Your Gut Bacteria Are What You Eat: A Science-Backed Guide to the Gut Microbiome
Discover how diet shapes your gut microbiome and impacts immunity, metabolism, and brain health. Learn evidence-based ways to improve gut bacteria naturally.
NUTRITION
Dr. T.S. Didwal, M.D.(Internal Medicine)
5/3/202613 min read
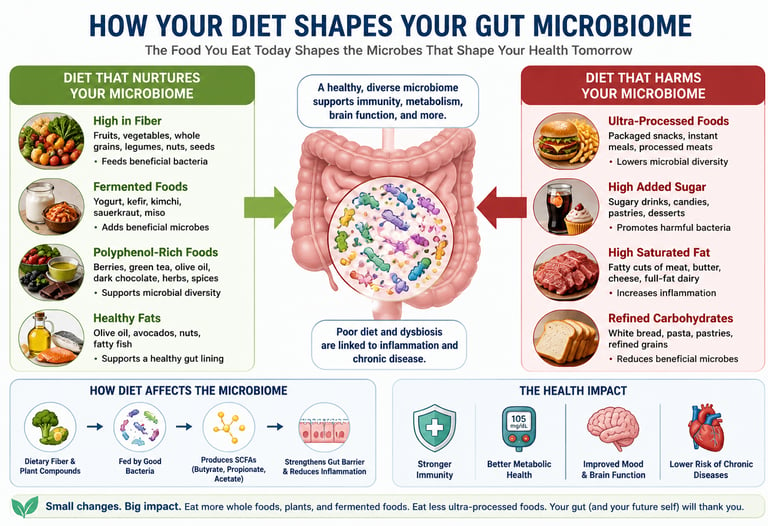
Diet is the most powerful modifiable factor shaping the gut microbiome. High-fibre, plant-rich, and Mediterranean-style diets increase beneficial bacteria and short-chain fatty acid production, improving immune, metabolic, and brain health. In contrast, ultra-processed foods and high saturated fat diets reduce microbial diversity and promote inflammation. Even small dietary changes can alter the microbiome within days.
Diet vs Microbiome Impact
Mediterranean Diet
Promotes high microbial diversity
Increases short-chain fatty acid (SCFA) production
Associated with lower systemic inflammation
Plant-Based Diet
Supports rich and diverse gut microbiota
Enhances SCFA production through high fiber intake
Linked to reduced inflammatory markers
Western Diet
Leads to reduced microbial diversity (dysbiosis)
Decreases beneficial SCFA production
Promotes chronic low-grade inflammation
Key Takeaway
Diet quality directly determines microbiome health:
Whole, fiber-rich diets → diversity + anti-inflammatory effects
Ultra-processed, high-fat diets → dysbiosis + inflammation
What is the fastest way to improve your gut microbiome?
Increase dietary fiber to 25–38 g/day
Eat 30+ plant foods per week
Add fermented foods daily
Reduce ultra-processed foods
Maintain consistent meal timing
Strength of Evidence
Strong evidence: dietary fiber, Mediterranean diet
Moderate evidence: fermented foods, probiotics
Emerging evidence: personalized microbiome-based diets
Clinical Takeaways
Diet shapes microbiome more than genetics in most adults
Fiber intake is the strongest predictor of microbial diversity
Ultra-processed foods consistently reduce beneficial bacteria
Microbiome changes precede metabolic disease
Inside your gut lives a dense, dynamic ecosystem of nearly 38 trillion microorganisms—collectively known as the gut microbiome—that functions almost like a hidden metabolic organ. Far from being passive passengers, these microbes actively regulate immune signaling, influence glucose and lipid metabolism, synthesize essential vitamins, and produce bioactive compounds that communicate with the brain and other organs (Sanz et al., 2025; Ross et al., 2024). Emerging research shows that disruptions in this ecosystem—often termed dysbiosis—are associated with a wide spectrum of chronic conditions, including obesity, type 2 diabetes, cardiovascular disease, and inflammatory disorders (Barb & Wallen, 2025).
