Your Gut Microbiome Explained: How It Controls Immunity, Metabolism, and Disease
The gut microbiome controls metabolism, immunity, and inflammation. Learn how to optimize it with simple, research-backed strategies.
NUTRITION
Dr. T.S. Didwal, M.D.(Internal Medicine)
5/4/202616 min read
What is the gut microbiome and why is it important?
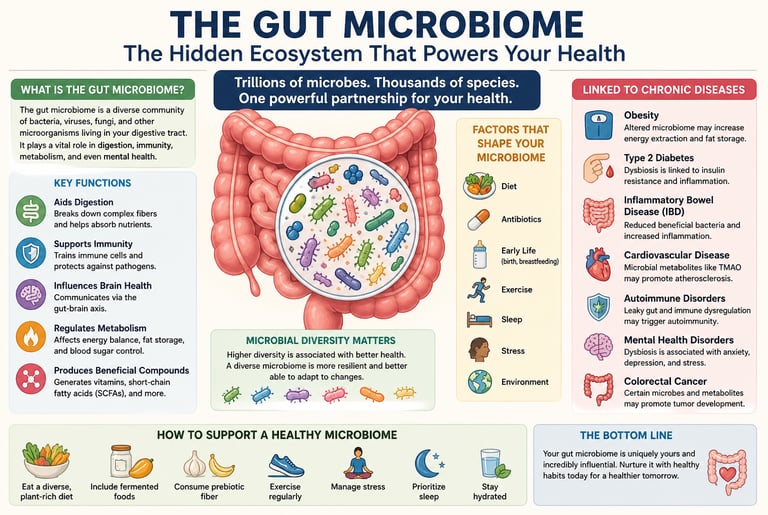
The gut microbiome is a community of trillions of microorganisms living in the digestive tract that regulate digestion, metabolism, immune function, and brain health. These microbes produce bioactive compounds like short-chain fatty acids that reduce inflammation, strengthen the gut barrier, and influence insulin sensitivity. An imbalanced microbiome (dysbiosis) is linked to obesity, diabetes, cardiovascular disease, and autoimmune disorders.
What You Need to Know About Your Gut Microbiome
The gut microbiome is a community of trillions of microorganisms that regulate digestion, metabolism, immunity, and even brain function.
Diet is the most powerful modulator: fiber-rich, plant-based, and fermented foods increase beneficial bacteria and anti-inflammatory metabolites.
Gut bacteria produce bioactive compounds (postbiotics) like short-chain fatty acids that influence insulin sensitivity, inflammation, and gut barrier integrity.
Dysbiosis (microbial imbalance) is linked to obesity, type 2 diabetes, cardiovascular disease, and autoimmune disorders.
Lifestyle factors—including sleep, exercise, stress, and antibiotic use—significantly shape microbiome diversity.
Emerging science focuses on microbial function (metabolomics, gene richness) rather than just bacterial composition.
Bottom line: A diverse, fiber-rich diet and healthy lifestyle can rapidly improve your gut microbiome and long-term health.
How to improve gut health naturally
Eat 30+ different plant foods per week to increase microbial diversity
Include fiber-rich foods (vegetables, legumes, whole grains) daily
Add fermented foods like yogurt, kefir, or kimchi
Exercise regularly to support beneficial gut bacteria
Sleep 7–9 hours to maintain microbiome balance
Manage stress to reduce gut inflammation
Avoid unnecessary antibiotics and ultra-processed foods
Advanced Concepts
Metabolomics & Postbiotics
Metabolomics studies the full spectrum of small molecules (metabolites) produced by the microbiome and host.
These metabolites—especially short-chain fatty acids (SCFAs), bile acid derivatives, and indoles—are key mediators of microbiome–host interactions.
Postbiotics are bioactive microbial products (not live bacteria) that can exert anti-inflammatory, metabolic, and immunomodulatory effects.
Clinical relevance: Potential for targeted therapies without variability seen in probiotic colonization.
Microbial Gene Richness
Refers to the total number and diversity of microbial genes in the gut microbiome.
Higher gene richness is consistently associated with:
Better metabolic health
Lower inflammation
Reduced risk of obesity and insulin resistance
Low gene richness correlates with dysbiosis and increased risk of chronic disease.
