Why Do Muscles Weaken With Age? The Mitochondria Link Explained
Discover why muscles weaken with age and how mitochondria drive it. Learn science-backed ways to rebuild strength with exercise and nutrition
EXERCISESARCOPENIA
Dr. T.S. Didwal, M.D.(Internal Medicine)
4/26/202615 min read
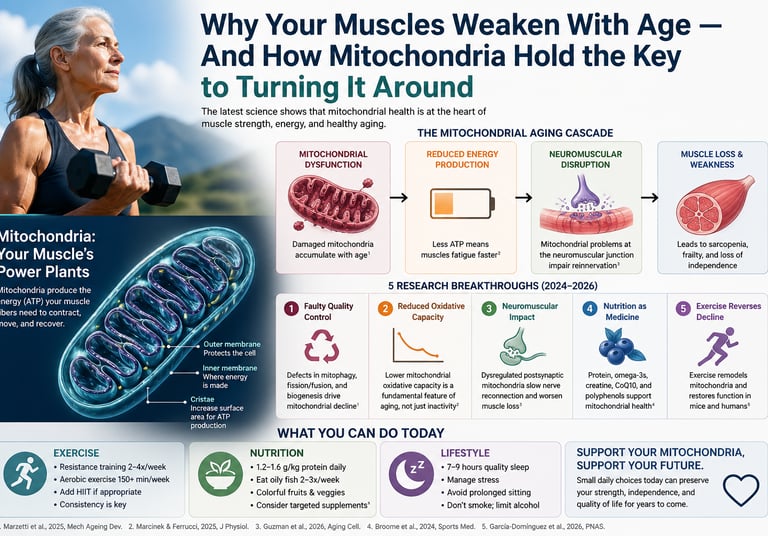
Key Causes of Muscle Weakness with Age
Decline in mitochondrial function and energy production
Reduced the oxidative capacity of muscle cells
Impaired neuromuscular junction (nerve–muscle signalling)
Loss of muscle mass and quality (sarcopenia)
Clinician’s Perspective: What This Means for You
Muscle weakness is not just “aging”—it’s biology you can influence.
What we now understand clinically is that declining strength is largely driven by reduced cellular energy production (mitochondrial dysfunction), not simply loss of muscle size. This means decline is modifiable, not inevitable.Early action matters more than late correction.
Mitochondrial changes begin as early as midlife. Patients who start regular exercise and adequate protein intake in their 40s and 50s tend to maintain better strength and independence later.Exercise is your most effective “medicine.”
From a clinical standpoint, no drug matches the mitochondrial benefits of exercise. A combination of resistance training (for muscle mass) and aerobic activity (for mitochondrial function) provides the strongest protection.Strength is more important than weight.
Many patients focus only on body weight, but muscle strength and quality are far better predictors of longevity, metabolic health, and fall risk.Protein intake is often underestimated.
In practice, most older adults consume less protein than needed. Spreading protein intake evenly across meals (rather than one large meal) improves muscle protein synthesis and mitochondrial signaling.Fatigue is a clinical signal—not just “tiredness.”
Persistent fatigue during routine activities may reflect declining mitochondrial efficiency and should prompt evaluation of physical activity, nutrition, sleep, and metabolic health.Neuromuscular health is equally important.
Maintaining the connection between nerves and muscles requires regular movement and resistance exercise. Periods of inactivity (e.g., illness, bed rest) can accelerate decline.Supplements can help—but only as adjuncts.
Creatine, omega-3 fatty acids, and possibly CoQ10 may support mitochondrial function, but they do not replace exercise and diet.Small, consistent habits outperform extreme efforts.
Clinically, patients who adopt sustainable routines—walking daily, strength training twice weekly, improving sleep—achieve the best long-term outcomes.It is never too late to improve.
Even patients in their 70s and 80s show measurable gains in strength, energy, and function when these principles are applied consistently.
"You don't have to accept muscle loss as an inevitable part of getting older. The science is catching up — and it starts deep inside your cells."
Muscle weakness is often dismissed as an inevitable consequence of aging—but modern science is telling a far more precise and hopeful story. What if the real driver of declining strength, slower recovery, and reduced endurance isn’t just “getting older,” but a gradual breakdown in the microscopic energy systems that power every muscle contraction? At the center of this shift are mitochondria—the tiny, dynamic organelles responsible for generating the fuel your muscles depend on to function.