What determines whether this internal ecosystem thrives or deteriorates? Among all modifiable factors, diet stands out as the most powerful and consistent driver of gut microbiome composition and function. Large-scale analyses and controlled trials demonstrate that dietary patterns rich in fiber, plant diversity, and fermented foods enhance microbial diversity and promote the production of short-chain fatty acids—key metabolites linked to reduced inflammation and improved metabolic health (Aslam et al., 2026; Muigano et al., 2025). In contrast, Western-style diets high in ultra-processed foods and saturated fats rapidly shift the microbiome toward a pro-inflammatory profile.
In essence, every meal you consume is not just nutrition for you—it is a biological signal shaping your microbiome and, in turn, your long-term health.
38T Microbial cells estimated in the human gut
1,000+ Bacterial species identified in the human gut1
2–4 Days dietary changes begin altering the microbiome6
80+ Controlled clinical trials reviewed in recent systematic analysis
Your gut microbiome: a brief introduction
The human gut microbiome refers to the collective genome of all the microorganisms residing in the gastrointestinal tract. It is one of the most complex ecosystems on Earth — and it is yours personally. No two people share an identical microbial fingerprint, shaped as it is by genetics, birth mode, infant feeding, early antibiotic exposure, environment, and crucially, diet.
A healthy, diverse microbiome is associated with robust immune function, better metabolic regulation, lower inflammatory markers, and even improved mental health through what researchers call the gut-brain axis. Conversely, reduced microbial diversity — a state called dysbiosis — has been linked to conditions ranging from irritable bowel syndrome and inflammatory bowel disease to obesity, type 2 diabetes, cardiovascular disease, and depression.
"The gut microbiome connects nutrition and human health in ways that are far more intricate than previously understood — it acts as a metabolic organ in its own right."— Sanz et al., Nature Reviews Gastroenterology & Hepatology (2025)
Science is evolving rapidly. As Asnicar et al. (2026) demonstrated in a landmark study, specific gut microbial species can be identified as robust biomarkers not just of disease risk, but of dietary exposure and overall nutritional status — meaning your microbiome may one day serve as a living readout of how well you are truly eating.
Dietary patterns that nourish your gut bacteria
The Mediterranean diet
Perhaps no dietary pattern has been more thoroughly studied in relation to gut health than the Mediterranean diet — rich in olive oil, legumes, whole grains, fish, vegetables, and fruits, with minimal processed foods and red meat. Multiple studies in this review confirm it is associated with higher microbial diversity, greater abundance of short-chain fatty acid (SCFA)-producing bacteria, and lower gut permeability.
SCFAs, particularly butyrate, propionate, and acetate, are produced when gut bacteria ferment dietary fibre. They are the gut lining's primary fuel source and have anti-inflammatory effects throughout the body. Muigano et al. (2025) highlight that fibre-rich dietary patterns dramatically increase SCFA production, which in turn supports the integrity of the intestinal barrier — reducing the risk of what is colloquially known as "leaky gut."
Plant-based and whole-food diets
The data consistently show that plant-rich diets — including vegetarian and vegan approaches — are associated with more favourable microbial profiles. Ross et al. (2024) emphasize that the interplay between diet and the gut microbiome is bidirectional: plant polyphenols, for example, reshape the microbiome in ways that then enhance the bioavailability of those same polyphenols. This creates a virtuous cycle where healthy eating literally upgrades your ability to benefit from healthy eating.
Wang (2025) points out that dietary habits can alter microbial composition within days — meaning the microbiome is surprisingly responsive to change. A single week of significantly increased vegetable and legume intake has been shown to measurably shift bacterial populations toward more health-promoting species.
