3 Hidden Types of Insulin Resistance: Why Thin People, Obese Individuals, and South Asians Develop Diabetes Differently
Discover the 3 hidden types of insulin resistance—lean, obese, and the South Asian metabolic phenotype. Learn early warning signs, biomarkers, and prevention strategies.
DIABETES
Dr. T.S. Didwal, M.D.(Internal Medicine)
3/14/202616 min read

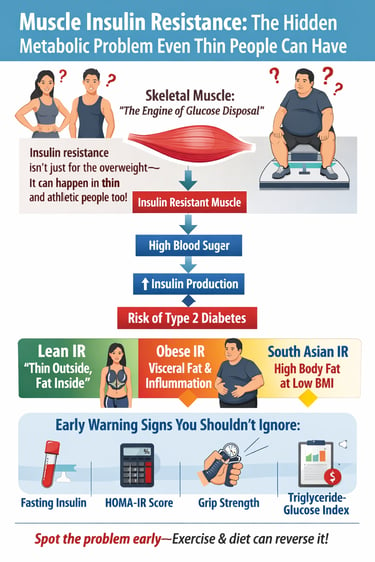
Insulin resistance is not only a problem for people who are overweight. Research shows that skeletal muscle—the body’s largest metabolic organ—controls about 70–80% of insulin-stimulated glucose uptake, meaning problems in muscle metabolism can lead to insulin resistance even in lean individuals. Early markers such as fasting insulin, HOMA-IR, grip strength, and triglyceride-glucose index can help detect metabolic dysfunction years before type 2 diabetes develops (Ahn, 2025; Kosmas et al., 2024).
Most people think insulin resistance happens only when someone becomes overweight or develops obesity. But modern metabolic research shows that this belief is incomplete. In reality, insulin resistance can develop in thin individuals, athletes with low muscle mass, and people who appear metabolically healthy during routine health checkups.
The key reason lies in an organ many people rarely think about when discussing diabetes: skeletal muscle.
Skeletal muscle is not just responsible for movement or physical strength. It is also the primary site where the body clears glucose from the bloodstream after a meal. When insulin works properly, muscle cells absorb glucose and store it as glycogen to be used for energy later. But when muscle cells become resistant to insulin signals—a condition known as muscle insulin resistance—glucose remains in the bloodstream, forcing the pancreas to produce more insulin.
Over time, this process can lead to hyperinsulinemia, chronic inflammation, metabolic syndrome, and eventually type 2 diabetes (Choo et al., 2026).
Researchers now recognize that insulin resistance appears in several distinct clinical phenotypes. Some people develop lean insulin resistance, sometimes called “thin outside, fat inside.” Others experience obesity-related insulin resistance, driven by visceral fat and inflammatory signals. A third pattern, especially common in South Asian populations, involves lower muscle mass, higher body fat percentage at lower body weight, and early metabolic dysfunction (Ahn, 2025).
Understanding these different forms of insulin resistance is critical because early warning signs often appear years before diabetes develops. Identifying these signals early allows lifestyle interventions—especially exercise, muscle strengthening, and dietary changes—to reverse metabolic dysfunction before permanent damage occurs (Kosmas et al., 2024).
In this article, we will explore the three major phenotypes of muscle-based insulin resistance, the early biomarkers doctors use to detect it, and the science-based strategies that can restore metabolic health.
Clinical pearls
1. The "TOFI" Paradox (Thin Outside, Fat Inside)
Scientific Perspective: Lean insulin resistance is often driven by ectopic lipid deposition. Even with a normal BMI, lipids can accumulate within the myocytes (intramyocellular) and around visceral organs, disrupting the PI3K/AKT signaling pathway required for glucose uptake.
Being "skinny" doesn't mean you are metabolically healthy. You can have "hidden fat" stored inside your muscles and around your organs that acts like a clog in your metabolism, making it hard for your body to process sugar.
2. The South Asian "Phenotype" Gap
Scientific Perspective: South Asian populations exhibit "sarcopenic obesity"—a higher body fat percentage and lower skeletal muscle mass at lower BMI levels. This necessitates lower diagnostic thresholds for metabolic syndrome (e.g., a BMI of 23 kg/m² vs. the global 25 kg/m²).