Key insight: Functional capacity (what microbes can do) may be more important than taxonomy (which microbes are present).
Enterotypes Debate
Enterotypes are proposed microbiome “clusters” dominated by genera like Bacteroides, Prevotella, or Ruminococcus.
Early research suggested stable, distinct categories, but newer evidence shows:
Microbiome composition exists on a continuum, not fixed types
Strongly influenced by diet, geography, and lifestyle
Current consensus: Enterotypes are useful conceptual models, but have limited clinical utility.
What if one of the most powerful regulators of your health isn’t an organ you can see—but a living ecosystem inside your gut? The human gut microbiome, composed of trillions of microorganisms, is now recognized as a central player in metabolism, immunity, and even brain function (Zhang et al., 2024). Far from being passive passengers, these microbes actively produce bioactive compounds that influence inflammation, insulin sensitivity, and gut barrier integrity—key processes underlying most chronic diseases (Ding et al., 2026).
Recent research has linked disruptions in the gut microbiome—known as dysbiosis—to conditions ranging from obesity and type 2 diabetes to cardiovascular disease, autoimmune disorders, and depression (Khalil et al., 2024; Zhang et al., 2024). What makes this discovery especially compelling is how rapidly the microbiome responds to change. Within just days, dietary patterns can reshape microbial composition and function, altering metabolic and immune outcomes (Rondinella et al., 2025).
Even more striking, emerging evidence suggests that it’s not just which microbes are present, but what they do—through their metabolites and gene activity—that determines their impact on health (Ding et al., 2026). This marks a shift toward a more dynamic, systems-level understanding of human biology.
In essence, your gut microbiome is not just part of your body—it is a critical driver of your long-term health trajectory.
What Exactly Is the Gut Microbiome?
Your gastrointestinal tract is home to an estimated 38 trillion microbial cells, a number that rivals or even surpasses the total number of human cells in your body. Collectively, these organisms — their genes, metabolic products, and interactions — make up the gut microbiome.
According to Zhang et al. (2024), the gut microbiota encompasses a diverse array of bacteria, archaea, viruses, and fungi that co-evolved with their human hosts over millennia, forming a deeply symbiotic relationship. This relationship is so fundamental that disruptions to the microbiome — a state called dysbiosis — have been linked to a growing list of chronic conditions, including obesity, type 2 diabetes, cardiovascular disease, inflammatory bowel disease, and even depression (Zhang et al., 2024).
The microbiome is not static. It changes throughout your lifetime, shaped by factors including birth method, breastfeeding, diet, antibiotic use, stress, sleep quality, and environmental exposures. Understanding how these factors influence microbial communities is key to unlocking personalized, microbiome-informed healthcare.
How Microbial Enzymes Drive Your Metabolism
One of the most groundbreaking recent discoveries concerns the role of microbial enzymes — biological catalysts produced by gut bacteria that directly influence human metabolism. Ding et al. (2026) published a landmark review in Nature Reviews Gastroenterology & Hepatology showing that the microbiome regulates host metabolic health and diseases specifically through these enzymatic pathways.
These microbial enzymes are responsible for a wide range of metabolic functions that human cells simply cannot perform on their own:
Breaking down complex dietary fibers into short-chain fatty acids (SCFAs) like butyrate, propionate, and acetate, which serve as energy sources for colonocytes (gut lining cells) and anti-inflammatory signaling molecules throughout the body.
Metabolizing bile acids, transforming primary bile acids from the liver into secondary bile acids that regulate fat digestion, cholesterol metabolism, and glucose homeostasis.
Biotransforming dietary phytochemicals and polyphenols (found in foods like berries, green tea, and olive oil) into bioactive compounds with antioxidant and anti-inflammatory properties.
Synthesizing vitamins, including vitamin K and several B vitamins, that are essential for blood clotting, neurological function, and energy metabolism.
When the balance of microbial enzyme activity is disrupted, the downstream consequences can be profound — contributing to metabolic syndrome, fatty liver disease, insulin resistance, and systemic inflammation (Ding et al., 2026).