Why fibre is the gut microbiome's favourite food
Dietary fibre is indigestible by human enzymes but fermentable by gut bacteria. When bacteria break down fibre, they produce SCFAs that protect the gut lining, reduce systemic inflammation, and may lower the risk of colorectal cancer. Yet most adults in high-income countries consume only half the recommended 25–38g of fibre per day.
Fermented foods
Fermented foods — yogurt, kefir, kimchi, sauerkraut, miso, tempeh — represent a direct delivery route for live microorganisms into the gut. Sanz et al. (2025) note that regular fermented food consumption is linked to enhanced microbial diversity and reduced circulating inflammatory proteins. Aslam et al. (2026), in their systematic review of 80 controlled clinical trials, found that probiotic and fermented food interventions produced the most consistent improvements in gut microbial outcomes across study populations.
Dietary patterns that harm the microbiome
The evidence cuts both ways. Just as some dietary patterns nurture gut health, others actively disrupt it.
Ultra-processed foods and added sugars
Ultra-processed foods — packaged snacks, ready meals, sugary drinks, refined bread products — are associated with reduced microbial diversity, increased inflammatory Proteobacteria, and depleted populations of beneficial Bifidobacterium and Lactobacillus species. Ray & Shankaran (2026) describe the gut microbiome and nutrition as being in a "symbiotic dialogue" — and note that ultra-processed foods fundamentally disrupt this conversation, leaving the microbiome impoverished and more susceptible to pathogenic colonization.
High saturated fat diets
Diets high in saturated fat, particularly those associated with Western eating patterns, tend to promote bile acid-tolerant bacteria while suppressing fibre-fermenting species. This shift is linked to increased gut permeability, systemic endotoxemia (where bacterial lipopolysaccharides leak into the bloodstream), and downstream metabolic dysfunction, including insulin resistance and non-alcoholic fatty liver disease.
The Western diet warning
Barb & Wallen (2025) note that the emerging research on diet-microbiome relationships is particularly urgent in the context of Western dietary patterns — high in saturated fat, refined carbohydrates, and ultra-processed foods, and low in fibre — which dominate food environments in many high-income countries and are now spreading globally through nutrition transition.
Antibiotic overuse and gut disruption
While not a dietary pattern per se, antibiotic exposure is a critical modifier of gut microbiome health. A single course of broad-spectrum antibiotics can reduce microbial diversity by 25–50% and drive long-term compositional shifts. The microbiome often fails to fully recover to its pre-antibiotic state — a finding that reinforces the importance of antibiotic stewardship and of supporting gut recovery through diet after necessary antibiotic use.
The gut-brain, gut-immune, and gut-metabolic axes
The microbiome does not work in isolation. It communicates with virtually every organ system through an elaborate network of chemical signals, nerve pathways, immune intermediaries, and circulating metabolites.
The gut-brain axis — a bidirectional communication network involving the vagus nerve, the enteric nervous system, and microbially produced neurotransmitters like serotonin and GABA — has attracted enormous scientific interest. Over 90% of the body's serotonin is produced in the gut, and gut bacteria play a direct role in its synthesis. This may help explain why gut dysbiosis is increasingly associated with anxiety, depression, and cognitive impairment.
The gut-immune axis is equally consequential. Approximately 70% of immune cells reside in the gut-associated lymphoid tissue. The microbiome trains these immune cells, helps distinguish self from non-self, and calibrates inflammatory responses. Ross et al. (2024) emphasise that dietary modulation of the microbiome may offer one of the most accessible routes to managing chronic inflammatory diseases.
On the metabolic front, Asnicar et al. (2026) identified specific microbial species strongly associated with beneficial cardiometabolic markers — lower triglycerides, better blood glucose regulation, healthier BMI — and traced these associations back to specific dietary exposures, particularly high fibre and fermented food intake.