If you are of South Asian descent, "normal" weight standards don't apply to you. You may face a higher risk of diabetes at a much lower weight than someone of European descent because your body tends to carry less muscle and more deep-seated fat.
3. Grip Strength as a Metabolic Mirror
Scientific Perspective: Handgrip strength is a validated proxy for mitochondrial function and overall muscle quality. Low force production correlates with reduced GLUT4 translocation, the process that actually pulls sugar out of the blood and into the muscle.
Your handshake is a window into your health. Weak grip strength is often a sign that your muscles aren't "breathing" or burning fuel efficiently. Strengthening your muscles doesn't just make you stronger; it makes your blood sugar easier to control.
4. The 48-Hour "Exercise Window"
Scientific Perspective: A single bout of resistance or aerobic exercise induces a non-insulin-dependent glucose uptake mechanism that persists for 24–48 hours. This occurs through the activation of AMPK, which bypasses damaged insulin receptors to move glucose into the cell.
Think of exercise as a "natural dose of insulin" that lasts for about two days. You don't have to train like an athlete every day, but you shouldn't go more than two days without moving, or you lose that protective "glitch" that helps your body clear out sugar.
5. Post-Prandial (After-Meal) "Glucose Sponges"
Scientific Perspective: Skeletal muscle accounts for approximately 70–80% of post-meal glucose disposal. Utilizing the "muscle pump" via light activity (like walking) immediately after eating leverages mechanical-induced glucose uptake, significantly flattening the insulin spike.
Your muscles are like sponges for sugar. If you sit down right after a big meal, the "sponges" stay dry, and sugar sits in your blood. If you take a 10-minute walk, you "wring out" the sponges, allowing them to soak up the sugar before it can cause damage.
6. HOMA-IR vs. Fasting Glucose
Scientific Perspective: Fasting glucose is a lagging indicator of metabolic health; it only rises once the pancreas can no longer hyper-insulinate to compensate for resistance. HOMA-IR (calculating the ratio of insulin to glucose) identifies "compensated" insulin resistance years before the patient becomes pre-diabetic.
Relying only on a standard blood sugar test is like checking your bank balance only after you’ve gone bankrupt. A fasting insulin test (used to calculate HOMA-IR) is like checking your spending habits—it tells you if your body is working too hard to keep appearances "normal" before a crisis hits.
What Is Insulin Resistance?
Insulin is a hormone produced by your pancreas that acts like a "key" — it unlocks cells so that glucose (sugar from food) can enter and be used as energy. When cells, particularly muscle cells, become resistant to this key, the pancreas has to produce more and more insulin to compensate. Over time, this overproduction leads to elevated blood sugar levels, chronic inflammation, and, eventually, type 2 diabetes.
At the molecular level, insulin resistance in muscle is driven by impaired signalling through the PI3K/AKT/mTOR pathway, mitochondrial dysfunction, lipid accumulation within muscle fibres (known as intramyocellular lipid deposition), and chronic low-grade inflammation. A landmark 2025 review by Ahn highlighted these disruptions as key drivers of skeletal muscle insulin resistance, emphasising that multiple molecular pathways converge to impair how muscle tissue handles glucose (Ahn, 2025).
Key fact: Skeletal muscle accounts for approximately 70–80% of insulin-stimulated glucose disposal
in the human body. When muscle becomes insulin-resistant, the entire metabolic system is affected.
Three distinct types of insulin resistance
1. Lean Insulin Resistance (The "TOFI" Phenotype)
The Concept: Known as "Thin Outside, Fat Inside," this affects individuals who appear healthy on the scale but have internal metabolic dysfunction.
Key Drivers:
Ectopic Fat: Fat is stored in dangerous locations where it doesn't belong, such as the liver and directly inside muscle fibres (intramyocellular lipids).
Muscle Deficiency: Low overall muscle volume means there are fewer "sinks" available to soak up glucose from the blood.
Typical Body Type: Normal BMI. These individuals often have thin limbs and no obvious abdominal bulge.