Practical Implication: Eating a diet rich in prebiotic fibers (found in foods like garlic, onions, oats, bananas, and legumes) directly fuels the microbial enzyme activity that protects your metabolic health.
The Microbiome, Energy, and Body Weight
Have you ever wondered why some people seem to gain weight easily while others don't, even when eating similar diets? The answer may lie partly in the gut.
Blok and Bozadjieva-Kramer (2025) argue in a compelling review that the gut microbiome plays a central, underappreciated role in energy metabolism — influencing how efficiently calories are extracted from food, how fat is stored, and how hunger hormones like ghrelin and GLP-1 are regulated.
Research has shown that the microbiome composition of individuals with obesity differs significantly from that of lean individuals. Specifically, studies have found that people with obesity tend to have:
Reduced microbial diversity, a marker consistently associated with poorer health outcomes.
Higher ratios of Firmicutes to Bacteroidetes, two dominant bacterial phyla, though this finding remains nuanced and context-dependent.
Lower levels of SCFA-producing bacteria, which limits the protective effects of butyrate on the gut lining and systemic inflammation.
The gut microbiome also communicates directly with the brain via the gut-brain axis — a bidirectional communication network involving neural, hormonal, and immune pathways. This means that microbial imbalances can affect not just digestion and metabolism but also appetite regulation, mood, and cognitive function (Blok & Bozadjieva-Kramer, 2025).
Key Takeaway: Microbiome-targeted interventions — including dietary change, targeted probiotics, and prebiotics — represent a promising complementary strategy for weight management and metabolic health, though they are best combined with other evidence-based approaches.
Diet: The Single Most Powerful Tool to Shape Your Microbiome
If there is one message from modern microbiome research that deserves to be shouted from the rooftops, it is this: what you eat profoundly and rapidly changes your gut microbial community.
Rondinella et al. (2025) provide a comprehensive overview of how different dietary patterns impact microbiome composition, with striking implications for immune function. Their research, published in Clinical Immunology Communications, found that:
Mediterranean diets — rich in vegetables, fruits, whole grains, legumes, olive oil, and fish — are consistently associated with higher microbial diversity, increased SCFA production, and reduced markers of systemic inflammation.
Ultra-processed food diets — high in refined sugars, artificial additives, saturated fats, and low in fiber — rapidly deplete beneficial bacterial species and promote the growth of pro-inflammatory microbes.
Plant-based diets support the growth of Bifidobacterium, Lactobacillus, and Faecalibacterium prausnitzii — species associated with gut barrier integrity, anti-inflammatory activity, and reduced disease risk.
High-fat, low-fiber diets have been shown to reduce microbial diversity within just a few days, highlighting how quickly dietary choices translate into microbial changes.
Barb and Wallen (2025) further emphasize in Nutrients that emerging research links specific dietary components — including polyphenols, omega-3 fatty acids, fermented foods, and dietary fiber — to targeted improvements in microbiome diversity and host health outcomes. Their review highlights the potential for precision nutrition — tailoring dietary recommendations to an individual's unique microbiome profile — as a frontier in personalized medicine.
Practical Dietary Strategies for a Healthier Microbiome
Prebiotic Fibers
Examples: Garlic, onions, leeks, asparagus, bananas, and oats.
Microbiome Benefit: Acts as a specialized "fertilizer" to nourish and grow your existing beneficial bacteria.
Probiotic Foods
Examples: Yogurt, kefir, kimchi, sauerkraut, miso, and tempeh.
Microbiome Benefit: Introduces live, active beneficial microbes directly into your digestive ecosystem.
Polyphenol-Rich Foods
Examples: Berries, green tea, dark chocolate, and olive oil.
Microbiome Benefit: These antioxidant compounds selectively promote the growth of diverse and protective bacterial species.
Omega-3 Sources
Examples: Fatty fish (salmon, sardines), flaxseeds, walnuts, and chia seeds.
Microbiome Benefit: Helps soothe the gut environment by reducing microbial-induced inflammation.
Whole Grains
Examples: Brown rice, quinoa, barley, and whole wheat.