"Diet-microbiome interactions are not simply about what we eat — they are about how our gut bacteria transform what we eat into signals that regulate the entire body."— Ross et al., Nature Reviews Microbiology (2024)
Personalised nutrition: the microbiome as a health compass
One of the most exciting frontiers in this field is personalised nutrition — the idea that individuals respond differently to the same foods, partly because of differences in their microbiome. This was compellingly illustrated by research showing that two people eating identical meals can have dramatically different blood glucose responses, predicted in part by their gut microbial composition.
Asnicar et al. (2026) found that gut microbial profiles could predict dietary exposure and health outcomes with remarkable accuracy, suggesting that microbiome profiling may soon become a clinical tool for tailoring dietary recommendations. Sanz et al. (2025) echo this vision, arguing that integrating microbiome data with nutritional guidance represents the next frontier in preventive medicine.
For now, while personalised microbiome testing is not yet standard clinical practice, the broad dietary principles that support a diverse and healthy microbiome apply across populations — even if the exact magnitude of response varies between individuals.
Practical applications: what you can start doing today
The science is clear, and the good news is that the microbiome is remarkably plastic — meaning it responds to dietary change relatively quickly. Here are evidence-based, practical steps grounded in the research reviewed here.
Eat the rainbow of plants
Aim for 30+ different plant foods per week. Diversity in plants drives diversity in gut bacteria. Include vegetables, fruits, legumes, whole grains, nuts, seeds, and herbs.
Prioritise dietary fibre
Target 25–38g of fibre daily from whole food sources: oats, lentils, beans, broccoli, berries, apples, and wholegrains. Fibre is the primary prebiotic that feeds your beneficial bacteria.
Add fermented foods daily
Incorporate plain yogurt, kefir, kimchi, sauerkraut, miso, or kombucha. Even small daily servings have been shown to increase microbial diversity within weeks.
Reduce ultra-processed foods
Replace packaged snacks, sugary drinks, and refined carbohydrates with whole food alternatives. Even partial replacement improves gut microbial composition.
Embrace polyphenol-rich foods
Berries, dark chocolate (70%+), green tea, olive oil, red wine (in moderation), and colourful vegetables are rich in polyphenols that selectively feed beneficial microbes.
Maintain a consistent eating rhythm
Circadian disruption from erratic meal timing negatively affects gut microbial composition. Consistent mealtimes support both microbiome health and metabolic regulation.
Start small: the 1-per-week rule
Research suggests you do not need to overhaul your diet overnight. Adding just one new plant food per week — a new legume, vegetable, or whole grain — creates meaningful microbial diversity over months. Small, sustainable steps outperform dramatic short-term changes.
Frequently asked questions
How quickly can diet change my gut microbiome?
Surprisingly quickly. Wang (2025) and other researchers have shown that measurable changes in gut microbial composition can appear within 2 to 4 days of a significant dietary shift. However, more stable, lasting changes — particularly increases in microbial diversity — typically require consistent dietary change over several weeks to months. Think of it like exercise: one workout matters, but consistent training changes your physiology.
Do I need to take probiotic supplements?
Not necessarily. While probiotic supplements can be beneficial in specific contexts — particularly after antibiotic use or in certain gastrointestinal conditions — food-based sources of probiotics (yogurt, kefir, kimchi, sauerkraut) are generally preferred by researchers. Aslam et al.'s (2026) systematic review of 80 clinical trials found that whole-food dietary interventions produced robust and consistent gut microbiome improvements. More importantly, prebiotics — the fibre that feeds your existing bacteria — may be even more impactful than probiotics for most healthy adults.
Is there one "best" diet for gut health?
No single diet is universally optimal, because gut microbiome composition varies significantly between individuals. However, the common thread across all the research reviewed here is dietary diversity and plant richness. Mediterranean-style, whole-food-based eating patterns consistently produce the most favourable gut microbial profiles. Emerging personalised nutrition science (Asnicar et al., 2026) suggests that microbiome profiling may one day allow truly individualised dietary recommendations.
Can gut health affect my mental health?