Major Risk: Undiagnosed Diabetes. Because they don't "look" the part, they are often excluded from early screening, allowing metabolic damage to progress silently for years.
2. Obese Insulin Resistance
The Concept: This is the most common form, where a significant excess of body fat creates a state of chronic, low-grade inflammation.
Key Drivers:
Visceral Adiposity: Deep belly fat acts like an endocrine organ, pumping out inflammatory signals (cytokines) that "jam" insulin receptors.
Lipid Overflow: High levels of free fatty acids in the blood interfere with how muscles process energy.
Typical Body Type: High BMI. Characterized by a high waist-to-hip ratio and visible central weight gain.
Major Risk: Metabolic Syndrome. A cluster of conditions, including high blood pressure, high triglycerides, and low "good" cholesterol (HDL), leading to significant cardiovascular risk.
3. South Asian Insulin Resistance
The Concept: A specific metabolic profile prevalent in South Asian populations where insulin resistance occurs much earlier and at lower weights.
Key Drivers:
Sarcopenic Obesity: A unique combination of low muscle mass and high body fat percentage, even in people who are not "heavy."
Thin-Fat Phenotype: High levels of abdominal fat relative to a smaller skeletal frame.
Typical Body Type: Lower BMI. Often appearing "lean" by Western standards but having a BMI above the ethnicity-specific cutoff of 23 kg/m².
Major Risk: Early-Onset Diabetes. Individuals in this group often develop Type 2 Diabetes 10–15 years earlier than other ethnic groups and at much lower weight thresholds.
Clinical Phenotype 1: Lean Insulin Resistance
Perhaps the most misunderstood presentation of insulin resistance is the lean phenotype — where an individual with a normal body mass index (BMI) and no visible signs of excess weight develops significant metabolic dysfunction. This is sometimes called "TOFI" — Thin Outside, Fat Inside.
What Drives Lean Insulin Resistance?
Lean insulin resistance is typically driven by:
• Ectopic fat accumulation: Visceral fat deposits inside the liver, pancreas, and within muscle fibres — even when subcutaneous (under-skin) fat is minimal.
• Genetic predisposition: Some individuals have an inherited tendency toward impaired insulin signalling in muscle cells.
• Physical inactivity with low muscle mass: Low muscle volume means reduced glucose storage capacity, even at a healthy weight.
• Mitochondrial inefficiency: Poor mitochondrial function in muscle cells impairs the ability to oxidise (burn) both glucose and fat.
Research published in Physiology by Nakhoul and Abdulnour-Nakhoul (2025) introduced a novel "Ohmic" model of insulin resistance — drawing an analogy from electrical engineering — to distinguish lean and obese insulin resistance patterns. Their model demonstrated that lean individuals can show insulin resistance characterised by different resistance-conductance profiles compared to obese subjects, underscoring that these are biologically distinct conditions that require tailored diagnostic approaches.
Signs You May Have Insulin Resistance Even If You Are Thin
Example:
• Persistent fatigue after meals
• Strong sugar cravings
• Elevated fasting insulin
• Increased waist circumference despite normal weight
• Family history of diabetes
• Low grip strength
Why Lean Insulin Resistance Is Often Missed
Because conventional screening tools like BMI and fasting blood glucose often appear normal in lean-resistant individuals, this phenotype is systematically underdiagnosed. A person may feel fine, look healthy, and pass a standard check-up — yet their muscle tissue is already struggling to absorb glucose efficiently. By the time fasting glucose rises to diabetic levels, years of silent metabolic damage may have already occurred.
Clinical pearl: A normal BMI does NOT rule out insulin resistance. Lean individuals with
a family history of diabetes, sedentary lifestyles, or unexplained fatigue should consider
asking their doctor about fasting insulin levels and HOMA-IR testing.
Clinical Phenotype 2: Obese Insulin Resistance
Obesity-related insulin resistance is the most widely studied and most commonly recognised form. Here, excess adipose (fat) tissue — particularly visceral fat around the abdominal organs — acts as a major driver of systemic metabolic dysfunction.