Emerging research shows that age-related muscle decline, or sarcopenia, is not simply a matter of losing muscle mass. It is increasingly understood as a problem of muscle quality and cellular energy failure, driven in large part by mitochondrial dysfunction (Marzetti et al., 2025). As mitochondrial quality control systems falter, damaged mitochondria accumulate, energy production declines, and muscle fibers become less efficient, more fatigable, and ultimately more prone to atrophy.
But the story goes even deeper. Mitochondrial dysfunction doesn’t just affect the muscle cell—it disrupts the neuromuscular connection itself, impairing the ability of nerves to communicate effectively with muscle fibres and accelerating functional decline (Guzman et al., 2026). At the same time, studies confirm that a reduction in mitochondrial oxidative capacity is a fundamental biological feature of aging, not merely a consequence of inactivity (Marcinek & Ferrucci, 2025).
Yet, there is a powerful and clinically important twist: these changes are not entirely irreversible. A growing body of evidence demonstrates that targeted interventions—particularly exercise and nutrition—can remodel mitochondrial function and restore aspects of muscle performance, even later in life (García-Domínguez et al., 2026; Broome et al., 2024).
In other words, the key to preserving strength, independence, and metabolic health with age may lie not just in your muscles—but deep within the mitochondria that power them.
What Is Sarcopenia — and Why Should You Care?
Before we dive into the mitochondria research, it helps to know what's at stake. Sarcopenia is the medical term for the progressive loss of skeletal muscle mass and function that occurs with aging. It affects an estimated 10–40% of older adults and is directly linked to:
Increased risk of falls and fractures
Loss of independence and mobility
Reduced quality of life
Higher risk of metabolic diseases like type 2 diabetes
Sarcopenia doesn't happen overnight. It's a slow, decades-long process — and mitochondria sit at the very heart of it.
The Five Research Breakthroughs You Need to Know
1. Faulty Mitochondrial Quality Control Is Accelerating Muscle Aging
In a comprehensive 2025 review, Marzetti et al. outlined how aging muscles are plagued by defective mitochondrial quality control (MQC). Think of MQC as your cells' internal housekeeping system — a set of processes that continuously monitor mitochondria, repair damaged ones, recycle broken parts, and build new, healthy ones.
When MQC works properly, your muscle cells maintain a robust pool of high-functioning mitochondria. When it breaks down — as it does progressively with age — damaged mitochondria accumulate, energy production plummets, and muscle cells begin to deteriorate and die.
The review identified three core failures that drive this process:
Mitophagy breakdown: Mitophagy is the process by which cells identify and destroy damaged mitochondria. In aging muscles, this "selective recycling" becomes sluggish and inefficient, allowing dysfunctional mitochondria to build up like uncollected rubbish.
Fission and fusion imbalance: Healthy mitochondria constantly split apart (fission) and merge (fusion) to maintain quality and distribute energy. In aging muscles, this dynamic balance is disrupted, leading to fragmented, inefficient networks.
Impaired biogenesis: The capacity to build fresh, new mitochondria also declines with age, meaning the supply of healthy replacements dwindles just when it is needed most.
Together, these three failures create a vicious cycle: poor quality control → damaged mitochondria → reduced energy → muscle wasting → further decline in MQC.
2. Reduced Oxidative Capacity: A Fundamental — Not Optional — Consequence of Aging
A pointed 2025 commentary by Marcinek and Ferrucci challenges a debate that has persisted in physiology for years: Is the decline in mitochondrial energy production in older muscles simply because older people exercise less? Or is it an unavoidable biological consequence of aging itself?
Their answer, backed by rigorous evidence: both factors matter, but reduced mitochondrial oxidative capacity is a fundamental feature of adult aging, independent of physical activity levels.
Oxidative capacity refers to your muscles' ability to use oxygen to generate energy — the slow-burn, sustainable fuel system that powers everything from walking to gardening to climbing stairs. As this capacity declines, muscles fatigue more quickly, recover more slowly, and ultimately begin to waste.
Critically, the authors argue that failing to recognize this as a true biological consequence — rather than merely a lifestyle consequence — leads clinicians and researchers to underestimate how proactive interventions need to be. Waiting for someone to become sedentary before addressing mitochondrial decline may already be too late. This insight has profound implications for how we approach preventive medicine, exercise prescription, and nutrition in middle age and beyond.
