe for future clinical trials.
For Carers and Family MembersIf you care for an older adult with declining muscle strength, consider discussing a multidisciplinary approach with their GP or geriatrician — combining a physiotherapist (for exercise prescription), a dietitian (for protein optimisation), and regular cognitive screening. The evidence reviewed here supports this integrated model.
Gaps in Current Research and What Comes Next
Science is never finished, and intellectual honesty requires acknowledging what we do not yet know. Most studies reviewed here — including the cross-sectional designs of Liu et al. (2026) and Shiratsuchi et al. (2026) — capture a single point in time, making it difficult to determine which came first: the muscle loss or the cognitive decline. While Mendelian randomization studies (Zhang et al., 2025; Sha et al., 2025) and the longitudinal work of Hou et al. (2026) provide stronger causal evidence, large-scale, randomised controlled trials are still needed to confirm that treating sarcopenia directly reduces dementia incidence.
Additionally, most studies have been conducted predominantly in East Asian populations — the AWGS criteria reflect this — raising questions about generalisability to South Asian, African, and Western European populations with differing body composition norms. Diverse population research is urgently needed.
Frequently Asked Questions
What exactly is sarcopenia and how is it diagnosed?
Sarcopenia is a progressive skeletal muscle disorder characterised by low muscle mass combined with either low muscle strength or low physical performance. The most current diagnostic standard — the Asian Working Group for Sarcopenia (AWGS) 2025 criteria — uses grip strength (measured with a dynamometer), a five-times sit-to-stand test, and muscle mass estimated by DEXA scan or bioimpedance analysis. A GP or geriatrician can arrange these assessments. Shiratsuchi et al. (2026) specifically used AWGS 2025 criteria and found meaningful associations even in middle-aged adults, suggesting screening should not wait until old age.
Does sarcopenia directly cause cognitive decline, or are they just correlated?
The evidence is moving strongly toward causality, though the picture is not yet completely definitive. Two independent studies — Zhang et al. (2025) in Frontiers in Aging Neuroscience and Sha et al. (2025) in JMIR Aging — both used Mendelian randomization, a method that mimics a randomised trial using genetic data. Both found evidence supporting a causal link, not mere association. Shared genetic pathways involving inflammation, mitochondrial dysfunction, and cellular senescence appear to drive both conditions simultaneously. The meta-analysis by Huang et al. (2025) further confirms that the association is robust across multiple study designs and populations.
At what age should I start worrying about sarcopenia and its cognitive effects?
Earlier than most people think. Muscle mass peaks in our late 20s to early 30s and begins declining by around 1–2% per year from the 4th decade onward. Shiratsuchi et al. (2026) — one of the first major studies to apply AWGS 2025 criteria in middle-aged adults — found cognitive associations even in this younger group. This does not mean you should panic in your 40s, but it does mean the best time to invest in resistance training and a protein-rich diet is before significant loss has occurred — ideally starting in your 30s or 40s. Prevention is far more effective than rehabilitation.
Can reversing sarcopenia improve cognitive function?
This is the critical question that existing research has not yet fully answered. Current studies establish that sarcopenia is associated with worse cognition and predicts faster decline (Hou et al., 2026), but randomised controlled trials specifically testing whether treating sarcopenia improves cognitive outcomes are still limited. However, the biology is highly encouraging: resistance training raises irisin, BDNF, and cerebral blood flow; reduces systemic inflammation; and improves insulin sensitivity — all mechanisms that benefit the brain. Evidence from exercise intervention trials in older adults consistently shows cognitive benefits, suggesting that reversing sarcopenia through structured exercise and nutrition may well protect cognition, even if definitive proof awaits.
Does this apply to people with Parkinson's disease or other neurological conditions?
Yes — and perhaps especially so. Liu et al. (2026) found that in Parkinson's disease patients, the coexistence of sarcopenia was associated with significantly lower cognitive function compared to Parkinson's patients without sarcopenia. This creates a compounding double burden. If you or a loved one has Parkinson's, stroke, multiple sclerosis, or another neurological condition that limits mobility, proactively discussing sarcopenia screening and management with a neurologist or geriatrician is particularly worthwhile. Physiotherapy, adapted exercise programmes, and protein-optimised nutrition remain relevant even in these more complex clinical contexts.
What dietary changes are most evidence-supported for preventing sarcopenia and protecting cognition?