The Adipose-Muscle Crosstalk
Obese insulin resistance involves a complex interplay between expanded fat tissue and skeletal muscle. Enlarged adipocytes (fat cells) release excessive amounts of pro-inflammatory cytokines such as TNF-alpha and IL-6, as well as elevated free fatty acids (FFAs). These molecules flood the bloodstream and impair insulin receptor signalling within muscle cells — essentially "jamming" the insulin key so it can no longer open the glucose-entry lock.
A comprehensive 2026 review by Choo, Ravi, and Subramaniyan detailed these mechanisms extensively, noting that obesity-induced insulin resistance involves lipid oversupply to muscles, reduced GLUT4 transporter activity (the protein that actually moves glucose into cells), and hyperactivation of inflammatory pathways including NF-kB and JNK signalling (Choo et al., 2026). These disruptions compound over time, creating a vicious cycle where insulin resistance worsens adiposity and adiposity deepens insulin resistance.
Beyond BMI: The Importance of Fat Distribution
Not all obesity carries the same metabolic risk. Research distinguishes between:
• Metabolically unhealthy obesity (MUO): High visceral fat, elevated triglycerides, low HDL cholesterol, and active insulin resistance in muscle.
• Metabolically healthy obesity (MHO): Higher BMI but preserved insulin sensitivity, often due to favourable fat distribution (more subcutaneous, less visceral fat).
Measuring waist circumference, waist-to-hip ratio, and body fat percentage — rather than BMI alone — gives a far more accurate picture of metabolic risk. Choo et al. (2026) emphasised that therapeutic strategies targeting visceral adiposity and muscle lipid accumulation are more effective than those focused simply on total weight reduction.
Clinical Phenotype 3: The South Asian Phenotype — A Special Concern
Why Indians and South Asians Develop
Diabetes at Lower Body Weight
For readers in South Asia — including India, Pakistan, Bangladesh, Sri Lanka, and Nepal — this section is particularly relevant. South Asians face a disproportionately high burden of type 2 diabetes globally, developing the condition at younger ages, at lower BMIs, and with less obesity than Western populations. Understanding why requires looking at the South Asian metabolic phenotype.
Defining Characteristics of the South Asian Phenotype
• Higher body fat percentage at the same BMI compared to White European populations.
• Greater tendency for central (abdominal) adiposity even in lean individuals.
• Lower skeletal muscle mass relative to body fat — a pattern sometimes called "metabolic obesity in the lean" or "sarcopenic obesity."
• Elevated fasting insulin and insulin resistance markers appearing at lower body weight thresholds.
• A genetic predisposition toward reduced beta-cell function and hepatic insulin resistance.
These characteristics mean that standard Western BMI cutoffs (overweight: 25 kg/m², obese: 30 kg/m²) are inappropriate for South Asians. The World Health Organization and Indian Council of Medical Research recommend using lower cutoffs of 23 kg/m² for overweight and 25 kg/m² for obesity in South Asian populations.
Important for South Asian readers: If your BMI is above 23 kg/m², you may already be at
significant risk for insulin resistance — even if you appear lean by global standards.
Early testing with HOMA-IR and fasting insulin is strongly advised.
Muscle Quality in South Asians
Research has shown that South Asians tend to have lower muscle glycogen storage capacity and reduced oxidative capacity in skeletal muscle fibres compared to other ethnic groups. Combined with dietary patterns higher in refined carbohydrates and often lower in high-quality protein, this creates a metabolic environment highly conducive to muscle-based insulin resistance. The review by Ahn (2025) noted that impaired mitochondrial function and increased intramyocellular lipid are particularly prominent features in insulin-resistant muscle tissue — patterns consistent with what is observed in South Asian populations.
Early Detection: Biomarkers You and Your Doctor Should Know
One of the most powerful aspects of modern metabolic research is the growing toolkit of early biomarkers for insulin resistance — markers that can detect dysfunction years before fasting blood glucose rises to a diabetic threshold.
1. HOMA-IR (Homeostatic Model Assessment of Insulin Resistance)
HOMA-IR is calculated from a simple fasting blood test using the formula: (fasting insulin x fasting glucose) / 405. It provides an estimate of insulin resistance based on how much insulin the pancreas must produce to maintain normal fasting glucose.