3. The Nerve-Muscle Connection: Mitochondria at the Neuromuscular Junction
One of the most fascinating — and underappreciated — aspects of age-related muscle loss is what happens at the neuromuscular junction (NMJ): the tiny gap where nerve fibers talk to muscle fibers.
For your muscles to contract, your nervous system must send electrical signals down motor neurons to your muscle fibers. This signal transmission depends on healthy mitochondria at the NMJ. When those mitochondria become dysregulated with age, the nerve-muscle conversation breaks down.
A landmark 2026 study by Guzman et al. showed that age-associated mitochondrial dysregulation at postsynaptic sites (the muscle side of the NMJ) directly disrupts the kinetics of nerve reinnervation — that is, the muscle's ability to be "re-wired" after injury, disuse, or normal age-related denervation.
As muscles age, motor neurons periodically lose their connection to muscle fibers and need to re-establish contact — a process called reinnervation. If postsynaptic mitochondria aren't working correctly, this reconnection is delayed or incomplete, leaving muscle fibers permanently "orphaned" from their nerve supply. Over time, these denervated fibers simply atrophy and disappear.
This research opens a new front in our understanding of sarcopenia: it's not just about the muscle cell itself, but about the entire neuromuscular circuit — and mitochondria are central to keeping that circuit intact.
4. Nutrition as Medicine for Your Mitochondria
If the previous studies paint a sobering picture of age-related mitochondrial decline, the 2024 review by Broome et al. offers a genuinely encouraging counterpoint: what you eat can meaningfully influence your mitochondrial health.
This comprehensive review, published in Sports Medicine, systematically examined the evidence for nutritional strategies that target mitochondrial function in aging muscles. The findings are both scientifically compelling and practically empowering.
Key nutritional targets identified by the research include:
Protein and amino acids: Adequate dietary protein — particularly leucine, a branched-chain amino acid — stimulates mitochondrial biogenesis (the creation of new mitochondria) and helps preserve muscle mass. The review emphasizes that older adults typically need more dietary protein than current general guidelines suggest, often in the range of 1.2–1.6 g per kilogram of body weight per day.
Creatine: One of the most extensively studied sports supplements, creatine supports ATP (energy) regeneration in muscles, reduces mitochondrial oxidative stress, and may help buffer the age-related decline in mitochondrial function. Evidence supports its use in older adults, particularly when combined with resistance exercise.
Omega-3 fatty acids: Found in oily fish like salmon, mackerel, and sardines, omega-3s help maintain mitochondrial membrane integrity and reduce chronic low-grade inflammation — a key driver of mitochondrial dysfunction in aging muscles.
Polyphenols and antioxidants: Plant compounds such as resveratrol, quercetin, and those found in berries and green tea activate cellular pathways (notably SIRT1 and AMPK) that stimulate mitophagy and mitochondrial biogenesis.
Coenzyme Q10 (CoQ10): A critical component of the mitochondrial electron transport chain, CoQ10 levels naturally decline with age. Supplementation may support energy production in aging muscles, though the evidence is still developing.
The overarching message from Broome et al. is that nutrition is not an afterthought in mitochondrial health — it is a primary lever that individuals can begin pulling today.
5. Exercise Actually Reverses Mitochondrial Aging
Perhaps the most exciting finding in the entire field comes from a 2026 study by García-Domínguez et al., published in the Proceedings of the National Academy of Sciences. The researchers demonstrated something remarkable: exercise-induced mitochondrial remodeling in skeletal muscle can actually reverse age-associated functional decline — not just slow it, but genuinely turn it back.
The study examined both aged mice and older human participants who underwent structured exercise programs. In both cases, exercise triggered profound changes in the mitochondrial landscape of skeletal muscle:
Mitochondrial biogenesis was upregulated — muscles began building more new mitochondria
Mitophagy was enhanced — the cellular garbage collection system was reactivated, clearing out damaged mitochondria
Mitochondrial network dynamics were restored — the fission-fusion balance that breaks down with aging was significantly improved
Oxidative capacity increased — muscles became measurably better at generating energy through aerobic pathways
Most importantly, these mitochondrial changes translated directly into improved physical function: older participants showed measurable gains in muscle strength, aerobic capacity, and exercise performance that paralleled what was seen in their younger counterparts.
This study is a watershed moment. It confirms that the mitochondrial decline of aging is not fixed or inevitable — it is, at least in significant part, reversible through exercise, even when started later in life.