Yes — and this is one of the most actively researched areas in gut-brain science. The gut and brain communicate through the vagus nerve, immune signals, and gut-produced neurotransmitters including serotonin (over 90% of which is made in the gut) and GABA. Gut dysbiosis is increasingly associated with anxiety, depression, and cognitive decline. While the science is still developing and dietary change alone should not replace mental health treatment, supporting a healthy microbiome through diet is considered a meaningful complementary strategy by researchers including Sanz et al. (2025).
How much fibre should I eat per day?
Current guidelines recommend 25g of dietary fibre per day for women and 38g for men. Most adults in Western countries consume roughly half that — around 15g. The research reviewed here consistently links higher fibre intake (from whole foods, not supplements) with greater microbial diversity and SCFA production. Practical targets: one serving of legumes (lentils, beans, chickpeas), one serving of whole grains, at least five portions of vegetables and fruit, and a small handful of nuts or seeds daily will put most people in the right range.
What happens to the gut microbiome when we take antibiotics?
Antibiotics are life-saving medicines, but they do not discriminate between harmful and beneficial bacteria. A single course of broad-spectrum antibiotics can reduce gut microbial diversity by 25–50%, with some studies showing incomplete recovery of certain species even months later. To support gut recovery after necessary antibiotic use: increase fermented foods and fibre-rich plant foods, consider a clinician-recommended probiotic, and avoid unnecessary follow-up antibiotic courses. Ross et al. (2024) discuss the post-antibiotic gut recovery period in detail as part of the broader diet-microbiome interaction story.
Are gut microbiome tests worth it?
Consumer-level microbiome tests are improving but are not yet considered clinically actionable by most healthcare guidelines. They can provide interesting insight and may motivate dietary change, but they cannot yet reliably diagnose conditions or prescribe specific interventions. The research field is moving rapidly — Asnicar et al. (2026) and Sanz et al. (2025) both describe a near-future where microbiome data integrates meaningfully with clinical nutrition. For now, the broad dietary principles supporting microbiome diversity are far more evidence-based than personalised test-and-treat approaches. If you are interested in testing, discuss it with your healthcare provider first.
Clinical pearls
1. The "Plasticity" of the Microbiome
Scientific Perspective: Metabolic and compositional shifts in gut microbiota occur rapidly. Research (Wang, 2025) indicates that dietary interventions can alter microbial gene expression and population abundance within 48 to 96 hours.
You aren't stuck with the gut you have today. Your "inner city" is incredibly responsive; if you start eating better on Monday, your microbiome is already beginning to remodel itself by Thursday.
2. Fiber as a Metabolic Fuel (SCFAs)
Scientific Perspective: Dietary fiber is the precursor to Short-Chain Fatty Acids (SCFAs) like butyrate. These metabolites are ligands for G-protein coupled receptors that regulate systemic inflammation and maintain the integrity of the intestinal epithelial barrier.
Think of fiber not just as "roughage" for regularity, but as the actual fuel your good bacteria need to produce "anti-inflammatory medicine" for your body. Without fiber, these bacteria starve, and your gut lining can become weak.
3. The "30-Plant" Diversity Rule
Scientific Perspective: Microbial alpha-diversity (the variety of species) is a primary marker of ecosystem resilience. The "American Gut Project" and subsequent studies (Asnicar et al., 2026) show that individuals consuming 30 or more types of plants per week have significantly more diverse microbiomes than those consuming fewer than ten.
Don't just eat "healthy"—eat "diverse." Instead of just buying spinach every week, rotate through kale, arugula, and chard. Diversity in your grocery cart leads to diversity in your gut.
4. The Gut-Brain Communication Hub
Scientific Perspective: The gut produces over 90% of the body’s serotonin. Through the vagus nerve and the production of neurotransmitters like GABA, the microbiome modulates the hypothalamic-pituitary-adrenal (HPA) axis, directly influencing stress responses and mood.