• Normal HOMA-IR: Less than 1.0 is optimal; below 1.9 is acceptable.
• Elevated HOMA-IR: Values above 2.5–3.0 indicate significant insulin resistance.
A 2026 review by Ibrahim highlighted HOMA-IR as one of the most clinically useful and cost-effective early biomarkers for predicting insulin resistance and progression to type 2 diabetes mellitus, noting its strong correlation with muscle-level glucose uptake impairment (Ibrahim, 2026).
2. Fasting Insulin
Fasting insulin is a more sensitive early marker than fasting blood glucose alone. Because the pancreas compensates for insulin resistance by producing more insulin, fasting insulin can become elevated years before blood glucose levels rise abnormally. Unfortunately, many routine health panels do not include fasting insulin — you may need to specifically request it from your doctor.
• Optimal fasting insulin: 2–5 mIU/L
• Borderline elevated: 6–10 mIU/L
• Significantly elevated: Above 10 mIU/L — warrants further metabolic assessment
3. Grip Strength — A Surprising Muscle-Metabolic Marker
Grip strength measured by hand dynamometry has emerged as a powerful, non-invasive proxy for overall skeletal muscle metabolic health. Multiple large studies have demonstrated that low grip strength correlates strongly with insulin resistance, poor glucose tolerance, and risk of type 2 diabetes — independent of total body weight.
The reasoning is physiological: grip strength reflects the functional quality of skeletal muscle. Low grip strength signals reduced muscle mass and poor mitochondrial function — the same muscle-level deficiencies that underlie insulin resistance. A 2024 review by Kosmas and colleagues on biomarkers of insulin sensitivity confirmed that reduced muscle strength, including grip strength, is an emerging and clinically relevant early indicator of metabolic dysfunction (Kosmas et al., 2024).
Practical tip: Ask your doctor about a simple hand dynamometry test at your next check-up.
For women, grip strength below 20 kg and for men below 30 kg may signal metabolic risk.
Resistance training to build muscle can improve both grip strength and insulin sensitivity simultaneously.
Other Emerging Biomarkers
Ibrahim (2026) and Kosmas et al. (2024) also discussed several other promising markers, including:
• Triglyceride-Glucose (TyG) Index: A simple calculation from fasting triglycerides and glucose that strongly predicts insulin resistance.
• Fasting C-peptide: Reflects pancreatic insulin secretion and can help differentiate types of insulin resistance.
• Adiponectin levels: Low adiponectin is associated with insulin resistance and visceral fat accumulation.
• High-sensitivity CRP (hsCRP): Elevated values indicate the chronic inflammation that drives and perpetuates insulin resistance.
Can Muscle-Based Insulin Resistance Be Reversed?
The encouraging answer is yes, and skeletal muscle is one of the most adaptable tissues in the human body. Research-backed interventions that improve muscle-based insulin resistance include:
Resistance and Aerobic Exercise
Exercise is the most powerful intervention for skeletal muscle insulin resistance. Resistance training increases GLUT4 transporter expression in muscle cells, improves mitochondrial density, and reduces intramyocellular lipid accumulation. Even a single bout of moderate-intensity exercise can temporarily improve insulin sensitivity for 24–48 hours. Ahn (2025) noted exercise as one of the most robust non-pharmacological modifiers of molecular insulin resistance pathways.
Dietary Strategies
• Reducing refined carbohydrate and added sugar intake to lower chronic insulin demand.
• Increasing high-quality protein to support muscle protein synthesis and mass.
• Including anti-inflammatory fats (omega-3 fatty acids) to reduce cytokine-driven insulin resistance.
• Time-restricted eating patterns that align food intake with circadian rhythm and reduce insulin secretion burden.
Pharmacological Options
When lifestyle interventions are insufficient, metformin remains the cornerstone pharmacological therapy, improving hepatic and peripheral insulin sensitivity. Newer agents, including GLP-1 receptor agonists and SGLT-2 inhibitors, have demonstrated significant improvements in insulin resistance markers in both lean and obese phenotypes, though optimal dosing strategies for lean individuals remain an active area of research (Ahn, 2025; Choo et al., 2026).
Key insights patients should know
• Insulin resistance can occur even in thin people.