Practical Applications: What You Can Do Starting Today
The research reviewed above points to a clear, actionable framework for protecting — and even improving — your mitochondrial health and muscle function as you age. Here's how to put the science into practice:
Exercise: Your Most Powerful Mitochondrial Tool
Resistance training (2–4 sessions per week): Lifting weights, using resistance bands, or doing bodyweight exercises like squats and push-ups is the single most effective stimulus for maintaining muscle mass and mitochondrial function. Start with lighter weights if needed and progress gradually.
Aerobic exercise (150+ minutes per week): Brisk walking, swimming, cycling, or dancing directly stimulates mitochondrial biogenesis and oxidative capacity — the very processes that decline with age. Aim for moderate intensity (you can talk, but not sing).
High-Intensity Interval Training (HIIT): Short bursts of higher-intensity effort — such as 30-second fast walks alternating with 90-second slow recovery walks — are particularly potent triggers for mitochondrial biogenesis, even in older adults. Always consult your doctor before starting HIIT.
Consistency over intensity: The research shows that regular, sustained exercise produces the most meaningful mitochondrial adaptations. Three times a week, every week, beats one heroic weekend session.
Nutrition: Feed Your Mitochondria
Prioritize protein at every meal: Aim for 25–40 g of high-quality protein per meal — eggs, Greek yogurt, chicken, fish, legumes, or quality protein supplements. Don't skip breakfast protein.
Eat oily fish 2–3 times per week: Salmon, mackerel, sardines, and trout are rich in omega-3 fatty acids that support mitochondrial membrane health.
Fill half your plate with colourful vegetables and fruits: Polyphenols in berries, leafy greens, tomatoes, and onions activate key mitochondrial pathways.
Consider targeted supplements (with your doctor's guidance): Creatine monohydrate (3–5 g/day), omega-3 fish oil (2–3 g EPA+DHA/day), and CoQ10 (100–300 mg/day) have the strongest evidence base for mitochondrial support in older adults.
Stay well hydrated: Mitochondrial reactions depend on adequate cellular water balance. Aim for 6–8 glasses of water daily.
Lifestyle Factors That Protect Mitochondria
Prioritize sleep: Mitochondrial repair and mitophagy are upregulated during deep sleep. Aim for 7–9 hours of quality sleep per night.
Manage chronic stress: Sustained high cortisol levels suppress mitochondrial biogenesis and accelerate muscle wasting. Mindfulness, breathing exercises, and social connection all help.
Avoid prolonged sitting: Break up sedentary time every 30–60 minutes with even brief movement — a short walk, some gentle stretching, or standing for a few minutes can make a meaningful difference.
Don't smoke, and limit alcohol: Both directly impair mitochondrial function and accelerate muscle loss.
Frequently Asked Questions
Q1: At what age does mitochondrial decline in muscles typically begin?
Research suggests that mitochondrial function in skeletal muscle begins to decline gradually from as early as the mid-30s to early 40s, though the rate of decline accelerates significantly after age 60. This is why prevention strategies — particularly regular exercise and adequate protein intake — are most effective when started well before significant muscle loss has occurred.
Q2: Is muscle loss with aging inevitable, or can it truly be reversed?
The landmark 2026 study by García-Domínguez et al. provides compelling evidence that exercise-induced mitochondrial remodeling can reverse — not just slow — age-associated functional decline in muscles, even in older individuals. While some degree of structural change is biological, a substantial portion of what most people experience as "inevitable" muscle loss is actually driven by preventable factors like inactivity and poor nutrition.
Q3: How much protein do older adults actually need for muscle health?
Current research, including the review by Broome et al. (2024), suggests that older adults typically need significantly more protein than the general recommended dietary allowance — approximately 1.2–1.6 grams per kilogram of body weight per day, distributed across meals. For a 70 kg (154 lb) person, that equates to roughly 84–112 g of protein daily.
Q4: What is the best type of exercise for mitochondrial health in older adults?
Both resistance training and aerobic exercise are valuable and complementary. Resistance training directly builds and maintains muscle mass, while aerobic exercise is the most potent driver of mitochondrial biogenesis and oxidative capacity. The evidence most strongly supports a combination of both, with HIIT adding particular benefit for mitochondrial adaptation where health status permits.
Q5: Are mitochondrial supplements like CoQ10 worth taking?