Microbiome Benefit: Fuels the production of Short-Chain Fatty Acids (SCFAs) like butyrate, which repair the gut lining and regulate metabolism.Source: Adapted from Rondinella et al. (2025) and Barb & Wallen (2025)
The Microbiome and Immune-Mediated Diseases
Perhaps nowhere is the microbiome's influence more clinically striking than in its relationship with the immune system. The gut houses approximately 70–80% of the body's immune cells, making it the largest immune organ in the human body.
Khalil et al. (2024) provide an extensive analysis of how the gut microbiome acts as a critical educator and regulator of both innate and adaptive immunity. Key findings include:
Gut bacteria train immune cells to distinguish between harmful pathogens and harmless substances, preventing both infections and inappropriate immune reactions (such as allergies and autoimmunity).
Microbial metabolites — particularly SCFAs like butyrate — directly regulate the development and function of regulatory T cells (Tregs), which suppress excessive immune responses.
Dysbiosis has been linked to increased intestinal permeability (the so-called "leaky gut"), which allows bacterial products to enter the bloodstream and trigger systemic inflammation, contributing to conditions including rheumatoid arthritis, multiple sclerosis, lupus, and type 1 diabetes (Khalil et al., 2024).
Rondinella et al. (2025) extend this analysis specifically to immune-mediated diseases, noting that dietary interventions that restore microbiome diversity have demonstrated measurable benefits in conditions like inflammatory bowel disease (IBD), celiac disease, and psoriasis. While this research is still maturing, the therapeutic potential is enormous.
Gut Microbiome and Common Chronic Diseases: A Summary
Obesity
Reduced microbial diversity
Increased energy extraction from food
Evidence: Blok & Bozadjieva-Kramer (2025)
Type 2 Diabetes
Impaired production of short-chain fatty acids (SCFAs)
Disruption of gut barrier integrity → metabolic inflammation
Evidence: Ding et al. (2026)
Inflammatory Bowel Disease (IBD)
Gut dysbiosis
Reduced levels of Faecalibacterium prausnitzii (anti-inflammatory bacterium)
Evidence: Khalil et al. (2024)
Cardiovascular Disease
Increased production of trimethylamine N-oxide (TMAO) by gut bacteria
Links to atherosclerosis and vascular inflammation
Evidence: Zhang et al. (2024)
Autoimmune Disorders
Increased intestinal permeability (“leaky gut”)
Dysregulated immune system education
Evidence: Rondinella et al. (2025)
Mental Health Disorders
Disruption of the gut–brain axis
Altered microbial influence on neurotransmitters and inflammation
Evidence: Blok & Bozadjieva-Kramer (2025)
Colorectal Cancer
Reduced protective microbial species
Increased production of carcinogenic metabolites
Evidence: Khalil et al. (2024)
Clinical Insight
Most associations are correlative rather than strictly causal, but collectively highlight the microbiome as a central player in chronic disease pathophysiology.
Lifestyle Factors That Impact Your Microbiome
Diet is the most powerful lever, but it is far from the only one. A growing body of evidence points to several lifestyle factors that profoundly influence microbiome health:
Sleep Quality: Poor sleep has been shown to reduce microbial diversity and increase pro-inflammatory bacteria. Prioritizing 7–9 hours of quality sleep per night supports microbiome resilience (Zhang et al., 2024).
Physical Activity: Regular moderate-intensity exercise increases microbial diversity and promotes SCFA-producing bacteria. Even 30 minutes of brisk walking most days of the week can have measurable microbiome benefits (Barb & Wallen, 2025).
Antibiotic Use: While lifesaving when necessary, antibiotics cause significant collateral disruption to the microbiome, sometimes with effects lasting months to years. Taking antibiotics only when clinically necessary and supporting recovery with probiotic-rich foods afterward is advisable.
Chronic Stress: Psychological stress activates the hypothalamic-pituitary-adrenal (HPA) axis, releasing cortisol and other hormones that alter gut motility, immune function, and microbial composition. Mind-body practices like meditation, yoga, and deep breathing may help protect the microbiome by reducing chronic stress responses.
Artificial Sweeteners: Emerging research suggests that certain artificial sweeteners — including saccharin and sucralose — may alter microbiome composition in ways that paradoxically impair glucose tolerance, though this area remains under active investigation (Zhang et al., 2024).