A growing body of research describes a condition sometimes called “lean insulin resistance” or thin outside, fat inside (TOFI). In these individuals, fat accumulates around organs and inside muscle tissue despite a normal body weight, impairing insulin signaling.
• Muscle health strongly influences metabolic health.
When skeletal muscle becomes resistant to insulin, glucose cannot enter cells efficiently. The pancreas compensates by producing more insulin, eventually leading to hyperinsulinemia, metabolic syndrome, and type 2 diabetes (Choo et al., 2026).
• South Asians face a unique metabolic risk.
People of South Asian ancestry often develop insulin resistance at lower body weights and younger ages. Lower muscle mass, higher visceral fat, and genetic susceptibility contribute to this distinctive metabolic phenotype.
• Early warning signs appear years before diabetes develops.
Important early indicators include:
Elevated fasting insulin levels
Increased HOMA-IR score
Higher triglyceride-glucose (TyG) index
Reduced muscle strength, including low grip strength (Kosmas et al., 2024)
• The encouraging news: muscle is highly adaptable.
Regular resistance training, physical activity, adequate protein intake, and healthy sleep can significantly improve insulin sensitivity and restore metabolic balance.
The bottom line
Insulin resistance is not simply a disease of excess weight. It is often a disease of impaired muscle metabolism. Recognizing this earlier — and strengthening skeletal muscle through lifestyle interventions — may be one of the most powerful strategies for preventing type 2 diabetes in modern populations.
Frequently Asked Questions (FAQs)
FAQ 1: I am thin and exercise regularly — can I still have insulin resistance?
Yes. Lean insulin resistance is a well-documented clinical phenotype where individuals with normal BMI develop significant muscle-level insulin dysfunction. This is especially common in people with low muscle mass (even at healthy body weight), a family history of diabetes, sedentary lifestyles despite not being overweight, or those who are of South Asian ancestry. A fasting insulin test and HOMA-IR calculation can reveal this even when fasting glucose appears normal.
FAQ 2: What is the difference between insulin resistance and type 2 diabetes?
Insulin resistance is the underlying root cause, while type 2 diabetes is a later-stage consequence. In insulin resistance, your pancreas compensates by producing more insulin — blood glucose may remain normal or slightly elevated. Type 2 diabetes develops when the pancreas can no longer keep up with the demand, and blood glucose rises to diabetic thresholds. Early detection of insulin resistance — through HOMA-IR and fasting insulin — can allow intervention before diabetes develops.
FAQ 3: Are South Asians more likely to develop insulin resistance even without obesity?
Yes, and this is one of the most important public health findings of the past two decades. South Asians tend to have higher body fat percentage at the same BMI, lower muscle mass, greater central adiposity, and a genetic predisposition toward reduced insulin sensitivity. These factors mean South Asians develop insulin resistance and type 2 diabetes at younger ages and lower BMIs than European populations. Ethnicity-adjusted screening cutoffs are essential for this demographic.
FAQ 4: How does grip strength relate to insulin resistance?
Grip strength is a functional marker of overall skeletal muscle quality and mass — the very tissue primarily responsible for insulin-stimulated glucose uptake. Studies have shown that low grip strength is independently associated with higher HOMA-IR, impaired glucose tolerance, and increased diabetes risk. It is inexpensive, non-invasive, and can be measured in any clinical setting. Improving grip strength through resistance training simultaneously improves insulin sensitivity.
FAQ 5: What is HOMA-IR and how is it calculated?
HOMA-IR stands for Homeostatic Model Assessment of Insulin Resistance. It is calculated from a simple formula using two fasting blood tests: HOMA-IR = (Fasting Insulin [mIU/L] x Fasting Glucose [mg/dL]) / 405. A value below 1.0 is optimal; values above 2.5 indicate moderate insulin resistance, and above 3.0 indicates significant resistance. It is one of the most widely used, validated, and cost-effective tools for detecting early insulin resistance in clinical practice.
FAQ 6: Can insulin resistance in the muscle be reversed without medication?