CoQ10 levels naturally decline with aging and are important for mitochondrial energy production. The evidence for supplementation is promising — particularly for people already showing signs of mitochondrial-related fatigue — but is not yet conclusive enough for universal recommendations. Creatine and omega-3 fatty acids currently have stronger evidence bases for mitochondrial muscle support in older adults. Always discuss supplements with your healthcare provider first.
Q6: Does the neuromuscular junction really play a role in sarcopenia?
Yes, and this is an area of rapidly growing research interest. The 2026 study by Guzman et al. demonstrated that mitochondrial dysfunction at the neuromuscular junction disrupts the nerve-muscle reconnection process, contributing directly to the loss of muscle fibers with age. This means that protecting neuromuscular health — through exercise, anti-inflammatory nutrition, and adequate protein — is an important dimension of sarcopenia prevention.
Q7: Can someone in their 70s or 80s still meaningfully improve their mitochondrial health?
Absolutely. Multiple studies reviewed here involved older participants in their 60s, 70s, and beyond, and demonstrated meaningful improvements in mitochondrial function, oxidative capacity, and physical performance. The biology of mitochondrial adaptation responds to exercise and nutrition stimuli at any age. It is never too late to start — though the sooner, the better.
Clinical Pearls: Mitochondrial Health and Aging Muscle
1. The "Cellular Trash" Accumulation (Mitophagy)
Scientific Perspective: Age-related sarcopenia is partially driven by a decline in mitophagy—the selective autophagy of damaged mitochondria. When this "quality control" fails, dysfunctional organelles accumulate, increasing reactive oxygen species (ROS) production and triggering myofiber apoptosis.
Patient Perspective: Think of your muscle cells like a kitchen. For the kitchen to run well, you have to take the trash out every night. As we age, that "trash pickup" gets lazy. Damaged parts build up inside your muscles, making them sluggish and weak. Exercise is what forces the "trash pickup" to start working again.
2. The "Wiring" and "Spark Plug" Connection (NMJ)
Scientific Perspective: Mitochondrial dysregulation at the postsynaptic neuromuscular junction (NMJ) perturbs reinnervation kinetics. Even if the motor neuron is healthy, mitochondrial failure at the muscle interface prevents the fiber from being "re-wired," leading to permanent denervation and atrophy.
Patient Perspective: Your nerves are like electrical wires and your muscles are the engine. The mitochondria are the "spark plugs" where the wire meets the engine. If the spark plugs are dirty, the engine won't start, no matter how much the nerve tries to "flip the switch." Keeping mitochondria healthy ensures the connection stays strong.
3. Oxidative Capacity as a "Use It or Lose It" Factor
Scientific Perspective: A decline in mitochondrial oxidative capacity (the ability to use oxygen for ATP production) is a fundamental biological consequence of aging. However, chronic physical inactivity exacerbates this, creating a "secondary aging" effect that is largely preventable through aerobic stimuli.
Patient Perspective: Your muscles have two engines: a "sprint" engine and a "marathon" engine. The marathon engine (mitochondria) is what keeps you going during a long walk. Aging naturally slows this engine down, but sitting still for too long turns it off completely. Moving regularly keeps the "marathon" engine tuned up and ready.
4. Mitochondrial Biogenesis: Building New Factories
Scientific Perspective: High-Intensity Interval Training (HIIT) and resistance exercise act as potent stimulators of PGC-1α, the master regulator of mitochondrial biogenesis. This pathway allows for the expansion of the mitochondrial reticulum, effectively "remodeling" aged muscle tissue.
Patient Perspective: Exercise doesn't just make your current muscles bigger; it actually tells your body to build new power plants inside your cells. You aren't stuck with the "energy factories" you have now—you can actually renovate and expand them by challenging your muscles with weights or brisk movement.
5. The Anabolic Threshold and Protein Timing
Scientific Perspective: Older adults often exhibit anabolic resistance, requiring higher per-meal doses of leucine-rich protein (25–40g) to stimulate the mTOR pathway and mitochondrial protein synthesis. Sub-threshold protein intake fails to trigger the necessary "building signals" for mitochondrial repair.
Patient Perspective: When you’re younger, your body is "loud" and hears even small signals to build muscle. As you age, your body becomes "hard of hearing." You need a "louder" signal—meaning a larger, high-quality dose of protein at breakfast and dinner—to get your muscle factories to start their shift and begin repairs.