Practical Applications: What You Can Start Doing Today
You don't need a prescription or a lab test to begin nurturing your microbiome. Here are evidence-based steps you can take right now:
Eat 30+ Different Plant Foods Per Week. Research consistently shows that people who eat the widest variety of plant foods have the most diverse microbiomes. Count every different vegetable, fruit, whole grain, legume, nut, seed, and herb you eat in a week — and aim to diversify.
Incorporate Fermented Foods Daily Yogurt with live cultures, kefir, kimchi, sauerkraut, miso soup, and kombucha, all of which introduce beneficial live microorganisms into your gut. Start with one serving daily and gradually increase.
Prioritize Prebiotic Fiber at Every Meal. Include a prebiotic-rich food at every meal — garlic or onions in cooking, a sliced banana over oatmeal, or legumes added to salads and soups. These fibers feed and sustain your existing beneficial bacteria.
Move Your Body Regularly. Aim for at least 150 minutes of moderate-intensity aerobic activity per week. Activities like walking, cycling, swimming, and dancing all support microbiome diversity.
Stay Well Hydrated. Adequate water intake supports mucus production in the gut, which forms an important habitat and protective layer for beneficial bacteria. Aim for 8–10 glasses of water daily.
Protect Your Sleep. Consistent sleep schedules, dark sleeping environments, and reduced screen time before bed all support the circadian rhythms that influence gut microbial activity.
Manage Stress Proactively. Even 10 minutes of mindfulness meditation daily has been shown to reduce stress hormones with downstream benefits for gut health. Apps, guided sessions, or simply quiet breathing exercises can help.
Use Antibiotics Wisely. Only take antibiotics when prescribed by a healthcare professional for a confirmed bacterial infection. Always complete the full course, and consider discussing probiotic support with your doctor during and after treatment.
Emerging Frontiers: The Future of Microbiome Medicine
The science of the microbiome is advancing at a breathtaking pace. Several exciting therapeutic approaches are on the horizon:
Fecal Microbiota Transplantation (FMT): The transfer of gut microbiota from a healthy donor to a recipient has shown remarkable success in treating recurrent Clostridioides difficile infection and is being investigated for IBD, obesity, and neurological conditions (Khalil et al., 2024).
Precision Probiotics: Next-generation probiotics are being engineered to colonize the gut more effectively, deliver specific therapeutic compounds, or target particular disease states.
Postbiotics: Beyond live bacteria, researchers are investigating the therapeutic potential of microbial metabolites (like specific SCFAs or bacterial cell wall components) administered directly as treatments.
Microbiome-Based Diagnostics: Stool-based microbiome profiling is moving closer to clinical use as a non-invasive diagnostic tool for conditions including colorectal cancer, IBD, and metabolic syndrome (Ding et al., 2026).
Dietary Precision Medicine: Algorithms that combine an individual's microbiome profile, genetics, blood sugar responses, and lifestyle data to generate personalized dietary recommendations are already in early clinical trials (Barb & Wallen, 2025).
Frequently Asked Questions (FAQs)
Q1. What is the difference between probiotics and prebiotics — and do I need both?
Probiotics are live beneficial microorganisms found in fermented foods (yogurt, kefir, kimchi) or supplements. Prebiotics are non-digestible dietary fibers (found in garlic, oats, bananas, etc.) that serve as food for your existing gut bacteria. Both are important — probiotics introduce beneficial microbes, while prebiotics sustain them. Think of prebiotics as the fertilizer and probiotics as the seeds (Barb & Wallen, 2025).
Q2. How quickly can my diet change my microbiome?
Remarkably quickly. Research shows that significant shifts in microbiome composition can occur within 24 to 72 hours of a major dietary change. However, lasting, beneficial changes require consistent dietary habits over weeks to months. The good news: the microbiome is highly responsive to improvement (Rondinella et al., 2025).
Q3. Can stress really affect my gut bacteria?
Yes — significantly. The gut-brain axis is a two-way street. Chronic psychological stress alters gut motility, immune function, and microbial composition, reducing diversity and promoting dysbiosis. Conversely, an imbalanced microbiome can amplify stress responses and contribute to anxiety and depression (Blok & Bozadjieva-Kramer, 2025).