Your gut is essentially a "second brain." If you are feeling anxious or foggy, it might not just be in your head—it could be a reflection of an imbalanced gut. Eating for your microbes is a legitimate form of mental health self-care.
5. Post-Antibiotic Recovery Strategy
Scientific Perspective: A single course of broad-spectrum antibiotics can deplete microbial diversity by up to 50%, creating a "vacant niche" that opportunistic pathogens can exploit. Recovery to baseline is often incomplete without targeted nutritional support.
If you must take antibiotics, think of it as a "forest fire" in your gut. Once the medicine is finished, you need to "replant" the forest immediately using fermented foods (probiotics) and "fertilize" it with fiber (prebiotics) to prevent weeds from growing back.
Author’s Note
This article synthesizes current evidence from high-quality peer-reviewed research to explain how dietary patterns shape the gut microbiome and influence systemic health. The science of diet–microbiome interactions is evolving rapidly, with new insights emerging from multi-omics, longitudinal cohorts, and controlled clinical trials. While the associations between diet, microbial diversity, and health outcomes are increasingly robust, it is important to recognize that causality is still being clarified, and individual responses to dietary interventions can vary significantly.
From a clinical perspective, the most consistent and actionable finding is that diet quality—particularly fiber intake, plant diversity, and minimization of ultra-processed foods—remains central to maintaining a healthy microbiome. However, microbiome science should not yet be overinterpreted to support highly restrictive or commercially driven dietary claims. Personalized nutrition based on microbiome profiling is a promising frontier, but it is not yet a standard of care.
This content is intended for educational purposes and to support informed decision-making. Patients with existing medical conditions—especially gastrointestinal, metabolic, or immunological disorders—should consult a qualified healthcare professional before making significant dietary changes
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Individual circumstances vary, and treatment decisions should always be made in consultation with qualified healthcare professionals.
Related Articles
Your Gut Microbiome Explained: How It Controls Immunity, Metabolism, and Disease | DR T S DIDWAL
Feed Your Gut, Fuel Your Health: Diet, Microbiota, and Systemic Health
References
Muigano, M. N., Liu, J., Liu, X., Luo, P., Li, Z., & Li, J. (2025). The impact of dietary patterns on the human gut microbiome and its health significance: A review. The FASEB Journal, 39(19), e71072. https://doi.org/10.1096/fj.202502040R
Asnicar, F., Manghi, P., Fackelmann, G., et al. (2026). Gut micro-organisms associated with health, nutrition and dietary interventions. Nature, 650, 450–458. https://doi.org/10.1038/s41586-025-09854-7
Ross, F. C., Patangia, D., Grimaud, G., et al. (2024). The interplay between diet and the gut microbiome: Implications for health and disease. Nature Reviews Microbiology, 22, 671–686. https://doi.org/10.1038/s41579-024-01068-4
Sanz, Y., Cryan, J. F., Deschasaux-Tanguy, M., et al. (2025). The gut microbiome connects nutrition and human health. Nature Reviews Gastroenterology & Hepatology, 22, 534–555. https://doi.org/10.1038/s41575-025-01077-5
Ray, S., & Shankaran, P. (2026). Nutrition and the gut microbiome: A symbiotic dialogue influencing health and disease. Frontiers in Nutrition, 13, 1761992. https://doi.org/10.3389/fnut.2026.1761992
Barb, J. J., & Wallen, G. R. (2025). Emerging research on the relationship between diet, gut microbes, and human health. Nutrients, 17(16), 2627. https://doi.org/10.3390/nu17162627
Wang, X. (2025). Unveiling the gut microbiota: How dietary habits shape health through microbiome modulation. Nutrients, 17(10), 1650. https://doi.org/10.3390/nu17101650
Aslam, H., Trakman, G., Dissanayake, T., et al. (2026). Dietary interventions and the gut microbiota: A systematic literature review of 80 controlled clinical trials. Journal of Translational Medicine, 24, 39. https://doi.org/10.1186/s12967-025-07428-9