6. The Lipid Membrane and Omega-3s
Scientific Perspective: Mitochondrial function depends on the fluidity and integrity of the inner mitochondrial membrane, which is highly susceptible to lipid peroxidation. Omega-3 fatty acids incorporate into these membranes, enhancing mitochondrial sensitivity and reducing "electron leakage."
Patient Perspective: Your mitochondria have a "skin" or membrane that protects them. As we age, this skin can get stiff or leaky, causing energy to "leak out" like a rusty battery. Healthy fats from fish (Omega-3s) act like a sealant, keeping the membrane flexible and making sure the energy goes where it’s supposed to.
Author’s Note
This article was written to bridge an important gap between rapidly evolving science and everyday clinical practice. In recent years, our understanding of muscle aging has shifted dramatically—from a simplistic view of “muscle loss with age” to a far more nuanced model centered on mitochondrial health, neuromuscular integrity, and metabolic resilience. As a clinician, I find that many patients are still unaware of how profoundly these microscopic processes influence their strength, energy, and independence.
The goal here is not only to summarize current evidence but to translate complex physiology into practical, actionable insights that can be applied in real life. Every recommendation discussed—whether related to exercise, nutrition, or lifestyle—is grounded in peer-reviewed research and aligned with what we see in clinical settings. At the same time, it is important to acknowledge that individual responses can vary based on age, comorbidities, medications, and baseline fitness levels.
A key message I hope readers take away is this: muscle decline is not purely inevitable—it is, to a meaningful extent, modifiable. The biology of aging is real, but so is the body’s capacity to adapt, even later in life. Exercise, in particular, remains one of the most powerful and underutilized interventions in medicine—capable of influencing mitochondrial function, preserving neuromuscular connections, and improving overall metabolic health.
Finally, this article is intended for educational purposes and should complement—not replace—personalized medical advice. Patients are encouraged to discuss any significant changes in exercise, diet, or supplementation with their healthcare provider, especially in the presence of chronic conditions.
The science is advancing rapidly—but the fundamentals remain clear: move regularly, nourish wisely, and support your body at the cellular level.
This article is intended for educational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider before beginning a new exercise or nutrition program, especially if you have an existing medical condition.
Related Articles
Why Obesity Isn’t Just Fat: How Muscle Quality Controls Fat Loss and Metabolism
Neuromuscular Aging Explained: How Nerve–Muscle Breakdown Causes Weakness After 40
The mTOR Switch: Why Your Cells Stopped Listening to Insulin | DR T S DIDWAL
Can Resistance Training Reverse Diabetes? The Muscle–mTOR Link | DR T S DIDWAL
References
Broome, S. C., Whitfield, J., Karagounis, L. G., et al. (2024). Mitochondria as nutritional targets to maintain muscle health and physical function during ageing. Sports Medicine, 54, 2291–2309. https://doi.org/10.1007/s40279-024-02072-7
García-Domínguez, E., García-Domínguez, C., Cabrera-Alarcón, J. L., Muñoz-Hernández, M. D. M., Hernansanz-Agustín, P., Curtabbi, A., Domenech-Fernandez, J., Calvo, E., Vázquez, J., Serrano, A. L., Muñoz-Cánoves, P., Olaso-González, G., Enríquez, J. A., & Gómez-Cabrera, M. C. (2026). Mitochondrial remodeling in skeletal muscle underlies exercise-induced reversal of age-associated functional decline in mice and humans. Proceedings of the National Academy of Sciences of the United States of America, 123(14), e2508286123. https://doi.org/10.1073/pnas.2508286123
Guzman, S. D., Fraczek, P. M., Itsani, K., et al. (2026). Age-associated dysregulation of postsynaptic mitochondria perturbs reinnervation kinetics. Aging Cell, 25(1), e70355. https://doi.org/10.1111/acel.70355
Marcinek, D. J., & Ferrucci, L. (2025). Reduced oxidative capacity of skeletal muscle mitochondria IS a fundamental consequence of adult ageing. The Journal of Physiology, 603(1), 17–20. https://doi.org/10.1113/JP285040
Marzetti, E., Calvani, R., Coelho-Junior, H. J., Landi, F., & Picca, A. (2025). Defective mitochondrial quality control in the aging of skeletal muscle. Mechanisms of Ageing and Development, 228, 112112. https://doi.org/10.1016/j.mad.2025.112112