Q4. Are probiotic supplements worth taking?
For generally healthy individuals eating a varied diet, high-quality probiotic foods likely provide as much benefit as supplements. Supplements may be particularly valuable after antibiotic treatment, during travel, or for specific conditions like IBS. Always choose products with clinically studied strains and discuss with a healthcare provider if you have a health condition (Zhang et al., 2024).
Q5. Does the microbiome affect mental health and brain function?
Increasingly, yes. The gut-brain axis involves neural, hormonal, and immune pathways, and gut bacteria produce neurotransmitters and neuroactive compounds including serotonin precursors, GABA, and dopamine. Dysbiosis has been associated with higher rates of anxiety, depression, and even cognitive decline, though this field is still developing (Blok & Bozadjieva-Kramer, 2025).
Q6. What are the warning signs of an unhealthy gut microbiome?
Common signs of dysbiosis include: chronic bloating, gas, diarrhea, or constipation; frequent infections or illness; persistent fatigue; unexplained skin issues like eczema or acne; food intolerances; and mood disturbances. If you experience several of these symptoms persistently, a healthcare evaluation is warranted (Khalil et al., 2024).
Q7. Is it possible to completely restore the microbiome after damage from antibiotics or poor diet?
The microbiome shows remarkable resilience and capacity for recovery. With consistent dietary improvement — especially increased dietary fiber, fermented foods, and plant variety — most people can significantly restore microbial diversity within weeks to months. Severe or prolonged disruptions may take longer, and some researchers suggest that truly "resetting" the microbiome may require sustained lifestyle changes over one to two years (Zhang et al., 2024; Khalil et al., 2024).
Clinical pearls that bridge high-level metabolic science with actionable patient-facing advice.
1. The "Chemical Factory" Concept
Scientific Perspective: The gut microbiome functions as an auxiliary endocrine and metabolic organ. Microbial enzymes facilitate pathways human DNA cannot, such as the deconjugation of bile acids and the biotransformation of polyphenols into bioavailable metabolites like urolithin A.
Think of your gut as a personal chemical factory. You don't just "digest" food; your bacteria take raw materials (like fiber) and manufacture "medicines" (vitamins and anti-inflammatories) that your own body isn't capable of making on its own.
2. Diversity as a Marker of Resilience
Scientific Perspective: Alpha-diversity (within-sample variety) is a primary proxy for microbiome health. Higher diversity correlates with a more robust "functional redundancy," meaning if one microbial species is suppressed (e.g., by antibiotics), others are available to maintain essential metabolic niches and immune signaling.
Your gut is like a rainforest. A forest with only one type of tree is fragile; a forest with thousands of species can survive a storm. Eating 30+ different plants a week ensures you have a diverse "crew" ready to handle any health challenge.
3. The "Postbiotic" Payoff
Scientific Perspective: The clinical benefit of fiber is largely mediated by Short-Chain Fatty Acids (SCFAs), specifically butyrate. Butyrate serves as the primary energy source for colonocytes and acts as a histone deacetylase (HDAC) inhibitor, which regulates gene expression related to inflammation and cell growth.
Fiber isn't just "bulk" to help you go to the bathroom. It is the fuel that allows your bacteria to poop out butyrate—a super-fuel that repairs your gut lining and tells your immune system to "calm down."
4. The Speed of Microbial Plasticity
Scientific Perspective: The microbiome exhibits rapid temporal dynamics; significant shifts in the "metatranscriptome" (what the bacteria are doing) can occur within 24–48 hours of a dietary intervention. However, long-term "engraftment" of new species requires consistent substrate availability (prebiotics) to prevent a return to the baseline dysbiotic state.
Your gut is incredibly forgiving and fast. You can start changing your internal chemistry with your very next meal. While one salad won't fix everything, your bacteria start shifting their behavior the moment the fiber hits your system.
5. The Barrier-Immunity Link
Scientific Perspective: Intestinal permeability ("Leaky Gut") is often a consequence of a depleted mucus layer. Certain bacteria, like Akkermansia muciniphila, are specialized "mucus-munchers" that stimulate the body to produce more mucus, thereby strengthening the physical barrier against systemic endotoxemia (LPS entry into the blood).