The most consistently supported dietary approach combines adequate protein intake (1.2–1.6g per kilogram of body weight per day, distributed evenly across meals), omega-3 fatty acids (from oily fish like salmon, mackerel, or sardines, or high-quality supplements), vitamin D (optimised through sun exposure and supplementation if deficient), and an overall anti-inflammatory dietary pattern such as the Mediterranean diet. The shared inflammatory pathways identified by Zhang et al. (2025) and the longitudinal data from Hou et al. (2026) both point to chronic inflammation as a key modifiable driver — and diet is one of the most powerful anti-inflammatory tools available without a prescription.
Are there any medications or supplements proven to prevent sarcopenia-related cognitive decline?
Currently, no single medication has regulatory approval specifically for preventing sarcopenia-related cognitive decline. Creatine monohydrate shows modest benefits for muscle mass in combination with resistance training. Vitamin D supplementation is strongly recommended for those who are deficient. Leucine-enriched protein supplements (including whey protein) may augment muscle protein synthesis in older adults who struggle to meet dietary protein needs from food alone. Some research explores the potential of myostatin inhibitors and testosterone therapy for severe sarcopenia, but these are not standard treatments. Always discuss any supplementation with a qualified healthcare professional, as individual needs, contraindications, and interactions vary significantly.
Clinical Pearls
1. The "Muscle-Brain" Secretion Pearl
Skeletal muscle functions as an endocrine organ, secreting myokines such as irisin during contraction. Irisin crosses the blood-brain barrier to upregulate Brain-Derived Neurotrophic Factor (BDNF), which is essential for hippocampal neurogenesis and synaptic plasticity.
Think of your muscles as a "pharmacy" for your brain. When you exercise, your muscles release natural chemicals that travel to your head and act like "brain fertilizer," helping your brain grow new connections and stay sharp.
2. The "Anabolic Resistance" Pearl
Aging is characterized by anabolic resistance, where the muscle's protein synthetic response to dietary amino acids is blunted. To overcome this, the leucine threshold must be met; clinical evidence suggests a minimum of 25–30g of high-quality protein per meal is required to stimulate maximal muscle protein synthesis in older adults.
:As we get older, our bodies become a bit "deaf" to the protein we eat. To make sure your muscles actually hear the message to stay strong, you can't just snack on protein. You need a solid "dose" of it—roughly the amount in a large chicken breast or a cup of Greek yogurt—at every meal.
3. The "Inflamm-aging" Pearl
Sarcopenia promotes a state of chronic systemic inflammation (elevated IL-6 and TNF-α). These pro-inflammatory cytokines can compromise the blood-brain barrier, leading to neuroinflammation, which is a primary driver in the progression of Alzheimer’s and other neurodegenerative diseases.
Weak, wasting muscles aren't just quiet; they get "cranky" and release inflammation into your bloodstream. This inflammation can leak into your brain, causing "brain fog" and speeding up memory loss. Keeping muscle mass keeps the "fire" of inflammation down.
4. The "Metabolic Sink" Pearl
Skeletal muscle is the primary site for glucose disposal (approx. 80% of postprandial glucose). Sarcopenia reduces the available "metabolic sink," leading to insulin resistance. Brain insulin resistance, in turn, impairs neuronal glucose metabolism and accelerates the accumulation of amyloid plaques.
Your muscles are like a giant sponge that soaks up sugar from your blood. If that sponge gets too small (muscle loss), the sugar stays in your system and causes damage. Since your brain also needs to process sugar to think, a "clogged" system makes it much harder for your brain cells to get the energy they need.
5. The "Grip Strength" Screening Pearl
Handgrip strength is a highly validated proxy for global muscular quality and is strongly correlated with executive function. A decline in grip strength often precedes clinical cognitive impairment, serving as a "biomarker" for the integrity of the central nervous system.
The strength of your handshake is a window into the health of your brain. If you find it’s getting harder to open jars or carry groceries, it’s a warning sign that your brain might be at risk, too. Testing your grip is one of the easiest ways for a doctor to check your "biological age."
6. The "Velocity over Volume" Pearl
While hypertrophy (muscle size) is important, muscle power (force x velocity) declines faster than muscle strength with age and has a stronger association with functional independence and cognitive processing speed. Training should include a high-velocity component to target Type II (fast-twitch) muscle fibers.