Absolutely. Skeletal muscle is highly plastic — it responds dramatically to lifestyle interventions. Regular resistance and aerobic exercise, a protein-adequate diet with reduced refined carbohydrates, weight management (even modest 5–10% reductions in body weight significantly improve insulin sensitivity), better sleep quality, and stress reduction have all been shown to substantially improve HOMA-IR, fasting insulin, and muscle metabolic function. Medication should be considered as a complement to, not a replacement for, lifestyle changes.
FAQ 7: Which blood tests should I ask my doctor for to screen for insulin resistance?
For a comprehensive early screening panel, request: (1) fasting insulin levels, (2) fasting blood glucose, (3) HOMA-IR calculation, (4) fasting lipid panel (especially triglycerides and HDL), (5) high-sensitivity CRP (hsCRP) for inflammation, and optionally (6) HbA1c for a 3-month blood sugar average. In South Asian patients, these tests are recommended starting in the late 20s or early 30s — earlier than standard guidelines suggest for other ethnic groups.
Author’s Note
As a physician working in metabolic medicine, I have increasingly observed that insulin resistance is often misunderstood — both by patients and sometimes even within routine clinical practice. Many people believe that diabetes and metabolic disease develop only in individuals who are visibly overweight. However, emerging research and real-world clinical experience show that insulin resistance frequently begins much earlier and in far more diverse body types than most people realize.
In particular, I have seen many patients who appear lean, active, and otherwise healthy, yet show clear biochemical evidence of metabolic dysfunction when evaluated with tests such as fasting insulin, HOMA-IR, or lipid markers. This is especially relevant in South Asian populations, where individuals may develop insulin resistance and type 2 diabetes at younger ages and at lower body weights compared with Western populations.
One of the central messages of this article is that skeletal muscle plays a critical role in metabolic health. Muscle is not simply responsible for movement and strength; it is one of the body’s most important metabolic organs, responsible for the majority of insulin-mediated glucose uptake. When muscle health declines — due to inactivity, low muscle mass, poor diet, or chronic inflammation — insulin resistance can develop silently years before diabetes appears.
The goal of this article is therefore not to alarm readers, but to encourage earlier awareness and proactive metabolic screening. Simple measures such as maintaining muscle mass through resistance exercise, improving diet quality, prioritising sleep, and monitoring key metabolic markers can dramatically reduce long-term risk.
Metabolic diseases develop slowly, but the encouraging reality is that early intervention is remarkably powerful. With the right knowledge and lifestyle strategies, insulin resistance can often be identified early and meaningfully reversed.
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Is Your Muscle Insulin Resistant? 2026 Update | DR T S DIDWAL
Activate Your Brown Fat: A New Pathway to Longevity and Metabolic Health | DR T S DIDWAL
Leptin vs. Adiponectin: How Your Fat Hormones Control Weight and Metabolic Health | DR T S DIDWAL
References
Ahn, B. (2025). Advances in Insulin Resistance—Molecular Mechanisms, Therapeutic Targets, and Future Directions. International Journal of Molecular Sciences, 26(6), 2574. https://doi.org/10.3390/ijms26062574
Choo, Y. N., Ravi, R. N., & Subramaniyan, V. (2026). Insulin resistance induced by obesity: Mechanisms, metabolic implications and therapeutic approaches. Molecular Biology Reports, 53(1), 357. https://doi.org/10.1007/s11033-026-11509-3
Ibrahim, A. Y. (2026). Clinical significance of emerging metabolic biomarkers in early prediction of insulin resistance and type 2 diabetes mellitus: A review study. International Journal of Science and Research Archive, 18(2), 345–352. https://doi.org/10.30574/ijsra.2026.18.2.0247
Kosmas, C. E., Sourlas, A., Oikonomakis, K., Zoumi, E. A., Papadimitriou, A., & Kostara, C. E. (2024). Biomarkers of insulin sensitivity/resistance. The Journal of International Medical Research, 52(10), 03000605241285550. https://doi.org/10.1177/03000605241285550
Nakhoul, N., & Abdulnour-Nakhoul, S. (2025). New and simple Ohmic definition of insulin resistance in lean and obese subjects. Physiology, 40(Suppl 1). https://doi.org/10.1152/physiol.2025.40.S1.0863