Your gut has a protective "coat" of mucus that keeps toxins out of your blood. When you eat the right foods (like polyphenols and fiber), you encourage "guardian" bacteria to keep that shield thick and strong, preventing the "leaks" that lead to fatigue and inflammation.
Call to Action: Take Charge of Your Gut Health Today
Your gut microbiome is one of the most powerful and responsive systems in your body — and the choices you make every single day shape it profoundly. The science is clear: a diverse, balanced microbiome is not a luxury; it is a foundation of lifelong health.
Here is your 3-step action plan to get started:
✅ Step 1 — Audit your plate this week. Track how many different plant foods you eat in 7 days. Aim to add at least 5 new plant varieties next week.
✅ Step 2 — Add one fermented food daily. Start with a daily serving of live-culture yogurt, kefir, or a small portion of kimchi or sauerkraut.
✅ Step 3 — Book a check-in with your healthcare provider if you are experiencing digestive symptoms, fatigue, or recurrent illness. Ask about microbiome-informed approaches to your care.
Author’s Note
The science of the gut microbiome has evolved rapidly over the past decade, transitioning from descriptive microbiology to a more integrated understanding of human physiology. As a clinician in internal medicine, I have observed a growing gap between emerging microbiome research and its practical application in everyday patient care. This article was written to bridge that gap—translating complex, evolving evidence into clinically meaningful, actionable insights.
While the associations between the gut microbiome and chronic diseases such as obesity, diabetes, cardiovascular disease, and autoimmune conditions are compelling, it is important to recognize that much of the current evidence remains correlative rather than strictly causal. The microbiome is not an isolated system but part of a dynamic network involving genetics, diet, environment, and lifestyle. Therefore, microbiome-targeted strategies should be viewed as complementary—not replacement—approaches within a broader framework of evidence-based medicine.
Importantly, the field is shifting from identifying “which microbes are present” to understanding what they do—their metabolic functions, gene activity, and interaction with the host. Concepts such as postbiotics, metabolomics, and microbial gene richness represent the next frontier in precision medicine, though their routine clinical use is still evolving.
My goal is to empower both clinicians and patients with science-backed clarity, while maintaining a critical, evidence-based perspective. As research advances, our understanding will continue to refine—and so should our recommendations.
This article is intended for educational purposes only and does not constitute medical advice. Always consult a qualified healthcare professional before making significant changes to your diet, lifestyle, or supplement regimen.
Related Articles
Your Gut Bacteria Are What You Eat: A Science-Backed Guide to the Gut Microbiome | DR T S DIDWAL
Feed Your Gut, Fuel Your Health: Diet, Microbiota, and Systemic Health
References
Barb, J. J., & Wallen, G. R. (2025). Emerging research on the relationship between diet, gut microbes, and human health. Nutrients, 17(16), 2627. https://doi.org/10.3390/nu17162627
Blok, N. B., & Bozadjieva-Kramer, N. (2025). Unlocking the gut: Microbiome's role in energy metabolism. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, 329(2), R326–R328. https://doi.org/10.1152/ajpregu.00158.2025
Ding, Y., Zhang, Z., Wang, K., et al. (2026). The microbiome regulates host metabolic health and diseases through microbial enzymes. Nature Reviews Gastroenterology & Hepatology. https://doi.org/10.1038/s41575-026-01195-8
Khalil, M., Di Ciaula, A., Mahdi, L., Jaber, N., Di Palo, D. M., Graziani, A., Baffy, G., & Portincasa, P. (2024). Unraveling the role of the human gut microbiome in health and diseases. Microorganisms, 12(11), 2333. https://doi.org/10.3390/microorganisms12112333
Rondinella, D., Margarita, E., Raoul, P. C., Gasbarrini, A., & Cammarota, G. (2025). The impact of diet on gut microbiome composition: Implications for immune-mediated diseases. Clinical Immunology Communications, 9, 1–11. https://doi.org/10.1016/j.clicom.2025.12.001
Zhang, Y., Wang, H., Sang, Y., et al. (2024). Gut microbiota in health and disease: Advances and future prospects. MedComm, 5, e70012. https://doi.org/10.1002/mco2.70012