It’s not just about how much you can lift; it’s about how fast you can move. Doing movements with a bit of "snap"—like standing up quickly from a chair—specifically trains the "fast" muscles that are most closely linked to how quickly your brain processes information.
Author’s Note
This article synthesizes emerging evidence on the relationship between Sarcopenia and Cognitive Impairment, drawing on recent peer-reviewed studies (2025–2026), including meta-analyses, longitudinal cohorts, and Mendelian randomization research. While the convergence of epidemiological, genetic, and mechanistic data strengthens the hypothesis of a causal muscle–brain connection, it is important to recognize that this field is still evolving.
Many of the biological pathways discussed—such as Chronic Inflammation, Insulin Resistance, and myokine signaling—are supported by experimental and clinical evidence but remain areas of active investigation. Additionally, most current data are derived from observational and genetic inference studies; large-scale randomized controlled trials are still needed to confirm whether targeted treatment of sarcopenia can directly reduce the incidence or progression of cognitive decline.
Clinicians and readers are encouraged to interpret these findings within the broader context of individualized patient care. The recommendations provided reflect current best evidence but should complement—not replace—clinical judgment, patient preferences, and local practice guidelines.
As research advances, our understanding of the muscle–brain axis will continue to refine prevention and treatment strategies in both metabolic and neurodegenerative health.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and exercise decisions should always be made in consultation with qualified healthcare professionals.
.Related Articles
The Neurobiology of Fitness: How Aerobic Capacity Shapes Neuroplasticity and Brain Aging
The Science of Healthy Brain Aging: Microglia, Metabolism & Cognitive Fitness | DR T S DIDWAL
The Aging Muscle Paradox: How Senescent Cells Cause Insulin Resistance and The Strategies to Reverse It | DR T S DIDWAL
References
Hou, Y., Long, R., Sun, Q., et al. (2026). Longitudinal associations of sarcopenia and biological age acceleration with rate of cognitive decline. BMC Public Health, 26, 761. https://doi.org/10.1186/s12889-026-26389-2
Huang, J., Li, M., Luo, Q., & Li, J. (2025). The association of sarcopenia, possible sarcopenia and cognitive impairment: A systematic review and meta-analysis. PLOS ONE, 20(5), e0324258. https://doi.org/10.1371/journal.pone.0324258
Liu, Q., Zhang, K., Tang, Y., et al. (2026). Association of sarcopenia with cognitive function in patients with Parkinson's disease: A cross-sectional study. Brain and Behavior, 16(5), e71460. https://doi.org/10.1002/brb3.71460
Sha, T., Zhang, Y., Wei, J., Li, C., Zeng, C., Lei, G., & Wang, Y. (2025). Sarcopenia and risk of cognitive impairment: Cohort study and Mendelian randomization analysis. JMIR Aging, 8, e66031. https://doi.org/10.2196/66031
Shiratsuchi, D., Makizako, H., Tabira, K., Miyake, Y., Kubozono, T., & Ohishi, M. (2026). Association between sarcopenia defined by the Asian Working Group for Sarcopenia 2025 criteria and cognitive function in middle-aged community-dwelling adults. Geriatrics & Gerontology International, 26(3), e70426. https://doi.org/10.1111/ggi.70426
Zhang, K., Huang, S., Liu, M., Zhang, Y., Li, W., Luo, B., & He, P. (2025). Sarcopenia and cognitive impairment: A multidimensional study of clinical associations, shared genetics, and causal links. Frontiers in Aging Neuroscience, 17, 1708170. https://doi.org/10.3389/fnagi.2025.1708170
Choe, Y.H., Cho, EJ., Choi, Y. et al. Effects of exercise modalities on cognitive and muscle function in older adults with cognitive impairment: a systematic review and meta-analysis. Sci Rep (2026). https://doi.org/10.1038/s41598-026-48294-9
Singh, B., Bennett, H., Miatke, A., Dumuid, D., Curtis, R., Ferguson, T., Brinsley, J., Szeto, K., Petersen, J. M., Gough, C., Eglitis, E., Simpson, C. E., Ekegren, C. L., Smith, A. E., Erickson, K. I., & Maher, C. (2025). Effectiveness of exercise for improving cognition, memory and executive function: a systematic umbrella review and meta-meta-analysis. British journal of sports medicine, 59(12), 866–876. https://doi.org/10.1136/bjsports-2024-108589